Abstract
In diseased hearts, impaired muscle within the hearts is passively stretched by contractions of the more viable neighboring muscle during the contraction phase. We investigated whether in the myocardium with nonuniform contraction such passive stretch regionally generates ROS within the stretched region and exacerbates arrhythmias. In trabeculae from rat hearts, force, intracellular Ca2+, and membrane potential were measured. To assess regional ROS generation, the slope of the change in the 2′,7′-dichlorofluorescein fluorescence (DCFslope) was calculated at the each pixel position along the long axis of trabeculae using DCF fluorescence images. Ca2+ waves and arrhythmias were induced by electrical stimulation. A H2O2 (1 mmol/L) jet regionally increased the DCFslope within the jet-exposed region. A blebbistatin (10 μmol/L) jet caused passive stretch of the muscle within the jet-exposed region during the contraction phase and increased the DCFslope within the stretched region, the velocity of Ca2+ waves, and the number of beats after electrical stimulation (0.2 μmol/L isoproterenol), while 3 μmol/L diphenyleneiodonium (DPI), NADPH oxidase inhibitor, decreased them. A jet of a solution containing 0.2 mmol/L H2O2 in addition to 10 µmol/L blebbistatin also increased them. A H2O2 jet within the region where Ca2+ waves propagated increased their velocity. In the myocardium with nonuniform contraction, passive stretch of the muscle by contractions of the neighboring muscle regionally increases ROS within the stretched region, and the regional ROS exacerbates arrhythmias by activating the propagation of Ca2+ waves.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In patients with heart failure and myocardial infarction, reactive oxygen species (ROS) is increased [16, 17], probably due to an increase in NADPH oxidase activity [13, 22] or a decrease in hydrogen peroxide scavenging enzyme catalase activity [2]. This increase in ROS is involved in the exacerbation of heart failure [37] as well as in the occurrence of arrhythmias [7, 19, 46] by increasing Ca2+ release from the sarcoplasmic reticulum (SR) [43, 47]. Actually, in patients with diseased hearts, the occurrence of lethal arrhythmias is an important determinant of their prognosis [29, 30].
In a diseased heart, impaired muscle is widely distributed throughout the heart, causing nonuniform muscle contraction [34, 45]. In such myocardium with nonuniform contraction, impaired muscle with weaker contractile strength is stretched by contractions of the more viable neighboring muscle during the contraction phase. Conversely, during the relaxation phase, the impaired muscle is passively shortened and dissociates Ca2+ from the myofilaments within the region due to a decrease in myofilament Ca2+ sensitivity [20], thereby inducing Ca2+ waves [27, 40] and arrhythmias [25]. Additionally, it has been reported that stretch of cardiac muscle increases ROS generation in isolated single myocytes [32, 33] and trabeculae [27, 28] and further increases the frequency of Ca2+ sparks [18, 31, 33] and the velocity of Ca2+ waves [28]. It has not yet been established, however, whether in the myocardium with nonuniform contraction stretch of the impaired muscle by contractions of the neighboring muscle also increases ROS generation within the stretched region. Furthermore, it has not yet been established whether such an increase in regional ROS affects the propagation velocity of Ca2+ waves and the occurrence of arrhythmia.
Therefore, in the present study, we focused on regional changes in ROS generation in the myocardium with nonuniform contraction, investigating whether ROS is regionally increased within its stretched region and affects the propagation velocity of Ca2+ waves and the occurrence of arrhythmias. Our results indicate that in the myocardium with nonuniform contraction, passive stretch of the muscle by contractions of the neighboring muscle regionally increases ROS generation within the stretched region and exacerbates arrhythmias by increasing the velocity of Ca2+ waves.
Materials and methods (see expanded materials and methods in the Online Data Supplement)
Measurements of force, sarcomere length, membrane potential, [Ca2+]i, and ROS
All animal procedures were performed according to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996). All experimental protocols were approved by the Ethics Review Board of Tohoku University (approval reference number 2014-004, 2015-023). After rats had been adequately anesthetized, trabeculae were obtained from their right ventricles. Force, sarcomere length, membrane potential, and [Ca2+]i were measured as previously described [23,24,25,26,27,28, 40]. To estimate regional changes in ROS, trabeculae were loaded with 2′,7′-dichlorofluorescein (DCF) as previously described [27, 28]. As shown in Fig. 1a, regional change in the DCF fluorescence (DCFslope) was calculated at each pixel along the long axis of trabeculae using the DCF fluorescence images before and after exposure to a H2O2 jet or a blebbistatin jet, and the profile of DCFslope along the trabeculae was then obtained. To create a nonuniform contraction model, trabeculae were regionally exposed to a jet of a solution containing 10 μmol/L blebbistatin, as previously described [25, 27, 40]. When a blebbistatin jet was used, measurements were performed a few minutes after the stoppage of the blebbistatin jet because blebbistatin has fluorescent properties [10, 11].
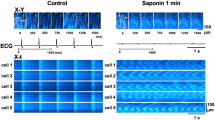
a Analysis of 2′,7′-dichlorofluorescein (DCF) fluorescence images. DCF fluorescence images were recorded before (DCFpre) and after (DCFpost) exposure to a H2O2 jet without stimulation or a blebbistatin jet with/without electrical stimulation (4 Hz for 30 s) (a). A region of interest (ROI; 50 × 720 pixels) was set along the long axis of a trabecula (T), and the profile of DCF fluorescence along the trabecula was calculated by vertically averaging the values of pixels within the ROI across the trabecula (b). To obtain the slope of the changes in the DCF fluorescence (DCFslope) along the trabecula, the difference in the profile of DCF fluorescence between the DCFpre and DCFpost was calculated pixel by pixel at the identical position along the trabecula and was divided by ΔTime. b Effect of a H2O2 jet on DCF fluorescence within trabeculae. Representative recordings of the profile of the DCFslope along a trabecula with (light blue) and without (black) exposure to a 1 mmol/L H2O2 jet (a). The trabecula was exposed to the jet in the region of X. Y indicates the region 0.5 mm apart from X (Exp. 170529). Summary data concerning the effect of a H2O2 jet on the DCFslope (n = 6) (b). Exposure to a H2O2 jet increased the DCFslope within X compared to that within Y (right panel). #p < 0.01 vs Y
Experimental protocol with trabeculae
Ca2+ waves were induced by electrical stimulation (400-ms stimulus intervals for 7.5 s), and arrhythmias were induced by electrical stimulation (250-ms stimulus intervals for 15 s) in the presence of 0.2 μmol/L isoproterenol. All measurements were performed at 24 °C.
Statistics
All measurements were expressed as mean ± SEM. Statistical analysis was performed with a paired t test for two-group comparisons and one-way repeated-measures ANOVA with Tukey-Kramer for multiple comparisons when the data were normally distributed. Otherwise, the Wilcoxon signed-ranks test was used for two-group comparisons, unless otherwise mentioned. These analyses were performed using software for statistical analysis (Ekuseru-Toukei 2012, Social Survey Research Information Co., Ltd., Tokyo, Japan). Values of p < 0.05 were considered to be significant.
Results
Effect of a H2O2 jet on ROS generation
To confirm whether the DCFslope calculated in the present study actually reflects regional changes in ROS generation, trabeculae were regionally exposed to a 1 mmol/L H2O2 jet. As shown in Fig. 1b, regional exposure to a H2O2 jet for 30 s increased the DCFslope within the jet-exposed region (X) compared with that within the region 0.5 mm apart from the jet-exposed region (Y), whereas the DCFslope showed no regional changes within trabeculae without exposure to a H2O2 jet. These results suggest that the DCFslope within trabeculae reflects regional changes in ROS.
Effect of a blebbistatin jet on ROS generation
Regional exposure of trabeculae to a jet of a solution that reduces muscle contraction causes regional stretch within the jet-exposed region by contractions of the neighboring muscle during the contraction phase, as previously reported [27, 40]. As shown in Fig. 2a, the sarcomere was stretched within the region exposed to a 10 μmol/L blebbistatin jet (stretched region: X), whereas it was shortened within the region apart from the jet-exposed region (contracting region: Y) during the contraction phase, representing nonuniform contraction. Regional changes in the DCF fluorescence along the long axis of trabeculae were then recorded when trabeculae contracted nonuniformly in response to regional exposure to the 10 μmol/L blebbistatin jet. As shown in Fig. 2b, c (a), electrical stimulation for 30 s increased the DCFslope within the region exposed to the blebbistatin jet (X) compared with that within the region 0.5 mm apart from the jet-exposed region (Y). This regional increase in the DCFslope was not detected without electrical stimulation, as shown in Fig. 2c (b). Besides, this regional increase was not detected after superfusion with 3 μmol/L diphenyleneiodonium (DPI), NADPH oxidase inhibitor, for 1 h (Fig. 2c (b)). These results suggest that when cardiac muscle contracts nonuniformly, ROS is regionally increased within the stretched region, at least in part, due to the activation of NADPH oxidase.
Regional effect of a blebbistatin jet on the DCFslope. a The upper panel shows force, and the lower panel shows changes in sarcomere length (SL). The trabecula was exposed to a 10 μmol/L blebbistatin jet in the region of X. Y indicates the region apart from X. The sarcomere within X (red line) was stretched, while the sarcomere within Y (purple line) was shortened by electrical stimulation (ST; 2-s stimulus intervals, 0.7 mmol/L [Ca2+]o; Exp. 151214). b Representative recordings of the profile of DCFslope along a trabecula exposed to a 10 μmol/L blebbistatin jet in the absence (black line) and presence (red line) of 3 μmol/L DPI. X indicates the region exposed to a blebbistatin jet, and Y indicates the region 0.5 mm apart from X (Exp. 150521). c Summary data concerning the effect of a blebbistatin jet on the DCFslope with 4 Hz electrical stimulation (n = 5) (a). Exposure to a blebbistatin jet increased the DCFslope within X compared to that within Y with electrical stimulation. *p < 0.01 vs Y. Summary data concerning the effect of a blebbistatin jet on the DCFslope without electrical stimulation (left panel) and that after superfusion with 3 μmol/L diphenyleneiodonium (DPI, n = 5, right panel) (b)
Roles of ROS within the stretched region in Ca2+ waves and arrhythmias
In trabeculae with nonuniform contraction, Ca2+ waves are initiated from the border zone between a contracting region and a stretched region due to Ca2+ dissociation from the myofilaments and propagate along trabeculae by Ca2+-induced Ca2+ release (CICR) from the SR, as previously reported [25, 27, 40]. To investigate whether ROS generation within the stretched region affects Ca2+ wave propagation and arrhythmias, we examined the effect of DPI on the propagation features of Ca2+ waves and the occurrence of arrhythmias in trabeculae exposed to a 10 μmol/L blebbistatin jet. As shown in Fig. 3a, electrical stimulation induced Ca2+ waves arising around the region exposed to a blebbistatin jet. Within the jet-exposed region, the peak [Ca2+]i of the Ca2+ waves ([Ca2+]CW) was higher than that within the region 0.4 mm apart from the jet-exposed region (Fig. 3b (a)). Besides, superfusion with DPI decreased the [Ca2+]CW within the jet-exposed region and the velocity of Ca2+ waves (Fig. 3a, b (b)), suggesting that ROS generation within the stretched region enhances Ca2+ release from the SR induced by the Ca2+ dissociated from the myofilaments and increases the velocity of Ca2+ waves even outside the jet-exposed region. Concerning the occurrence of arrhythmias, we have previously reported that in the presence of isoproterenol, electrical stimulation induces arrhythmias due to acceleration of Ca2+ waves in the myocardium with nonuniform contraction [25, 26, 36]. Also in the present study, electrical stimulation induced arrhythmias in the presence of 0.2 μmol/L isoproterenol, as shown in Fig. 3c. Superfusion with DPI decreased the number of beats induced by electrical stimulation (Fig. 3c, d), suggesting that ROS generation within the stretched region is involved with the occurrence of arrhythmias.
Effect of DPI on Ca2+ waves and arrhythmias in the myocardium exposed to a blebbistatin jet. a Representative recordings of force (upper panels) and regional changes in [Ca2+]i (lower panels) during the last three electrical stimuli (ST; 400-ms stimulus intervals for 7.5 s) and Ca2+ waves in the absence (left panels) and presence (right panels) of 3 μmol/L DPI. White arrows indicate the first Ca2+ waves. Yellow dotted lines a and b indicate the jet-exposed region and the region 0.4 mm apart from the jet-exposed region where the peaks of [Ca2+]i of Ca2+ waves ([Ca2+]CW) were calculated, respectively. In the left panel, Ca2+ waves appeared around the region exposed to a 10 μmol/L blebbistatin jet and propagated along the trabecula. In the right panel, the velocity of the Ca2+ wave was decreased in the presence of DPI (2.0 mmol/L [Ca2+]o; Exp. 140528). b Summary data concerning the [Ca2+]CW within the region indicated by lines a and b in the left panel of a. #p < 0.01 vs line a (a). Summary data concerning the effect of DPI on the [Ca2+]CW within the region indicated by lines a in the panels of a (left panel) and the velocity of Ca2+ waves (right panel; n = 7). *p < 0.05 vs (–) (b). c Representative recordings of membrane potential (upper panels) and force (lower panels) after the last three electrical stimuli (ST; 250-ms stimulus intervals for 15 s) in the absence (left panels) and presence (right panels) of 3 μmol/L DPI in a trabecula exposed to a blebbistatin jet (2.0 mmol/L [Ca2+]o, 0.2 μmol/L isoproterenol; Exp. 150129). d Summary data concerning the effect of DPI on the number of beats induced by electrical stimulation (n = 5; 2.0 ± 0.5 mmol/L [Ca2+]o). *p < 0.05 vs (–)
In order to further examine whether ROS within the stretched region increased the velocity of Ca2+ waves outside the jet-exposed region and induced arrhythmias, we added H2O2 to a solution used for a blebbistatin jet. Electrical stimulation induced a Ca2+ wave arising around the region exposed to a 10 μmol/L blebbistatin jet (Fig. 4a). Addition of 0.2 mmol/L H2O2 to the blebbistatin jet increased the [Ca2+]CW within the jet-exposed region and the velocity of the Ca2+ wave (Fig. 4a, b). Furthermore, addition of H2O2 increased the number of beats induced by electrical stimulation (Fig. 4c, d). Taken together, these results suggest that a regional increase in ROS within the stretched region enhances Ca2+ release from the SR within the region and that this enhanced Ca2+ release works as an enhanced initiator of CICR for propagation of Ca2+ waves and induces arrhythmias.
Effect of a jet of a solution containing H2O2 in addition to blebbistatin on Ca2+ waves and arrhythmias. a Representative recordings of force (upper panels) and regional changes in [Ca2+]i (lower panels) during the last three electrical stimuli (ST; 400-ms stimulus intervals for 7.5 s) and a Ca2+ wave. The left panels show changes in a trabecula exposed to a 10 μmol/L blebbistatin jet, and the right panels show changes in the trabecula exposed to a jet of a solution containing 0.2 mmol/L H2O2 in addition to 10 μmol/L blebbistatin. White arrows indicate the first Ca2+ waves. In the left panel, a Ca2+ wave appeared around the jet-exposed region and propagated along the trabecula. In the right panel, the velocity of the Ca2+ wave was increased to 4.7 mm/s. Yellow dotted line a in both panels indicates the regions where the [Ca2+]CW were calculated (2.0 mmol/L [Ca2+]o; Exp. 151015). b Summary data concerning the effect of addition of H2O2 to the blebbistatin jet on the [Ca2+]CW within the region indicated lines a in the panels of a (left panel) and the velocity of Ca2+ waves (right panel; n = 6). *p < 0.05 vs blebbistatin jet. c Representative recordings of membrane potential (upper panels) and force (lower panels) after the last three electrical stimuli (ST; 250-ms stimulus intervals for 15 s) before (left panels) and after (right panels) addition of 0.2 mmol/L H2O2 to the blebbistatin jet. In the right panels, addition of H2O2 to the blebbistatin jet increased the number of beats induced by electrical stimulation (2.0 mmol/L [Ca2+]o, 0.2 μmol/L isoproterenol; Exp. 151026). d Summary data concerning the effect of addition of H2O2 to the blebbistatin jet on the number of beats induced by electrical stimulation (n = 6; 1.9 ± 0.1 mmol/L [Ca2+]o). *p < 0.05 vs bleb jet
It is possible, however, that addition of H2O2 to the blebbistatin jet may have affected the regional contractile strength [12], thereby increasing the velocity of Ca2+ waves and the number of beats after electrical stimulation. We thus examined the effect of H2O2 on the developed force. The bath superfusate containing both 0.2 mmol/L H2O2 and 10 μM blebbistatin decreased the developed force to the level similar to that in the superfusate containing only 10 μmol/L blebbistatin (data not shown), meaning that the addition of H2O2 to a blebbistatin jet does not affect the contractile features within the stretched region.
Roles of ROS in Ca2+ wave propagation
Finally, to examine whether ROS affected the CICR mechanism, trabeculae were exposed to a 0.2 mmol/L H2O2 jet during propagation of Ca2+ waves. To minimize the effect of H2O2 on the contractile strength, the bath was superfused with a solution containing 10 μmol/L blebbistatin. This bath superfusion with blebbistatin decreased the force developed by electrical stimulation to 8.7 ± 1.4% of its initial value. To induce Ca2+ waves due to Ca2+ leak from the SR, trabeculae were exposed to a 10 mmol/L Ca2+ jet. As shown in Fig. 5a, electrical stimulation induced spontaneous increases in [Ca2+]i (white arrowheads) within the jet-exposed region just before electrical stimulation and induced Ca2+ waves arising from the jet-exposed region after electrical stimulation. When a H2O2 jet was directed to the region where Ca2+ waves propagated, it increased the velocity of Ca2+ waves (Fig. 5a). Figure 5b shows the summary data. A H2O2 jet increased the velocity of Ca2+ waves, suggesting that ROS accelerates Ca2+ waves probably activating the CICR mechanism.
Effect of a H2O2 jet on Ca2+ waves induced by a 10 mmol/L Ca2+ jet. a Representative recordings of force (upper panels) and regional changes in [Ca2+]i (lower panels) during the last three electrical stimuli (ST; 400-ms stimulus intervals for 7.5 s) in the absence (left panels) and presence (right panels) of a 0.2 mmol/L H2O2 jet. Muscle contractions were minimized by the bath superfusion with 10 μmol/L blebbistatin, and Ca2+ waves were induced by a 10 mmol/L Ca2+ jet. White arrows indicate the first Ca2+ waves, and white arrowheads indicate spontaneous increases in [Ca2+]i just before electrical stimulation. In the left panel, Ca2+ waves appeared within the region exposed to the 10 mmol/L Ca2+ jet and propagated along the trabecula. In the right panel, exposure to the H2O2 jet increased the velocity of the first Ca2+ wave (2.0 mmol/L [Ca2+]o; Exp. 140910). b Summary data concerning the effect of the 0.2 mmol/L H2O2 jet on the velocity of Ca2+ waves (n = 6). *p < 0.05 vs (–)
Discussion
The present study characterized the effect of regional muscle stretch on ROS generation, Ca2+ waves, and arrhythmias using the cardiac muscle model representing nonuniform contraction. To the best of our knowledge, it shows for the first time that in the myocardium with nonuniform contraction, passive stretch of the muscle by contractions of the neighboring muscle regionally generates ROS within the stretched region and that such regional ROS generation exacerbates arrhythmias by activating the propagation of Ca2+ waves, as discussed below.
Regional ROS generation within the stretched region
It has been reported that in cardiac muscle, stretch of the muscle increases ROS [27, 28, 32, 33], the frequency of Ca2+ sparks [18, 31, 33], and the velocity of Ca2+ waves [28]. In the present study, the DCFslope was increased within the region stretched by contractions of the neighboring muscle during the contraction phase in trabeculae exposed to a blebbistatin jet (Fig. 2c (a)). We assume that this regional increase in the DCFslope reflects a regional increase in ROS generation for the following reasons. First, the blebbistatin jet regionally increased the DCFslope in the manner similar to the H2O2 jet (Fig. 1b). Second, the DCFslope was not increased within the blebbistatin jet-exposed region after superfusion with DPI (Fig. 2c (b)), suggesting that the DCFslope was increased due to the activation of NADPH oxidase although DPI inhibits the synthesis of both oxygen- and nitrogen-derived reactive species and many other flavoproteins depending on the concentration [1]. Third, the DCFslope was measured a few minutes after the stoppage of a blebbistatin jet because blebbistatin has fluorescent properties by itself. Fourth, the DCFslope was not increased within the blebbistatin jet-exposed region without electrical stimulation (Fig. 2c (b)). Thus, the results in the present study suggest that in the myocardium with nonuniform contraction, passive muscle stretch during the contraction phase regionally increases ROS within the stretched region, at least in part, due to the activation of NADPH oxidase.
Roles of ROS in Ca2+ waves and arrhythmias
ROS increases Ca2+ release from the SR [39, 43, 47] by oxidizing ryanodine receptors (RyRs) [6] or activating calcium/calmodulin-dependent protein kinase II (CaMKII) [8, 9]. It further increases the velocity of Ca2+ waves [18] and exacerbates arrhythmias [7, 19]. Likewise, H2O2 causes triggered arrhythmias [42] by directly activating RyRs [39] or by impairing Na+ current inactivation [35] through activation of CaMKII [39] or protein kinase C [41]. In addition, H2O2 changes force and Ca2+ transients [12] through the modulation of the Ca2+ current [15], HERG [3], and the sodium-calcium exchange current [14, 21].
As for the initiation mechanism of Ca2+ waves, two mechanisms have been proposed [27]. One is Ca2+ leak from the SR due to Ca2+ overload [23, 28], and the other is Ca2+ dissociation from the myofilaments in the myocardium with nonuniform contraction [25, 40]. In the latter mechanism, regional differences in contractile strength causes stretching of muscle by contractions of the more viable neighboring muscle. During the relaxation phase, Ca2+ is dissociated from the myofilaments due to the passive shortening and initiates Ca2+ waves from the border zone between the contracting and stretched region [40]. As for the propagation mechanism, CICR has been believed to underlie both the Ca2+ waves. In the present study, the blebbistatin jet caused nonuniform contraction (Fig. 2a) and induced Ca2+ waves from the jet-exposed region (Figs. 3a and 4a), suggesting that Ca2+ waves in Figs. 3a and 4a were initiated by Ca2+ dissociation from the myofilaments, while Ca2+ waves using a high Ca2+ jet in Fig. 5 were initiated by SR Ca2+ leak.
Concerning an increase in the propagation velocity of Ca2+ waves outside the jet-exposed region in Figs. 3 and 4, we assume that an increase in [Ca2+]i due to ROS generation within the stretched region works as an enhanced initiator of CICR for propagation of Ca2+ waves for the following reasons. First, the [Ca2+]CW within the stretched region was higher than that outside the blebbistatin jet-exposed region (Fig. 3b (a)). Second, superfusion with DPI decreased the [Ca2+]CW within the jet-exposed region and the velocity of Ca2+ waves (Fig. 3b (b)), while addition of H2O2 to a blebbistatin jet increased them (Fig. 4b). Third, we have previously reported that the velocity of Ca2+ waves increases depending on the [Ca2+]CW in trabeculae [23]. Fourth, we have also reported that the velocity of Ca2+ waves increases depending on the Ca2+ dissociated from the myofilaments within the jet-exposed region when trabeculae are shortened [24].
In the present study, superfusion with DPI decreased the number of beats after electrical stimulation (Fig. 3), and addition of H2O2 to a blebbistatin jet increased it (Fig. 4). Besides, as shown in Fig. 5, a H2O2 jet directed to the region where Ca2+ waves propagated increased the velocity of Ca2+ waves. Furthermore, we have previously reported that an increase in the velocity of Ca2+ waves enhances the amplitude of delayed after depolarizations and cause arrhythmias [36]. Taken together, these results suggest that in the myocardium with nonuniform contraction, an increase in ROS within the stretched region increases the velocity of Ca2+ waves by activating CICR [5, 43, 47] and thereby induces arrhythmias.
Clinical implications
In patients with heart failure and myocardial infarction, lethal arrhythmias frequently occur [29, 30]. Within such diseased hearts, impaired muscle is widely distributed, and thus, the hearts exhibit nonuniform contraction due to the regional difference in contractile strength. Results of the present study suggest that in patients with diseased hearts, stretch of the impaired muscle by contractions of the more viable neighboring muscle increases ROS, especially within the stretched region and that such an increase in ROS causes arrhythmias by activating Ca2+ waves, which is induced by the Ca2+ dissociated from the myofilaments due to the difference in contractile strength.
Study limitations
In diseased hearts, abnormal Ca2+ handling frequently occurs, especially within the impaired muscle, causing ROS generation [44] and arrhythmias [4, 38]. In the present study, however, the region showing stretch by exposure to a blebbistatin jet was not impaired but was just paralyzed.
Conclusion
In the myocardium with nonuniform contraction, passive stretch of the muscle by contractions of the neighboring muscle regionally increases ROS generation within the stretched region, and the regional ROS exacerbates arrhythmias by activating the propagation of Ca2+ waves.
References
Aldieri E, Riganti C, Polimeni M, Gazzano E, Lussiana C, Campia I, Ghigo D (2008) Classical inhibitors of NOX NAD(P)H oxidases are not specific. Curr Drug Metab 9:686–696
Bäumer AT, Flesch M, Wang X, Shen Q, Feuerstein GZ, Böhm M (2000) Antioxidative enzymes in human hearts with idiopathic dilated cardiomyopathy. J Mol Cell Cardiol 32:121–130. https://doi.org/10.1006/jmcc.1999.1061
Bérubé J, Caouette D, Daleau P (2001) Hydrogen peroxide modifies the kinetics of HERG channel expressed in a mammalian cell line. J Pharmacol Exp Ther 297:96–102
Beuckelmann DJ, Näbauer M, Erdmann E (1992) Intracellular calcium handling in isolated ventricular myocytes from patients with terminal heart failure. Circulation 85:1046–1055
Bogeski I, Kappl R, Kummerow C, Gulaboski R, Hoth M, Niemeyer BA (2011) Redox regulation of calcium ion channels: chemical and physiological aspects. Cell Calcium 50:407–423. https://doi.org/10.1016/j.ceca.2011.07.006
Bovo E, Lipsius SL, Zima AV (2012) Reactive oxygen species contribute to the development of arrhythmogenic Ca2+ waves during β-adrenergic receptor stimulation in rabbit cardiomyocytes. J Physiol 590:3291–3304. https://doi.org/10.1113/jphysiol.2012.230748
Burgoyne JR, Mongue-Din H, Eaton P, Shah AM (2012) Redox signaling in cardiac physiology and pathology. Circ Res 111:1091–1106. https://doi.org/10.1161/CIRCRESAHA.111.255216
Dries E, Bito V, Lenaerts I, Antoons G, Sipido KR, Macquaide N (2013) Selective modulation of coupled ryanodine receptors during microdomain activation of calcium/calmodulin-dependent kinase II in the dyadic cleft. Circ Res 113:1242–1252. https://doi.org/10.1161/CIRCRESAHA.113.301896
Erickson JR, Joiner MLA, Guan X, Kutschke W, Yang J, Oddis CV, Bartlett RK, Lowe JS, O’Donnell SE, Aykin-Burns N, Zimmerman MC, Zimmerman K, Ham AJL, Weiss RM, Spitz DR, Shea MA, Colbran RJ, Mohler PJ, Anderson ME (2008) A dynamic pathway for calcium-independent activation of CaMKII by methionine oxidation. Cell 133:462–474. https://doi.org/10.1016/j.cell.2008.02.048
Farman GP, Tachampa K, Mateja R, Cazorla O, Lacampagne A, de Tombe PP (2008) Blebbistatin: use as inhibitor of muscle contraction. Pflugers Arch 455:995–1005
Fedorov VV, Lozinsky IT, Sosunov EA, Anyukhovsky EP, Rosen MR, Balke CW, Efimov IR (2007) Application of blebbistatin as an excitation-contraction uncoupler for electrophysiologic study of rat and rabbit hearts. Heart Rhythm 4:619–626. https://doi.org/10.1016/j.hrthm.2006.12.047
Goldhaber JI, Liu E (1994) Excitation-contraction coupling in single Guinea-pig ventricular myocytes exposed to hydrogen peroxide. J Physiol 477:135–147
Heymes C, Bendall JK, Ratajczak P, Cave AC, Samuel JL, Hasenfuss G, Shah AM (2003) Increased myocardial NADPH oxidase activity in human heart failure. J Am Coll Cardiol 41:2164–2171
Hinata M, Matsuoka I, Iwamoto T, Watanabe Y, Kimura J (2007) Mechanism of Na+/Ca2+ exchanger activation by hydrogen peroxide in Guinea-pig ventricular myocytes. J Pharmacol Sci 103:283–292
Hudasek K, Brown ST, Fearon IM (2004) H2O2 regulates recombinant Ca2+ channel α1c subunits but does not mediate their sensitivity to acute hypoxia. Biochem Biophys Res Commun 318:135–141
Ide T, Tsutsui H, Kinugawa S, Suematsu N, Hayashidani S, Ichikawa K, Utsumi H, Machida Y, Egashira K, Takeshita A (2000) Direct evidence for increased hydroxyl radicals originating from superoxide in the failing myocardium. Circ Res 86:152–157
Ide T, Tsutsui H, Hayashidani S, Kang D, Suematsu N, Nakamura K, Utsumi H, Hamasaki N, Takeshita A (2001) Mitochondrial DNA damage and dysfunction associated with oxidative stress in failing hearts after myocardial infarction. Circ Res 88:529–535
Iribe G, Ward CW, Camelliti P, Bollensdorff C, Mason F, Burton RAB, Garny A, Morphew MK, Hoenger A, Lederer WJ, Kohl P (2009) Axial stretch of rat single ventricular cardiomyocytes causes an acute and transient increase in Ca2+ spark rate. Circ Res 104:787–795. https://doi.org/10.1161/CIRCRESAHA.108.193334
Jeong EM, Liu M, Sturdy M, Gao G, Varghese ST, Sovari AA, Dudley SC Jr (2012) Metabolic stress, reactive oxygen species, and arrhythmia. J Mol Cell Cardiol 52:454–463. https://doi.org/10.1016/j.yjmcc.2011.09.018
Kentish JC, ter Keurs HEDJ, Ricciardi L, Bucx JJ, Noble MI (1986) Comparison between the sarcomere length-force relations of intact and skinned trabeculae from rat right ventricle. Influence of calcium concentrations on these relations. Circ Res 58:755–768
Liu T, O’Rourke B (2013) Regulation of the Na+/Ca2+ exchanger by pyridine nucleotide redox potential in ventricular myocytes. J Biol Chem 288:31984–31992. https://doi.org/10.1074/jbc.M113.496588
Maack C, Kartes T, Kilter H, Schäfers HJ, Nickenig G, Böhm M, Laufs U (2003) Oxygen free radical release in human failing myocardium is associated with increased activity of rac1-GTPase and represents a target for statin treatment. Circulation 108:1567–1574. https://doi.org/10.1161/01.CIR.0000091084.46500.BB
Miura M, Boyden PA, ter Keurs HEDJ (1999) Ca2+ waves during triggered propagated contractions in intact trabeculae. Determinants of the velocity of propagation. Circ Res 84:1459–1468
Miura M, Wakayama Y, Endoh H, Nakano M, Sugai Y, Hirose M, ter Keurs HEDJ, Shimokawa H (2008) Spatial non-uniformity of excitation-contraction coupling can enhance arrhythmogenic delayed afterdepolarizations in rat cardiac muscle. Cardiovasc Res 80:55–61. https://doi.org/10.1093/cvr/cvn162
Miura M, Nishio T, Hattori T, Murai N, Stuyvers BD, Shindoh C, Boyden PA (2010) Effect of nonuniform muscle contraction on sustainability and frequency of triggered arrhythmias in rat cardiac muscle. Circulation 121:2711–2717. https://doi.org/10.1161/CIRCULATIONAHA.109.907717
Miura M, Hattori T, Murai N, Nagano T, Nishio T, Boyden PA, Shindoh C (2012) Regional increase in extracellular potassium can be arrhythmogenic due to nonuniform muscle contraction in rat ventricular muscle. Am J Physiol Heart Circ Physiol 302:H2301–H2309. https://doi.org/10.1152/ajpheart.01161.2011
Miura M, Murai N, Hattori T, Nagano T, Stuyvers BD, Shindoh C (2013) Role of reactive oxygen species and Ca2+ dissociation from the myofilaments in determination of Ca2+ wave propagation in rat cardiac muscle. J Mol Cell Cardiol 56:97–105. https://doi.org/10.1016/j.yjmcc.2012.12.011
Miura M, Taguchi Y, Nagano T, Sasaki M, Handoh T, Shindoh C (2015) Effect of myofilament Ca2+ sensitivity on Ca2+ wave propagation in rat ventricular muscle. J Mol Cell Cardiol 84:162–169. https://doi.org/10.1016/j.yjmcc.2015.04.027
Myerburg RJ, Interian A, Mitrani RM, Kessler KM, Castellanos A (1997) Frequency of sudden cardiac death and profiles of risk. Am J Cardiol 80:10F–19F
Packer M (1985) Sudden unexpected death in patients with congestive heart failure: a second frontier. Circulation 72:681–685
Petroff MGV, Kim SH, Pepe S, Dessy C, Marbán E, Balligand JL, Sollott SJ (2001) Endogenous nitric oxide mechanisms mediate the stretch dependence of Ca2+ release in cardiomyocytes. Nat Cell Biol 3:867–873. https://doi.org/10.1038/ncb1001-867
Pimentel DR, Amin JK, Xiao L, Miller T, Viereck J, Oliver-Krasinski J, Baliga R, Wang J, Siwik DA, Singh K, Pagano P, Colucci WS, Sawyer DB (2001) Reactive oxygen species mediate amplitude-dependent hypertrophic and apoptotic responses to mechanical stretch in cardiac myocytes. Circ Res 89:453–460
Prosser BL, Ward CW, Lederer WJ (2011) X-ROS signaling: rapid mechano-chemo transduction in heart. Science 333:1440–1445. https://doi.org/10.1126/science.1202768
Siogas K, Pappas S, Graekas G, Goudevenos J, Liapi G, Sideris DA (1998) Segmental wall motion abnormalities alter vulnerability to ventricular ectopic beats associated with acute increases in aortic pressure in patients with underlying coronary artery disease. Heart 79:268–273
Song Y, Shryock JC, Wagner S, Maier LS, Belardinelli L (2006) Blocking late sodium current reduces hydrogen peroxide-induced arrhythmogenic activity and contractile dysfunction. J Pharmacol Exp Ther 318:214–222. https://doi.org/10.1124/jpet.106.101832
Sugai Y, Miura M, Hirose M, Wakayama Y, Endoh H, Nishio T, Watanabe J, ter Keurs HE, Shirato K, Shimokawa H (2009) Contribution of Na+/Ca2+ exchange current to the formation of delayed after depolarizations in intact rat ventricular muscle. J Cardiovasc Pharmacol 53:517–522. https://doi.org/10.1097/FJC.0b013e3181a913f4
Terentyev D, Györke I, Belevych AE, Terentyeva R, Sridhar A, Nishijima Y, de Blanco EC, Khanna S, Sen CK, Cardounel AJ, Carnes CA, Györke S (2008) Redox modification of ryanodine receptors contributes to sarcoplasmic reticulum Ca2+ leak in chronic heart failure. Circ Res 103:1466–1472. https://doi.org/10.1161/CIRCRESAHA.108.184457
ter Keurs HEDJ, Boyden PA (2007) Calcium and arrhythmogenesis. Physiol Rev 87:457–506
Wagner S, Ruff HM, Weber SL, Bellmann S, Sowa T, Schulte T, Anderson ME, Grandi E, Bers DM, Backs J, Belardinelli L, Maier LS (2011) Reactive oxygen species-activated ca/calmodulin kinase IIδ is required for late INa augmentation leading to cellular Na and Ca overload. Circ Res 108:555–565. https://doi.org/10.1161/CIRCRESAHA.110.221911
Wakayama Y, Miura M, Stuyvers BD, Boyden PA, ter Keurs HEDJ (2005) Spatial nonuniformity of excitation-contraction coupling causes arrhythmogenic Ca2+ waves in rat cardiac muscle. Circ Res 96:1266–1273
Ward CA, Giles WR (1997) Ionic mechanism of the effects of hydrogen peroxide in rat ventricular myocytes. J Physiol 500:631–642
Xie LH, Chen F, Karagueuzian HS, Weiss JN (2009) Oxidative-stress-induced afterdepolarizations and calmodulin kinase II signaling. Circ Res 104:79–86. https://doi.org/10.1161/CIRCRESAHA.108.183475
Xu L, Eu JP, Meissner G, Stamler JS (1998) Activation of the cardiac calcium release channel (ryanodine receptor) by poly-S-nitrosylation. Science 279:234–237
Yan Y, Wei CL, Zhang WR, Cheng HP, Liu J (2006) Cross-talk between calcium and reactive oxygen species signaling. Acta Pharmacol Sin 27:821–826. https://doi.org/10.1111/j.1745-7254.2006.00390.x
Young AA, Dokos S, Powell KA, Sturm B, McCulloch AD, Starling RC, McCarthy PM, White RD (2001) Regional heterogeneity of function in nonischemic dilated cardiomyopathy. Cardiovasc Res 49:308–318
Zhang H, Gomez AM, Wang X, Yan Y, Zheng M, Cheng H (2013) ROS regulation of microdomain Ca2+ signalling at the dyads. Cardiovasc Res 98:248–258. https://doi.org/10.1093/cvr/cvt050
Zima AV, Blatter LA (2006) Redox regulation of cardiac calcium channels and transporters. Cardiovasc Res 71:310–321
Conflict of interest
The authors declare that they have no conflict of interest.
Funding
This work was supported by Grant-in-Aid for Scientific Research (C) from Japan Society for the Promotion of Science (M. Miura, No. 26460288; C Shindoh No. 16K08485).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miura, M., Taguchi, Y., Handoh, T. et al. Regional increase in ROS within stretched region exacerbates arrhythmias in rat trabeculae with nonuniform contraction. Pflugers Arch - Eur J Physiol 470, 1349–1357 (2018). https://doi.org/10.1007/s00424-018-2152-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-018-2152-x