Abstract
Purpose
Severe bleeding after trauma frequently leads to a poor outcome. Prehospital fluid replacement therapy is considered an important primary treatment option. We conducted a retrospective matched pair analysis to assess the influence of prehospital fluid replacement volume on the clinical course of patients with solid abdominal organ trauma.
Methods
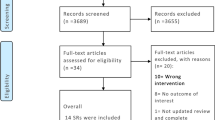
Data were analyzed from 51,425 patients in TraumaRegister DGU® of the German Trauma Society. Inclusion criteria were as follows: injury severity score ≥16 points, primary admission, age ≥16 years, no isolated brain injury, transfusion of at least one unit of packed red blood cells (pRBCs), and systolic blood pressure ≥20 mmHg at the accident site. The patients were divided into “low-volume” (0–1000 ml) and “high-volume” (≥1500 ml) groups according to the matched pair criteria. In each group, 68 patients met the inclusion criteria.
Results
Higher volume in fluid replacement was associated with increased need for transfusion (pRBCs: low-volume: 7.71 units, high-volume: 9.16 units; p = 0.074) and with by trend reduced clotting ability (prothrombin time: low-volume: 71.47 %, high-volume: 66.47 %; p = 0.27). The percentage of patients in shock (systolic blood pressure <90 mmHg) upon admission was equal in the two groups (25.0 %; p = 1). The mortality rate was discretely higher in the high-volume group (low-volume: 11.8 %, high-volume: 19.1 %; p = 0.089).
Conclusions
Excessive prehospital fluid replacement is able to lead in an increased mortality rate in patients with solid abdominal organ injury. Our results support the concept of restrained fluid replacement in the preclinical treatment of severe trauma patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Bleeding as a result of severe trauma correlates with high initial mortality rates and with secondary complications [1–4]. Blunt trauma is the most frequent form of severe trauma in Europe, accounting for 95 % of severe trauma cases in Germany according to the TraumaRegister DGU® annual report 2010. Blunt trauma that causes bleeding into the large thoracic and/or abdominal body cavities is especially difficult to assess diagnostically. Furthermore, such injuries are related to increased mortality rates [5–7].
At first glance, it seems reasonable to replace the lost blood with fluids as quickly as possible, ideally at the accident site [8]. However, only a few studies have confirmed that the immediate administration of fluids is beneficial to abdominal trauma patients with internal bleeding. In contrast to assessments of blunt trauma patients, the influence of prehospital fluid replacement on patients with penetrating injuries has been investigated more thoroughly. Follow-up examinations of soldiers wounded in the Falklands War indicated that patients with hypotensive circulation and simultaneous hyperpyrexia prior to hospital treatment had better outcomes [9]. Additional studies of patients who suffered penetrating injuries showed that excessive replacement volume (>2000 ml) resulted in longer delays from injury to hospital and correlated with increased mortality rates after trauma in most cases [10–12]. Bickell et al. also found positive associations between moderate fluid replacement and permissive hypotension (90 mmHg) in patients with penetrating injuries. Moderate fluid replacement also reduces the time from injury to hospital and is supported by several studies [13–16].
Currently, there are few evidence-based recommendations regarding the prehospital treatment of patients with hemorrhaging after blunt trauma to the solid abdominal organs. Anecdotal reports suggest that minimizing the injury-to-hospital time and direct delivery to a level one trauma center are the recommended course of action for penetrating injuries. In a systematic review, Butler presents a decision tree based on the Tactical Combat Casualty Care (TCCC) guidelines that takes into account the presence of hemorrhagic shock. However, that study concludes that it is not possible to provide definite recommendations, since most of the guidelines are based on animal experiments and the evidence levels in humans are too low [17]. In addition, the study did not specifically consider blunt trauma but instead provides more general recommendations. With regard to blunt abdominal trauma, recent studies recommend minimal treatment at the accident site with the goal of maintaining patient vital signs and providing rapid transport to a higher-level trauma center [18–20]. On the other hand, some reports continue to recommend extensive volume replacement as the best treatment option [21, 22]. Turner et al. found no relationship between mortality or outcome and the infused volume in patients with blunt trauma [23]. However, that study focused on patients who were less severely injured (>75 % had ISS <16).
An examination of the current literature raises questions: does the fluid replacement volume have consequences for hemorrhagic shock in the posttraumatic course, including multiple organ failure (MOF), sepsis, outcome, and mortality? The hypothesis of this study was that greater prehospital fluid replacement volume could have a negative impact on patient outcome. We addressed these questions and the hypothesis by analyzing a patient cohort selected from TraumaRegister DGU®. These patients had suffered severe injuries (abbreviated injury scale (AIS) >3) that resulted in hemorrhaging.
Methods
The TraumaRegister DGU® was founded in 1993. The aim of this multi-center database is an anonymous and standardized documentation of severely injured patients.
Data are collected prospectively in four consecutive time phases from the site of the accident until discharge from hospital: (a) prehospital phase, (b) emergency room and initial surgery, (c) intensive care unit, and (d) discharge. The documentation includes detailed information on demographics, injury pattern, comorbidities, pre- and in-hospital management, course on intensive care unit, relevant laboratory findings including data on transfusion, and outcome of each individual. The inclusion criterion is admission to hospital via emergency room with subsequent ICU/ICM care or reach the hospital with vital signs and die before admission to ICU.
The infrastructure for documentation, data management, and data analysis is provided by Akademie der Unfallchirurgie (AUC)—Academy for Trauma Surgery (GmbH), a company affiliated to the German Trauma Society. The scientific leadership is provided by the Committee on Emergency Medicine, Intensive Care and Trauma Management (Sektion NIS) of the German Trauma Society. The participating hospitals submit their data anonymously into a central database via a web-based application. Scientific data analysis is approved according to a peer review procedure established by Sektion NIS.
The participating hospitals are primarily located in Germany (90 %), but a rising number of hospitals of other countries contribute data as well (at the moment from Austria, Belgium, China, Finland, Luxembourg, Slovenia, Switzerland, The Netherlands, and the United Arab Emirates). Currently, approx. 25,000 cases from more than 600 hospitals are entered into the database per year. Participation in TraumaRegister DGU® is voluntary. For hospitals associated with TraumaNetzwerk DGU®, however, the entry of at least a basic data set is obligatory for reasons of quality assurance.
Only patients from Germany and Austria were included in this study to minimize variations due to different rescue systems. All patients were attended by a physician prior to hospital admission. Records that were collected between 1993 and 2009 (51,425 patients) were considered for this study. Use of data from TraumaRegister DGU® was approved by the Ethics Committee of the University of Witten/Herdecke, Campus Cologne-Merheim, Germany. The present study is in line with the publication guidelines of the TraumaRegister DGU® and registered as TR-DGU project ID 2013–041.
Patients were selected for this study according to the following criteria:
-
Primary admission to a trauma center (no transfers)
-
Injury Severity Score (ISS) ≥16
-
Age ≥16 years
-
Infusion of at least one unit of pRBCs
-
Systolic blood pressure ≥20 mmHg at the accident site
-
Data available for the administered prehospital fluid replacement volume, hemoglobin concentration upon hospital admission, and blood pressure at the accident site and upon hospital admission
Based on the prehospital fluid replacement volume (crystalloids and colloids), patients were divided into “low-volume” (0–1000 ml) and “high-volume” (≥1500 ml) groups. This classification system was based on the mean fluid replacement value for all patients that met the inclusion criteria. To raise the accuracy of this study, patients with a prehospital fluid replacement volume between >1000–1500 were not included at all.
To evaluate the effect of the prehospital fluid replacement volume, patients with low- and high-volume fluid replacement were matched according to the following criteria:
-
The pattern of injury for the following abdominal organs: liver, spleen, kidney, and pancreas, where matching criteria were AIS severity ≥3 points
-
The date of the injury (to account for changes in treatment over time): (I) 2002–2005, (II) 2006–2009, and (III) 2010–2012
-
Systolic blood pressure at the accident site, which had to be at least 20 mmHg: (I) 20–60 mmHg, (II) 61–90 mmHg, and (III) ≥91 mmHg
-
Age: (I) 16–54, (II) 55–69, and (III) ≥70 years
Because the three following characteristics depend on and correlate with the administered fluid replacement volume, the patient cohort was also matched with respect to the following characteristics:
-
Intubation (yes/no)
-
Method of rescue transport (air vs. ground transportation)
-
Time from injury to hospital ±30 min (i.e., the difference in the time from injury to hospital in matched patients did not exceed 30 min)
Sepsis was defined according to the criteria of Bone, which are close to the American College of Chest Physicians/Society of Critical Care Medicine (ACCP-SCCM) consensus conference definition [24]. Single organ failure was defined as a value ≥3 for the sequential organ failure assessment (SOFA) score [25]. The hospitals participating in TraumaRegister DGU® entered this value as the total value in the registry, and no conclusions about individual patient management or intervention can be drawn. MOF was listed if simultaneous organ failure was recorded for at least two organs. Preclinical parameters, length of hospital stay, and coagulation ability were examined separately in each group. Prothrombin time is a parameter that is commonly used as a measure of coagulation ability in Germany and corresponds to the international normalized ratio (INR). To evaluate the ISS within groups with sufficiently complete data, prognosis estimation was performed using the Revised Injury Severity Classification (RISC) [26]. The prognosis was then compared to the observed mortality rate in the corresponding group. Prognoses were also calculated according to the Trauma and Injury Severity Score (TRISS). The probability of surviving over time was analyzed using Kaplan-Meier statistics.
Statistics
Data were analyzed with the Statistical Package for the Social Sciences software (SPSS1; version 17, Chicago, IL, USA). Incidences are represented as number of cases and percentages, and measured values are represented as means and standard deviations. Differences between the two groups with low and high prehospital replacement volumes were evaluated using the chi-square test in cases of categorical variables and using the t test in cases of continuous variables. In cases of obvious deviation from normality, continuous variables were tested with a non-parametric rank test (Mann-Whitney). We applied a significance level of 5 % to all statistical tests.
Results
Using data from TraumaRegister DGU®, 68 patients with severe abdominal injuries with a high prehospital replacement volume were matched with 68 patients from the low-volume replacement group. The average patient age in the entire group (n = 136) was 35.31 (15.17) years; p = 0.35. The mean ISS was the same in both groups (low-volume: 34.97 (12.36), high-volume: 34.65 (11.17); p = 0.99). As expected, most injuries were blunt trauma injuries (88.8 %). The injury severity scores in the abdominal organs had identical distributions in the groups (Tables 1 and 2). The injury causes are shown according to subgroup in Table 3. The similarity of the characteristics of the patients in this study supports the idea that the groups receiving low- or high-volume replacement therapy were similar and comparable.
Preclinical and emergency department treatment
Less fluid was infused prior to arrival at the hospital in the low-volume group (mean volume, 818.38 ml) than in the high-volume group (mean volume, 2101.84 ml). As shown in Table 4, patients in the high-volume group received proportionally greater (combined) volumes of crystalloid and colloidal infusion solutions. Patients in the high-volume group had lower mean systolic blood pressure at the accident site (low-volume: 112.12 (26.58) mmHg, high-volume: 108.25 (22.46) mmHg; p = 0.36). Upon arrival at the hospital, there was no significant difference in the mean systolic blood pressure between the two groups. The percentage of patients in the two groups with systolic blood pressure <90 mmHg was the same at the accident site and upon arrival at the hospital. Overall, the clinical parameters for circulation were similar in both the low- and high-volume patient groups.
Hemoglobin concentration, base excess, and coagulation values were measured during treatment in the emergency department. Patients who received high fluid volumes showed lower blood values. Namely, the hemoglobin concentration was lower in the high-volume group (low-volume: 11.38 (2.42) mg/dl, high-volume: 10.08 (3.30) mg/dl; p = 0.03). Similar results were observed for the prothrombin time (low-volume: 71.47 (22.69) %, high-volume: 66.47 (21.04) %; p = 0.27) and platelet counts (low-volume: 219,326.53/nl, high-volume: 181,081.63/nl; p = 0.008). Patients in the high-volume group received more units of pRBCs (low-volume: 7.71 (9.76) units, high-volume: 9.16 (9.88) units; p = 0.074). A higher percentage of patients in the high-volume group received more than 10 units of pRBCs (low-volume: 27.9 %, high-volume: 33.8 %; p = 0.458), and high-volume patients were perfused with more fresh-frozen plasma (low-volume: 4.81 (7.26) units, high-volume: 6.64 (7.85) units; p = 0.047). As shown in Table 4, patients in the high-volume fluid replacement group required additional preclinical therapeutic measures, including catecholamine supplementation, sedation, and insertion of a chest tube at the accident site. There was no significant difference between the low- and high-volume groups in terms of the percentages of patients who required cardiopulmonary resuscitation at the accident site.
Clinical course and outcome
The number of days spent in the intensive care unit (ICU), the total length of the hospital stay, and the total number of days of intubation was similar in both groups (Table 5). The occurrence of sepsis, organ failure, and MOF did not differ significantly between the two groups. In this context, it must be noted that in the high-volume group, more patients died before experiencing sepsis, multiple organ failure, or organ failure. The residual cohort then consisted of patients with better outcomes.
There was no significant difference in TRISS prognosis between the low- and high-volume groups, but the RISC prognosis showed a higher probability by trend of death for patients in the high-volume group (low-volume: 10.44 %, high-volume: 13.85 %; p = 0.089; Table 5). The RISC prognosis is based on data that are collected in the hospital, including prothrombin time, hemoglobin concentration, and administered pRBCs. However, these values are directly influenced by the administered prehospital fluid replacement volume. In contrast, the TRISS prognosis is not influenced by the administered prehospital fluid replacement volume. The percentage of patients who died was higher in the high-volume group (19.1 %) versus the low-volume group (11.8 %). The difference of 7.3 % is not statistically significant (p = 0.089) and should therefore be only evaluated by trend.
Discussion
Our study reveals a possible connection between increased prehospital fluid replacement volume and impairment of the coagulation system and hemoglobin concentration upon arrival at the hospital. This finding was also illustrated by the number of units of pRBCs transfused into patients who received either low or high volumes of prehospital replacement fluid and was especially apparent and clinically relevant in patients who received more than 10 units of pRBCs. This connection is supported by studies of blunt trauma patients conducted by Turner and Trunkey as well as by Geeraedts [27].
The decision to initially administer enhanced therapy (thoracic drainage, intubation, central venous catheter, etc.) at the accident site must be made on a case-by-case basis. A comprehensive standard protocol cannot be established for this situation. However, our findings support the idea that recommendations for the preclinical treatment of patients with penetrating trauma also apply to the preclinical treatment of patients with blunt trauma. These recommendations include limiting preclinical therapy to stabilization of the patient’s cardiovascular and pulmonary systems and prioritizing rapid transport to a level one trauma center.
The question of why patients in the high-volume group received a much larger volume of fluid (approximately 1300 ml more, on average) than patients in the low-volume group remains unanswered. Patients were classified so that their initial hemodynamic conditions were approximately the same, as was the injury severity in each part of the body. This helped minimize possible bias due to differences in injury severity. In addition, there was no significant difference in the mean ISS between the groups.
As mentioned before, the attending personnel make the decision of how much fluid should be administered to a given patient. With regard to hemodynamic stability, only patients with systolic blood pressure ≥20 mmHg at the accident site were included in this study. It is assumed that patients with systolic blood pressure <20 mmHg receive larger volumes of fluid at the accident site; thus, these patients were not investigated due to lack of a matching control group. Therefore, it was not possible to investigate the effects of prehospital fluid volume in these patients. Nonetheless, the results show that patients were reanimated at the accident site. It should be noted that the systolic blood pressure data that were used for matching referred to the blood pressure values that were initially measured at the accident site. Patient assessment and subsequent therapy decisions are made continuously during emergency treatment according to the patient’s condition. However, no conclusions about individual decisions can be drawn in a retrospective statistical analysis such as this. The present data support the idea that once a medical team opts for extended therapy, this extended therapy is related to increased replacement volume. Injury severity does not always seem to be a determining factor in this context. For example, the significantly higher number of chest tubes used in the higher-volume group cannot be explained by more severe injury to the thorax. It is possible that the determining factors are the education and experience of the attending medical personnel. This is supported by the findings of a review by Oestern concerning the medical assistance provided to severely injured patients in emergency trauma departments [28]. In addition, the matched pair criteria of time from injury to hospital, intubation, and rescue means of transport did not explain the higher fluid replacement volume in the high-volume group. The individual assessments by ambulance service personnel may play a role in determining the replacement volume. This is speculative, however, and cannot be assessed due to a lack of relevant studies at this time. However, this question merits further investigation in the future.
One finding of this study was that higher replacement volume was related to higher mortality rate by trend. The increase of approximately 9 % in the mortality rate in the high-volume group appeared within 6 h after arrival at the hospital, so the higher mortality was not due to patients who were brought to the hospital because of higher fluid replacement volume. However, these findings are not statistically significant. Rather, the elevated mortality rate appeared in the first operative phase or the first intensive care phase of patient treatment. It can be assumed, as indicated by this study, that a higher prehospital replacement volume was responsible for the higher mortality rate after trauma because, for example, this impaired the coagulation system. The possible treatments for such impaired conditions seem to be limited to the initial phase of treatment after trauma. The present study refutes the notion that if a patient survives the first operative phase or the first intensive care phase, then the mortality rate does not change significantly in subsequent treatment phases. There were no significant differences in the rates of sepsis, organ failure, or MOF because of the higher mortality rate in the high-volume group. The results were similar for both groups in terms of the total days spent in the ICU and the number of days of intubation. For these parameters, the higher mortality by trend in the high-volume group needs to be taken into consideration. The best course of action appears to be lower-volume fluid replacement that minimizes additional impairment in coagulation and dilution of oxygen carriers, plus rapid transport to a level one trauma center for definitive surgical and intensive medical therapy. As mentioned above, Geeraedts and Turner drew similar conclusions.
Interestingly, the RISC score confirmed the influence of fluid volume on mortality as this score was directly influenced by the administered prehospital volume, e.g., by the values for coagulation, hemoglobin concentration, and pRBC transfusion. The mortality as predicted by the RISC scores was almost as expected.
Our study does not support Turner’s conclusion that the replacement volume does not influence the mortality rate. Rather, the present study does not support aggressive volume replacement after blunt trauma of the abdomen and bleeding. Dutton et al. reported similar results for penetrating and blunt trauma injuries in the USA [29], as did Haut, also in a study conducted in the US. Haut postulates that the routine use of prehospital volume replacement should be avoided since it is associated with increased mortality. As a caveat, it must be noted that the emergency system in the US is different from the emergency system in our study. Whilst the ISS was split into four groups, no organ-specific matching (e.g., using the AIS) was performed. Lastly, the average time from injury to hospital and the volumes of the administered solutions were not reported in that study [30]. We conclude that permissive hypotension represents a therapeutic option, as has been shown for patients with penetrating trauma.
Limitations
-
1.
The TRISS calculations could only be performed for 46 % of the participating trauma centers, whereas the RISC methodology was available for 88 % of the cases. Thus, the data might be biased, as TRISS was not calculated for the majority of the trauma cases. However, this indicates that RISC is much easier to calculate than TRISS. This may be because RISC does not include determination of the prehospital respiratory rate, which was documented by physicians at the accident site in only 60 % of cases.
-
2.
Regarding the coagulation analysis, only the prothrombin ratio, prothrombin time, and platelet counts are documented and available for analysis in TraumaRegister DGU®. Other laboratory values that are relevant to coagulation, such as fibrinogen and protein C, are not documented in TraumaRegister DGU®. Furthermore, the precise pharmacological composition of the administered solutions was not documented in the registry. As a rule, Ringer’s solution or 0.9 % NaCl are used as crystalloid solutions, HAES1 is used as colloidal solution, and HyperHAES1 as a hyperoncotic solution.
-
3.
All patients were treated by a physician at the accident site. However, the specialty of the physician at the accident site (e.g., (trauma) surgeon, anesthesiologist, or internist) was not recorded. For example, in Scandinavian countries, only anesthesiologists are allowed to work as physicians at an accident site. In German-speaking countries, any physician with an additional certificate in “emergency medicine” is authorized to work as an emergency physician at an accident site. This certificate is not comparable to “emergency physician” certifications in most European countries, in the USA, or in Australia. In these countries, “emergency physician” represents a separate specialty. Furthermore, the individual decisions of the emergency doctors remain unclear due to lack of data in TraumaRegister DGU®.
-
4.
The matched pair analysis is dependent on the quality of the matching criteria. When patients are matched, not all patients in TraumaRegister DGU® are included, since patients without a “match partner” are not included. The advantage of the comparison of patients included in the matched pair analysis, however, is based on the fact that small differences can be demonstrated.
-
5.
Finally, this was a retrospective analysis, so only associations (not causalities) can be ascribed to the data. In the future, a prospective randomized study will be crucial for clarifying the advantages or disadvantages of a particular volume therapy for the most severely injured, bleeding patients at accident sites.
Conclusions
Due to registered limitations and the small group sizes, statistical analyses could hinder to show significant differences between the two study groups. Therefore, interpretation of the mentioned study data needs to be performed carefully and were indicated clearly, whether there are trends or significant differences.
However, if not indicated, aggressive volume replacement may lead by trend to increased mortality and could be related to early traumatic coagulopathy. The results of this study show that permissive hypotension and limited volume replacement during rescue have a positive impact on patients suffering from trauma and severe bleeding.
References
Heuer M, Hussmann B, Lefering R et al (2011) Pancreatic injury in 284 patients with severe abdominal trauma: outcome, course and treatment algorithm. Langenbecks Arch Surg 396:1067–1076
Sauaia A, Moore FA, Moore EE, Moser KS, Brennan R, Read RA et al (1995) Epidemiology of trauma deaths: a reassessment. J Trauma 38:185–193
Holcomb JB (2004) Methods for improved hemorrhage control. Crit Care 8:57–60
Kauvar DS, Wade CE (2005) The epidemiology and modern management of traumatic hemorrhage: US and international perspectives. Crit Care 9:1–9
Heuer M, Taeger G, Kaiser GM et al (2009) Prognostic factors of liver injury in polytraumatic patients. Results from 895 severe abdominal trauma cases. J Gastrointest Liver Dis 18:197–203
White CE, Hsu JR, Holcomb JB (2009) Haemodynamically unstable pelvic fractures. Injury 40:1023–1030
Gonzalez EA, Moore FA, Holcomb JB, Miller CC, Kozar RA, Todd SR et al (2007) Fresh frozen plasma should be given earlier to patients requiring massive transfusion. J Trauma 62:112–119
Kreimeier U, Lackner CK, Prueckner S, Ruppert M, Peter K (2003) Neue Strategien in der Volumenersatztherapie beim Polytrauma. Notfall Rettungsmed 6:77–88
Williams JG, Riley TR, Moody RA (2007) Resuscitation experience in the Falkland Islands campaign. J R Army Med Corps 153:70–72
Bickell WH, Stern S (1998) Fluid replacement for hypotensive injury victims: how, when and what risks? Curr Opin Anaesthesiol 11:177–180
Bickell WH, Barrett SM, Romine-Jenkins M, Hull SS Jr, Kinasewitz GT (1994) Resuscitation of canine hemorrhagic hypotension with large-volume isotonic crystalloid: impact on lung water, venous admixture, and systemic arterial oxygen saturation. Am J Emerg Med 12:36–42
Bickell WH (1993) Are victims of injury sometimes victimized by attempts at fluid resuscitation? Ann Emerg Med 22:155–163
Yaghoubian A, Lewis RJ, Putnam B, De Virgilio C (2007) Reanalysis of prehospital intravenous fluid administration in patients with penetrating truncal injury and field hypotension. Am Surg 73:1027–1030
Kreimeier U, Messmer K (2002) Small-volume resuscitation from experimental evidence to clinical routine. Advantages and disadvantages of hypertonic solutions. Acta Anaesthesiol Scand 46:625–638
Hussmann B, Lefering R, Waydhas C et al (2013) Does increased prehospital replacement volume lead to a poor clinical course and an increased mortality? A matched-pair analysis of 1896 patients of the Trauma Registry of the German Society for Trauma Surgery who were managed by an emergency doctor at the accident site. Injury 44:611–617
Sapsford W (2008) Should the ‘C’ in ‘ABCDE’ be altered to reflect the trend towards hypotensive resuscitation? Scand J Surg 1:4–11
Butler F (2011) Fluid resuscitation in tactical combat casualty care: brief history and current status. J Trauma 70:11–12
Geeraedts LM Jr, Kaasjager HA, van Vugt AB, Froelke JP (2009) Exsanguination in trauma: a review of diagnostics and treatment options. Injury 40:10–20
Hussmann B, Taeger G, Lefering R, Waydhas C, Nast-Kolb D, Ruchholtz S et al (2011) Lethality and outcome in multiple injured patients after severe abdominal and pelvic trauma: influence of preclinical volume replacement—an analysis of 604 patients from the trauma registry of the DGU. Unfallchirug 114:705–712
Hussmann B, Lefering R, Taeger G, Waydhas C, Ruchholtz S, Lendemans S (2011) Influence of prehospital fluid resuscitation on patients with multiple injuries in hemorrhagic shock—a matched-pairs analysis of 2,702 patients from the DGU Trauma Registry. J Emerg Trauma Shock 4:465–471
Malvestio MA, de Sousa RM (2010) Clinical and prehospital survival indicators in blunt trauma: a multivariate analysis. Rev Esc Enferm USP 44:352–359
Bernhard M, Helm M, Aul A, Gries A (2004) Preclinical management of multiple trauma. Anaesthesist 53:887–904
Turner J, Nicholl J, Webber L, Cox H, Dixon S, Yates D (2000) A randomized controlled trial of prehospital intravenous fluid replacement therapy in serious trauma. Health Technol Assess 4:1–57
Bone RC (1991) Let’s agree on terminology: definition of sepsis. Crit Care Med 19:973–976
Vincent JL, Moreno R, Takala J, Willatts S, De Mendonca A, Bruining H et al (1996) The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the working group on sepsis-related problems of the European Society of Intensive Care Medicine. Intensive Care Med 22:707–710
Lefering R (2009) Development and validation of the Revised Injury Severity Classification (RISC) score for severely injured patients. Eur J Trauma Emerg Surg 35:437–447
Trunkey DD (2001) Prehospital fluid resuscitation of the trauma patient. An analysis and review. Emerg Med Serv 30:93–95
Oestern HJ (1999) Management of polytrauma patients in an international comparison. Unfallchirurg 102:80–91
Dutton RP, Mackenzie CF, Scalea TM (2002) Hypotensive resuscitation during active hemorrhage: impact on in-hospital mortality. J Trauma 52:1141–1146
Haut ER, Kalish BT, Cotton BA, Efron DT, Haider AH, Stevens KA et al (2011) Prehospital intravenous fluid administration is associated with higher mortality in trauma patients: a national trauma data bank analysis. Ann Surg 253:371–377
Acknowledgments
We would like to thank the members of the Polytrauma Study Group of the German Trauma Society for their many years of hard work on TraumaRegister DGU®. At the beginning of 2008, the Polytrauma Study Group was integrated into the Committee on Emergency Medicine, Intensive Care and Trauma Management of the German Trauma Society (Sektion NIS).
Participating hospitals
The clinics that provided to the TraumaRegister DGU® data for this analysis can be found at www.traumaregister.de. We thank the clinics for their cooperation.
Conflicts of interest
The authors declare that there are no conflicts of interests.
Author contributions
Matthias Heuer, MD, Ph.D, is responsible in the study conception and design; acquisition, analysis, and interpretation of data; and drafting of the manuscript. Björn Hussmann, MD, contributed in the critical revision of manuscript. Rolf Lefering contributed in the acquisition, analysis, and interpretation of data and critical revision of the manuscript. Gernot M. Kaiser, MD, and Christoph Eicker, MD, contributed in the critical revision of the manuscript. Olaf Guckelberger, MD, and Sven Lendemans, MD, helped in the study conception and design, analysis and interpretation of data, and critical revision of the manuscript.
Author information
Authors and Affiliations
Consortia
Corresponding author
Rights and permissions
About this article
Cite this article
Heuer, M., Hussmann, B., Lefering, R. et al. Prehospital fluid management of abdominal organ trauma patients—a matched pair analysis. Langenbecks Arch Surg 400, 371–379 (2015). https://doi.org/10.1007/s00423-015-1274-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-015-1274-2



