Abstract
Introduction
Efficient postoperative pain control is important after hemorrhoidal surgery. Although several locally applied medications have been used, current evidence regarding the optimal strategy is still conflicting. This network meta-analysis assessed analgesic efficacy and safety of the various topical medications in patients submitted to excisional procedures for hemorrhoids.
Methods
The present study followed the Cochrane Handbook for Systematic Reviews of Interventions and the PRISMA guidelines. The last systematic literature screening was performed at 15 June 2023. Comparisons were based on a random effects multivariate network meta-analysis under a Bayesian framework.
Results
Overall, 26 RCTs and 2132 patients were included. Regarding postoperative pain, EMLA cream (surface under the cumulative ranking curve (SUCRA) 80.3%) had the highest ranking at 12-h endpoint, while aloe vera cream (SUCRA 82.36%) scored first at 24 h. Metronidazole ointments had the highest scores at 7 and 14 days postoperatively. Aloe vera had the best analgesic profile (24-h SUCRA 84.8% and 48-h SUCRA 80.6%) during defecation. Lidocaine (SUCRA 87.9%) displayed the best performance regarding overall morbidity rates.
Conclusions
Due to the inconclusive results and several study limitations, further RCTs are required.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rationale
Hemorrhoidal disease (HD) is among the most common benign anorectal disorders, with a notable impact on patient quality of life (QoL) [1,2,3]. Current estimations suggest that a significant proportion of the population will be diagnosed with hemorrhoids and, ultimately, submitted to surgical management of HD [2]. Moreover, it is expected that the demand for outpatient HD treatment will increase over 23% the next two decades [2].
Contemporary HD management includes a staged approach that incorporates both conservative measures, interventional modalities, and surgical procedures [4, 5]. Excision of hemorrhoids is considered the gold standard treatment for high-grade HD, since it is associated with minimal recurrence risk [4, 5]. However, the main drawback of such an approach is the increased rate of postoperative pain that debilitates the patient, prolongs hospitalization, and postpones the return to normal daily routines [5]. Despite multiple technical variations, including closure of hemorrhoidectomy incision and the use of energy sources and staplers, postoperative pain remains a major issue after HD excisional procedures [6, 7].
Therefore, it becomes apparent that an efficient postoperative pain control strategy is of paramount importance to achieve optimization of HD management under a day-case surgery framework [8, 9]. More specifically, failure of ambulatory management ranges from 0 to 61% in some studies, with unsatisfactory pain control being among the most common causes of hospitalization prolongation [9]. Several approaches to minimize postoperative pain have been described, including multimodal analgesia, local anesthetics, electroacupuncture, anal infiltration with botulinum toxin, and topically applied pharmaceutic agents [10,11,12,13,14].
Theoretically, the local application of ointments allows the maximization of the analgesic effect of the active agent, while in parallel minimizing the risk of systematic adverse events [11, 13]. Several studies compared the various locally applied medications regarding the analgesic efficiency and agent-associated morbidity, with conflicting results [15,16,17]. This was mainly attributed to the inconsistency in the experimental and control arms and assessed endpoints [15,16,17]. Finally, recent pooled analyses attempted to provide an overall evaluation of the various strategies without reaching definitive conclusions [18, 19]. Recent meta-analyses by Jin et al. [18] and Yang et al. [19] compared a minimal sample of the reported ointment-based medications with interventions such as acupuncture and botulinum toxin without conclusive results, thus contributing to the lack of firm evidence [18, 19]. Additionally, these studies focused mainly on pain outcomes, without addressing the safety profile of the various local agents. Finally, the publication of several recent trials highlights the need for updated pooled data regarding the optimal local agent after excisional operations for HD [20, 21].
Objectives
Subsequently, the aim of this study was to compare the postoperative pain management efficacy and safety of the various ointment-based medications in patients submitted to excisional procedures for HD.
Methods
Study protocol
The present study adhered to the Cochrane Handbook for Systematic Reviews of Interventions [22] and the PRISMA guidelines [23].
Endpoints
The primary endpoint of our network meta-analysis (NMA) was the comparison of the various local agents in terms of pain management efficacy at 12 h postoperatively. Postoperative pain measurements were based on the Visual Analog Scale (VAS). Secondar outcomes included VAS measurements at 24 h, 48 h, 7 days, and 14 days postoperatively. Perianal pain during defecation was also assessed during the above-mentioned time endpoints. Postoperative opioid consumption and overall and medication-specific complications were also examined.
Search strategy
A systematic screening of the electronic scholar databases (Medline, Scopus, Web of Science, and CENTRAL) was performed by two researchers (P.K., C.C.). The last search date was 15 June 2023. The following keywords combined with Boolean logic nexuses were introduced: “hemorrhoid”, “hemorrhoidectomy”, “postoperative”, “post”, and “pain”. The references of all relevant manuscripts were also reviewed.
Eligibility and exclusion criteria
All prospective randomized controlled trials (RCTs) that compared at least two ointment-based agents or a combination of them, after hemorrhoidectomy, and provided relevant endpoint data were considered eligible. There was no restriction in terms of manuscript language or hemorrhoidectomy surgical technique; Milligan-Morgan, Ferguson, stapled, or energy device hemorrhoidectomies were all included.
The following exclusion criteria were introduced: (1) non-human studies, (2) non-RCT study design, (3) reviews or meta-analyses, (4) no outcome of interest, (5) no comparison group, (6) irretrievable or duplicate data, (7) non-excisional management of HD (e.g., rubber band ligation, hemorrhoidal artery ligation, radio frequency ablation), (8) all arms not linked to other eligible studies’ groups, and (7) articles in the form of letters, abstracts, editorials, or conference abstracts.
Data collection and quality assessment
In addition to the above-mentioned endpoints, the following data were also extracted from all eligible trials: first author’s name, study methodology, study center information, publication year, follow-up period, and patients’ gender, age, and body mass index (BMI). Moreover, information regarding patient characteristics, anesthetic technique, surgical procedure details, and local treatment regimen was, also, recorded.
Quality evaluation of the included RCTs was based on Cochrane’s Risk of Bias 2 assessing tool [24]. All data extraction and quality assessment were performed in duplicate and blindly by two independent researchers (C.C., P.K.). In case of discrepancy that was not resolved through mutual revision, the opinion of a third researcher (B.I.) was considered.
Statistical analysis
Continuous and categorical variables were presented in the form of mean (standard deviation (SD)) and N (percentage), respectively. The corresponding effect estimates were provided in the form of mean difference (MD) and odds ratio (OR). All pooled effect sizes were reported with the respective 95% confidence intervals (95%CI). In case that the mean or SD of an outcome was not provided, then they were estimated from the corresponding median, range, and interquartile range (IQR), based on the formula described by Hozo et al. [25].
Multiple arm comparisons were performed through the implementation of a random effects multivariate network meta-analysis under a Bayesian framework. Network geometry plot consisted of nodes and edges that represented the interventions and direct comparisons, respectively. The weight of the nodes and the thickness of the edges represented the number of studies that examined a treatment and compared two given interventions, respectively.
Local medication ranking was completed through the introduction of Markov chain Monte Carlo simulations. More specifically, the analysis model performed 25,000 sample iterations. The surface under the cumulative ranking curve (SUCRA) ranking and the respective ranking probabilities were also calculated. Ranking summary was depicted in Litmus Ran-o-Gram plots [26,27,28]. Subgroup analysis included the re-estimation of all endpoints’ SUCRA values for each surgical procedure respectively (Milligan-Morgan, Ferguson, energy device, and stapled hemorrhoidectomy).
Publication bias estimation was based on the visual inspection of the primary endpoint funnel plot. Statistical significance was considered at the level of p < 0.05.
Results
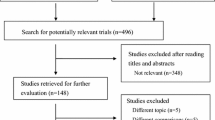
The application of the literature screening algorithm (Fig. 1) resulted to the retrieval of 6534 entries (Medline 2562, Scopus 2167, Web of Science 801, CENTRAL 984). After the removal of 2681 duplicate records, 3853 titles and abstracts were screened. Application of the eligibility criteria resulted to the exclusion of 3808 entries during this phase. Overall, 45 studies were submitted to a full text review, with 20 reports considered non-eligible (1 non-RCT [29], 2 studies with not linked study arms [30, 31], 3 conference abstracts/ editorials, and 14 irrelevant records). Moreover, 1 study was identified through reference list hand searching. Subsequently, 26 RCTs [15,16,17, 20, 21, 32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52] were included in the present meta-analysis.
Table 1 summarizes the characteristics of the eligible studies. Only two RCTs [15, 48] were performed in multiple centers. The publication year ranged from 2004 [51] to 2021 [20]. In total, 2132 patients were included in this pooled analysis. Gender, age, and BMI allocations in the various study subgroups were also provided in Table 1. Mean postoperative follow-up spanned from 1 [36] to 378 days [38]. Data regarding patient characteristics, anesthetic technique, surgical procedure details, and the local treatment regimen were provided in Supplementary Material Tables.
Quality evaluation of the eligible studies (Supplementary Material Figures) confirmed that most of them had minor methodological deficits and thus were associated with a low or moderate risk of bias. Kohen’s k statistic (k 80.2%, p < 0.001) confirmed a substantial level of interrater agreement.
Statistical analyses were available for all time endpoints of postoperative pain VAS measurements. Figure 2 summarizes the network geometry of the respective outcomes. Regarding the primary endpoint (VAS at 12 h), EMLA cream (combination of lidocaine and prilocaine) had the highest probability of ranking first (44%) and achieved an 80.3% SUCRA value (Fig. 3, Supplementary Material Tables). At 24 h, an aloe vera based cream ranked first (SUCRA 82.36%), while at 48 h, glyceryl trinitrate (SUCRA 89.6%) and aloe vera (SUCRA 77.7%) regimens had the highest probability of reducing postoperative pain. Similar results were estimated for Milligan-Morgan hemorrhoidectomies (Supplementary Material Tables), while diltiazem ranked first at 24- and 48-h VAS endpoint in the Ferguson subgroup. Metronidazole including ointments consistently had the highest scores at 7 (SUCRA 74.2%) and 14 days (SUCRA 91.6%) postoperatively.
Postoperative pain network plots a at 12 h, b at 24 h, c at 48 h, d at 7 days, e at 14 days, f during defecation at 24 h, g during defecation at 48 h, and h during defecation at 14 days; The weight of the nodes (orange) and the thickness of the edges (grey) represent the number of studies that examined a treatment and compared two given interventions, respectively
Ranking of treatments using Litmus Rank-O-Gram. Higher SUCRA values (green) correspond to higher ranking and better performance over lower SUCRA estimations (yellow and red). Postoperative pain Visual Analog Scale assessments: a at 12 h, b at 24 h, c at 48 h, d at 7 days, e at 14 days, f during defecation at 24 h, g during defecation at 48 h, and h during defecation at 14 days
Results regarding postoperative pain during defecation were provided for the 24-h, 48-h, and 14-day endpoints (Fig. 3, Supplementary Material Tables). In the immediate postoperative period, aloe vera ointments had the best analgesic profile (24-h SUCRA 84.8% and 48-h SUCRA 80.6%). At 14 days postoperatively, atorvastatin had the highest probability (SUCRA 82.5%) of ranking first.
Diclofenac-containing creams (Supplementary Material Tables, Supplementary Material Figures) were associated with the highest probability of reducing postoperative opioid consumption at 12 (SUCRA 85.6%) and 24 h (SUCRA: 85.2%). Cholestyramine ointments systematically had the worst opioid-sparing performance.
Lidocaine-based ointments (SUCRA 87.9%), followed by lidocaine and nifedipine combinations (SUCRA 85.6%), displayed the best performance regarding overall morbidity rates (Supplementary Material Tables, Supplementary Material Figures). Similar performance for lidocaine was noted in the Milligan-Morgan and stapled subgroup (Supplementary Material Tables), whereas placebo ranked first in the Ferguson and energy device arm. Furthermore, lidocaine ointments received the higher score in terms of postoperative itching (SUCRA 72.4%) and headache (SUCRA 92.2%), while glyceryl trinitrate (GTN) (SUCRA 16.5%) and its combinations (SUCRA 21.6%) had the worst performance. In terms of postoperative nausea and vomiting, EMLA (SUCRA 66.4%) creams had the highest ranking.
Visual inspection of the primary outcome funnel plot did not reveal the presence of publication bias (Supplementary Material Figures) since a symmetrical distribution of the eligible studies over the combined effect size line was identified.
Discussion
Summary of evidence
Hemorrhoidectomy is the standard of care for grade III and IV hemorrhoidal disease [53]. However, the main disadvantage of excisional approaches is the increased rate of severe postoperative pain [53]. Efficient analgesia is an important step in the effective application of a day-case hemorrhoidal surgery protocol [53]. Inadequate pain control after hemorrhoidectomy is directly associated with prolonged length of hospital stay, elevated patient inconvenience, extension of sick leave period, further need for medication regimens, and increased overall hospitalization costs [54]. According to Xia et al. [55], the inpatient conversion rate of day-case hemorrhoidectomies was 11.2%, with non-manageable pain being the leading cause of readmission.
A recent procedure-specific postoperative pain management guideline group [53] recommended that analgesia after hemorrhoidectomy should be based on a multimodal approach. However, the topical application of a medication after hemorrhoidectomy displays multiple advantages, such as higher local concentration of the active agent, avoidance of potentially serious adverse events, and increased patient compliance [56]. However, despite several available options, the local medication with the optimal analgesic profile is still not found [19].
In our pooled analysis, EMLA had the highest efficacy during immediate postoperative period. This is in parallel with the results of Shiau et al. [34] and Rahimi et al. [36], where the application of the local anesthetic combination showed a significant analgesic effect. Although aloe vera ranked first at 24 h, glyceryl trinitrate had the best probability of optimal analgesia at 2 days after hemorrhoidectomy. GTN increases anorectal blood flow and reduces the posthemorrhoidectomy sphincter spasm, thus providing a notable analgesic effect [37]. Accordingly, Karanlik et al. [37] reported a significant reduction in mean VAS scores of the GTN arm during the whole postoperative period after hemorrhoid excision.
Interestingly, metronidazole ointments had the highest SUCRA values in the medium- and long-term postoperative pain endpoints. The analgesic effect of metronidazole is still unclear [57]. Possible explanations include the prevention of wound infection by gut anaerobic bacteria and the anti-inflammatory properties of metronidazole [57]. On a clinical level, multiple studies reported an analgesic role for metronidazole after hemorrhoidal surgery [51, 52]. A recent meta-analysis by Lyons et al. [57] estimated a significant reduction of postoperative pain on days 1, 2, and 7. On the contrary, Wanis et al. [58] questioned the analgesic role of metronidazole, since after a sensitivity analysis and the exclusion of a low-quality study, the overall effect was not significant.
Due to the postoperative presence of scars in a well-innervated anatomical area [56], the evaluation of analgesia efficacy during defecation is pivotal in selecting the optimal local regimen. We estimated that aloe vera creams had the highest probability of reducing VAS scores during defecation in the first 48 h. Similarly, Eshghi et al. [43] reported that aloe vera had a significant effect on pain during bowel movement in the early postoperative period; this effect was not apparent, though, at the rest of the follow-up period [43]. Current literature supports that aloe vera preparations have multiple biological effects, including immunomodulatory, anti-inflammatory, antioxidant, and wound healing properties [43]. These could possibly be the pathophysiological basis of these significant clinical effects of aloe vera [43].
However, atorvastatin preparations outperformed aloe vera in terms of defecation analgesia at the 2-week endpoint. These findings are supported by a recent RCT from Ala et al. [49], where atorvastatin emulgel significantly reduced pain during defecation at 7 and 14 days postoperatively. Atorvastatin is a 3-hydroxy-3-methylglutaryl coenzyme A (HMG CoA) reductase inhibitor that has several pleiotropic effects [49]. Animal studies showed that atorvastatin-based analgesia is due to the attenuation of the neuropathic pain through inhibition of oxidative stress in the sciatic nerve, spinal cord, and brain parenchyma, while subsequent trials confirmed that this effect becomes apparent after 48 h [49].
Opioids are considered a major pillar of postoperative analgesia [59]. However, opioids display a major adverse event risk that affects their overall safety profile [59]. Due to the increased level of postoperative pain, opioid-based rescue analgesia is quite common in patients submitted to hemorrhoidectomy [60]. As a result, reduction of opioid requirements is an important efficacy endpoint for local preparations. Our network meta-analysis validated that diclofenac has the highest potential of reducing opioid consumption at 12 and 24 h postoperatively. In a 3-arm RCT by Rahimi et al. [36], diclofenac surpassed EMLA regarding overall opioid dosage. Diclofenac is a NSAID that acts through the inhibition of prostaglandin release [36]. Although previous studies describe a sustained sphincter spasm relieving effect of diclofenac, we did not provide long-term results due to the scarcity of relevant data.
The ideal topical regimen should also be characterized by minimal adverse effects. More specifically, the active agent that will be locally applied after HD surgery should reduce the risk for local and systemic complications, thus ensuring the safety of the treatment protocol. Although pairwise comparisons regarding the adverse event risk are abundant [15, 32, 35], this is not the case for pooled analyses. In a recent network meta-analysis by Yang et al. [19], the authors failed to identify overall morbidity data and thus to estimate the respective treatment rankings. In our study, lidocaine and lidocaine combined with nifedipine creams had the lowest risk of local and systematic complications. Similarly, in the RCT by Perrotti et al. [15], the addition of nifedipine to a lidocaine-based cream did not significantly increase the risk of adverse events (1.5% vs 0.7%).
Local adverse events, such as skin irritation and itching may be the result of multiple factors, including the surgical technique, the presence of contact allergens in the composition of the ointment, and the irritation potential of the active agent itself [15]. Although this complication is rarely reported, the clinical impact may be considerable in some cases, resulting to a substantial decrease in the postoperative quality of life [44, 52]. We confirmed that topical application of lidocaine was associated with the lowest risk for itching symptoms during the treatment regimen.
GTN-based regimens are traditionally associated with a significant risk of headache that may result to dose reduction or treatment cessation after HD surgery [39]. Although the exact pathway of GTN-induced headache is not still clear, current evidence suggests that NO release from blood vessels or brain parenchyma acts as a trigger mechanism for headache [61]. Our pooled analyses were on par with these findings; GTN-based ointments had the worst performance while lidocaine regimens ranked first, in terms of headache risk reduction.
Limitations
First, methodological deficits and small sample size of the included RCTs may have impacted the validity of the analyses’ outcomes. Furthermore, the discrepancies in terms of patient characteristics, anesthesia, and technical details were another considerable source of bias. Regarding the latter, although a subgroup ranking analysis was performed for all procedure types, the reported results should be critically acclaimed since comparisons for all interventions were scarcely available in most endpoints. Thus, in most cases, SUCRA ranking was reported for only a small proportion of the initial treatment pool. Moreover, the reported alterations in the dosage schemes may have impacted the clinical significance of the final outcomes. It must also be mentioned that the considerable variations in terms of follow-up period could have significantly affected time-related outcomes. Finally, the inconsistency in the examined local agents and time endpoints prohibited the safe extrapolation of conclusions regarding the ranking of all available treatments in terms of postoperative morbidity and perianal pain scores.
Conclusions
The present network meta-analysis confirmed that the optimal ointment-based medication for topical application after excisional procedures for HD is not yet established. The examined regimens showed an optimal performance in some outcomes, but no agent displayed superiority in all endpoints. The ideal topical medication should be associated with an acceptable safety profile, while in parallel, it should provide a consistent and long-term analgesic effect and minimize symptoms during defecation. Subsequently, and given several study limitations, further high-quality RCTs are required to provide paired comparisons and elucidate the efficacy and safety of the various topical medications in the posthemorrhoidectomy setting.
Data availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
References
Sheikh P, Régnier C, Goron F, Salmat G (2020) The prevalence, characteristics and treatment of hemorrhoidal disease: results of an international web-based survey. J Comp Eff Res 9:1219–1232. https://doi.org/10.2217/CER-2020-0159
Sandler RS, Peery AF (2019) Rethinking what we know about hemorrhoids. Clin Gastroenterol Hepatol 17:8. https://doi.org/10.1016/J.CGH.2018.03.020
Rørvik HD, Styr K, Ilum L et al (2019) Hemorrhoidal Disease Symptom Score and Short Health ScaleHD: new tools to evaluate symptoms and health-related quality of life in hemorrhoidal disease. Dis Colon Rectum 62:333–342. https://doi.org/10.1097/DCR.0000000000001234
van Tol RR, Bruijnen MPA, Melenhorst J et al (2018) A national evaluation of the management practices of hemorrhoidal disease in the Netherlands. Int J Colorectal Dis 33:577–588. https://doi.org/10.1007/S00384-018-3019-5
Gallo G, Martellucci J, Sturiale A et al (2020) Consensus statement of the Italian society of colorectal surgery (SICCR): management and treatment of hemorrhoidal disease. Tech Coloproctol 24:145–164. https://doi.org/10.1007/S10151-020-02149-1
Simillis C, Thoukididou SN, Slesser AAP et al (2015) Systematic review and network meta-analysis comparing clinical outcomes and effectiveness of surgical treatments for haemorrhoids. Br J Surg 102:1603–1618. https://doi.org/10.1002/BJS.9913
Balciscueta Z, Balciscueta I, Uribe N (2021) Post-hemorrhoidectomy pain: can surgeons reduce it? A systematic review and network meta-analysis of randomized trials. Int J Colorectal Dis 36:2553–2566. https://doi.org/10.1007/S00384-021-04013-6
Ng KS, Holzgang M, Young C (2020) Still a case of “no pain, no gain”? An updated and critical review of the pathogenesis, diagnosis, and management options for hemorrhoids in 2020. Ann Coloproctol 36:133–147. https://doi.org/10.3393/AC.2020.05.04
Vinson-Bonnet B, Higuero T, Faucheron JL et al (2015) Ambulatory haemorrhoidal surgery: systematic literature review and qualitative analysis. Int J Colorectal Dis 30:437–445. https://doi.org/10.1007/S00384-014-2073-X
Ye S, Zhou J, Guo X, Jiang X (2022) Three acupuncture methods for postoperative pain in mixed hemorrhoids: a systematic review and network meta-analysis. Comput Math Methods Med 2022. https://doi.org/10.1155/2022/5627550
Huang YJ, Chen CY, Chen RJ et al (2018) Topical diltiazem ointment in post-hemorrhoidectomy pain relief: a meta-analysis of randomized controlled trials. Asian J Surg 41:431–437. https://doi.org/10.1016/J.ASJSUR.2017.06.002
Fu H, Guo W, Zhou B et al (2022) Efficacy and safety of micronized purified flavonoid fractions for the treatment of postoperative hemorrhoid complications: a systematic review and meta-analysis. Phytomedicine 104. https://doi.org/10.1016/J.PHYMED.2022.154244
Rudiman R, Hanafi RV, Evan C, Halim F (2023) The efficacy of topical sucralfate in improving pain and wound healing after haemorrhoidectomy procedure: a systematic review, meta-analysis, and meta-regression of randomised clinical trials. Int Wound J 20:543–553. https://doi.org/10.1111/IWJ.13901
Lie H, Lukito PP, Sudirman T et al (2023) Utility of botulinum toxin injection for post-operative pain management after conventional hemorrhoidectomy: a systematic review and meta-analysis of clinical trials. Scand J Gastroenterol 58:116–122. https://doi.org/10.1080/00365521.2022.2116292
Perrotti P, Dominici P, Grossi E et al (2010) Topical nifedipine with lidocaine ointment versus active control for pain after hemorrhoidectomy: results of a multicentre, prospective, randomized, double-blind study. Can J Surg 53:17–24
Tan K-Y, Sng KK, Tay K-H et al (2006) Randomized clinical trial of 0.2 per cent glyceryl trinitrate ointment for wound healing and pain reduction after open diathermy haemorrhoidectomy. Br J Surg 93:1464–1468. https://doi.org/10.1002/bjs.5483
Sugimoto T, Tsunoda A, Kano N et al (2013) A randomized, prospective, double-blind, placebo-controlled trial of the effect of diltiazem gel on pain after hemorrhoidectomy. World J Surg 37:2454–2457. https://doi.org/10.1007/s00268-013-2124-4
Jin J, Unasa H, Bahl P et al (2023) Can targeting sphincter spasm reduce post-haemorrhoidectomy pain? A systematic review and meta-analysis. World J Surg 47:520–533. https://doi.org/10.1007/s00268-022-06807-3
Yang Y, Feng K, Lei Y et al (2023) Comparing the efficacy and safety of different analgesic strategies after open hemorrhoidectomy: a systematic review and network meta-analysis. Int J Colorectal Dis 38:4. https://doi.org/10.1007/s00384-022-04294-5
Abidi SS, Bakhtiar N, Kerawala AA, Awan S (2021) Topical diltiazem ointment for post- hemorrhoidectomy pain. J Ayub Med Coll Abbottabad 33:125–128
Vejdan AK, Khosravi M, Amirian Z et al (2020) Evaluation of the efficacy of topical sucralfate on healing haemorrhoidectomy incision wounds and reducing pain severity: a randomised clinical trial. Int Wound J 17:1047–1051. https://doi.org/10.1111/iwj.13369
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA (editors) (2023) Cochrane Handbook for Systematic Reviews of Interventions version 6.4 (updated August 2023). Cochrane. Available from https://www.training.cochrane.org/handbook
Hutton B, Salanti G, Caldwell DM et al (2015) The PRISMA extension statement for reporting of systematic reviews incorporating network meta-analyses of health care interventions: checklist and explanations. Ann Intern Med 162:777. https://doi.org/10.7326/M14-2385
Sterne JAC, Savović J, Page MJ et al (2019) RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 366:l4898. https://doi.org/10.1136/bmj.l4898
Hozo SP, Djulbegovic B, Hozo I (2005) Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol 5:13. https://doi.org/10.1186/1471-2288-5-13
Salanti G, Ades AE, Ioannidis JPA (2011) Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol 64:163–171. https://doi.org/10.1016/J.JCLINEPI.2010.03.016
Kossmeier M, Tran US, Voracek M (2020) Charting the landscape of graphical displays for meta-analysis and systematic reviews: a comprehensive review, taxonomy, and feature analysis. BMC Med Res Methodol 20. https://doi.org/10.1186/S12874-020-0911-9
Veroniki AA, Straus SE, Fyraridis A, Tricco AC (2016) The rank-heat plot is a novel way to present the results from a network meta-analysis including multiple outcomes. J Clin Epidemiol 76:193–199. https://doi.org/10.1016/J.JCLINEPI.2016.02.016
Kashif M, Ch TS, Bayazeed DA et al (2020) Effectiveness of 10% sucralfate ointment in the prevention of postoperative pain in patients undergoing open hemorrhoidectomy. Pakistan J Med Health Sci 14:1932–1934
Basso MP, Christiano AB, Marciano MR et al (2011) Comparison of effect of topical metronidazole 10% associated with nifedipine 0.3% versus nifedipine 0.3% alone in postoperative hemorrhoidectomy by hybrid technique; [Comparação do efeito do metronidazol tópico 10% associado à nifedipina tópica 0,3% versu. Revista Brasileira de Coloproctologia 31:131–138. https://doi.org/10.1590/S0101-98802011000200004
Bader QA, Mohammed HR, Hatef ZS (2020) Stability and analgesic efficacy study for a mixture of diltiazem, lidocaine and diclofenac gel formulations in post hemorrhoidectomy pain reduction in Iraqi patients. Int J Pharm Res 12:2792–2800. https://doi.org/10.31838/ijpr/2020.SP2.333
Vahabi S, Beiranvand S, Karimi A, Moradkhani M (2019) Comparative study of 0.2% glyceryl trinitrate ointment for pain reduction after hemorrhoidectomy surgery. Surg J (N Y) 5:e192–e196. https://doi.org/10.1055/s-0039-3400532
Albatanony A (2016) Sucralfate ointment reduces pain and improves healing following haemorrhoidectomy: a prospective, randomized, controlled and double-blinded study. Egypt J Surg 35:102. https://doi.org/10.4103/1110-1121.182783
Shiau J-M, Su H-P, Chen H-S et al (2008) Use of a topical anesthetic cream (EMLA) to reduce pain after hemorrhoidectomy. Reg Anesth Pain Med 33:30–35. https://doi.org/10.1016/j.rapm.2007.07.012
Amoli HA, Notash AY, Shahandashti FJ et al (2011) A randomized, prospective, double-blind, placebo-controlled trial of the effect of topical diltiazem on posthaemorrhoidectomy pain. Colorectal Dis 13:328–332. https://doi.org/10.1111/j.1463-1318.2009.02138.x
Rahimi M, Kazemeini AR, Pourtabatabaei N, Honarmand AR (2012) Comparison of topical anesthetic cream (EMLA) and diclofenac suppository for pain relief after hemorrhoidectomy: a randomized clinical trial. Surg Today 42:1201–1205. https://doi.org/10.1007/s00595-012-0222-9
Karanlik H, Akturk R, Camlica H, Asoglu O (2009) The effect of glyceryl trinitrate ointment on posthemorrhoidectomy pain and wound healing: results of a randomized, double-blind, placebo-controlled study. Dis Colon Rectum 52:280–285. https://doi.org/10.1007/DCR.0b013e31819c98a7
Cross T, Bartlett L, Mushaya C et al (2012) Glyceryl trinitrate ointment did not reduce pain after stapled hemorrhoidectomy: a randomized controlled trial. Int Surg 97:112–119. https://doi.org/10.9738/CC92.1
Mari FS, Nigri G, Dall’Oglio A et al (2013) Topical glyceryl trinitrate ointment for pain related to anal hypertonia after stapled hemorrhoidopexy: a randomized controlled trial. Dis Colon Rectum 56:768–773. https://doi.org/10.1097/DCR.0b013e31828b282c
Gupta PJ, Heda PS, Kalaskar S, Tamaskar VP (2008) Topical sucralfate decreases pain after hemorrhoidectomy and improves healing: a randomized, blinded, controlled study. Dis Colon Rectum 51:231–234. https://doi.org/10.1007/s10350-007-9092-4
Rodríguez-Wong U, Ocharán-Hernández ME, Toscano-Garibay J (2016) Topical diltiazem for pain after closed hemorrhoidectomy. Rev Gastroenterol Mex 81:74–79. https://doi.org/10.1016/j.rgmx.2016.02.001
Yadav S, Khandelwal RG, Om P et al (2018) A prospective randomized double-blind study of pain control by topical calcium channel blockers versus placebo after Milligan-Morgan hemorrhoidectomy. Int J Colorectal Dis 33:895–899. https://doi.org/10.1007/s00384-018-3067-x
Eshghi F, Hosseinimehr SJ, Rahmani N et al (2010) Effects of Aloe vera cream on posthemorrhoidectomy pain and wound healing: results of a randomized, blind, placebo-control study. J Altern Complement Med 16:647–650. https://doi.org/10.1089/acm.2009.0428
Ala S, Saeedi M, Eshghi F et al (2013) Efficacy of 10% sucralfate ointment in the reduction of acute postoperative pain after open hemorrhoidectomy: a prospective, double-blind, randomized, placebo-controlled trial. World J Surg 37:233–238. https://doi.org/10.1007/s00268-012-1805-8
Silverman R, Bendick PJ, Wasvary HJ (2005) A randomized, prospective, double-blind, placebo-controlled trial of the effect of a calcium channel blocker ointment on pain after hemorrhoidectomy. Dis Colon Rectum 48:1913–1916. https://doi.org/10.1007/s10350-005-0135-4
Ala S, Eshghi F, Enayatifard R et al (2013) Efficacy of cholestyramine ointment in reduction of postoperative pain and pain during defecation after open hemorrhoidectomy: results of a prospective, single-center, randomized, double-blind, placebo-controlled trial. World J Surg 37:657–662. https://doi.org/10.1007/s00268-012-1895-3
Franceschilli L, D’Ugo S, de Luca E et al (2013) Role of 0.4% glyceryl trinitrate ointment after haemorrhoidectomy: results of a prospective randomised study. Int J Colorectal Dis 28:365–369. https://doi.org/10.1007/s00384-012-1544-1
Khan KI, Waqas A, Akmal M et al (2014) Efficacy of combination of 0.2% GTN and lignocaine ointments in wound healing and pain relief after Milligan Morgan hemorrhoidectomy–a comparison with lignocaine and 0.2% GTN ointments separately. Int J Surg 12:329–333. https://doi.org/10.1016/j.ijsu.2014.01.013
Ala S, Alvandipour M, Saeedi M et al (2017) Effects of topical atorvastatin (2 %) on posthemorrhoidectomy pain and wound healing: a randomized double-blind placebo-controlled clinical trial. World J Surg 41:596–602. https://doi.org/10.1007/s00268-016-3749-x
Ala S, Alvandipour M, Saeedi M et al (2020) Effect of topical baclofen 5% on post-hemorrhoidectomy pain: randomized double blind placebo-controlled clinical trial. J Gastrointest Surg 24:405–410. https://doi.org/10.1007/s11605-019-04147-7
Nicholson TJ, Armstrong D (2004) Topical metronidazole (10 percent) decreases posthemorrhoidectomy pain and improves healing. Dis Colon Rectum 47:711–716. https://doi.org/10.1007/s10350-003-0129-z
Ala S, Saeedi M, Eshghi F, Mirzabeygi P (2008) Topical metronidazole can reduce pain after surgery and pain on defecation in postoperative hemorrhoidectomy. Dis Colon Rectum 51:235–238. https://doi.org/10.1007/s10350-007-9174-3
Sammour T, Barazanchi AWH, Hill AG et al (2017) Evidence-based management of pain after excisional haemorrhoidectomy surgery: a PROSPECT review update. World J Surg 41:603–614. https://doi.org/10.1007/S00268-016-3737-1
Roche B, Marti MC (2000) Outpatient proctological surgery: an analysis of 3725 cases. Tech Coloproctol 4:35–38. https://doi.org/10.1007/S101510050052/METRICS
Xia W, Barazanchi AWH, MacFater W et al (2019) Day case versus inpatient stay for excisional haemorrhoidectomy. ANZ J Surg 89:E5–E9. https://doi.org/10.1111/ANS.14838
Sammour T (2022) Pain after hemorrhoidectomy. Dis Colon Rectum 65:951–952
Lyons NJR, Cornille JB, Pathak S et al (2017) Systematic review and meta-analysis of the role of metronidazole in post-haemorrhoidectomy pain relief. Colorectal Dis 19:803–811. https://doi.org/10.1111/codi.13755
Wanis KN, Emmerton-Coughlin HM, Coughlin S et al (2017) Systemic metronidazole may not reduce posthemorrhoidectomy pain: a meta-analysis of randomized controlled trials. Dis Colon Rectum 60:446–455. https://doi.org/10.1097/DCR.0000000000000792
Homsi J, Brovman EY, Rao N et al (2019) The association between potential opioid-related adverse drug events and outcomes in colorectal surgery. J Laparoendosc Adv Surg Tech A 29:1436–1445. https://doi.org/10.1089/LAP.2019.0408
Lu PW, Fields AC, Andriotti T et al (2020) Opioid prescriptions after hemorrhoidectomy. Dis Colon Rectum 63:1118–1126. https://doi.org/10.1097/DCR.0000000000001570
Thomsen LL, Olesen J (2001) Nitric oxide in primary headaches. Curr Opin Neurol 14:315–321. https://doi.org/10.1097/00019052-200106000-00009
Author information
Authors and Affiliations
Contributions
Conception and design of the study: PK. Acquisition of data: CC and PK. Drafting the article: CC and PK. Critical revision: SD, TK, and BI. Final approval: TG.
Corresponding author
Ethics declarations
Ethics approval
This is a systematic review and meta-analysis. The institutional Research Ethics Committee has confirmed that no ethical approval is required.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Perivoliotis, K., Chatzinikolaou, C., Symeonidis, D. et al. Comparison of ointment-based agents after excisional procedures for hemorrhoidal disease: a network meta-analysis of randomized controlled trials. Langenbecks Arch Surg 408, 401 (2023). https://doi.org/10.1007/s00423-023-03128-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00423-023-03128-4