Abstract
Recent studies in the field of task switching have shown that humans can base expectancies for tasks on temporal cues. When tasks are predictable based on the duration of the preceding pre-target interval, humans implicitly adapt to this predictability, indicated by better performance in trials with validly compared to invalidly predicted tasks. Yet, it is not clear which internal timing mechanisms are involved. Previous research has suggested that intervals from the sub- and supra-second range are processed by distinct cognitive timing systems. As earlier studies on temporally predictable task switching have used predictive intervals stemming from both ranges, it was not yet clear if the time-based expectancy effect was driven by just one of the two internal timing systems. The present study used clearly sub-second intervals (10 ms and 500 ms) in Experiment 1, while clearly supra-second intervals (1500 ms and 3000 ms) were used in Experiment 2. Substantial adaptation effects were observed in both experiments, showing that sub- as well as supra-second timing systems are involved in time-based expectancies for tasks. The present findings offer important implications for our theoretical understanding of the internal timing mechanisms involved in time-based task expectancy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In many real-life multitasking scenarios, the time we have to wait for a task predicts to some degree which type of task will be required. Indeed, recent research has demonstrated that participants are able to learn time–task contingencies and to form time-based expectancies when time predicts the upcoming task (Aufschnaiter, Kiesel, Dreisbach, Wenke, & Thomaschke, 2018a; Aufschnaiter, Kiesel, & Thomaschke, 2018b). The studies by Aufschnaiter and colleagues showed that the predictive value of interval durations plays a crucial role for the cognitive processing of tasks in a task-switching scenario. However, until now, it is not yet clear, which internal timing mechanisms are involved in time-based expectancy for tasks. There is, for example, accumulating evidence from neuroscience, neuropharmacology, and behavioral psychology that sub-second and supra-second intervals are processed by different cognitive timing systems (Lewis & Miall, 2003a; Wiener, Turkeltaub, & Coslett, 2010). As the previous studies on time-based task expectancy included intervals from both ranges, it was not clear until now whether the time-based expectancy effect was driven by just one of the internal timing systems. The present study, in which a pair of clearly sub-second intervals (10 ms and 500 ms) was used in the first experiment, while a pair of clearly supra-second intervals (1500 ms and 3000 ms) was used in the second experiment, investigated, if time-based task expectancy is possible in the sub-second, as well as in the supra-second range.
Time-based expectancy in the task-switching paradigm
Recently, it could be shown, that not just mere stimulus–response events can be expected based on a preceding predictive interval (Thomaschke, Kiesel, & Hoffmann, 2011; Thomaschke, Wagener, Kiesel, & Hoffmann, 2011a, Thomaschke, Wagener, Kiesel, & Hoffmann, 2011b), but also cognitively more demanding tasks in a task-switching scenario (Aufschnaiter et al., 2018a). In contrast to time-based expectancy, which is the focus of the present study, and which means expecting what will happen at a certain point in time, time expectancy means expecting when something will happen, without knowing what will happen at this point in time. Time expectancy is usually investigated by manipulating the duration between warning signal and target stimulus (e.g., Los, & Agter, 2005; Los, Knol, & Boers, 2001; Steinborn, & Langner, 2012; Steinborn, Rolke, Bratzke, & Ulrich, 2008). Thus, time expectancy is conceptually different and can be defined as a prediction of the duration of an interval prior to an event (Thomaschke, Kunchulia, & Dreisbach, 2015). However, time expectancy is not the focus of the present study, and thus will not be further discussed here.
Time-based expectancy means expecting a certain event at a certain point in time. Several studies on time-based expectancy could show that when time predicts an upcoming stimulus–response event, participants are able to form time-based expectancies (Thomaschke et al., 2015). In the laboratory, time-based expectancy is investigated by applying an adapted variant of the foreperiod (FP) paradigm (e.g., Los & Horoufchin, 2011; Schröter, Birngruber, Bratzke, Miller, & Ulrich, 2015; Steinborn & Langner, 2011), namely, the time–event correlation paradigm, which was initially introduced by Wagener & Hoffmann (2010). In this paradigm, two targets and two pre-target intervals occur equally often, but one target is frequently (80%) combined with the short interval, while the other target is frequently (80%) combined with the long interval. Typically, a time-based expectancy effect can be observed, with faster responses in trials with frequent combinations of interval and target, compared to trials with infrequent combinations of interval and target (Wagener & Hoffmann, 2010; Thomaschke et al., 2011a, 2011b).
Recent studies revealed empirical evidence for time-based expectancy in various contexts: Time-based expectancy could be shown for motor responses (Thomaschke & Dreisbach, 2013; Volberg & Thomaschke, 2017), response conflict (Wendt & Kiesel, 2011), language processing (Roberts & Francis, 2013; Roberts, Margutti, & Tarkano, 2011), stimulus form (Thomaschke, Hoffmann, Haering, & Kiesel, 2016), and stimulus location (Rieth & Huber, 2013).
Importantly, as already mentioned above, recent studies revealed that not only events in single-task scenarios (see above), but also cognitively more demanding tasks in the task-switching paradigm can be expected based on preceding predictive intervals (Aufschnaiter et al., 2018a, b). Time-based expectancy for tasks is typically investigated by applying a combination of the above-mentioned time–event correlation paradigm (Wagener & Hoffmann, 2010) and the standard task-switching paradigm (for a review, see Kiesel et al., 2010).
Current theories on time-based expectancy assume associative learning of correlations between interval duration and target features as the central cognitive mechanism underlying the formation of time-based expectancy (see Thomaschke & Dreisbach, 2015). Thomaschke and Dreisbach (2015) recently developed an associative learning model, explaining the formation of the time-based expectancy effect in humans. The model is based on Machado’s (1997) and Los and colleagues’ (2001) accounts of temporal associative learning (for a detailed model description, see Thomaschke & Dreisbach, 2015). The model explains time-based expectancy by a cascade of temporal representations (i.e., successive neural activation states), which is triggered by the onset of a warning interval preceding a target stimulus. Whenever the requirement to execute Task A occurs shortly after a specific temporal representation has been passed, this entails the strengthening of the connection between this specific temporal representation and that neural population which generates expectancy for the requirement to execute Task A. Yet, we currently do not know whether these connections between temporal states and the expectancy-generating neural populations can account for the formation of time-based expectancy in the sub-second, as well as in the supra-second time range.
However, there is another model from the field of research on temporal cognition, which may also account for the time-based expectancy effect. The model implies that temporal processing may rely on time-dependent changes in neural network dynamics and is, therefore, called state-dependent network model (Buonomano, 2007; Karmarkar & Buonomano, 2007). It assumes that stimulus representations change over time, such that there is a time-dependent (temporal) signature for each processed stimulus. According to this model, the temporal signature of a preceding stimulus is responsible for whether neural networks fire in response to a second stimulus or not (cf. Buonomano, 2014). Thus, the state-dependent network model can account for the typical empirical finding in research on time-based expectancy, namely, that participants react significantly faster to targets frequently presented after a certain time interval, compared to targets infrequently presented after that time interval (cf. Aufschnaiter et al., 2018a). The encoding of each sensory stimulus feature (i.e., the target stimulus in the present study) is dependent on the temporal context of the previous encoding of sensory stimulus features (i.e., the duration of the fixation cross in the present study). Taking into consideration that time-based expectancy is often explained by the above-mentioned associative learning mechanism (cf. Thomaschke et al., 2011a; Thomaschke & Dreisbach, 2015), the learned association between the temporal context of the fixation cross, which serves as an implicit task cue, and the activation of specific neural networks, responding to one specific task, might lead to the effect of time-based task expectancy.
However, regarding temporal cognition involved in time-based expectancy, it could be only shown so far that time-based expectancy employs a relative, rather than an absolute representation of time (Aufschnaiter et al., 2018a, 2018b; Thomaschke et al., 2015). Until now, it is not yet clear, which internal timing mechanisms are involved in time-based expectancy for tasks, as there is empirical evidence pointing towards distinct timing mechanisms for the cognitive processing of intervals in the sub-second and supra-second range (cf. Rammsayer & Troche, 2014).
Neural mechanisms of timing
There are several theoretical approaches in the timing literature concerning the cognitive processing of intervals stemming from different time ranges. The perhaps most influencing theoretical model of timing, referred to as the internal clock model, was introduced in the early 1960s and relies on an internal, single pacemaker–accumulator clock (Creelman, 1962; Treisman, 1963; cf. Rammsayer & Ulrich, 2001). This notion of a single timing mechanism, based on neural counting, was long time thought to be underlying temporal processing of intervals irrespective of their duration (cf. Grondin, 2010). To date, however, little evidence has supported the view that biological clocks in the human brain rely on the counting of pulses or tics of a pacemaker. Instead, there is ongoing research in the area of psychophysical investigations on interval timing, trying to answer the question if one common timing mechanism or rather distinct timing mechanisms underlie the cognitive processing of interval timing in the sub-second and supra-second range (cf. Buonomano, 2014; Rammsayer & Troche, 2014).
In this context, several studies provided evidence for the distinct timing hypothesis. This hypothesis assumes that the processing of brief interval durations in the sub-second range is based upon an automatic, sensory mechanism, whereas the processing of longer interval durations in the supra-second range seems to be cognitively mediated (Hayashi, Kantele, Walsh, Carlson, & Kanai, 2014; Lewis & Miall, 2003b; Rammsayer & Lima, 1991; however, see Rammsayer, & Ulrich, 2005). The distinct timing hypothesis is further supported by neuroimaging studies (Lewis & Miall, 2003a, 2003b) and by neuropharmacological studies (Rammsayer, 2008, 2009; Wiener, Lohoff, & Coslett, 2011).
However, research has failed until now to reveal where and how temporal information is exactly processed in the human brain and empirical findings point towards the fact that the cognitive processing of time seems to be a rather complex interaction of many cortical and subcortical structures (cf. Merchant, & de Lafuente, 2014). Moreover, as already mentioned, psychological and neuroscientific research suggests that timing in the range of tens to hundreds of milliseconds relies on distinct neural mechanisms compared to timing in the range of seconds. Thus, there seem to be distinct neural timing mechanisms across different time scales (Buonomano, 2007), which, however, may be functionally related (Gooch, Wiener, Hamilton, & Coslett, 2011; Lewis & Miall, 2006; Merchant, Harrington, & Meck, 2013; Rammsayer & Troche, 2014; Wiener et al., 2010).
Research question
There is manifold empirical evidence for distinct neural systems processing sub-second and supra-second intervals in the human brain. These findings, stemming from the domain of behavioral, pharmacological and neuroscientific research, lead to the question if time-based expectancy in the task-switching paradigm can benefit equally well from different distinct neural timing mechanisms in the cognitive system.
Previous studies on temporally predictable task switching included predictive intervals from both the sub-second and the supra-second range (for example, 500 ms from the sub-second and 1500 ms from the supra-second range in the study by Aufschnaiter et al., 2018a). Consequently, it was not yet clear, whether the time-based expectancy effect was driven by just one of the two timing systems. In previous experiments, we mainly observed time-based expectancy effects for the short (sub-second) as well as for the long (supra-second) interval. Yet, based on this finding, we cannot conclude that participants formed time-based expectancies for both intervals. Instead, it might be that participants formed the expectancy for a specific task occurring after a specific time interval only for one interval (either the sub- or the supra-second one) and formed a negation for all other events, that is they expected “not this task” at “not this interval”.
In two experiments with a pair of clearly sub-second intervals (10 ms and 500 ms), and a pair of clearly supra-second intervals (1500 ms and 3000 ms), the present study investigated if the time-based task expectancy effect can still be found if both predictive pre-target intervals in a task-switching experiment either stem only from the sub-second (Experiment 1), or only from the supra-second time range (Experiment 2). Furthermore, the present study investigated, if the cognitive system still can use time-based expectancy in task switching, when the sub-second time range is narrowed (to 10 ms in Experiment 1), or, respectively, when the supra-second time range is broadened (up to 3 s in Experiment 2), compared to earlier studies on time-based task expectancy.
Experiment 1
To examine the question whether time-based expectancy for tasks can be observed in sub-second time ranges, a combination of the standard task-switching paradigm (cf. Kiesel et al., 2010) and the time–event correlation paradigm (Wagener & Hoffmann, 2010) was used. The procedure involved two different intervals (10 ms and 500 ms) and two different tasks (magnitude judgement task and parity judgement task). Each of the intervals predicted one of the tasks with 90% probability. We expected that participants learn the associations between interval and the task, thereby building up time-based task expectancies. These time-based expectancies should be observable by faster responses in trials with frequent combinations of interval and task, compared to trials with infrequent combinations of interval and task.
Method
Participants
34 participants (22 females; mean age = 24.15, SD = 5.03, range = 20–40 years; 32 right-handed) took part in the experiment in exchange for course credits or monetary compensation for their participation. All participants had normal or corrected-to-normal vision and were naïve concerning the hypotheses. Participants provided written informed consent prior to the experiment and were treated according to the ethical standards of the American Psychological Association. Sample size was determined according to the effect size reported in previous studies on time-based expectancy. These were on average medium (d = 0.5). Power analyses (1 − β = 0.8) yielded a minimum number of 27 participants.
Apparatus and stimuli
Participants responded by key presses on two response buttons on a serial response box (Psychology Software tools), which was centrally aligned in front of the computer screen. Target stimuli were yellow or blue numbers between 1 and 9, except 5, presented against a black background at a viewing distance of 50 cm. The size of the stimuli was approximately 8 × 5 mm. The fixation cross was the “+” symbol (Arial typeface, approximately 6 × 6 mm). All stimuli were presented centrally on the screen.
Procedure
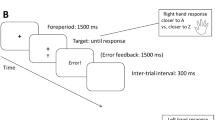
Each trial started with a blank screen for 300 ms (inter-trial interval, ITI), which was followed by the presentation of a fixation cross for a variable interval of either 10 ms or 500 ms. After this interval, the target stimulus was presented. Each stimulus occurred with equal probability. Depending on the color of the target stimulus, participants had to judge whether the displayed number was smaller or larger than 5, or whether it was odd or even. The mapping of colors to tasks was counterbalanced across participants. Responses were given with the same two buttons for both tasks. For the parity judgement task, the mapping of responses to keys was counterbalanced across participants. For the magnitude judgement task, responses to numbers smaller than five were always given with the left response button, while responses for numbers bigger than five were always given with the right response button (cf. we chose a SNARC compatible mapping; Dehaene, Bossini, & Giraux, 1993; Wood, Willmes, Nuerk, & Fischer, 2008). Participants were instructed to respond as fast and as correct as possible. After the detection of an error, the word Fehler! (German for “Error!”) was displayed in red on a black screen for 1500 ms. After correct responses, no explicit feedback was given. The duration of the interval predicted the upcoming task type in the current trial with 90% probability, which means that the short interval was followed by one task with 90% probability, while the long interval was followed by the other task with 90% probability. Both intervals and tasks appeared with same overall frequencies and the mapping of tasks to intervals was counterbalanced across participants. Participants were not informed that the intervals had different lengths, or that these interval lengths were correlated with tasks. Please note that the short interval was effectively undetectable for the participants, because the 10 ms duration of the fixation cross was too short to be consciously visible. Thus, participants experienced either a 500 ms fixation cross, or no fixation cross at all. The 10 ms fixation cross was included merely for reasons of methodological analogy between Experiments 1 and 2 (see below).
The experiment consisted of two sessions of 30 min each, which were tested on consecutive days. Both sessions of the experiment consisted of four blocks each: one learning block, and three test blocks. The only difference between learning blocks and test blocks was that after the detection of an error, the instruction was once again presented in silver font color on a black screen for 8000 ms in the learning blocks, before the next trial started with the presentation of the ITI. Each block comprised 120 trials. Given that both tasks occurred equally often, each task type appeared in 60 trials of one block. As the duration of the pre-target interval predicted the upcoming task type with 90% probability, each task type was validly predicted by the pre-target interval in 54 of the 60 trials and was invalidly predicted by the pre-target interval in 6 trials. Between blocks, participants could take a break, which they could terminate themselves by pressing the spacebar. After the second session of the experiment, participants were asked by the experimenter if they had noticed any temporal regularities in the experiment.
Results
Following earlier studies on time-based expectancy, we analyzed only the second session (e.g., Thomaschke & Dreisbach, 2013). Data from the learning blocks, from the first three trials of each test block, as well as trials with number repetitions and trials following an error trial were excluded from analyses. In addition, we excluded trials with reaction times (RTs) < 100 ms from analyses and we removed trials, in which RTs deviated more than 3 standard deviations from the corresponding cell mean, computed separately for each participant, block and experimental condition (Bush, Hess, & Wolford, 1993). Furthermore, trials with errors were removed from the RT analyses. Three-factor repeated measures ANOVAs with the factors interval (10 vs. 500 ms), transition (switch vs. repetition), and predictability of interval–task combination (predictable vs. unpredictable; referred to as predictability from hereon) were conducted separately for RTs and error rates.
One participant was removed from the sample due to high error rates (mean percentage of errors: 21.71%; mean percentage of errors for all participants: 4.39%). This resulted in a final sample of 33 participants.
RTs and error rates are shown in Fig. 1. For mean RTs and SD of each factor combination, see Table 1. With regard to RTs, the main effects for transition and predictability were significant. Responses to task repetitions (M = 765 ms, SD = 112) were faster than to task switches (M = 900 ms, SD = 156), F (1, 32) = 53.895, p < .001, ηp2 = 0.627. Furthermore, responses in trials with predictable combinations of interval and task (M = 807 ms, SD = 120) were significantly faster than in trials with unpredictable combinations of interval and task (M = 858 ms, SD = 149), F (1, 32) = 8.032, p = .008, ηp2 = 0.201. The only significant interaction for RTs was between interval and transition, F (1, 32) = 8.977, p = .005, ηp2 = 0.219, meaning that switch costs were higher in trials with the short interval (Δ = 166 ms) compared to trials with the long interval (Δ = 105 ms). The interaction between interval and predictability, F (1, 32) = 1.424, p = .241, ηp2 = 0.043, as well as the interaction between transition and predictability did not reach significance, F < 1. For a complete overview of the statistical results of the reported ANOVA, see Table 2.
Main results of Experiment 1: Mean reaction times (RTs in ms; lines) and percentages of errors (PEs in %; bars), depending on predictability of interval–task combination, are displayed separately for task transition (task repetition vs. task switch) and interval duration (10 ms vs. 500 ms). Error bars represent 1 standard error of the mean
Given the non-significant interaction between transition and predictability, we next performed a Bayesian repeated measures ANOVA with default prior scales using JASP (JASP team, version 0.8.1.1). The Bayesian approach is a model selection procedure that indicates the likelihood ratio of two or more hypotheses based on the given data. Thus, Bayesian analysis provides the possibility of evaluating evidence in favor of the (null-) hypothesis. In this context, the Bayes-factor (BF) indicates how strong the data is in favor of the (null-) hypothesis, with the convention that a BF between 1 and 3 indicates anecdotal evidence, a BF between 3 and 10 moderate evidence, and a BF above 10 strong evidence for a (null-)hypothesis (Lee & Wagenmakers, 2013). Our Bayesian analyses showed that the null-hypothesis model for the interaction between transition and predictability (no difference of time-based task expectancy between-task repetitions and task switches) was > 4 times as likely as an interaction (BF = 4.684). Furthermore, Bayesian analysis remained inconclusive regarding the null-hypothesis model for the interaction between interval and predictability (no difference of time-based task expectancy between short and long interval) (BF in favor of the null-hypothesis = 2.97; BF in favor of the alternative hypothesis = 0.33).
With regard to error rates, no main effect or interaction reached significance. For mean error rates and SD of each factor combination, see Table 3.
Discussion
In Experiment 1, we investigated if time-based task predictability affects behavior in a task-switching scenario when predictive intervals from the sub-second time range are used. The results showed that participants responded significantly faster in trials with predictable combinations of interval and task compared to trials with unpredictable combinations of interval and task. This means that participants were able to form time-based task expectancies and to expect one task after the short interval (effectively 0 ms) and the other task after the long interval. Most importantly, this time-based task expectancy effect was independent from the absolute duration of the predictive interval and was even observed in trials with a predictive interval of 10 ms. One may conjecture that an interval of 10 ms - or effectively zero - is too short to build up expectations for a task. However, we assume that participants start building up expectancy before the beginning of the pre-target interval. Participants seem to use the ITI of 300 ms to start the trial with a high degree of expectancy for a specific task and thus the expectancy for the task which is associated with the short interval does not seem to require any additional time to be built up. Moreover, participants responded significantly faster in trials with a task repetition compared to trials with a task switch which reflects the typical switch costs in task switching (see also Kiesel et al., 2010; Koch, Poljac, Müller, & Kiesel, 2018).
The fact that we did not find any interaction between transition and predictability, suggests that cognitive processes seem to benefit from time-based expectancy, that take place in both switch and repetition trials (see also Dreisbach, Haider, & Kluwe, 2002; Koch, 2003, 2005). This finding was further supported by a Bayesian analysis.
Furthermore, none of the participants noticed any temporal regularity during the whole experiment.
Thus, the results of Experiment 1 confirmed that the effect of time-based expectancy in task switching can be observed with predictive intervals entirely from the sub-second time range (10 and 500 ms). In order to investigate if time-based expectancy can be also observed with predictive intervals from the supra-second time range, we conducted Experiment 2.
Experiment 2
The purpose of Experiment 2 was to test whether time-based predictability also affects behavior in a task-switching scenario when predictive intervals only from the supra-second time range are used (1500 and 3000 ms). Again, we expected faster responses in trials with predictable combinations of interval and task compared to trials with unpredictable combinations of interval and task.
Method
Participants
34 participants (29 females; mean age = 22.47, SD = 3.79, range = 19–37 years; 31 right-handed) took part in the experiment. Participants again received course credits or monetary compensation for their participation. All participants fulfilled the same criteria as in Experiment 1.
Apparatus, stimuli, and procedure
Apparatus, stimuli and procedure were the same as in Experiment 1, with the exception that now only intervals from the supra-second time range (1500 and 3000 ms) were used which predicted the upcoming task in the current trial with 90% probability.
Results
Data preprocessing was the same as in Experiment 1. RT and PE analysis were also conducted as in Experiment 1. RTs and error rates are shown in Fig. 2. For mean RTs and SD of each factor combination, see Table 4. With regard to RTs, only the main effects for transition and predictability were significant. Responses to task repetitions (M = 899 ms, SD = 214) were faster than to task switches (M = 960 ms, SD = 271), F (1, 33) = 19.168, p < .001, ηp2 = 0.367. In trials with predictable combinations of interval and task (M = 907 ms, SD = 235), responses were significantly faster than in trials with unpredictable combinations of interval and task (M = 952 ms, SD = 262), F (1, 33) = 4.147, p = .050, ηp2 = 0.112. The interaction between interval duration and predictability, F (1, 33) = 2.642, p = .114, ηp2 = 0.074, as well as the interaction between transition and predictability, F < 1, did not reach significance. For a complete overview of the statistical results of the reported ANOVA, see Table 5.
Main results of Experiment 2: Mean reaction times (RTs in ms; lines) and percentages of errors (PEs in %; bars), depending on predictability of interval–task combination, are displayed separately for task transition (task repetition vs. task switch) and interval duration (1500 ms vs. 3000 ms). Error bars represent 1 standard error of the mean
A Bayesian repeated measures ANOVA revealed that the null-hypothesis model for the interaction between transition and predictability (no difference of time-based task expectancy between-task repetitions and task switches) was > 4 times as likely as a difference hypothesis (BF = 4.727). Moreover, like in Experiment 1, Bayesian analysis remained inconclusive regarding the null-hypothesis model for the interaction between interval and predictability (no difference of time-based task expectancy between short and long interval) (BF in favor of the null-hypothesis = 1.88; BF in favor of the alternative hypothesis = 0.53). No other main effect or interaction reached significance.
With regard to error rates, no main effect or interaction was significant. For mean error rates and SD of each factor combination, see Table 6.
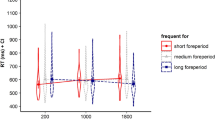
In order to investigate, if experiment interacted with interval duration, task transition or predictability, a mixed measures ANOVA with the between-subjects factor experiment and the within-subject factors interval duration, transition and predictability was conducted. As the absolute durations of the predictive intervals differed between experiments, we coded interval duration in relative terms either as “short” (10 ms in Experiment 1 and 1500 ms in Experiment 2) or “long” (500 ms in Experiment 1 and 3000 ms in Experiment 2). RTs and error rates are shown in Fig. 3. There was a significant interaction between transition and experiment, F (1, 65) = 10.450, p = .002, ηp2 = 0.138, meaning that switch costs were larger in Experiment 1 (Δ = 136 ms) than in Experiment 2 (Δ = 61 ms). Furthermore, results showed that neither the effect of interval duration, F (1, 65) < 1, nor the effect of predictability, F (1, 65) < 1, differed between experiments. Bayesian analyses revealed that the null-hypothesis model for the interaction between predictability and experiment (no difference of time-based task expectancy between the two experiments) was > 5 times as likely as a difference hypothesis (BF = 5.567). Moreover, our results revealed a significant three-way interaction between interval duration, predictability and experiment, F (1, 65) = 4.024, p = .049, ηp2 = 0.058. Visual inspection of Figs. 1 and 2 suggests that time-based task expectancy is larger for the long (500 ms) interval in Experiment 1 and the short (1500 ms) interval in Experiment 2 then for the resp. other intervals.
Main results of a cross-experiment analysis: Mean reaction times (RTs in ms; lines) and percentages of errors (PEs in %; bars), depending on predictability of interval–task combination, are displayed separately for experiment and task transition (task repetition vs. task switch). Error bars represent 1 standard error of the mean
Furthermore, the interaction between transition, predictability and experiment did not gain significance, F (1, 65) < 1. Bayesian repeated measures ANOVA revealed that the null-hypothesis for the interaction between transition, predictability and experiment (no difference of time-based task expectancy for task repetitions and task switches between the two reported experiments) was > 4 times as likely as a difference hypothesis (BF = 4.943). In addition, we found a main effect for experiment, F (1, 65) = 4.259, p = .043, ηp2 = 0.061, meaning that participants responded faster in Experiment 1 (M = 832 ms, SD = 274) compared to Experiment 2 (M = 929 ms, SD = 270).
With regard to error rates, no main effect or interaction gained significance in the cross-experiment analysis. Importantly, participants did not notice any temporal regularity in none of the reported experiments.
Discussion
Experiment 2 investigated if the effect of time-based task expectancy is also observable with predictive intervals from the supra-second range (1500 and 3000 ms). The results revealed a significant time-based expectancy effect, which means that participants responded significantly faster in trials with predictable combinations of interval and task compared to trials with unpredictable combinations of interval and task. Like in Experiment 1, participants responded again significantly faster in trials with a task repetition compared to trials with a task switch and thus showed the typical task-switching costs.
The results of the cross-experiment analysis revealed that the time-based task expectancy effect did not differ between predictive intervals from the sub-second range (see Experiment 1) and predictive intervals from the supra-second range (see Experiment 2). Bayesian analysis further supported this finding. Moreover, we did not find any interaction between expectancy, transition and experiment. Thus, it seems as if switches do not benefit more from time-based expectancy when the pre-target intervals are very short (see Experiment 1).
General discussion
The present study investigated whether the time-based task expectancy effect (Aufschnaiter et al., 2018a, b) can be observed for both sub-second (10 or 500 ms; Experiment 1) and supra-second (1500 or 3000 ms; Experiment 2) predictive intervals. Results clearly indicated that time-based expectancy for tasks is possible in the sub- as well as in the supra-second range.
Most importantly, the time-based task expectancy effect was independent from the absolute duration of the predictive interval in both experiments and was even observable in trials with a predictive interval of only 10 ms in Experiment 1 Thus, the results of Experiment 1 indicate that flexible task-specific time-based preparation does not require much time as the expectancy for the task associated with the short (10 ms) interval turns immediately into expectancy for the task associated with the long interval (500 ms) when no stimulus is presented at the beginning of the trial. We conjecture that participants build up their expectancy for the task associated with the 10 ms interval already during the ITI of 300 ms. However, in the present experiment we cannot exactly assess at which point in time during the interval of 500 ms expectancy changes towards the other task. But the fact that the expectancy effect is observable for both predictive intervals in Experiment 1 reflects a highly dynamic and fast updating of time-based expectancy. This implies rather flexible task-specific preparation during a very short and narrow temporal course of the pre-target interval. However, even though the longer absolute durations of the predictive intervals in Experiment 2 prevent perfect preparation for the upcoming task at both critical moments, which is mirrored in overall slower response times in Experiment 2 (compared to Experiment 1), we still find the time-based expectancy effect for both the short and the long interval duration in Experiment 2 This implies that an effective and dynamic updating of time-based expectancy towards the other task can also take place in the supra-second time range, if no target has been presented after 1500 ms.
Although no significant interaction between predictability and interval duration could be found in any of the reported experiments, a cross-experiment analysis revealed a significant interval × predictability × experiment interaction: Across the two reported experiments, the time-based expectancy effect seems smaller in trials with the very short interval of 10 ms in Experiment 1 (Δ = 34 ms), as well as in trials with a very long interval of 3000 ms in Experiment 2 (Δ = 14 ms) compared to predictive pre-target intervals of 500 ms in Experiment 1 (Δ = 67 ms) and 1500 ms in Experiment 2 (Δ = 76 ms). The finding that across the two experiments, the expectancy effect is initially small at 10 ms, becomes substantial at 500 and 1500 ms and then decreases again at 3000 ms might be interpreted in the light of time uncertainty. The aspect of time uncertainty, which may have caused the effect of time-based task expectancy to be (at least descriptively) small for the 3000 ms interval (cf. Klemmer, 1956), leads to an interesting implication regarding future research on the topic of time-based task expectancy in different timing ranges: What would happen if predictive intervals, stemming from the supra-second range, of an even longer duration (for example 5000 ms) would be used in the task-switching paradigm? Does a temporal limit exist, beyond which it is not possible any longer for the human brain to process the informative value of a predictive interval’s duration? Further research is needed to assess whether the descriptive difference on time-based expectancy is a valid, meaningful result in the first place. If so, it would be interesting to assess in which time limits the formation of time-based expectancy for tasks is possible. Moreover, regarding temporal conditions in the context of building up and making use of time-based task expectancy, it would be interesting to further investigate time-based task expectancy in the context of hierarchical task switching (see Schneider & Logan, 2006). Is it, for example, possible to expect entire task sequences based on predictive time intervals and to show improved performance at the task level given such validly predicted sequences? In order to investigate the functional relationship between cognitive processes on the sequence level and on the task level, it would be interesting to explore the impact of invalidly predicted task sequences on cognitive processes associated with transitions on the task level.Footnote 1
Additionally, the results of Experiment 1 of the present study can be seen as an extension of the literature on time-based expectancy in general by replicating and extending the finding of Thomaschke et al. (2011a), who revealed that time-based expectancy can be built with short inter-FP spans (200 ms in the study by Thomaschke et al., 2011a; 490 ms in Experiment 1 of the present study) in very short FP regions (300 ms in the study by Thomaschke et al., 2011a; 10 ms in Experiment 1 of the present study). Thomaschke et al. (2011a) revealed in their study that event-specific temporal expectancy exists for a broad range of predictive intervals, ranging from at least 200 ms up to 1500 ms. The results of the present study extend these findings by showing that time-based expectancy can be built for complete task sets within the temporal range of 10 ms up to 3000 ms.
Furthermore, the fact that we did not find any interaction between transition and predictability in either of the two reported experiments, suggests that cognitive processes in both switch and repetition trials seem to benefit equally from time-based expectancy (see also Aufschnaiter et al., 2018a; Dreisbach et al., 2002). This finding was further supported by a Bayesian analysis. Another interesting finding of the present experiments is the fact that switch costs differed significantly between experiments with larger switch costs in Experiment 1 (136 ms) compared to Experiment 2 (61 ms). This finding is consistent with previous studies on task switching, which found that switch costs decreased with increasing response–cue intervals (RCI) (Altmann, 2005; Koch, 2001). The finding of decreased switch costs in Experiment 2, in which pre-target interval durations from the supra-second time range were used, can, therefore, be interpreted in the context of a passive decay account, which means that the activation of the previous executed task may decay over time (however, see Rogers & Monsell, 1995).
Please note that the cross-experimental analysis did not reveal any interaction between predictability, transition and experiment, which means that switches do not benefit more from time-based expectancy when the pre-target intervals stem from the sub-second range (Experiment 1) than when they stem from the supra-second range (Experiment 2). This finding is rather surprising as previous studies could show that task switches benefit much more from extending the cue–stimulus interval (CSI) than task repetitions, which is in general explained by task preparation/task reconfiguration processes that are required to a larger extent for task switches than task repetitions (see Kiesel et al., 2010). So one could have predicted that switches benefit more from time-based expectancy in trials with very short pre-target intervals, compared to task repetitions. In contrast, another prediction could have been that the temporal window, in which the participant is sufficiently confident about the predictive interval being clearly short, or being clearly long might be too narrow for switch preparations and only wide enough for fast repetition preparations in Experiment 1. However, the present study revealed that there seems to be no difference concerning time-based task expectancy in switch trials between the two reported experiments.
Importantly, as the time-based task expectancy effect did not differ between the two reported experiments, it can be concluded that time-based expectancy for tasks in the task-switching paradigm does not seem to differ between sub- and supra-second time ranges. However, please note that the time-based task expectancy effect, at least descriptively, is smaller with increasing intervals, but nevertheless is still observable within a temporal range up to 3000 ms. This interesting finding can be interpreted in the context of studies from the field of time expectancy, which revealed that a shift of the warning signal modality facilitated preparation at short FPs (1000 ms) but not at long FPs (3000 ms; Steinborn, Rolke, Bratzke, & Ulrich, 2009, 2010). The authors concluded that time expectancy at short FPs is triggered by sensory aftereffects of the onset of the warning signal, whereas time expectancy at long FPs above 1000 ms seems to be conceptually driven. Future studies on time-based task expectancy should further investigate the reported attenuation of the time-based expectancy effect given increased pre-target intervals (up to 3000 ms in Experiment 2) in the context of sensory-driven vs. conceptually driven mechanisms underlying temporal expectations.
Please note that overall, RTs were slower in Experiment 2 compared to Experiment 1. This could have several explanations: First, the slower RTs in Experiment 2 could reflect that temporal conditions in Experiment 2 were less favorable than in Experiment 1. In Experiment 1, participants seem to use the ITI of 300 ms to prepare for the first critical moment of a potentially appearing task at 10 ms after the ITI (see Los & Schut, 2008) and, if no target is presented at the first critical moment, switch online to prepare for the other task at the second critical moment of 500 ms after the ITI. Please note that in both presented experiments, the first critical moment (i.e., the short pre-target interval) has to be cognitively represented to prompt task-set activation of the resp. task in case the target occurs, or it has to be available as a contextual cue to switch expectancy and to activate the resp. task-set for the other task, in case no target has occurred after the short interval. Importantly, the cognitive processing of the predictive intervals occurs without participants’ consciousness, as time-based expectancy is an implicit phenomenon. In this context, the temporal interval acts as an automatic retrieval cue, as the passing of a specific temporal state automatically activates the corresponding expectancy-generating neural populations (see associative learning model developed by Thomaschke & Dreisbach, 2015). This strengthened connection between specific temporal states and expectancy-generating neural populations might be stored in (non-declarative) long-term memory.
As the absolute durations of the intervals are much longer in Experiment 2, which leads to less favorable temporal conditions compared to Experiment 1, participants are not able to perform optimally at the two critical moments of target presentation (at 1500 ms and 3000 ms after the ITI; see also Näätänen, Muranen, & Merisalo, 1974). These less-than-optimal temporal conditions may lead to overall reduced RTs compared to Experiment 1. Second, the overall slower response times in Experiment 2, compared to Experiment 1, could also be due to failure-to-engage trials during preparatory processes, which become more likely with increasing interval duration (cf. Hohle, 1965). It is conceivable, that participants fail to prepare the task that is validly predicted by the duration of the pre-target interval in some trials. Consequently, response times in valid conditions would be a mixture of prepared (fast) and unprepared (slow) task performances (see De Jong, 2000; Nieuwenhuis & Monsell, 2002, for similar considerations on task switch costs). If such failures to engage are more likely for longer interval durations, an interesting question is how to counteract these failures. Future research should investigate, whether the increase of cognitive efficiency, for example via explicit instructions (cf. Steinborn, Langner, & Huestegge, 2017) can lead to faster overall response times in the time–event correlation paradigm with predictive intervals of long absolute durations from the supra-second time range. However, the overall slower response times in Experiment 2, compared to Experiment 1, could be also due to the fact that the targets were presented in a lower overall frequency in Experiment 2, compared to Experiment 1. In animal trace-conditioning research, it could be shown that the pecking responses of pigeons are facilitated in experimental blocks with high response frequencies, compared to experimental blocks with low response frequencies (cf. Smith, 1974). Thus, the response tendencies of the participants in Experiment 1 might have been increased given the overall higher frequencies of target presentation and response rates, due to the short pre-target intervals of 10 ms and 500 ms.
Regarding future research on the topic of time-based task expectancy in different timing ranges and considering state-dependent neural network models, the question arises if the effect of time-based expectancy in the task-switching paradigm would be still observable, if the fixation cross would not mark the predictive interval, but if only the ITI would be marked by a fixation cross, and the predictive interval would only be observable by an empty screen. In the latter case, participants would have to compare the duration between the fixation cross and the imperative target stimulus, whereas in the former case, participants have to estimate the duration of the fixation cross (as it is the case in the present experiments). In case of a fixation cross, which marks the predictive interval and, therefore, is continuously displayed before the presentation of the target stimulus, there is supposed to be a complex interaction between the current input pattern and the current activity of the neural network (cf. Buonomano, 2014). However, if only the ITI is marked by a fixation cross for 300 ms and the predictive interval is marked by an empty screen before target presentation, the incoming sensory information about the fixation cross and subsequently the incoming sensory information about the target stimulus may lead to the activation of distinct patterns of neuronal cycles, as the synaptic strengths may differ over time. Given these interesting different implications provided by the model of state-dependent neural networks, future research on time-based task expectancy in different timing ranges should determine whether the effect of filled or empty predictive intervals has an influence on the encoding of duration by neural networks.
To conclude, the results of the present study show that humans seem to be highly flexible concerning the expectation of tasks across different time ranges, stemming from both the sub- and the supra-second range. As the time-based task expectancy effect is still observable within a temporal range up to 3 s, the results of the present study can be interpreted as a replication and important extension of previous work on time-based task expectancy by revealing that time-based expectancy in a task-switching paradigm seems to benefit equally well from different distinct neural timing systems in the human brain.
Notes
Concerning time-based expectancy in the context of ordered task sequences, it should be noted that a recent study (Mittelstädt, Kiesel, Fischer, Rieger and Thomaschke, in revision) investigated time-based expectancy in a dual-task paradigm. The authors found that the backward-compatibility effect between tasks was reduced when incompatible dual-task trials were predicted by one of two possible FPs with a high degree of probability.
References
Altmann, E. M. (2005). Repetition priming in task switching: Do the benefits dissipate? Psychonomic Bulletin & Review, 12, 535–540.
Aufschnaiter, S., Kiesel, A., Dreisbach, G., Wenke, D., & Thomaschke, R. (2018a). Time-based expectancy in temporally structured task switching. Journal of Experimental Psychology: Human Perception and Performance, 44(6), 856–870.
Aufschnaiter, S., Kiesel, A., & Thomaschke, R. (2018b). Transfer of time-based task expectancy across different timing environments. Psychological Research, 82(1), 230–243.
Buonomano, D. V. (2007). The biology of time across different scales. Nature Chemical Biology, 3(10), 594–597.
Buonomano, D. V. (2014). The neural mechanisms of timing on short timescales. In V. Arstila, & D. Lloyd (Ed.), Subjective time: The philosophy, psychology, and neuroscience of temporality (pp. 329–342). Cambridge: MIT.
Bush, L. K., Hess, U., & Wolford, G. (1993). Transformations for within-subject designs: A Monte Carlo investigation. Psychological Bulletin, 113(3), 566–579.
Creelman, C. D. (1962). Human discrimination of auditory duration. The Journal of the Acoustical Society of America, 34(5), 582–593.
De Jong, R. (2000). An intention-activation account of residual switch costs. In S. Monsell & J. Driver (Eds.), Control of cognitive processes: Attention and performance XVIII (pp. 357–376). Cambridge: MIT.
Dehaene, S., Bossini, S., & Giraux, P. (1993). The mental representation of parity and number magnitude. Journal of Experimental Psychology: General, 122(3), 371–396.
Dreisbach, G., Haider, H., & Kluwe, R. H. (2002). Preparatory processes in the Task-Switching paradigm: Evidence from the use of probability cues. Journal of Experimental Psychology: Learning, Memory, and Cognition, 28, 468–483.
Gooch, C. M., Wiener, M., Hamilton, A. C., & Coslett, H. (2011). Temporal discrimination of sub-and suprasecond time intervals: A voxel-based lesion mapping analysis. Frontiers in Integrative Neuroscience, 5, 59.
Grondin, S. (2010). Timing and time perception: A review of recent behavioral and neuroscience findings and theoretical directions. Attention, Perception, & Psychophysics, 72(3), 561–582.
Hayashi, M. J., Kantele, M., Walsh, V., Carlson, S., & Kanai, R. (2014). Dissociable neuroanatomical correlates of subsecond and suprasecond time perception. Journal of Cognitive Neuroscience, 26(8), 1685–1693.
Hohle, R. H. (1965). Inferred components of reaction times as functions of foreperiod duration. Journal of Experimental Psychology, 69(4), 382–386.
Karmarkar, U. R., & Buonomano, D. V. (2007). Timing in the absence of clocks: Encoding time in neural network states. Neuron, 53(3), 427–438.
Kiesel, A., Steinhauser, M., Wendt, M., Falkenstein, M., Jost, K., Philipp, A. M., & Koch, I. (2010). Control and interference in task switching—A review. Psychological Bulletin, 136(5), 849–874.
Klemmer, E. T. (1956). Time uncertainty in simple reaction time. Journal of Experimental Psychology, 51(3), 179–184.
Koch, I. (2001). Automatic and intentional activation of task sets. Journal of Experimental Psychology: Learning, Memory, and Cognition, 27, 1474–1486.
Koch, I. (2003). The role of external cues for endogenous advance reconfiguration in task switching. Psychonomic Bulletin & Review, 10, 488–492.
Koch, I. (2005). Sequential task predictability in task switching. Psychonomic Bulletin & Review, 12, 107–112.
Koch, I., Poljac, E., Müller, H., & Kiesel, A. (2018). Cognitive structure, flexibility, and plasticity in human multitasking—An integrative review of dual-task and task-switching research. Psychological Bulletin, 144, 557–583.
Lee, M. D., & Wagenmakers, E. J. (2013). Bayesian data analysis for cognitive science: A practical course. New York: Cambridge University Press.
Lewis, P. A., & Miall, R. C. (2003a). Brain activation patterns during measurement of sub-and supra-second intervals. Neuropsychologia, 41(12), 1583–1592.
Lewis, P. A., & Miall, R. C. (2003b). Distinct systems for automatic and cognitively controlled time measurement: Evidence from neuroimaging. Current Opinion in Neurobiology, 13(2), 250–255.
Lewis, P. A., & Miall, R. C. (2006). A right hemispheric prefrontal system for cognitive time measurement. Behavioural Processes, 71(2–3), 226–234.
Los, S. A., & Agter, F. (2005). Reweighting sequential effects across different distributions of foreperiods: Segregating elementary contributions to nonspecific preparation. Perception and Psychophysics, 67(7), 1161–1170.
Los, S. A., & Horoufchin, H. (2011). Dissociative patterns of foreperiod effects in temporal discrimination and reaction time tasks. Quarterly Journal of Experimental Psychology, 64(5), 1009–1020.
Los, S. A., Knol, D. L., & Boers, R. M. (2001). The foreperiod effect revisited: Conditioning as a basis for nonspecific preparation. Acta Psychologica, 106, 121–145.
Los, S. A., & Schut, M. L. (2008). The effective time course of preparation. Cognitive Psychology, 57(1), 20–55.
Machado, A. (1997). Learning the temporal dynamics of behavior. Psychological Review, 104, 241–265.
Merchant, H., & de Lafuente, V. (2014). Introduction to the neurobiology of interval timing. In H. Merchant & V. de Lafuente (Eds.), Neurobiology of interval timing (pp. 33–47). New York: Springer.
Merchant, H., Harrington, D. L., & Meck, W. H. (2013). Neural basis of the perception and estimation of time. Annual Review of Neuroscience, 36, 313–336.
Mittelstädt, V., Kiesel, A., Fischer, R., Rieger, T., & Thomaschke, R. (in revision). Temporal predictability of between-task interference in dual-tasking. Foreperiods as contextual cues modulate the backward compatibility effect.
Näätänen, R., Muranen, V., & Merisalo, A. (1974). Timing of expectancy peak in simple reaction time situation. Acta Psychologica, 38(6), 461–470.
Nieuwenhuis, S., & Monsell, S. (2002). Residual costs in task switching: Testing the failure-to-engage hypothesis. Psychonomic Bulletin & Review, 9, 86–92.
Rammsayer, T. (2008). Neuropharmalogical approaches to human timing. In S. Grondin (Ed.), Psychology of time (pp. 295–320). Bingley: Emerald.
Rammsayer, T. (2009). Effects of pharmacologically induced dopamine-receptor stimulation on human temporal information processing. NeuroQuantology, 7(1), 103–113.
Rammsayer, T., & Ulrich, R. (2001). Counting models of temporal discrimination. Psychonomic Bulletin & Review, 8(2), 270–277.
Rammsayer, T., & Ulrich, R. (2005). No evidence for qualitative differences in the processing of short and long temporal intervals. Acta Psychologica, 120(2), 141–171.
Rammsayer, T. H., & Lima, S. D. (1991). Duration discrimination of filled and empty auditory intervals: Cognitive and perceptual factors. Perception & Psychophysics, 50(6), 565–574.
Rammsayer, T. H., & Troche, S. J. (2014). Elucidating the internal structure of psychophysical timing performance in the sub-second and second range by utilizing confirmatory factor analysis. In H. Merchant & V. de Lafuente (Eds.), Neurobiology of interval timing (pp. 33–47). New York: Springer.
Rieth, C. A., & Huber, D. E. (2013). Implicit learning of spatiotemporal contingencies in spatial cueing. Journal of experimental psychology: Human Perception and Performance, 39(4), 1165–1180.
Roberts, F., & Francis, A. L. (2013). Identifying a temporal threshold of tolerance for silent gaps after requests. The Journal of the Acoustical Society of America, 133(6), 471–477.
Roberts, F., Margutti, P., & Takano, S. (2011). Judgments concerning the valence of inter-turn silence across speakers of American English, Italian, and Japanese. Discourse Processes, 48(5), 331–354.
Rogers, R. D., & Monsell, S. (1995). Costs of a predictable switch between simple cognitive tasks. Journal of Experimental Psychology: General, 124, 207–231.
Schneider, D. W., & Logan, G. D. (2006). Hierarchical control of cognitive processes: Switching tasks in sequences. Journal of Experimental Psychology: General, 135(4), 623–640.
Schröter, H., Birngruber, T., Bratzke, D., Miller, J., & Ulrich, R. (2015). Task predictability influences the variable foreperiod effect: Evidence of task-specific temporal preparation. Psychological Research, 79(2), 230–237.
Smith, J. B. (1974). Effects of response rate, reinforcement frequency, and the duration of a stimulus preceding response-independent food. Journal of the Experimental Analysis of Behavior, 21(2), 215–221.
Steinborn, M. B., & Langner, R. (2011). Distraction by irrelevant sound during foreperiods selectively impairs temporal preparation. Acta Psychologica, 136(3), 405–418.
Steinborn, M. B., & Langner, R. (2012). Arousal modulates temporal preparation under increased time uncertainty: Evidence from higher-order sequential foreperiod effects. Acta Psychologica, 139(1), 65–76.
Steinborn, M. B., Langner, R., & Huestegge, L. (2017). Mobilizing cognition for speeded action: Try-harder instructions promote motivated readiness in the constant-foreperiod paradigm. Psychological research, 81(6), 1135–1151.
Steinborn, M. B., Rolke, B., Bratzke, D., & Ulrich, R. (2008). Sequential effects within a short foreperiod context: Evidence for the conditioning account of temporal preparation. Acta Psychologica, 129(2), 297–307.
Steinborn, M. B., Rolke, B., Bratzke, D., & Ulrich, R. (2009). Dynamic adjustment of temporal preparation: Shifting warning signal modality attenuates the sequential foreperiod effect. Acta Psychologica, 132(1), 40–47.
Steinborn, M. B., Rolke, B., Bratzke, D., & Ulrich, R. (2010). The effect of a cross-trial shift of auditory warning signals on the sequential foreperiod effect. Acta Psychologica, 134(1), 94–104.
Thomaschke, R., & Dreisbach, G. (2013). Temporal predictability facilitates action, not perception. Psychological Science, 24(7), 1335–1340.
Thomaschke, R., & Dreisbach, G. (2015). The time-event correlation effect is due to temporal expectancy, not to partial transition costs. Journal of Experimental Psychology: Human Perception and Performance, 41(1), 196–218.
Thomaschke, R., Hoffmann, J., Haering, C., & Kiesel, A. (2016). Time-based expectancy for task relevant stimulus features. Timing & Time Perception, 4, 248–270.
Thomaschke, R., Kiesel, A., & Hoffmann, J. (2011). Response specific temporal expectancy: Evidence from a variable foreperiod paradigm. Attention, Perception, & Psychophysics, 73, 2309–2322.
Thomaschke, R., Kunchulia, M., & Dreisbach, G. (2015). Time-based event expectations employ relative, not absolute, representations of time. Psychonomic Bulletin & Review, 22, 890–895.
Thomaschke, R., Wagener, A., Kiesel, A., & Hoffmann, J. (2011a). The scope and precision of specific temporal expectancy: Evidence from a variable foreperiod paradigm. Attention, Perception, & Psychophysics, 73, 953–964.
Thomaschke, R., Wagener, A., Kiesel, A., & Hoffmann, J. (2011b). The specificity of temporal expectancy: Evidence from a variable foreperiod paradigm. The Quarterly Journal of Experimental Psychology, 64, 2289–2300.
Treisman, M. (1963). Temporal discrimination and the indifference interval: Implications for a model of the “internal clock”. Psychological Monographs: General and Applied, 77(13), 1–31.
Volberg, G., & Thomaschke, R. (2017). Time-based expectations entail preparatory motor activity. Cortex, 92, 261–270.
Wagener, A., & Hoffmann, J. (2010). Temporal cueing of target-identity and target-location. Experimental Psychology, 57(6), 436–445.
Wendt, M., & Kiesel, A. (2011). Conflict adaptation in time: Foreperiods as contextual cues for attentional adjustment. Psychonomic Bulletin & Review, 18(5), 910–916.
Wiener, M., Lohoff, F. W., & Coslett, H. B. (2011). Double dissociation of dopamine genes and timing in humans. Journal of Cognitive Neuroscience, 23(10), 2811–2821.
Wiener, M., Turkeltaub, P., & Coslett, H. B. (2010). The image of time: A voxel-wise meta-analysis. Neuroimage, 49(2), 1728–1740.
Wood, G., Willmes, K., Nuerk, H.-C., & Fischer, M. H. (2008). On the cognitive link between space and number: A meta-analysis of the SNARC effect. Psychology Science, 50(4), 489–525.
Acknowledgements
This research was supported by a grant within the Priority Program, SPP 1772 from the German Research Foundation (Deutsche Forschungsgemeinschaft, DFG), Grant no TH 1554/3-1. We thank Sander Los and Michael Steinborn for many helpful comments on an earlier version of the article. Raw data of the reported experiments are available via the Open Science Framework: https://osf.io/z8mvj/, https://doi.org/10.17605/osf.io/z8mvj.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Research involving human and animal participants
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aufschnaiter, S., Kiesel, A. & Thomaschke, R. Humans derive task expectancies from sub-second and supra-second interval durations. Psychological Research 84, 1333–1345 (2020). https://doi.org/10.1007/s00426-019-01155-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00426-019-01155-9