Abstract
Purpose
Laparoscopy-assisted pylorus-preserving gastrectomy has been increasingly reported as a treatment for early gastric cancer located in the middle third of the stomach because of its low invasiveness and preservation of pyloric function. Advantages of a totally laparoscopic approach to distal gastrectomy, including small wound size, minimal invasiveness, and safe anastomosis, have been recently reported. Here, we introduce a new procedure for intracorporeal gastro-gastrostomy combined with totally laparoscopic pylorus-preserving gastrectomy (TLPPG).
Methods
The stomach is transected after sufficient lymphadenectomy with preservation of infrapyloric vessels and vagal nerves. The proximal stomach is first transected near the Demel line, and the distal side is transected 4 to 5 cm from the pyloric ring. To create end-to-end gastro-gastrostomy, the posterior wall of the anastomosis is stapled with a linear stapler and the anterior wall is made by manual suturing intracorporeally. We retrospectively assessed the postoperative surgical outcomes via medical records. The primary endpoint in the present study is safety.
Results
Sixteen patients underwent TLPPG with intracorporeal reconstruction. All procedures were successfully performed without any intraoperative complications. The mean operative time was 275 min, with mean blood loss of 21 g. With the exception of one patient who had gastric stasis, 15 patients were discharged uneventfully between postoperative days 8 and 11.
Conclusions
Our novel hybrid technique for totally intracorporeal end-to-end anastomosis was performed safely without mini-laparotomy. This technique requires prospective validation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastric cancer remains one of the most frequent causes of death from cancer in eastern Asia. Currently, many patients are now diagnosed with early-stage gastric cancer (EGC) because of advances in cancer screening, and the prognosis for EGC continues to improve [1]. Therefore, the focus of treatment for EGC has changed from extension of survival to improvement in patients’ quality of life after surgery. Laparoscopy-assisted pylorus-preserving gastrectomy (LAPPG), one of the types of limited gastrectomy for EGC, has been reported increasingly as a treatment of EGC located in the middle third of the stomach because of its low invasiveness and preservation of pyloric function. However, this method requires that a mini-laparotomy is made on the upper abdomen to enable reconstruction [2].
Here, we introduce a novel laparoscopic procedure for intracorporeal gastro-gastrostomy, a hybrid technique using a linear stapler and manual suturing. In addition, we describe the short-term surgical results from our initial experience with totally laparoscopic pylorus-preserving gastrectomy (TLPPG).
Patients and methods
Indications for TLPPG
The indications for TLPPG were cT1, cN0 gastric cancer with confirmed adenocarcinoma located in the middle third of the stomach with its distal margin located more than 6 cm proximal to the pyloric ring. Patients who had indications for endoscopic mucosal resection or endoscopic submucosal dissection were excluded from the study.
The diagnosis was based on preoperative examinations including gastrointestinal endoscopy, upper gastrointestinal series, and abdominal computed tomography. The disease stage was determined according to the third English edition of the Japanese Classification of Gastric Carcinoma [3].
Preoperative lesion marking
During the operation, the exact location of the tumor is usually difficult to identify by laparoscopic observation from the serosal side. Therefore, marking of the preoperative lesion by both endoscopic clipping of the safe resection margins and submucosal tattooing is performed to verify the location of the lesion and the safe resection margin.
Operative procedure
Under general anesthesia, the patient is placed in a modified lithotomy position. An initial trocar for the laparoscope is inserted through the umbilicus using an open method. After establishing a pneumoperitoneum with carbon dioxide, four operating trocars are placed as illustrated in Fig. 1. The extent of lymph node dissection is, in principle, D1+ according to the Japanese gastric cancer treatment guidelines from 2010 (version 3) [4]. The infrapyloric artery and vein are preserved to maintain the blood supply and prevent congestion of the antrum. The hepatic and celiac branches of the vagal nerve are also preserved. The lymph node dissection is performed as previously described [5].
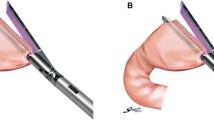
The proximal stomach is first transected by double 60-mm linear staplers at the Demel line vertical to the greater curvature. The distal margin is transected by a 60-mm linear stapler extracorporeally or intracorporeally at the proximal side 5 cm from the pylorus (Fig. 2). We remove the specimen from the umbilical port site with a 3- to 4-cm extension of the longitudinal incision. After the re-pneumoperitoneum, entry holes are made on both edges of the greater curvature corners, and the linear stapler is inserted at both the proximal and distal stomach through these entry holes. The posterior walls of both stumps are stapled by a 60-mm linear stapler, whereas both staple lines of the anterior wall are removed by laparoscopic coagulation shears. Stay sutures are placed at both sides of the greater and lesser curvatures, and the stitches are pulled to prevent contamination of gastric content within the operative field and, thus, maintain oncologic safety (Figs. 3 and 4).
Illustration and laparoscopic views of intracorporeal gastro-gastrostomy. a The entry holes are made on both edges of the greater curvature corners and the 60-mm linear stapler is inserted at both the proximal and distal stomach through these entry holes. b The posterior walls of both stumps of the stomach are stapled
The layer-to-layer hand-sewn anastomosis is made intracorporeally. We use the V-Loc (Covidien, Dublin, Ireland) for this procedure. The inner-layer closure starts from the greater curvature and extends toward the lesser curvature with continuous suturing. The second seromuscular layer is turned back toward the greater curvature. After the last stitch, the suture is simply cut without any knots. The intracorporeal end-to-end anastomosis is now complete (Figs. 5 and 6).
The detailed procedures are given in a video clip (Online Resource 1).
Results
The clinical characteristics, surgical outcomes, and pathological findings of the patients are presented in Table 1. From January 2013 to March 2015, 16 patients underwent TLPPG in the Department of Surgery at Iwate Medical University Hospital, Morioka, Japan. All patients had been diagnosed with mucosal or submucosal gastric cancer, without lymph node metastasis and located within the middle third of the stomach. Two patients were indicated for surgery after endoscopic submucosal dissection because of a histological diagnosis of submucosal invasion. We are prepared to change the operative procedure if we observe massive tumor invasion or visible lymph node metastasis by intraoperative findings. In the present series, all patients underwent D1+ lymph node dissection according to the Japanese treatment guidelines for gastric cancer (version 3) [4].
All patients underwent intracorporeal gastro-gastric reconstruction. The mean operative time was 275 min (range 217–341 min) and mean blood loss 21 g (range 6–52 g). Fifteen patients were discharged uneventfully between postoperative days 8 and 11 (mean length of postoperative hospital stay, 10 days); the remaining patient developed a gastric stasis and temporarily had to stop oral intake. This patient was discharged on postoperative day 36 with sufficient oral intake. No cases of recurrence were observed in this short follow-up period. Figure 7 shows an upper gastrointestinal examination on postoperative day 3. A good passage of Gastrografin was observed, without gastric stasis. Gastric fiberscopy, performed 1 year after surgery, revealed an intact anastomosis line and an absence of food residue in the remnant stomach (Fig. 8).
Discussion
Pylorus-preserving gastrectomy (PPG), a function-preserving procedure to prevent both dumping syndrome and alkaline reflux, has become one of the surgical options for EGC. LAPPG has several advantages over conventional PPG, especially regarding early surgical outcomes, namely, reduction of intraoperative blood loss and postoperative pain, and acceleration of recovery of bowel function and oral intake, with consequent shorter postoperative stay in hospital [5–7]. Recently, the usefulness of totally laparoscopic reconstruction relative to the conventional approach has been reported in terms of smaller wound size, minimal invasiveness, and safer anastomosis independently of the patient’s body habitus [8]. More recently, intracorporeal stapled anastomosis following TLPPG was reported by Lee et al. [9] and Kumagai et al. [10]. Their technique is a modification of the delta-shaped anastomosis that is one of the types of Billroth-I reconstruction reported by Kanaya et al. [11]. This method needs a side-to-side gastro-gastrostomy, and involves the remnant antrum via the first stapling. There is a concern when using this approach that gastric stasis may occur because of obstruction of antral gastric motility. Therefore, the question arises as to what extent an end-to-end gastro-gastrostomy can be performed through a totally laparoscopic approach. To answer this question, we have developed a hybrid technique that uses both a linear stapler and manual suturing performed intracorporeally. We first tried a purely manual anastomosis, whereby it was extremely difficult to achieve an anastomosis of sufficient size because of contraction of the stomach wall. By using a linear stapler, we are able to maintain an adequate length of posterior wall. Once the back wall is fixed, it is straightforward to create the front wall by manual suturing. The handsewn anastomosis enables us to reduce the cartridge number, resulting in lower operative costs. We adopted the V-Loc for suturing when closing the anterior wall. The safety and efficacy of the V-Loc for intracorporeal reconstruction after totally laparoscopic gastrectomy has been reported by Lee et al. [12]. This device enables the surgeon to work efficiently with both hands and to focus exclusively on subsequent stitch placement, without the need to maintain tension on preceding throws to prevent slippage.
One drawback of intracorporeal reconstruction is exposure of the gastric lumen in the operative field, leading to a concern for micro-dissemination of free cancer cells in the remnant gastric lumen [13]. To maintain oncologic safety, we place stay sutures on both sides of the greater and lesser curvature, and pull the stitches upward through the abdominal wall. The anastomosis is then turned along the ventral side, thus preventing the gastric contents being shed.
In terms of postoperative outcomes, one patient developed postoperative gastric stasis, requiring a postoperative stay in hospital of 36 days. Because this patient had diabetes, diabetic nephropathy, and a duodenal ulcer, we considered that the gastric motility and pyloric function of this patient had worsened before the operation. The remaining patients had no postoperative complications. Previous reports have stated that postoperative gastric stasis is the most frequent complication, occurring in 7.8–15 % of patients [5–7, 10, 11, 14], whereas the incidence in our series was only 6.3 %. However, our patient cohort was small; moreover, the preserved infrapyrolic artery and vein (which prevents gastric congestion) as well as an end-to-end hybrid anastomosis might have contributed to the low incidence of gastric stasis. We expect to improve the patient’s quality of life (QOL) by this procedure. We have already started to evaluate the postoperative QOL using a recently developed scale (PGSAS-45) [15].
In conclusion, our novel hybrid technique for totally intracorporeal end-to-end anastomosis can be safely performed and achieved without mini-laparotomy. Further clinical observations are required with larger cohorts and a prospective study to demonstrate the safety of this procedure, as well as the potential benefit over conventional techniques.
References
Yada T, Yokoi C, Uemura N (2013) The current state of diagnosis and treatment for early gastric cancer. Diagn Ther Endosc 2013:241320
Jiang X, Hiki N, Nunobe S, Fukunaga T, Kumagai K, Nohara K, Katayama H, Ohyama S, Sano T, Yamaguchi T (2011) Long-term outcome and survival with laparoscopy-assisted pylorus-preserving gastrectomy for early gastric cancer. Surg Endosc 25:1182–1186
Japanese Gastric Cancer Association (2011) Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer 14:101–112
Japanese Gastric Cancer Association (2011) Japanese gastric cancer treatment guidelines 2010 (ver.3). Gastric Cancer 14:113–123
Nunobe S, Hiki N, Fukunaga T, Tokunaga M, Ohyama S, Seto Y, Yamaguchi T (2007) Laparoscopy-assisted pylorus-preserving gastrectomy: preservation of vagus and infrapyloric blood flow induces less stasis. World J Surg 31:2335–2340
Morita S, Katai H, Saka T, Fukagawa T, Sano T, Sasako M (2008) Outcome of pylorus-preserving gastrectomy for early gastric cancer. Br J Surg 95:1131–1135
Hiki N, Shimoyama S, Yamaguchi H, Kubota K, Kaminishi M (2006) Laparoscopy-assisted pylorus-preserving gastrectomy with quality controlled lymph node dissection in gastric cancer operation. J Am Coll Surg 203:162–169
Ikeda O, Sakaguchi Y, Aoki Y, Harimoto N, Taomoto J, MasudaT OT, Adachi E, Toh Y, Okamura T, Baba H (2009) Advantages of totally laparoscopic distal gastrectomy over laparoscopically assisted distal gastrectomy for gastric cancer. Surg Endosc 23:2374–2379
Lee SW, Bouras G, Nomura E, Yoshinaka R, Tokuhara T, Nitta T, Tsunemi S, Tanigawa N (2010) Intracorporeal stapled anastomosis following laparoscopic segmental gastrectomy for gastric cancer: technical report and surgical outcomes. Surg Endosc 24:1774–1780
Kumagai K, Hiki N, Nunobe S, Sekikawa S, Chiba T, Kiyokawa T, Jiang X, Tanimura S, Sano T, Yamaguchi T (2015) Totally laparoscopic pylorus-preserving gastrectomy for early gastric cancer in the middle stomach: technical report and surgical outcomes. Gastric Cancer 18:183–187
Kanaya S, Gomi T, Momoi H, Tamaki N, Isobe H, Kitayama T, Wada Y, Ohtoshi M (2002) Delta-shaped anastomosis in totally laparoscopic Billroth I gastrectomy: new technique of intraabdominal gastroduodenostomy. J Am Coll Surg 195:284–287
Lee SW, Nomura E, Tokuhara T, Kawai M, Matsuhashi N, Yokoyama K, Fujioka H, Hiramatsu M, Okuda J, Uchiyama K (2011) Laparoscopic technique and initial experience with knotless, unidirectional barbed suture closure for staple-conserving, delta-shaped gastroduodenostomy after distal gastrectomy. J Am Coll Surg 213:e39–e45
Han TS, Kong SH, Lee HJ, Ahn HS, Hur K, Yu J, Kim WH, Yang HK (2011) Dissemination of free cancer cells from the gastric lumen and from perigastric lymphovascular pedicles during radical gastric cancer surgery. Ann Surg Oncol 18:2818–2825
Suh YS, Han DS, Kong SH, Kwon S, Shin CI, Kim WH, Kim HH, Lee HJ, Yang HK (2014) Laparoscopy-assisted pylorus-preserving gastrectomy is better than laparoscopy-assisted distal gastrectomy for middle-third early gastric cancer. Ann Surg 259:485–493
Namikawa T, Hiki N, Kinami S, Okabe H, Urushihara T, Kawashima H, Fukushima N, Kodera Y, Yumiba T, Oshio A, Nakada K (2014) Factors that minimize postgastrectomy symptoms following pylorus-preserving gastrectomy: assessment using a newly developed scale (PGSAS-45). Gastric Cancer 18:397–406
Acknowledgments
Authors’ contributions
Study conception and design: Koeda
Acquisition of data: Koeda, Chiba, Noda, Nishinari, Segawa, Akiyama, Iwaya
Analysis and interpretation of data: Koeda, Nishizuka, Nitta, Otsuka
Drafting of manuscript: Koeda
Critical revision: Sasaki
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded in part by a Health Labour Sciences Research Grant of Japan and by MEXT KAKENHI Grant of Japan (Number 15K10114).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
(MP4 200076 kb)
Rights and permissions
About this article
Cite this article
Koeda, K., Chiba, T., Noda, H. et al. Intracorporeal reconstruction after laparoscopic pylorus-preserving gastrectomy for middle-third early gastric cancer: a hybrid technique using linear stapler and manual suturing. Langenbecks Arch Surg 401, 397–402 (2016). https://doi.org/10.1007/s00423-016-1378-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-016-1378-3