Abstract
Background
Tumors arising from the esophagogastric junction (OGJ) are increasing in incidence in the west, but data from Asian populations are conflicting. Singapore has a mixed-Asian population with an international lifestyle. This study was designed to examine the changing trends in incidence of gastric cardia cancer (type III) within this population and to compare the clinicopathological characteristics and outcome of these tumors with gastric tumors.
Methods
Trends in cancer incidence were obtained from the Singapore Cancer Registry. Clinicopathological data were prospectively collected from patients undergoing surgery for gastric cancer who presented to the National University Hospital between 2000 and 2005. Patients underwent surgery with or without (neo)adjuvant therapy. Survival duration was analyzed.
Results
The incidence of cardia tumors has increased each decade since 1968 (1968–1982, 6.3%; 1983–1992, 7.6%; 1993–1997, 8.4%; 1998–2002, 9.1%; 2003–2007, 16.2%). Among the study population (n = 159) cardia tumors were associated with male sex (p < 0.01) and dysphagia (p < 0.01). Although R0 resection rates were similar, systemic recurrence rates were higher among patients with cardia cancer (p = 0.031) and survival was reduced compared with patients with non-cardia gastric cancer (median survival 26 vs. 69 months; p < 0.001). Cardia location of the tumor and metastatic lymph node ratio were identified as independent adverse prognostic indicators on multivariate analysis.
Conclusions
Similar to western societies, the incidence of proximal gastric cancer is increasing in Singapore. Cardia tumors are associated with poorer outcomes, suggesting that cardia cancer is a distinct disease from true gastric cancer requiring different management strategies to improve the outcome for these patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The incidence of adenocarcinoma arising at the esophagogastric junction (OGJ) continues to increase in the west. Whereas the global incidence of gastric cancer is gradually declining and the incidence of squamous cell carcinoma of the esophagus remain at a steady rate, OGJ tumors are increasing at the fastest rate of any solid malignancy in Europe and North America [1, 2]. The rising incidence of these tumors has been linked with increasing rates of Barrett’s metaplasia and the increasing prevalence of obesity and reflux disease, which are implicated in the pathophysiology [3, 4]. Epidemiological and clinical studies have led some authors to suggest that tumors located at the OGJ are distinct from other tumors located in the esophagus or distal stomach, with differing risk factors, tumor characteristics, and biological behavior [5, 6]. Siewert and Stein proposed a novel classification for tumors located within 5 cm of the OGJ, and this system has been widely adopted for the classification of these tumors [7]. It is proposed that the etiology and tumor behavior also may differ between the different subtypes of junctional tumors [8].
Junctional tumors are associated with adverse prognosis compared with other esophageal and gastric cancers. The anatomical site of these tumors potentially allows tumor spread to lymph nodes located above or below the diaphragm leading to difficulties in staging classifications and controversies in the optimal surgical strategy for these patients [9]. These difficulties have been reflected by the addition of new guidance for the staging of OGJ tumors in the latest edition of the TNM staging classification [10].
In contrast to data from the west, there are conflicting reports relating to the changing incidence of OGJ tumors among Asian populations. Studies from Japan, India, and China have all reported an increasing incidence of OGJ cancers during the past few decades [11–14]. Kusano et al. reported an increase in the proportion of junctional tumors from 2 to 10% in a high-volume cancer center in Japan during a 40-year interval [11]. Other groups from Korea, Taiwan, and Japan have not, however, identified any changes in the incidence of junctional tumors [15–17].
Singapore has a mixed-Asian population with an international lifestyle. The incidence rates of gastric cancer are lower than those of high-risk Asian populations, such as Korea and Japan, but are higher than those reported in the west [18]. In contrast, rates of esophageal cancer are lower than those recorded in Europe and North America. Singapore is uniquely placed to investigate the effects of an international lifestyle on the incidence of OGJ cancer within an Asian population. Rapidly increasing rates of obesity have been reported already [19]. This study was designed to examine the changing trends in incidence of OGJ cancer within this population and to investigate and compare the clinicopathological characteristics of type III junctional (cardia) cancers with more distally sited gastric cancers. In addition, survival duration will be investigated and potential prognostic variables will be identified.
Materials and methods
Cancer trends in Singapore
Information on the annual incidence and subsite location of gastric cancer in Singapore was obtained from the Singapore Cancer Registry based at the National Registry of Disease Office, Singapore [20]. The Registry was established in 1968, and the office collects demographic and pathological information on all cancers diagnosed within Singapore. Detailed anatomical localization of the tumor is a data requirement and tumor located at the gastric cardia is provided as an option. This information is published at regular intervals: 1968–1982 (15 years), 1983–1992 (10 years), 1993–1997 (5 years), 1998–2002 (5 years), and 2003–2007 (5 years). Notification of all new diagnoses of cancer is a requirement under the National Registry of Diseases Act in Singapore.
Study population
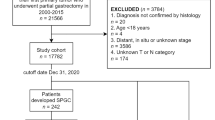
The study population was derived from a consecutive series of patients newly diagnosed with gastric cancer presenting to the National University Hospital between 2000 and 2005. Data on patients diagnosed with gastric and oesophageal cancer are prospectively collected into a combined database at our hospital. All patients diagnosed with gastric cancer and who were suitable for surgical resection were included in the present study. Patients were staged by a combination of computerized tomography (CT), endoscopic ultrasound (EUS), and laparoscopy according to the International Union Against Cancer (UICC 6th edition) [10]. Tumors located at the esophagogastric junction were classified according to Siewert and Stein [7]. The study population included type III (cardia) tumors, but type I and II tumors were excluded from this study. Type III tumors are located in the gastric cardia with the tumor epicenter within 2–5 cm of the esophagogastric junction and with tumor extension into the distal esophagus [7]. Demographic and clinical information was prospectively collected. All patients underwent treatment with curative intent after discussion at the unit multidisciplinary team meeting (MDT). Subjects underwent a total or subtotal radical gastrectomy with D2 lymphadenectomy in accordance with the principles outlined by the Japanese Gastric Cancer Society [21]. Routine splenectomy was not performed in all cases. Splenectomy was undertaken for tumors located on the proximal greater curve or for cases where there was concern regarding splenic hilar lymph node involvement. Patients underwent a combination of surgery alone or surgery in conjunction with neoadjuvant chemotherapy, perioperative chemotherapy, adjuvant chemotherapy, adjuvant radiotherapy, or adjuvant chemoradiotherapy. As a general principle, patients with early-stage tumors underwent surgery alone and patients with locally advanced disease (T2/3 N0/1) were offered adjuvant chemoradiotherapy similar to the treatment regime developed by the Intergroup trial (INT 0116) [22]. In more recent years, perioperative chemotherapy was offered to selected patients following the MAGIC study guidelines [23]. Surgical and pathological information, including total number of lymph nodes harvested, metastatic lymph node ratio, level of R resection, and final pathological stage, were recorded for each patient.
All patients were followed-up at regular intervals by the surgical team. Investigations were initiated during follow-up for clinical suspicion of disease recurrence. Routine investigations were not performed on asymptomatic patients. Disease recurrence was determined by clinical examination or radiological imaging. Tissue confirmation was obtained for selected patients. The presence of disease recurrence was documented and the pattern of recurrence was recorded in each case. Recurrence was recorded as local recurrence (defined as evidence of recurrent disease at the resection site, for example, at the site of anastomosis), peritoneal disease or lymph node recurrence (recurrent disease within the abdominal lymph nodes), or systemic recurrence (defined as evidence of distant metastatic disease, such as liver, bone, or pulmonary metastases). Survival duration, defined as time from histological confirmation of disease until death, was recorded for every patient.
Statistical analysis
All data were assumed to be nonparametric. Comparison of continuous variables was performed using the Mann–Whitney U test and categorical variables were tested by the chi-square test. Survival comparisons were made using the log-rank test. Cox’s proportional hazards model was used to determine the effects of clinical and investigative variables on survival duration in both univariate and multivariate analyses. Multivariate testing employed stepwise regression (using entry p value 0.05, removal p value 0.1) to determine the factors independently predictive of death.
Results
Changing trends in incidence of OGJ type III (cardia) cancer
The changing incidence rates of cancers arising from the gastric cardia within the population of Singapore between 1968 and 2007 are shown in Table 1. The proportion of tumors located at the gastric cardia has increased with each time period documented by the Singapore Cancer Registry since records began in 1968: 1968–1982, 6.3%; 1983–1992, 7.6%; 1993–1997, 8.4%; 1998–2002, 9.1%; 2003–2007, 16.2%. A higher proportion of tumors located at the gastric cardia were identified among patients presenting to the National University Hospital between 2000–2005 (20.1%; Table 1). This group of patients forms the population for the remainder of this study.
Study population
Patient demographics
A total of 159 patients underwent surgical resection for the treatment of gastric adenocarcinoma at the National University Hospital between 2000 and 2005. Tumors were located at the gastric cardia in 32 (20%) patients, gastric antrum/distal stomach in 84 (53%) patients, and the remaining 43 (27%) were located in the gastric fundus, body, or diffusely involving the stomach. Patient demographics are shown in Table 2. Cardia tumors were more commonly associated with male sex (p < 0.01) and with symptoms of dysphagia (p < 0.01), most likely due to tumor involvement of the OGJ. Non-cardia cancers were more likely to present with gastrointestinal bleeding or abdominal discomfort. There was no difference in patient age at the time of diagnosis. Most patients were of Chinese origin, and there was no difference in racial distribution between the two study groups. All tumors were adenocarcinoma and the tumor grade was similar between the two groups.
Treatment strategies
A large proportion of patients underwent surgery alone without adjuvant therapy as their definitive treatment; 18 of 32 for cardia tumors (56%), and 78 of 127 for non-cardia tumors (61%). Metastatic disease was discovered at the time of laparotomy in ten patients in the surgery alone group and these patients underwent a palliative bypass or biopsy and closure. The remaining patients received pre- or postoperative adjuvant treatment in the form of chemotherapy or radiotherapy, or both (Table 2). Treatment strategies were broadly similar between the two groups; however, patients with gastric cancer were more likely to receive postoperative chemoradiotherapy than patients with cardia cancer (20 vs. 15%, respectively) but less likely to undergo neoadjuvant therapy (2 vs. 13%, respectively; p = 0.056, chi-square test).
An R0 resection was achieved for most patients. There was no difference in R1 and R2 resection rates between the two patient groups (p = 0.887, chi-square test). After radical resection, the total number of harvested lymph nodes was similar between patients with cardia cancer [median 18 nodes (interquartile range 12–24)] and non-cardia cancer [median 16 nodes (interquartile range 17–28)] (p = 0.975, Mann–Whitney U test). Although the overall ratio of metastatic lymph nodes was similar between the two groups (cardia = 0.3 (interquartile range 0.1–0.6) vs. non-cardia = 0.2 (interquartile range 0–0.5), p = 0.144; Mann–Whitney U test), the final pathological stage differed between the two groups. Sixteen (50%) cardia tumors were locally advanced (stage III), whereas the stage group distribution for non-cardia tumors was more evenly distributed for each stage group (p = 0.034, chi-square test).
Patterns of recurrence
At the time of data analysis, 25 (78%) patients with cardia cancer and 59 (50%) patients with non-cardia cancer had evidence of disease recurrence (Table 2). Patterns of recurrence were different between the two groups with patients with cardia cancer more likely to recur with systemic disease (48 vs. 32%; p = 0.031; chi-square test). The liver was the most common site for systemic recurrence for patients with cardia cancer and gastric cancer (9 and 20 respectively). Metastases were detected in the lung in five patients with cardia cancer and seven patients with gastric cancer, and bone metastases were found in two patients with cardia cancer and five patients with gastric cancer. Two patients with cardia cancer and four patients with gastric cancer had evidence of systemic recurrence at multiple sites (liver, lung, or bone). Two patients with cardia cancer and nine patients with gastric cancer had evidence of both systemic and local disease recurrence.
Survival duration and prognostic variables
Patients were followed-up for an average 68 (range 44–110) months. At the time of censoring the data, 25 (78%) patients with cardia cancer and 68 (54%) patients with non-cardia cancer had died (Table 2). Two patients with non-cardia gastric cancer died from unrelated causes (intracranial bleeds). Information obtained from death certificates indicated that all other deaths were disease-related. Survival was compared for only those patients who underwent a curative (R0) resection. Median survival was significantly reduced for patients with cardia cancer compared with patients with non-cardia cancer (median survival 26 vs. 69 months, p < 0.001; log-rank test; Fig. 1).
Survival curves comparing survival duration of patients with cardia tumors (thick line) and patients with non-cardia tumors (thin line) from the time of diagnosis. Analysis shows only those patients who underwent a “curative” (R0) resection. Median survival cardia tumors = 26 months versus median survival non-cardia tumors = 69 months (p < 0.001; log-rank test). Survival duration (month). Cumulative survival, Survival duration (month)
Table 3 shows clinical and pathological prognostic variables identified on univariate analysis. Patient age at diagnosis, disease stage, ratio of metastatic lymph nodes, level of R resection, and the anatomical location of the primary tumor (cardia vs. non-cardia) were all associated with survival. Multiple regression analysis was performed to identify which of these variables retained prognostic value when co-analyzed. Metastatic lymph node ratio (hazard ratio 6.5; 95% confidence interval 3.18–13.39; p < 0.001) and the anatomical location of the primary tumor (hazard ratio 2.9; 95% confidence interval 1.71–4.9; p < 0.001) retained independent prognostic value (Table 4).
Discussion
The relative incidence of proximal gastric cancer in Singapore has steadily increased during the previous 40 years from 6 to 16% without a corresponding reduction in gastric cancer overall. Among our study population, the incidence of proximal gastric cancer was 20%. Cardia cancer demonstrated differing demographic characteristics to more distal gastric cancer. Although R0 resection rates were similar for cardia and distal gastric tumors, systemic recurrence rates were higher among cardia patients. Survival duration was markedly reduced for patients with cardia tumors, even when disease stage was taken into account. Tumors located at the gastric cardia and metastatic lymph node ratio were identified as independent adverse prognostic indicators on multivariate analysis.
Although the rising incidence of junctional carcinoma is well acknowledged in the west, data from Asia is less clear. Recent reports from Japan, India, and China have all suggested an increasing incidence of OGJ cancers during the past few decades [11–14]. Some high-volume cancer centers have reported an increase in the proportion of junctional tumor from 2 to 10% [11]. Other groups from Korea, Taiwan, and Japan have not identified any changes in the incidence of junctional tumor [15–17]. The reason for this disparity is not clear but may reflect differing methods of data collection among different units. In many Asian centers, gastric cancers and esophageal cancers are managed by separate departments. As such, some type I and possibly some type II tumor may be managed by thoracic surgeons leading to an underreporting of the true incidence. This may account for the different subtype distribution of junctional cancers between east and west. A recent national audit from the United Kingdom reported a relatively equal distribution of junctional subtypes (type I 42%, type II 27%, type III 31%) [24]. The pattern appears different in Japan where most junctional cancers are type II (56%) and type III (41%) and very few type I (3%) [17]. Once again, this may reflect different reporting practices but is more likely to represent a true difference between the populations. Within our own unit in Singapore, where esophageal and gastric cancer data are both collected, there are few type I tumors (3%). Type I cancers in particular are associated with Barrett’s metaplasia and rates of Barrett’s esophagus remain low among most Asian populations [25, 26]. This observation supports the theory that different subtypes of junctional tumor possess different risk factors and etiological influences [5, 6]. Therefore, not only are incidence rates of junctional cancers different between eastern and western populations, but also the subtype distribution varies between the two populations. There also is some difficulty and disparity in deciding between type II and type III tumor and the treatment option often is slightly clouded. Nonetheless, it was evident at the recent Japanese Gastric Cancer Association meeting that many units throughout Asia were reporting an increasing incidence in proximal gastric cancer (82nd Annual Meeting, Japanese Gastric Cancer Association, Niigata, Japan, 2010).
In Singapore, the incidence of junctional cancers has increased during the past 40 years. However, a personal series of 150 consecutive patients with gastric cancer operated on during an 11-year period in Singapore was published from our own unit approximately 20 years ago. In that report, 22% of tumors were located at the gastric cardia, which is similar to the present study [27]. However, that study reports a single surgeon’s experience and the proportion of cardia cancers was much higher than those published by the population-wide Singapore Cancer Registry. We restricted the present study to type III tumors only. This was decided due to the low numbers of type I and II tumors, making meaningful interpretation of data difficult and postulating that these different subtypes may possess different tumor characteristics. The incidence of cardia cancer in Singapore is higher than that reported in other Asian countries and is similar to that reported in western populations [24]. Cardia tumors were associated with male sex and different clinical characteristics. The different demographics of this population compared with patients with distal gastric cancer suggest that different processes are involved in the pathogenesis of these tumors, as previously suggested [5, 6]. Junctional cancer has been linked with obesity, reflux disease, smoking, and alcohol ingestion [3, 4, 28]. We did not investigate these risk factors in detail in the present study, but the published rates of obesity in Singapore are increasing and following those reported in western countries [19]. However, rates of Barrett’s metaplasia remain low in Singapore and this may account for the low incidence of type I tumors. Singapore has a predominantly Asian population, but the increasing trend in incidence of proximal gastric cancer may reflect the increasing “western lifestyle” among the population.
Patients with cardia cancer were more likely to have locally advanced disease on final staging (stage III). The association between tumor located at the esophagogastric junction and more advanced disease stage has been reported elsewhere and some authors have attributed these findings to more aggressive behavior of junctional tumor [29]. Relatively few patients received neoadjuvant or adjuvant therapy by current standards. There are several explanations for this. First, patients with early-stage disease (T1/2 N0) are not routinely offered adjuvant therapy in our department. Second, for some patients, the presence of comorbidities, such as ischemic heart disease, precluded the use of potentially cardiotoxic therapies. Finally, in Singapore patients are required to pay for their medical treatment and for some patients financial constraints may impact on treatment choices. Furthermore, overseas foreign nationals commonly seek their medical treatment in Singapore. These patients often decline neoadjuvant and adjuvant therapies preferring to proceed directly with surgery and an early return to their native country.
Recurrence was more common among patients with cardia cancer. Previous studies examining the pattern of lymphatic spread from junctional cancers suggest that type III cancers preferentially make their way to the coeliac axis and have a similar pattern of lymphatic spread to gastric body cancers [6, 30]. All patients underwent the same, standardized radical D2 lymphadenectomy and this may explain the similar rates of local recurrence between the two patient groups. However, systemic recurrence to distant organs, such as the liver, lung, and bone, was more common for patients with cardia tumors. Such spread may be considered via the hematogenous route and higher rates of hematogenous metastases from junctional tumors have been noted previously [29]. The explanation behind the different rates of haematogenous spread between cardia and non-cardia gastric cancers remains a matter of speculation, but may be explained by differing tumor behavior and biology, reaffirming once again that these tumors are different diseases. Increased usage of (neo)adjuvant systemic therapy may have a beneficial impact on these higher rates of systemic recurrence.
There was a large variation in the use of neoadjuvant and adjuvant treatments within the current study population. However, this variation affected both study groups and for each group the principle treatment modality was surgery alone. With these limitations in mind, survival among patients with cardia cancer was significantly reduced compared with survival among non-cardia cancer patients. The adverse prognosis associated with junctional tumors has been reported by other groups and has been attributed to higher tumor grade, more advanced disease stage, increased rates of lymphatic and blood vessel invasion, and increased lymph node metastases [6, 17, 29, 31]. We studied the prognostic role of many of these variables, such as disease stage, tumor grade, lymph node metastases, and R resection rates, as well as tumor site in a multivariate model. Tumors located at the gastric cardia retained independent prognostic value. Adenocarcinoma located at the gastric cardia, compared with gastric cancer located at other sites in the stomach, has been reported as an independent adverse prognostic variable by another group [29]. In that study, higher rates of systemic disease recurrence were noted among patients with cardia tumors. However, another study from Korea did not report any difference in survival duration for patients with cardia cancer compared with patients with true gastric cancer [32].
Similar to western societies, the incidence of proximal gastric cancer is increasing in Singapore. Compared with more distal gastric cancers, cardia tumors are associated with different clinicopathological characteristics and are associated with poorer outcomes and different patterns of disease recurrence. These findings suggest that type III cancers are a distinct disease entity, and these patients require different management strategies to those with true gastric cancer. The overall poorer prognosis reported for patients with cardia cancer in combination with the relatively higher rates of systemic recurrence are strong arguments for the wider use of adjuvant treatments for these patients. Published randomised trials have demonstrated a clear survival benefit for the use of perioperative chemotherapy for these patients, and it is this regimen that would be our preferred management strategy for such patients with locally advanced cardia tumors (i.e., T3 or node-positive patients) [23]. However, additional radiotherapy may be of benefit for selected patients, for example, those with high local nodal burden, to provide additional regional control [22]. The development of individualised treatment regimens based on tumor biology and host genotype may provide hope for improved outcomes in the future. This is currently a topic of research within our own institution.
References
Steevens J, Botterweck AA, Dirx MJ et al (2010) Trends in incidence of oesophageal and stomach cancer subtypes in Europe. Eur J Gastroenterol Hepatol 22(6):669–678
Blot W, Devesa SS, Kneller RW et al (1991) Rising incidence of adenocarcinoma of the oesophagus and gastric cardia. JAMA 265:1287–1289
Jankowski JA, Harrison RF, Perry I et al (2000) Barrett’s metaplasia. Lancet 356:2079–2085
Cameron AJ, Romero Y (2000) Symptomatic gastro-oesophageal reflux as a risk factor for oesophageal adenocarcinoma. Gut 46:745–755
Siewert JR, Stein HJ (1996) Adenocarcinoma of the gastroesophageal junction: classification, pathology and extent of resection. Dis Esoph 9:173–182
Siewert JR, Feith M, Werner M et al (2000) Adenocarcinoma of the esophagogastric junction: results of surgical therapy based on anatomical/topographic classification in 1,002 consecutive patients. Ann Surg 232(3):353–361
Siewert JR, Stein HJ (1998) Classification of carcinoma of the oesophago-gastric junction. Br J Surg 85:1457–1459
Siewert JR, Feith M, Stein HJ (2005) Biologic and clinical variations of adenocarcinoma at the esophago-gastric junction: relevance of a topographic-anatomic subclassification. J Surg Oncol 90(3):139–146
Peters CJ, Hardwick RH, Vowler SL et al (2009) Generation and validation of a revised classification for oesophageal and junctional adenocarcinoma. Br J Surg 96(7):724–733
Sobin LH, Gospodarowicz M, Wittekind C (2009) International Union Against Cancer (UICC), TNM classification of malignant tumors, 7th edn. Wiley
Kusano C, Gotoda T, Khor CJ et al (2008) Changing trends in the proportion of adenocarcinoma of the esophagogastric junction in a large tertiary referral center in Japan. Gastroenterol Hepatol 23(11):1662–1665
Tony J, Kumar SK, Thomas V (2007) Time trends and pathological profile of carcinoma lower oesophagus and gastro-oesophageal junction over the last 20 years—an experience from South India. Trop Gastroenterol 28(3):113–116
Zhou Y, Zhang Z, Zhang Z et al (2008) A rising trend of gastric cardia cancer in Gansu Province of China. Cancer Lett 269(1):18–25
Blaser MJ, Saito D (2002) Trends in reported adenocarcinomas of the oesophagus and gastric cardia in Japan. Eur J Gastroenterol Hepatol 14(2):107–113
Lee JY, Kim HY, Kim KH et al (2003) No changing trends in incidence of gastric cardia cancer in Korea. J Korean Med Sci 18(1):53–57
Chang SS, Lu CL, Chao JY et al (2002) Unchanging trend of adenocarcinoma of the esophagus and gastric cardia in Taiwan: a 15-year experience in a single center. Dig Dis Sci 47(4):735–740
Hasegawa S, Yoshikawa T, Cho H et al (2009) Is adenocarcinoma of the esophagogastric junction different between Japan and western countries? The incidence and clinicopathological features at a Japanese high-volume cancer center. World J Surg 33(1):95–103
Look M, Gao F, Low CH, Nambiar R (2001) Gastric cancer in Singapore. Gastric Cancer 4(4):219–222
National Health Survey (2004) Singapore Ministry of Health, Epidemiology and Disease Control Division. http://www.moh.gov.sg/mohcorp/publications.aspx?id=92
Singapore Cancer Registry, National Registry of Disease Office, Singapore, 2010: www.nrdo.gov.sg/page.aspx?id=76
Japanese Gastric Cancer Association (1998) Japanese classification of gastric carcinoma, 2nd English Edition. Gastric Cancer 1(1):10–24
Macdonald JS, Smalley SR, Benedetti J et al (2001) Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med 345:725–730
Cunningham D, Allum WH, Stenning SP et al (2006) Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer. N Engl J Med 355(1):11–20
Association of Upper Gastrointestinal Surgeons, UK (2009) www.augis.org/pdf/audits/nhs-ic-og-clinical-audit_2nd_Annual_Report_2009.pdf
Kim JH, Rhee PL, Lee JH et al (2007) Prevalence and risk factors of Barrett’s esophagus in Korea. J Gastroenterol Hepatol 22(6):908–912
Hongo M (2004) Review article: Barrett’s oesophagus and carcinoma in Japan. Aliment Pharmacol Ther 20(Suppl 8):50–54
Ti TK (1993) Pattern and surgical treatment of gastric cancer in Singapore. Br J Surg 80(7):886–889
Brown LM, Silverman DT, Pottern LM et al (1994) Adenocarcinoma of the esophagus and esophagogastric junction in white men in the United States: alcohol, tobacco, and socioeconomic factors. Cancer Causes Control 5(4):333–340
Saito H, Fukumoto Y, Osaki T et al (2006) Distinct recurrence pattern and outcome of adenocarcinoma of the gastric cardia in comparison with carcinoma of other regions of the stomach. World J Surg 30(10):1864–1869
Aikou T, Shimaza H (1989) Differences in main lymphatic pathways from the lower esophagus and gastric cardia. Jpn J Surg 19:290–295
Salvon-Harman JC, Cady B, Nikulasson S et al (1994) Shifting proportions of gastric adenocarcinomas. Arch Surg 129:381–389
An JY, Baik YH, Choi MG et al (2010) The prognosis of gastric cardia cancer after R0 resection. Am J Surg 199(6):725–729
Conflicts of interest
There are no potential or actual conflicts of interest to disclose.
Author information
Authors and Affiliations
Corresponding author
Additional information
Presented to the 82nd Annual Meeting of the Japanese Gastric Cancer Association, Niigata, Japan 2010.
Rights and permissions
About this article
Cite this article
Deans, C., Yeo, M.S.W., Soe, M.Y. et al. Cancer of the Gastric Cardia is Rising in Incidence in an Asian Population and is Associated with Adverse Outcome. World J Surg 35, 617–624 (2011). https://doi.org/10.1007/s00268-010-0935-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-010-0935-0