Abstract
Background
The appropriate extent of gastric resection for patients with proximal third gastric cancer is controversial. This study addresses whether the choice of surgical strategy (proximal gastrectomy [PG] versus total gastrectomy [TG]) influences the outcomes for proximal third gastric adenocarcinoma.
Materials and methods
Review of prospective database at Tata Memorial Hospital from January 2010 to December 2012 identified 343 patients diagnosed and treated for gastric cancer. Of these, 75 underwent curative resections with D2 lymphadenectomy for proximal third gastric adenocarcinoma, which entailed proximal gastrectomy in 43 and total gastrectomy in 32 patients, depending on the epicenter of the primary and its relation with the mid-body of the stomach. Morbidity, lymph node yield, resection margins, patterns of recurrence, and survival were compared between these two groups.
Results
41/75 tumors were pT3 (23 cases [53.4 %] in the PG and 18 cases [56.3 %] in the TG group). Thirty-six patients [83.7 %] in PG and 29 patients [90.6 %] in TG group received neoadjuvant chemotherapy (NACT). There were no significant differences with regard to median blood loss, general complication rates and length of hospitalization between the two groups. The lymph node yield was comparable between the two procedures [PG = 14; TG = 15]. Positive proximal resection margin rates were comparable between the two groups [PG = 4.7 %; TG = 9.4 %], and there was no statistical difference observed in the distal resection margin positivity rates [PG = 4.7 %; TG = 3.1 %]. Regarding the patterns of recurrence, local recurrence in PG was 4.7 % and there was no local recurrence in the TG group (p = 0.08). Distant recurrence rates was dominant in TG [PG = 30.2 % versus TG = 53.1 %]. The overall 2-year survival following PG and TG was 73.8 and 49.9 %, respectively, and not statistically different (p = 0.10).
Conclusions
The extent of resection for proximal third gastric cancer does not influence the clinical outcome. PG and TG have similar survival rates. Both procedures can be accomplished safely. Therefore, PG should be an alternative to TG, even in locally advanced proximal gastric cancers treated by NACT, provided that the tumor size and location permit preservation of adequate remnant of stomach without compromising oncological resection margins. Future QOL studies would further lend credence to the concept of PG for proximal third gastric cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastric cancer remains a cancer with a high mortality rate the world over [1]. Over the past decades, the predominant site of occurrence has shifted from the distal to the proximal stomach [2, 3]. In North American and some European countries, adenocarcinoma of the cardia is the primary type of gastric cancer. In Asia, a similar trend is observed [4]. The shift in gastric cancer subsite has renewed interest in the management of upper third gastric cancer with a focus on the optimal extent of resection, i.e., proximal (PG) or total gastrectomy (TG).
Gastric remnant preservation by reducing the extent of radical gastrectomy is an important goal of surgical treatment [5]. For distal gastric malignancy, consensus has been reached that curative R0 resection can be achieved by subtotal gastrectomy which is considered oncologically equivalent to TG even in locally advanced but resectable disease [6, 7]. Proximal gastric cancer can be treated either by TG or PG. The choice of either of the procedures is dictated by tumor size and epicenter, tumor stage, potential volume of the future remnant stomach, and surgeon’s preference. Proponents of TG have argued that complete resection can achieve a longer tumor-free distal resection margin along with a more radical lymphadenectomy which translates into better curative effect [8]. Alternatively, others argue that PG achieves a survival rate equivalent to that of TG, with added advantage of preservation of the gastric remnant [9].
The newly published “Japanese Gastric Cancer Treatment Guidelines 2010” recommends that PG is only suitable for some early stage diseases [10] i.e., for cT1cN0 tumors, where more than half of the distal stomach can be preserved. However, majority of published studies showed that there is no significant difference in survival rate between TG and PG [11–13].
Thus, the optimal extent of gastric resection for proximal gastric cancer remains controversial and there is still no general agreement on the choice of surgical extent.
The purpose of this study is to present our prospective 2-year experience with upper third gastric cancer treated with either radical PG or with TG and to evaluate the surgical outcome of these procedures in an attempt to define an optimal approach for upper third gastric cancer.
Materials and methods
Patients
Medical records of 343 patients (Fig. 1a) undergoing surgery for gastric cancer between January 2010 and December 2012, at the Division of Gastrointestinal and Hepato-Pancreato-Biliary Surgical Oncology, Tata Memorial Hospital, Mumbai, India were evaluated from a prospectively maintained database. Radical D2 gastrectomy was performed by or under the supervision of the consultant surgeons in the unit (SVS, MG). Among these, 75 patients (21.8 %) underwent curative gastric resection for upper third gastric cancer. Preoperatively, all patients were investigated with routine blood investigations, including blood counts, liver and renal functions, and an endoscopy to map the location of the disease and to obtain tissue diagnosis. A contrast-enhanced multi-detector computed tomography (MDCT) scan of the abdomen and pelvis was performed to stage the disease.
Eligibility criteria
Inclusion
Siewert’s classification is a well-established and an internationally accepted classification for adenocarcinomas of the esophagogastric junction (AEG). It helps to choose the appropriate surgical approach and to make results from different institutions comparable.
Type I tumors are a distinct entity that should be treated as a distal esophageal cancer. In contrast, type III tumors represent proximal gastric cancer and should be approached in accordance with gastric cancer guidelines. The characterization of type II tumors, however, remains controversial. Most evidence suggests that these tumors behave more like proximal gastric tumors than distal esophageal adenocarcinomas [14, 15].
In this series, upper third gastric cancer was defined as gastric cancer involving the GE junction exclusively and proximal gastric cancer involving the cardia, fundus, proximal body, or all the three subsites plus possible involvement of the GE junction as assessed by endoscopy/radiology/intraoperatively, i.e., Siewert–Stein type II and III.
Management of type II and III tumors, however, remains controversial. A vast variety of approaches for surgical resection of tumors of the esophagogastric junction has been proposed, including abdominothoracic en bloc esophagogastrectomy, subtotal esophagectomy with resection of the proximal stomach, total gastrectomy with transhiatal resection of the distal esophagus, and limited resection of the esophagogastric junction.
In accordance with the recently published French National recommendations [16], two surgical options are possible for Siewert’s type II: either a total gastrectomy with partial esophagectomy through an abdominal approach, or a proximal gastrectomy associated with distal esophagectomy through a transthoracic or transhiatal approach.
EORTC-gastrointestinal cancer group recommendations for surgery of resectable gastric/GE junction adenocarcinoma could be a free-margin surgery with at least D1 resection combined to removal of a minimum of 15 lymph nodes [17].
In general, it has been well-accepted that the selection of the surgical approach to GEJ cancers should be tailored based upon achieving macroscopic and microscopic tumor resection [18]. Hence transabdominal gastrectomy with less extensive esophageal resection is also an option in Siewert’s II.
Exclusion
Patients who had any one of the following were excluded: Siewert–Stein type I adenocarcinoma, stage IV gastric cancer, gastric remnant adenocarcinoma, anastomotic site recurrent carcinoma, gastrointestinal stromal tumors (GIST), neuroendocrine tumors, lymphomas, antro-pyloric lesions, synchronous primary, combined operation, cirrhotic in liver failure, and recent myocardial infarction within the previous 6 months.
Staging
We did not demonstrate the tumor size or invasion beyond the middle third of the stomach in the two groups. We have not done this, as in our clinical practice this did not influence our treatment approach, and since they are not an integral part of AJCC-TNM staging. Staging was done in accordance with the seventh edition of the AJCC for having a standard understanding.
Planning for neoadjuvant chemotherapy
All patients diagnosed with locally advanced but resectable gastric cancer received perioperative chemotherapy as per the MAGIC trial protocol. The imaging details of every patient were reviewed at the joint meeting of the Gastrointestinal Disease Management Group (GI-DMG) as per routine policy. The indications for directing patients towards NACT were as follows: (1) biopsy-proven gastric adenocarcinoma, (2) MDCT scans indicative of tumor stage ≥T3, i.e., circumferential wall thickening, perigastric fat stranding, with or without nodal metastases, and (3) no evidence of distant metastases on MDCT scan.
Re-staging/ response assessment after NACT
All patients were restaged with a MDCT scan 3 to 4 weeks after completion of the NACT, and a final decision was made for surgery if complete response or stable disease was recorded after NACT.
Surgical approach
A curative resection entailed proximal or total gastrectomy with a radical D2 lymphadenectomy. A gross margin of 5 cm on stretched specimens just after resection was desirable; however, inpatients treated with neoadjuvant chemotherapy, a negative margin of at least 1cm, as confirmed on intraoperative frozen section (IOFS), was deemed acceptable.
Total gastrectomy was performed to achieve sufficient margins when the epicenter of the primary extended beyond the confines of the mid-body and whenever there was a risk of close margins as perceived by the surgeon and when the remnant stomach was inadequate to achieve a safe tension-free esophagogastric anastomosis.
TG was performed in 32 patients followed by Roux-En-Y, end to side, mechanical circular stapled esophago-jejunostomy and jejuno-jejunostomy for biliary diversion (CDH 25, J &J, USA).
Proximal gastrectomies were performed when the epicenter of the cancer was limited to the gastro-esophageal junction, fundus, cardia, proximal or mid-body, in which at least half of the distal stomach could be preserved.
PG was performed in 43 patients. The operative procedure entailed surgical resection of the tumor-free distal esophagus and proximal stomach with adequate margins. Restoration of the digestive tract continuity following a curative proximal gastric resection was done by a single-layer, tension-free, mechanical circular stapled esophago-gastric anastomosis (CDH 29, J &J, USA) or hand-sewn by 4–0 PDS sutures in two layers.
Seven patients in PG underwent thoracoabdominal approach, when complete resection was less likely to be achieved with the abdominal approach alone, based on the patient factors such as body habitus (narrow costal margins), prior abdominal surgery, and hiatus hernia.
Proximal and distal resection margins were evaluated by intraoperative frozen section to confirm disease-free resection margins. Distal margins were measured from the distal-most part of the tumor bearing mucosa to distal-most part of uninvolved mucosa and not from the epicenter of the disease to uninvolved mucosa. Similarly, proximal margins were also measured.
All procedures were performed in a standardized manner and included proximal or total gastrectomy along with a D2 lymphadenectomy, as adopted from the technique practiced by the National Cancer Center, Tokyo, Japan, since 2002 [19, 20].
Prophylactic splenectomy with a total gastrectomy was not a routine practice at our institute in view of lack of level I evidence to support a survival benefit [21].
Multivisceral resection was performed whenever the gastric cancer mass had directly infiltrated the adjacent resectable organ or in the presence of extensive adhesions between the cancer and adjacent organs.
A standard D2 lymph node dissection was performed in all individuals which incorporated one to 12 nodal stations, excluding the suprapyloric and infrapyloric stations five and six in proximal gastrectomy.
All patients were followed up regularly as per standardized follow up protocol which consisted of regular physical examinations, hemogram, blood counts, biochemistry profile, tumor markers, chest X-ray, and ultrasonography of the abdomen and pelvis. Upper GI endoscopy and CECT was performed only when there was a high index of suspicion of recurrence or for patient symptoms.
Clinicopathological parameters such as age, gender, blood loss, complications, 30-day mortality, length of hospitalization, histological type, stage, resection margin status, lymph node yield, pathologically positive lymph nodes, recurrence, pattern of recurrence, and survival were compared and analyzed between the two groups based on information obtained from medical records.
Ethics
The data of the present study were collected in the course of common clinical practice and, accordingly, the signed informed consent was obtained from each patient for any surgical and clinical procedure. The study protocol conforms to the ethical guidelines of the “World Medical Association Declaration of Helsinki: Ethical Principles for Medical Research Involving Human Subjects” adopted by the 18th WMA General Assembly, Helsinki, Finland, June 1964, as revised in Tokyo 2004. No approval of the institutional review committee was needed.
Statistical analysis
All statistical analyses were carried out with the Statistical Package for the Social Sciences (SPSS) version 20. Kaplan–Meier method and log-rank test was used to compare the survival curves (in months) between the two groups. Cases lost to follow-up were considered to have died due to disease while analyzing the survival data. Categorical variables between the groups were compared by Chi-square test. A p value less than 0.05 was considered to be statistically significant.
Results
Clinical characteristics (Table 1)
Seventy-five patients underwent gastric resection and standardized D2 lymphadenectomy for proximal gastric cancer. PG was performed in 43 patients and 32 underwent TG. The median age in PG and TG group was 53 years (range 29–74) and 59 years (range 23–85), respectively. There was male predominance in both the groups (86 % in PG and 75 % in TG). Twenty-nine cases (67.4 %) in the PG cohort were GE junction tumors and 16 cases (50 %) in the TG were proximal body tumors. Majority of the tumors were pT3 and amounted for 23 cases (53.4 %) in the PG and 18 cases (56.3 %) in the TG group. Neoadjuvant chemotherapy was administered to 36 patients (83.7 %) in PG and 29 patients (90.6 %) in TG. Detailed clinicopathological characteristics of the study groups are listed in Table 1. Both the groups were comparable with regard to clinicopathological variables. A detailed endoscopic mapping of the disease extent, in relation to the mid-body of the stomach in the present study is described in Table 2. Twenty-five percent (08/32) of cases who underwent a TG had a disease extending from the GE junction to beyond the mid-body of the stomach, 15.6 % (05/32) had a disease extending from fundus to beyond mid-body, and 50 % (16/32) had disease extending from the proximal body to beyond the mid-body.
Pathological characteristics (Tables 1 and 3)
There were no significant differences in tumor differentiation and overall stage between the groups. The median number of lymph nodes retrieved from the surgical specimen was 15 for TG and 14 for PG which was not statistically significant (p = 0.82). The number of histological positive nodes between the groups was higher in TG as compared to PG (mean 0–9 versus 1–18); however, the median number of positive lymph nodes for both the cohort was 1. Following PG, proximal margin was involved by the disease in two patients (4.7 %) while three cases (9.4 %) had a positive proximal margin after TG. Distal margin positive rates were 4.7 and 3.1 %, respectively, for PG and TG. The surgical margin status rates were not significantly different between the groups. All patients who had margin positivity on final histopathological report (HPR) received adjuvant chemoradiotherapy (as a part of perioperative chemotherapy). Splenectomy rates in PG and TG groups were 2.3 and 3.1 %, respectively. Only one patient in the PG group underwent splenectomy along with multivisceral resection. Curative gastric resection was performed in all the 75 individuals. The distribution of the depth of tumor invasion, lymph node, and distant metastasis along with residual tumor were comparable between the two groups as shown in Table 3.
Morbidity and mortality
Median length of postoperative hospitalization following PG and TG was 9 and 10 days, respectively. Postoperative complications have been defined as per our previous publication (anastomotic and duodenal stump leaks were detected either by drainage of bilious contents in the tube drains placed in the abdominal cavity or signs and symptoms suggestive of intra-abdominal sepsis, or both) and delayed gastric emptying (DGE) as per the International Study Group of Pancreatic Surgery (ISGPS) [22]. All complications were graded according to the Clavien–Dindo classification [23]. The postoperative complication rates in PG group was 29.9 and 27.9 % in TG and there was no differences in complication rates including DGE, surgical site infections, anastomotic leaks, intestinal obstruction (Table 4). There was no 30-day mortality in the study group.
Recurrence
The median follow-up for patients undergoing curative gastric resection in the PG and TG group were 19 months (range 7–43 months) and 17 months (range 4–43 months), respectively. The pattern of recurrence was confirmed by clinicoradiological examination. In the PG group, two cases (4.7 %) had local recurrence and none amongst the TG. Distant recurrence was dominant in the TG group (Table 5).
Survival
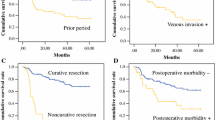
The estimated 2-year overall survival rate in the PG group was 73.8 and 49.9 % for TG (Fig. 2a). Stage-specific survival analysis between the groups was made by combining stage I with II and stage III-A with III-B and C. The 2-year overall survival for stage I and II was 75.7 % in PG and 63 % in TG (Fig. 2b) while the survival rates in stage III-A and III-B + C was 42.9 % in PG and 30.1 % in the TG group (Fig. 2c).
a. Cumulative survival curves of patients treated by proximal and total gastrectomy. p = 0.10 (log rank test). b Cumulative survival curves of patients treated with PG and TG for stage I and II. p = 0.37 (log rank test). c Cumulative survival curves of patients treated with PG and TG for stage III-A and III-B + C. p = 0.39 (log-rank test)
Discussion
In India, the second most common type of fatal cancer amongst men and women aged between 30 and 69 years is gastric cancer. It is 12.6 % amongst men and 14.1 % in women [24]. In the West, there has been a dramatic rise in the incidence of proximal gastric cancer [25]. The clinical relevance is that the overall prognosis of patients with proximal gastric cancer is worse than for those with distal tumors. A striking difference in the pathoanatomy of proximally located gastric cancer is observed between the East and the West. Siewert’s type I tumor is the most prevalent type in Europe [26, 27] while type III subcardia tumors are the most common type in Japan [28].
The extent of optimal gastric resection for proximal gastric cancer continues to remain controversial, and a final consensus is yet to be met. Skepticism about PG lingers amongst surgeons.
Although some concerns persist regarding the oncological adequacy and extent of radical lymphadenectomy of PG, the data published so far are promising. Previous retrospective studies that compared TG to PG had hardly showed superior results of TG in terms of long-term survival [29–31]. A 5-year overall survival analysis by Yoo CH et al., showed no significant difference between the PG (54.8 %) and TG (47.8 %). However, a major concern in the literature is the risk of reflux esophagitis as a result of direct anastomosis between the esophagus and the remnant stomach following proximal gastrectomy [32, 33].
This study is perhaps the largest series from the Indian subcontinent, and to the best of our knowledge, this is the first analysis comparing PG and TG in the post- neoadjuvant chemotherapy (NACT) era. The findings of this study support the equivalence of TG and PG in terms of lymphadenectomy, resection margins, morbidity, recurrence, and survival for both early and locally advanced proximal gastric adenocarcinoma.
The extent of lymphadenectomy is a consideration when TG and PG are compared. A pattern of lymph nodal metastasis in proximal gastric cancer is thought to flow from the nodal basin number 3 to nodal basin number 7 then to number 9 or number 16 [34]. Recent studies have shown that early proximal third gastric cancer is free of metastases to the lower perigastric lymph nodes [35, 36], and the newly published “Japanese Gastric Cancer Treatment Guidelines 2010” recommends that PG is only suitable for some early-stage diseases.
Maruyama et al. [37] and Kazuya et al. [38] Demonstrated that the incidence of LN metastasis increased with deeper tumor invasion. It is imperative that TG should be recommended for advanced proximal gastric cancer. However, several studies have reported that probability of the lymph nodal metastasis to the supra- and infrapyloric lymph nodes was very low in cases of upper third gastric cancer, and that resection of these nodes had little impact on survival [38–40].
Goto H. et al. [41] demonstrated potential possible involvement and difference with regard to lymph nodal station 5, amongst Siewert’s II and III but with no real impact on overall survival. Therefore, we have reasons to believe that function-preserving surgery such as well-done proximal gastrectomy, could still be a reasonable approach to Siewert’s III responders, in the era of perioperative chemotherapy.
Inarguably, TG enables a more complete nodal dissection and it is apparent from the number of lymph nodes retrieved in the current study that TG does result in a somewhat better lymph node yield. Although TG provides more complete lymphadenectomy, the clinical relevance of these factors is uncertain. In our series, median lymph nodes retrieved were 14 in the PG and 15 in the TG group. The range of lymph nodes retrieved was 1–27 in PG and 2–31 in TG. A subset of induviduals, 14 % in PG and 6.3 % in TG had complete pathological response at primary following neoadjuvant chemotherapy. Probably, the lymph nodal yield amongst this subset of patients was low in view of the response at nodal stations as well.
Wide tumor-free resection margins offer an advantage in terms of local control rates. Although the level of proximal resection margin is the same between TG and PG, TG naturally provides a longer distal tumor-free margin. In our series, 86 % in the PG and 87.5 % in the TG group had adequate proximal and distal tumor-free resection margins. In our study, the fresh specimen was opened longitudinally and the gross length of the margin was decided by the surgeon on the basis of visual and tactile sensation; in addition to this, all specimens were subjected to IOFS analysis for confirmation of margin adequacy and need for immediate revision of margins. IOFS guided in converting 6.9 % of planned PG to a TG to achieve adequate margins; however, the discordance rates between IOFS and final histopathological examination (HPE) was 9.4 % in the PG and 12.5 % in the TG group indicating the limitations (false negative) of IOFS.
Previous reports concerning operative morbidity and mortality related to PG are conflicting; some showed no difference and others showed high morbidity and mortality rates as compared with TG. Meta-analysis of five studies [30, 31, 42–44], regarding operative mortality and the accumulated mortality rates between TG and PG, did not show significant difference (OR = 0.61, 95 % CI = 0.18–2.07, p = 0.43). Information on postoperative complications was reported in four studies [30, 42, 44, 45]. Meta-analysis revealed that patients after PG procedure experienced significantly higher morbidity risk compared to TG (OR = 0.11, 95 % CI = 0.08–0.17, p < 0.00001); however, Peto OR estimates did not change the mortality and morbidity comparison results. Regardless of the extent of the resection, morbidity rates observed in the present study was similar, suggesting that both procedures can be performed safely with acceptable outcomes in centers experienced in gastric cancer surgery. NACT was administered in 65 patients, and as in our previous study, perioperative outcomes of gastrectomy with D2 lymphadenectomy for locally advanced resectable gastric cancer was not influenced by NACT [46].
Local recurrence following proximal gastrectomy is a cause for concern, and it is assumed that it might be prevented if total gastrectomy is performed. Three studies reported data on recurrence with heterogeneity among them [30, 31, 45]. Meta-analysis indicated a trend of increasing the risk of tumor recurrence by PG procedure but without significant difference by OR estimate. Moreover, on Peto OR estimate, PG procedure showed a significant correlation with increased risk of tumor recurrence (Peto OR = 0.53, 95 % CI = 0.35–0.82, p = 0.004). Yoo CH et al. [30] analyzed the risk factors for local recurrence following proximal gastrectomy and advocated to avoid PG for infiltrative or the diffuse-type tumor, greater than 5 cm in tumor size, and serosal invasion. However, in our study, significant subsets of patients were downstaged with NACT (83.7 % in PG and 90.6 % in TG). This possibly explains the low rate of local recurrence in patients with PG. Distant recurrence was more common following TG but not statistically significant. Our observations are persuasive that PG can be performed with an acceptable low local recurrence rates even amongst locally advanced proximal third gastric cancer individuals who show good response to NACT.
Meta-analysis on seven observational studies [30, 31, 42–45, 47] showed that PG had no significant difference from TG on a 5-year overall survival rate (OR = 0.89, 95 % CI = 0.61–1.29, p = 0.53). Subgroup analysis was performed by different subpopulations: early diseases or unselected resectable diseases (including both early and locally advanced gastric cancer). Both meta-analyses showed no significant differences between two groups (OR = 1.33, 95 % CI = 0.23–7.85 and OR = 0.87, 95 % CI = 0.60–1.27, respectively). However, the accumulated 5-year overall survival rates of TG and PG groups were 72.3 % (502/694) versus 60.7 % (193/318), respectively, with absolute survival rate increasing 11.6 %. The survival analysis of this study showed an estimated 2-year overall survival rate of 73.8 % following PG and 49.9 % following TG. It implied a trend of potential survival benefit of PG procedure for proximal gastric cancer; however, the p value was not statistically significant (p = 0.10). The PG group faired prognostically better than the TG, probably because tumors in TG had higher proportion of undifferentiated tumors (68 %), bulky primary despite chemotherapy (poor responders), and more distant failures as compared to PG.
All the 75 individuals diagnosed of proximal third gastric cancer, ie, Siewert’s type II and III, as per current standard treatment were potential candidates for TG; however, in a cohort of 43 (43/75), we judiciously offered PG whenever a distal resection margin was negative, and the remnant stomach was adequate to perform a safe esophago-gastric anastomosis, without compromising any of the oncological principles. Total gastrectomy was performed whenever there was a risk of close margins as perceived by the surgeon.
We believe that proximal gastrectomy is a feasible option in Siewert’s type II and III locally advanced resectable gastric cancer, as the oncological outcomes are comparable with total gastrectomy.
The present study has two limitations. First, it is retrospective, and the second limitation is that postoperative function and patient quality of life following organ-preserving surgery (PG) was neither evaluated nor documented well. High-quality randomized controlled trials are still expected to elucidate the real worth of these two procedures.
Conclusions
In Siewert’s type II–III gastric cancer, current standard of treatment is total gastrectomy. In a post-MAGIC trial era, our study supports the adequacy of PG in comparison to TG which was offered to a group marginally different from that of TG. PG was equivalent to TG with regards to lymphadenectomy, resection margins, morbidity, pattern of recurrence, and survival for early and locally advanced resectable proximal gastric cancer. Both procedures can be accomplished safely. Therefore, proximal gastrectomy could be an alternative to total gastrectomy even in locally advanced proximal gastric cancer which is adequately downstaged by NACT, and provided the tumor size and location permit preservation of an adequate remnant of stomach without compromising oncologic principles.
References
Parkin DM, Bray FI, Devesa SS (2001) Cancer burden in the year 2000. The global picture. Eur J Cancer 37(Suppl 8):S4–66
Crew KD, Neugut AI (2006) Epidemiology of gastric cancer. World J Gastroenterol 12:354–62
Shang J, Pena AS (2005) Multidisciplinary approach to understand the pathogenesis of gastric cancer. World J Gastroenterol 11:4131–9
Deans C, Yeo MSW, Soe MY, Shabbir A, Ti TK, So JBY (2011) Cancer of the gastric cardia is rising in incidence in an Asian population and is associated with adverse outcome. World J Surg 35:617–24. doi:10.1007/s00268-010-0935-0
Katai H (2006) Function-preserving surgery for gastric cancer. Int J Clin Oncol 11:357–66. doi:10.1007/s10147-006-0613-2
Gouzi JL, Huguier M, Fagniez PL, Launois B, Flamant Y, Lacaine F et al (1989) Total versus subtotal gastrectomy for adenocarcinoma of the gastric antrum. A French prospective controlled study. Ann Surg 209:162–6
Bozzetti F, Marubini E, Bonfanti G, Miceli R, Piano C, Gennari L (1999) Subtotal versus total gastrectomy for gastric cancer. Ann Surg 230:170
Papachristou DN, Fortner JG (1980) Adenocarcinoma of the gastric cardia. The choice of gastrectomy. Ann Surg 192:58–64
Jakl RJ, Miholic J, Koller R, Markis E, Wolner E (1995) Prognostic factors in adenocarcinoma of the cardia. Am J Surg 169:316–9
Japanese Gastric Cancer Association (2011) Japanese gastric cancer treatment guidelines (ver.3). Gastric Cancer 14:113–23
Erturk MS, Cicek Y, Ersan Y (2016) Analysis of clinicopathologicalprognostic parameters in adenocarcinoma of the gastric cardia. Acta Chir Belg 103:611–5
Smith JW, Brennan MF (1992) Surgical treatment of gastric cancer. Proximal, mid, and distal stomach. Surg Clin North Am 72:381–99
Stipa S, Di Giorgio A, Ferri M (1992) Surgical treatment of adenocarcinoma of the cardia. Surgery 111:386–93
Rüdiger Siewert J, Feith M, Werner M, Stein HJ (2000) Adenocarcinoma of the esophagogastric junction: results of surgical therapy based on anatomical/topographic classification in 1,002 consecutive patients. Ann Surg 232:353–61
Hosokawa Y, Kinoshita T, Konishi M, Takahashi S, Gotohda N, Kato Y et al (2012) Clinicopathological features and prognostic factors of adenocarcinoma of the esophagogastric junction according to Siewert classification: experiences at a single institution in Japan. Ann Surg Oncol 19:677–83. doi:10.1245/s10434-011-1983-x
Slim K, Blay JY, Brouquet A, Chatelain D, Comy M, Delpero JR et al (2009) Digestive oncology: surgical practices. J Chir (Paris) 146(Suppl 2):S11–80. doi:10.1016/S0021-7697(09)72398-1
Van Cutsem E, Van de Velde C, Roth A, Lordick F, Köhne C-H, Cascinu S et al (2008) Expert opinion on management of gastric and gastro-oesophageal junction adenocarcinoma on behalf of the European Organisation for Research and Treatment of Cancer (EORTC)-gastrointestinal cancer group. Eur J Cancer 44:182–94. doi:10.1016/j.ejca.2007.11.001
DeMeester SR (2006) Adenocarcinoma of the esophagus and cardia: a review of the disease and its treatment. Ann Surg Oncol 13:12–30. doi:10.1245/ASO.2005.12.025
Shrikhande SV, Shukla PJ, Qureshi S, Siddachari R, Upasani V, Ramadwar M et al (2006) D2 lymphadenectomy for gastric cancer in Tata Memorial Hospital: Indian data can now be incorporated in future international trials. Dig Surg 23:192–7. doi:10.1159/000094537
Japanese Gastric Cancer Association (1998) Japanese classification of gastric carcinoma—2nd English edition. Gastric Cancer 1:10–24. doi:10.1007/s101209800016
Yu W, Choi GS, Chung HY (2006) Randomized clinical trial of splenectomy versus splenic preservation in patients with proximal gastric cancer. Br J Surg 93:559–63. doi:10.1002/bjs.5353
Wente MN, Bassi C, Dervenis C, Fingerhut A, Gouma DJ, Izbicki JR et al (2007) Delayed gastric emptying (DGE) after pancreatic surgery: a suggested definition by the International Study Group of Pancreatic Surgery (ISGPS). Surgery 142:761–8. doi:10.1016/j.surg.2007.05.005
Dindo D, Demartines N, Clavien P-A (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–13
Dikshit R, Gupta PC, Ramasundarahettige C, Gajalakshmi V, Aleksandrowicz L, Badwe R et al (2012) Cancer mortality in India: a nationally representative survey. Lancet 379:1807–16. doi:10.1016/S0140-6736(12)60358-4
Borch K, Jönsson B, Tarpila E, Franzén T, Berglund J, Kullman E et al (2000) Changing pattern of histological type, location, stage and outcome of surgical treatment of gastric carcinoma. Br J Surg 87:618–26. doi:10.1046/j.1365-2168.2000.01425.x
Blomjous JG, Hop WC, Langenhorst BL, ten Kate FJ, Eykenboom WM, Tilanus HW (1992) Adenocarcinoma of the gastric cardia. Recurrence and survival after resection. Cancer 70:569–74
de Manzoni G, Pedrazzani C, Pasini F, Durante E, Gabbani M, Grandinetti A et al (2003) Pattern of recurrence after surgery in adenocarcinoma of the gastro-oesophageal junction. Eur J Surg Oncol 29:506–10
Kodera Y, Yamamura Y, Shimizu Y, Torii A, Hirai T, Yasui K et al (1999) Adenocarcinoma of the gastroesophageal junction in Japan: relevance of Siewert’s classification applied to 177 cases resected at a single institution. J Am Coll Surg 189:594–601
Kaibara N, Nishimura O, Nishidoi H, Kimura O, Koga S (1987) Proximal gastrectomy as the surgical procedure of choice for upper gastric carcinoma. J Surg Oncol 36:110–2
Yoo CH, Sohn BH, Han WK, Pae WK (2004) Long-term results of proximal and total gastrectomy for adenocarcinoma of the upper third of the stomach. Cancer Res Treat 36:50–5. doi:10.4143/crt.2004.36.1.50
Harrison LE, Karpeh MS, Brennan MF (1998) Total gastrectomy is not necessary for proximal gastric cancer. Surgery 123:127–30
Buhl K, Schlag P, Herfarth C (1990) Quality of life and functional results following different types of resection for gastric carcinoma. Eur J Surg Oncol 16:404–9
Hsu CP, Chen CY, Hsieh YH, Hsia JY, Shai SE, Kao CH (1997) Esophageal reflux after total or proximal gastrectomy in patients with adenocarcinoma of the gastric cardia. Am J Gastroenterol 92:1347–50
Kobayashi T, Sugimura H, Kimura T (2002) Total gastrectomy is not always necessary for advanced gastric cancer of the cardia. Dig Surg 19:15–21
Okamura T, Tsujitani S, Korenaga D, Haraguchi M, Baba H, Hiramoto Y et al (1988) Lymphadenectomy for cure in patients with early gastric cancer and lymph node metastasis. Am J Surg 155:476–80
Kitamura K, Nishida S, Yamamoto K, Ichikawa D, Okamoto K, Taniguchi H et al (1998) Lymph node metastasis in gastric cancer in the upper third of the stomach—surgical treatment on the basis of the anatomical distribution of positive node. Hepatogastroenterology 45:281–5
Maruyama K, Gunvén P, Okabayashi K, Sasako M, Kinoshita T (1989) Lymph node metastases of gastric cancer. General pattern in 1931 patients. Ann Surg 210:596–602
Kitamura K, Yamaguchi T, Nishida S, Yamamoto K, Ichikawa D, Okamoto K et al (1997) The operative indications for proximal gastrectomy in patients with gastric cancer in the upper third of the stomach. Surg Today 27:993–8
Shimada H, Suzuki T, Nakajima K, Hori S, Hayashi H, Takeda A et al (1999) Lymph node metastasis with adenocarcinoma of the gastric cardia: clinicopathological analysis and indication for D1 dissection. Int Surg 84:13–7
Moreaux J, Msika S (1988) Carcinoma of the gastric cardia: surgical management and long-term survival. World J Surg 12:229–35
Goto H, Tokunaga M, Miki Y, Makuuchi R, Sugisawa N, Tanizawa Y et al (2014) The optimal extent of lymph node dissection for adenocarcinoma of the esophagogastric junction differs between Siewert type II and Siewert type III patients. Gastric Cancer. doi:10.1007/s10120-014-0364-0
An JY, Youn HG, Choi MG, Noh JH, Sohn TS, Kim S (2008) The difficult choice between total and proximal gastrectomy in proximal early gastric cancer. Am J Surg 196:587–91. doi:10.1016/j.amjsurg.2007.09.040
Kondoh Y, Okamoto Y, Morita M, Nabeshima K, Nakamura K, Soeda J et al (2007) Clinical outcome of proximal gastrectomy in patients with early gastric cancer in the upper third of the stomach. Tokai J Exp Clin Med 32:48–53
Zhou Y, Pan J, Sheng Y, Liu H, Fan Z (2007) Surgical treatment effects in cancer of the cardia and esophagogastric junction. Chin Ger J Clin Oncol 6:P220–1. doi:10.1007/s10330-006-0040-x
Kim JH, Park SS, Kim J, Boo YJ, Kim SJ, Mok YJ et al (2006) Surgical outcomes for gastric cancer in the upper third of the stomach. World J Surg 30:1870–6. doi:10.1007/s00268-005-0703-8, discussion 1877–8
Shrikhande SV, Barreto SG, Talole SD, Vinchurkar K, Annaiah S, Suradkar K et al (2013) D2 lymphadenectomy is not only safe but necessary in the era of neoadjuvant chemotherapy. World J Surg Oncol 11:31. doi:10.1186/1477-7819-11-31
Shiraishi N, Adachi Y, Kitano S, Kakisako K, Inomata M, Yasuda K (2002) Clinical outcome of proximal versus total gastrectomy for proximal gastric cancer. World J Surg 26:1150–4. doi:10.1007/s00268-002-6369-6
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in the study were in accordance with the ethical standards of the institution.
Informed consent
Informed consent was obtained from all individual participants included in the study
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(SAV 57 kb)
Rights and permissions
About this article
Cite this article
Sugoor, P., Shah, S., Dusane, R. et al. Proximal gastrectomy versus total gastrectomy for proximal third gastric cancer: total gastrectomy is not always necessary. Langenbecks Arch Surg 401, 687–697 (2016). https://doi.org/10.1007/s00423-016-1422-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-016-1422-3