Abstract
Purpose
To examine the effect of separate and combined exposure to hypoxia [normoxia (FIO2 = 0.21) vs. moderate altitude (FIO2 = 0.13)] and temperature [thermoneutral (22 °C) vs. cold (5 °C)] on muscle fatigue development in the forearm, after repeated low-resistance contractions.
Methods
Eight males were exposed for 70 min to four separate conditions in a balanced order. Conditions were normoxic-thermoneutral (N), hypoxic-thermoneutral, normoxic-cold and hypoxic-cold. After 15-min seated rest, participants carried out intermittent dynamic forearm exercise at 15 % maximal isometric voluntary contraction (MVC) for eight consecutive, 5-min work bouts. Each bout was separated by 110 s rest during which MVC force was collected.
Results
When exposed to hypoxia and cold independently, the exercise protocol decreased MVC force of the finger flexors by 8.1 and 13.9 %, respectively, compared to thermoneutral normoxia. When hypoxia and cold were combined, the decrease in MVC force was 21.4 % more than thermoneutral normoxia, reflecting an additive effect and no interaction. EMG relative to force produced during MVC, increased by 2 and 1.2 μV per kg (36 and 23 % of N) for cold and hypoxia, respectively. When the stressors were combined the effect was additive, increasing to 3.1 μV per kg (56 % of N).
Conclusion
When compared to exercise in thermoneutral normoxic conditions, both cold and hypoxia significantly reduce brief MVC force output. This effect appears to be of mechanical origin, not a failure in muscle fibre recruitment per se. Additionally, the reduction in force is greater when the stressors are combined, showing an additive effect.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Passive cold exposure can reduce a muscle’s mechanical response (e.g. power) to a given electrophysiological excitation or descending voluntary drive (Ferretti 1992; Oksa et al. 2002). This is widely attributed to reductions in muscle temperature (Bergh and Ekblom 1979a) which reduces contractile function due to slowed intramuscular energetics and peripheral nerve conduction velocities (Kossler et al. 1987; Bigland-Ritchie et al. 1981; Bergh 1980; Faulkner et al. 1990; Sweitzer and Moss 1990; Ferretti 1992; De Ruiter and De Haan 2000; Allen et al. 2008; Racinais and Oksa 2010; Cahill et al. 2011; Cè et al. 2012). Several studies report that action potential propagation, ATP hydrolysis, Ca2+ handling and sensitively as well as cross-bridge force kinetics are adversely affected by lower tissue temperatures (Kossler et al. 1987; Sweitzer and Moss 1990; Mucke and Heuer 1989; Ferretti 1992; Oksa et al. 2002 Cè et al. 2012). However, the slowing of mechanical processes, as well as efferent and afferent nerve conduction, occur independently of exercise (present during passive cold exposure), and thus may even serve to attenuate metabolite production, and/or increase central drive, during prolonged isometric contractions (Ray et al. 1997; Segal et al. 1986; De Ruiter and De Haan 2000; Todd et al. 2005; Allen et al. 2008; Cahill et al. 2011; Lloyd et al. 2014). Nevertheless, during dynamic exercise in cooled muscle (i.e. active cold exposure), significant increases in skeletal muscle fatigue are reported (Bergh and Ekblom 1979b; Bergh 1980; Faulkner et al. 1990; Racinais and Oksa 2010). This is predominantly due to co-activation of the agonist–antagonist pair (Oksa et al. 1997) resulting in higher workload for the agonist muscle (Oksa et al. 2002) thereby reducing aerobic–mechanical efficiency (McArdle et al. 1976). Furthermore, dynamic exercise in cold muscle is likely affected by reductions in muscle blood flow (Yanagisawa et al. 2004; Gregson et al. 2011), which may hinder oxygen delivery (Amann and Calbet 2007) and diminish the removal metabolic by-products (Blomstrand et al. 1984).
Contrary to tissue cooling, passive exposure to hypoxia does not appear to affect maximal force generating capacity or action potential propagation (Perrey and Rupp 2009). However, increases in muscle fatigue during prolonged exercise in hypoxia have been observed during both whole-body (Amann and Calbet 2007) and repeated contractions of isolated muscle groups (Fulco et al. 1994, 1996; Katayama et al. 2007; Perrey and Rupp 2009; Millet et al. 2008, 2012; Christian et al. 2014a). The rise in muscle fatigue during hypoxia can be largely attributed to a shift of the relative exercise intensity, higher muscle fibre recruitment, and thereby increased intramuscular metabolic disturbance (Edwards 1981; Fulco et al. 1996; Amann et al. 2006a, b; 2007a, b; Fulco et al. 1994; Katayama et al. 2007; Christian et al. 2014a). Specifically, the increase in inorganic phosphate, reactive oxygen species and hydrogen ion production and their interference with the contractile proteins and sarcoplasmic Ca2+ release mechanisms are thought to be a major factor behind the increase in muscle fatigue development (Haseler et al. 1999; Hogan et al. 1999; Amann and Calbet 2007; Perrey and Rupp 2009). In hypoxia, evidence also suggests increased afferent feedback and decreased cerebral oxygenation can reduce voluntary drive to the muscle, exacerbating net fatigue (Amann and Dempsey 2007; Amann et al. 2006a, 2007b; Goodall et al. 2010; Millet et al. 2008, 2012). However, the relative contributions of afferent feedback and cerebral oxygenation, as well as the sense of effort, to changes in central drive during fatiguing exercise in hypoxia remains subject to on-going investigations (Millet et al. 2008, 2012; Goodall et al. 2010; Amann et al. 2013; Christian et al. 2014a, b).
While much research exists on these stressors separately, ascent to altitude often constitutes of exposure to both hypoxia and cold stress; however, the interactive effects of these stressors in combination are not well understood (Tipton 2012). Studies that have examined combined hypoxic-cold stress have focused largely on thermogenesis, skin blood flow and thermal sensitivity (Robinson and Haymes 1990; Johnston et al. 1996; Gautier et al. 1987, Wood 1991; Cipriano and Goldman 1975; Simmons et al. 2010, 2011), leaving fatigue development and human performance relatively unexamined (Tipton 2012). Given the potential for hypoxic-cold to severely compromise oxygen delivery to the active muscle—through simultaneous reductions in oxygen transport (muscle blood flow) and arterial oxygen content (hypoxemia) (Yanagisawa et al. 2004; Gregson et al. 2011; Amann and Calbet 2007)—as well as greatly increase metabolite production—through simultaneous rises in agonist–antagonist co-activation in the cold and type II recruitment in hypoxia (Edwards 1981; Fulco et al. 1996; Oksa et al. 2002; Amann et al. 2006a, b, 2007a, b; Fulco et al. 1994, Katayama et al. 2007; Christian et al. 2014a)—this study sought to investigate the independent and combined (interactive) effects of hypoxia and cold on forearm fatigue.
To investigate the interaction between hypoxia and cold on fatigue development an additive effects model (standard ANOVA) was used. Using the additive model, stressor (in fact all variable) interactions are categorised as either synergistic or antagonistic (Folt et al. 1999). Significant interactions suggest the effect size of one variable has been reduced (antagonistic) or accentuated (synergistic) by the presence (or effect) of the other, whereas additive effects are seen during net stressor independence, i.e. no interaction. Interactions are best illustrated using variable A and B:
Importantly, nullifying interactions (Eq. 4) are strong antagonistic interactions in which the influence of one variable has been entirely abolished by the presence (or effect) of the other; while multiplicative effects describe strong synergistic interactions (Eq. 5).
To examine the interaction between hypoxia and cold on fatigue development, we quantified changes in the relationship between electromyogram (EMG) and maximal isometric voluntary contraction (MVC) force in response to low-intensity, intermittent ‘gripping’ exercises. An isolated forearm model was used due to the importance of finger flexor function for climbing, mountaineering or those performing manual work at altitude, as well as due to the known exacerbation of fatigue during prolonged (>4 min) low-intensity (<30 % MVC) isolated muscle exercise in both cold and hypoxic environments (e.g. Oksa et al. 2002; Perrey and Rupp 2009). It was hypothesised that (1) independent exposure to hypoxia or cold will induce a significant increase in post-exercise neuromuscular fatigue, compared to control conditions; and (2) during combined hypoxic-cold exposure, a synergistic interaction on fatigue will occur, with reductions in muscle blood flow (Yanagisawa et al. 2004; Gregson et al. 2011), and increased metabolite production (co-activation) during cold (Oksa et al. 2002) synergistically accentuated by the reductions in oxygen delivery and higher type II muscle fibre activation observed in hypoxia (e.g. Amann and Calbet 2007).
Methods
Subjects
Eight healthy men volunteered as participants for this study. Their (mean ± SD) age was 21.9 ± 0.8 years and body mass index was 23.5 ± 1.8. Using the short International Physical Activity Questionnaire, a minimal activity level of 25 MET-hours per week was used when selecting volunteers. The average weekly exercise level was 41.5 ± 15.4 MET-hours per week. No participant was trained in a specific sport, but all participants were regularly participating in a range of physical activities, and thus appeared well accustomed to novel and strenuous exercise regimes. All participants were requested to abstain from caffeine, alcohol and exhaustive exercise 24 h prior to the experiment.
The experimental protocol was approved by the Loughborough University Ethical Advisory Committee and was conducted in accordance with the World Medical Associations Declaration of Helsinki for medical research using human participants. All participants were given an information sheet that outlined the procedure, risks and requirements for the experiment. Participants provided written informed consent and completed a questionnaire-based health screening.
Experimental protocol
Systemic, 70-min exposures to four conditions were performed in T.I.S.S. Peak Performance (Series 2009) Chambers at Loughborough University Environmental Ergonomic Research Centre. Participants were exposed once to each of four conditions; control/normoxic-thermoneutrality (N), hypoxic-thermoneutrality (H), normoxic-cold (C) and hypoxic-cold (HC). Thermoneutral conditions (N, H) were 22 °C (50 % RH) ambient temperature (T a) and subjects were dressed in shorts, a t-shirt, socks and trainers. In cold conditions (C, HC) T a was 5 °C (50 % RH) and participants wore the same clothing, minus any upper body insulation (t-shirt). 5 °C T a was selected in an attempt to reduce average skin temperature (T sk) by approximately 5–10 °C, which in turn was assumed to change forearm muscle temperature and cause an increase in fatigue (Oksa et al. 2002) when compared to thermoneutral conditions (N, H). Hypoxic exposures (H, HC) were 0.13 Fraction of Inspired Oxygen (FIO2: equivalent attitude = ~4000 m) aiming to reduce peripheral arterial oxygen saturation (SpO2) to approximately 85 %, a moderate level assumed high enough to influence fatigue during isolated muscle exercise (Millet et al. 2008, 2012; Perrey and Rupp 2009; Christian et al. 2014a). The selection of temperature and FIO2 also aimed to balance severity with ecological validity, in order to maintain relevance for those working or exercising at altitude. Normobaric hypoxia was achieved using an inbuilt chamber hypoxic air generator. Hypoxia was continuously monitored for consistency using a Servomex (570A, Sussex, UK) oxygen analyser as well as the inbuilt analyser on the T.I.S.S. Peak Performance (Series 2009) Chambers. Condition order was balanced and exposures were separated by at least 4 days to allow full recovery from the fatigue protocol. Participants were blinded to conditions prior to exposure.
Measurements of temperature, arterial O2 saturation, skin blood flow, heart rate and perceived exertion
During all conditions aural (T co) and local skin temperature (T sk) from 4 different sites on the exercising arm (bicep, midline of the posterior forearm, midline of the anterior forearm and posterior of hand), were collected. Grant International skin and aural thermistors were secured using Transpore 3M medical grade tape. Aural thermistors were also insulated using cotton wool and earmuffs. Data were recorded at 1-min intervals from 1-min pre-exposure using a Squirrel Data Logger (1000 series, Grant Instruments, UK).
Immediately after each MVC was performed, percentage saturation of peripheral arterial blood (SpO2) and heart rate (HR) were measured using a pulse oximeter attached to the middle finger of the non-exercising arm (Model 8500, Nonin Medical, Netherlands). HR and SpO2 were collected once every 5 s over the first 20 s of each 110-s rest period. Baseline measures of resting HR and SpO2 were collected pre-exposure to condition N, post-5-min supine rest.
Laser Doppler flowmetry [LDF; arbitrary perfusion units (AU; Flux)] of the index finger on the non-exercising hand (Server, Satellite and Optic Probe, MoorLAB, Moor Instruments, UK) and Borg’s Rate of Perceived Exertion (RPE) scores were also recorded immediately after each MVC was performed. LDF was calibrated prior to exposure using the Brownian motion of polystyrene microspheres diluted in water. This LDF device has been used in previous physiological studies to assess skin blood flow (Thompson et al. 2005) and is precise to ±3 % and accurate to ±10AU as determined by the manufacturer (Moor Instruments 2004).
Fatigue protocol and force measurement
Upon exposure to the test conditions, each participant was secured into a restraint system that maintained 90° flexion of the elbow, with the palm and anterior forearm facing vertically, while restricting any movement of the wrist. The system was used to isolate the working forearm muscles and maintain consistent muscle dynamics during exercise. Participants remained secured throughout the experiment. Once in the environmental conditions, participants undertook a 15-min rest period, allowing time for body heat to decline in cold (not reaching steady state) and arterial oxygenation to stabilise in hypoxia. After the rest period, participants performed a fatigue protocol that consisted of eight 5-min work bouts, each separated by 110-s rest/data collection periods, timed using a standard digital stopwatch. During each 5-min exercise bout, dynamic grip clenches (Fit66 Adjustable Grip Exerciser) were performed every 2 s (timed using an audio/visual metronome), at a workload of 15 % of the MVC recorded at the start of the exposure (MVCbaseline) on their first experimental day. Given all subjects were healthy, regularly active, and accustomed to performing a wide range of physical activities, no separate familiarisation was deemed necessary; however, to ensure an accurate prediction of workload was made, practise attempts were available during the MVC used to predict the workload. In all other circumstances, only one MVC was performed. Based on pilot studies, the fatigue protocol was designed to induce an estimated workload that was approximately equal to or greater than, ‘hard’ or 15 on Borg’s RPE Scale, when conducted in thermoneutral normoxic conditions.
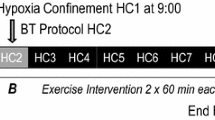
MVC force (3-s contraction) using a Grip Dynamometer (Takei™ No. 1857) was collected at the start of each exposure (MVCbaseline) and after every second work bout (MVC1, MVC2, MVC3, MVC4). A change in MVC, in conjunction with the corresponding EMG data was used to quantify fatigue. Baseline measures of EMG (EMGbaseline) and MVC were collected on immediate exposure to the experimental condition. A schematic overview of the experimental protocol and interventions is shown in Fig. 1.
Schematic representation of the experimental protocol, measurements taken and interventions. T co aural temperature, T sk skin temperature, MVC maximal isometric voluntary contraction, EMG electromyography, LDF laser Doppler blood flow, SpO 2 peripheral oxygen saturation percentage, HR heart rate, RPE rating of perceived exertion. Straight arrows indicate the timing of data collection interventions
Electromyography and fatigue index
To evaluate the myoelectrical activity of the working muscles, surface EMG (Biometrics Ltd, UK) was measured on flexor carpi radialis (FCR), flexor digitorum superficialis (FDS) and extensor digitorum (ED). Other studies analysing forearm muscle fatigue have utilised similar muscle groups (West et al. 1995; Oksa et al. 2002). Flexor group (FCR and FDS) contractions were dynamic during grip clench exercises, and isometric during MVC. The extensor digitorum acts as a fixator and was isometric during both contraction manoeuvres.
The placement of each electrode sensor followed the recommendations outlined by SENIAM (Hermens et al. 2000). The skin was cleaned with alcohol and shaved when necessary. To ensure accurate placement, participants were positioned with the hand supinated and elbow flexed (FDS and FCR) or with the hand pronated and the elbow extended (ED). Each electrode (Biometric Ltd EMG pre amplifier type no. SX230) was placed over the belly of the muscle, identified using palpation over the centre line between the origin and insertion of the muscle. Finger and grip movements were used to tense the appropriate muscle, inducing visible muscle tone, and aiding the accuracy and consistency of EMG placement. The placement of the FDS (1), the FCR (2) and ED (3) electrodes anatomically corresponded to: (1) 2/3 medial of the lateral border of the ventral forearm (in line with the 2nd middle phalanx) and 2/3 distal from antecubital fossa to the wrist–palm intersection; (2) medial of the lateral border of the ventral forearm (in line with the first middle phalanx) and 1/3 distal from the antecubital fossa to wrist–palm intersection; and finally (3) one half medial of the lateral border of the dorsal forearm (in line with the second middle phalanx) and 1/3 distal from the line of the antecubital fossa to the ulna styloid process. All sensors and wires were secured using double-sided tape with the two 2-cm spaced probe contacts running in parallel with the muscle fibres. The reference/ground electrode (Biometrics Ltd earthing strap type no. R200) was placed above the ulna styloid process (inactive tissue) on the opposite wrist. All signals were zeroed prior to measurement and MVC data were logged on a DataLog P3X8 (Biometrics Ltd, UK).
EMG was recorded during all MVC measurements. The EMG sample rate used was 1000 Hz, and was amplified 1000 times to minimise noise on the connecting cable. A signal band of 15–450 Hz, minus the unwanted line frequency of 50 or 60 Hz, including the harmonics of this frequency, was measured. Root mean square (RMS; μV) amplitude values were averaged over a 400-ms running average for the duration of the MVC. Analysis was carried out on the corresponding DataLog Software (Biometrics Ltd, UK) then confirmed and stored using Microsoft Excel 2007. For EMG analysis, a 1-s manually selected sample to include the steadiest values either side of the highest point in the EMG signal was used. Each muscle EMGrms amplitude was calculated using the mean over each 1-s trace. To calculate the average combined muscles EMGrms amplitude, FDS FCR and ED were weighted as 1:1:2 representing a value equal in its contributions from flexors and extensors compartments.
The variable used in this study to define the level of mechanical fatigue was the fatigue index (FI; Oksa et al. 2002). The FI quantifies fatigue using changes in MVC force and EMG amplitude of both the flexor and extensor muscles (mean flexor and extensor weighted 1:1; see above), relative to start and finish of the exercise protocol. FI is calculated as:
If FI = 1.0 then fatigue is not apparent, however, the higher the value above 1.0, the greater the mechanical failure (force independent of excitation). FI differed in the present study from Oksa et al. (2002), because EMG data were collected during the MVC manoeuvre, not during submaximal (fatiguing) exercise; although FI in this case still represents electromechanical transmission failure as well as fatigue though intramuscular metabolic disturbance of the mechanical contraction. To represent the electromechanical ratio over time, EMGrms relative to force produced during MVC (μV per kg) was used; this variable is analogous to FI.
Data analysis
Independent variables for this study were (1) FIO2; (2) T a; and (3) experimental time. Dependent variables were MVC, EMGrms amplitude, RPE, HR, LDF flux, T co, T sk, and FI. All LDF data were corrected for individuals occluded baseline value using a 2-min vascular occlusion (rubber band applied to the finger base) during the 15-min rest period in condition N. MVC and EMG were calculated to represent a change from baseline for each condition. In the figures, EMG data and FI were normalised to the control condition. The following calculation was used to express the variables in condition H, C, and HC, as a percentage change from condition N:
Significance between conditions across the whole exposure was tested using a three-way [2 × 2 × 5; FIO2 × T a × time (fatigue)], repeated measures ANOVA. Significance was tested at a 95 % confidence level (p < 0.05). To test FI and specific time point data for significance between FIO2 and T a, two-way (2 × 2) repeated measures ANOVA was used. When no significant interaction was observed, the effect of cold and hypoxia were reported as C and HC collapsed or H and HC collapsed, respectively. All results are displayed as mean ± SEM.
Results
Temperature, arterial O2 saturation, skin blood flow, rate of perceived exertion and heart rate
Table 1 shows body temperature, SpO2, skin blood flow, RPE and HR across conditions at each MVC intervention. A significant (p = 0.001) reduction in pre-exposure to MVC4 ∆T co was observed during cold conditions (−0.46 ± 0.18 °C), whereas a significant increase (p = 0.007) in ∆T co was observed in thermoneutral conditions (0.19 ± 0.05 °C). The results showed a significant effect of cold T a (p = 0.003), but no significant effect of hypoxia (p = 0.6) and no interaction (p = 0.5) between stressors. At MVC4 T sk of the posterior and anterior forearm was also significantly (p < 0.05) lower during cold exposures (25.6 ± 0.5 °C) than thermoneutral (32.7 ± 0.3 °C), therefore achieving the aimed reduction in local T sk of 5–10 °C. Forearm T sk increased slightly at exercise commencement (t = 15-min) in all conditions but remained significantly (p < 0.05) lower in cold. Mean skin blood flow (LDF flux) over all time points was also significantly (p = 0.001) lower in the cold (−178 ± 63 AU). HR varied similarly with temperature (p = 0.03), reducing from 85 ± 2 bpm in thermoneutral conditions to 78 ± 2 bpm in cold. We observed no significant effects of hypoxia on forearm T sk (p = 0.8), HR (p = 0.2) or LDF flux (p = 0.3). Mean SpO2 measurements across the four interventions showed a significant (p = 0.001) reduction from 97.5 ± 0.3 % SpO2 in normoxic conditions to 85.5 ± 0.6 % SpO2 in hypoxic conditions. While the effect of temperature on SpO2 was significant (p = 0.01) the mean change was minimal (~1 %).
RPE increased significantly (p = 0.03) over time (RPE first set 12.6 ± 0.7; RPE last set 14.0 ± 0.7), but no participants reached task failure during the fatigue protocol in any conditions. There was no main effect over time for temperature (p = 0.4) and hypoxia (p = 0.1). However, a significant effect of temperature was observed at MVC3 (p = 0.021) and MVC4 (p = 0.014) and a trend for the effect of hypoxia (p = 0.074) was observed at MVC1 (Table 1).
Maximal isometric voluntary contraction
Figure 2 shows MVC force produced at each intervention in each condition. MVC decreased significantly (p < 0.05) over time and varied significantly (p < 0.05) between conditions at various MVC time points (see Fig. 2). However, when combined there was no significant statistical interaction between cold and hypoxia (independent contributions) on the total decrease at MVC4 (p = 0.8) or at the MVC time points, i.e. the combined effect of cold and hypoxia was additive. Figure 2, panel b, also shows MVC plotted as a percentage decline from the baseline MVC.
The effect of fatigue on maximal voluntary contraction (MVC) across conditions. Panel b shows maximal voluntary contraction force normalised to baseline for each intervention. N, thermoneutral (22 °C T a) normoxic (0.21 FIO2); H, thermoneutral (22 °C T a) hypoxic (0.13 FIO2); C, cold (5 °C T a) normoxic (0.21 FIO2); HC, cold (5 °C T a) hypoxic (0.13 FIO2). Symbols for effects within the same time point: *Significant for the effect of temperature to p < 0.05 level. #Significant for the effect of FIO2 to p < 0.05 level
Electromyography and fatigue index
EMGrms amplitude during MVC for the combined forearm muscles decreased incrementally with time (p = 0.03); however, the effects of temperature (p > 0.2) and hypoxia (p > 0.5) were not significant between interventions (Fig. 3, Panel a), except during MVC1 where the normalised combined EMGrms was higher (p = 0.05) during cold conditions (C + HC) by 32 ± 13 μV (Fig. 3, Panel b).
The effect of fatigue on root mean squared electromyogram amplitude (EMG) across conditions. Panel b shows EMGrms normalised to baseline for each intervention. N, thermoneutral (22 °C T a) normoxic (0.21 FIO2); H, thermoneutral (22 °C T a) hypoxic (0.13 FIO2); C, cold (5 °C T a) normoxic (0.21 FIO2); HC, cold (5 °C T a) hypoxic (0.13 FIO2). Symbols for effects within the same time point: *Significant for the effect of temperature to p < 0.05 level (data significant when normalised percentage of baseline only)
The FI of the forearm muscles increased to 1.25 in cold and 1.10 in hypoxia. Variance was significant for effects of temperature (p = 0.003) and hypoxia (p = 0.01), however, there was no stressor interaction (p = 0.9): FI was equal 1.45 in combined conditions (Fig. 4, Panel a). Expressed as a percentage of condition N (FI %), exercise resulted in 24 ± 7 % and 39 ± 9 % higher fatigue in hypoxia and cold, respectively. The combined effect was additive resulting in 62 ± 11 % increase in FI, i.e. there was no interaction (Fig. 4, Panel b). EMGrms relative to force produced during MVC (μV per kg) was also significantly increased, with main effects for cold (p = 0.02) over time and a strong trend for hypoxia (p = 0.06) over time. By MVC4 the effect was significant for both cold (p = 0.003) and hypoxia (p = 0.008), increasing by 2.0 and 1.2 μV per kg (36 and 23 % of condition N), respectively. The combined effect was additive (3.1 μV per kg and 56 % of condition N) showing no interaction (p = 0.9) between stressors (Fig. 5, Panel a).
The effect of condition on fatigue index (panel a) and condition normalised (∆) fatigue index (panel b); N, thermoneutral (22 °C T a) normoxic (0.21 FIO2); H, thermoneutral (22 °C T a) hypoxic (0.13 FIO2); C, cold (5 °C T a) normoxic (0.21 FIO2); HC, cold (5 °C T a) hypoxic (0.13 FIO2). *Significant for the effect of temperature to p < 0.005 level. #Significant for the effect of FIO2 to p < 0.05 level
Shows the EMG/MVC relative to baseline, and normalised to the thermoneutral condition for each time point in hypoxia (H), cold (C) and hypoxic-cold (HC). The right hand axis on both panels shows each variable as percentage change of thermoneutral normoxic condition. Symbols for effects within the same time point: @trend for effects of temperature (p < 0.08). *Significant for effects of temperature (p < 0.05); **significant for effects of temperature (p < 0.01). +Trend for effects of FIO2 (p < 0.08). #Significant for effects of FIO2 (p < 0.05); ##significant for effects of FIO2 (p < 0.01)
Discussion
Summary of main findings
This study quantified forearm muscle fatigue development during independent and combined reductions in ambient temperature and inspired oxygen concentration, during repeated low-resistance exercise. The results demonstrate an additive, not interactive, effect on fatigue when humans are exposed to combined hypoxia and cold. Additive effects were observed for both MVC force output and electromechanical ratio (FI and μV per kg) at various time points.
Maximal voluntary contraction in hypoxic-cold
The decline in maximal voluntary force over time can be used to quantify net (overall) neuromuscular fatigue (Gandevia 2001).
In this study, independent exposure to hypoxia and cold significantly reduced MVC force at various time points (Fig. 2). In hypoxia, the decline in MVC force (Fig. 2, Panel b) occurred early in the fatigue protocol, before plateauing at a lower level than thermoneutral normoxic conditions (Fig. 2, Panel b). This differed from cold stress, which continuously reduced MVC over time, resulting in a greater impact of cooling [compared to hypoxia] in the later stages of the exercise protocol (Fig. 2, Panel b). The differences in the temporal decline in MVC may be explained by the stabilisation of arterial oxygenation early in the hypoxic protocol (Table 1), contrary to a more progressive cold penetration through peripheral tissue with cold exposure.
When hypoxia and cold were combined, the temporal (shape of) decline in MVC force (Fig. 2, Panel b) reflected equal (additive) contributions from both stressors, with neither hypoxia nor cold taking clear precedence in the combined condition (Fig. 2, Panel b), i.e. during hypoxic-cold, MVC was subject to both an early (hypoxic) and progressive (cold) decrease over time. This is reflected in the final MVC (MVC4), which reduced in force to a value equal to the summative effect of each stressor individually. Specifically during independent cold and hypoxic stress, MVC force declined by 8.1 and 13.9 % more than thermoneutral normoxia, while MVC force decreased by 21.4 % more during combined hypoxic-cold, closely matching the additive value of hypoxia and cold individually (22 %).
Electromechanical mechanisms during fatigue
Force and EMG assessment has been widely used to understand central and peripheral contributions to fatigue (for reviews see: Gandevia 2001; Amann and Calbet 2007). However, EMG is perhaps better suited to subdividing electrophysiological excitation and mechanical fatigue further downstream from the neuromuscular junction—to a point within the muscle fibre itself (Allen et al. 2008). Importantly under this definition, mechanical fatigue still encompasses many of the intramuscular factors usually associated with peripheral fatigue (Enoka and Stuart 1992; Fitts 1994; Allen et al. 2008), while the changes in motor unit excitation remain partially representative of descending voluntary drive (Gandevia 2001).
Mechanical fatigue in hypoxic-cold
In the present study, mechanical fatigue was disassociated from electrophysiological changes using the electromechanical ratio. These include EMGrms relative to MVC force (in μV per kg) and the FI (Figs. 4, 5). EMG/MVC and the FI measure the direct mechanical response to net fibre excitation and can be attributed on an individual fibre basis to both electromechanical transmission failure and a reduced mechanical response (e.g. force or power) per unit of excitation (Allen et al. 2008).
In this study, independent cold exposure resulted in an increase in both ∆EMG/MVC and the FI (Figs. 4, 5). A similar yet smaller effect was observed during independent exposure to hypoxia. By MVC4 (post-exercise), ∆EMG/MVC was significantly increased by 1.2 and 2 μV per kg in hypoxia and cold, respectively, representing a 23 and 36 % change from N (Fig. 5). This corresponded to a 24 and 39 % increase in FI, respectively (Fig. 4). FI and ∆EMG/MVC reflected the effect of hypoxia and cold on MVC force (Fig. 2), suggesting fatigue was predominantly of mechanical origin in this study, i.e. a failure distal of electrophysiological processes. This is supported by the time course of mechanical fatigue, which is similar to those observed in electrically stimulated muscle fibres in vitro; an early increase, a plateau, then a late increase (Allen et al. 2008; Marcora and Staiano 2010).
Mechanical failure independent of excitation during cold exposure can be attributed to number of factors, such as increases in the relative exercise intensity due to co-activation of the agonist–antagonist pair (Oksa et al. 1997, 2002; Racinais and Oksa 2010) and reduced muscle blood flow (Yanagisawa et al. 2004; Gregson et al. 2011). It may also result from progressive cold penetration through the muscle tissue (Oksa et al. 2002) gradually increasing the number of muscle fibres affected by slowed intramuscular energetics (Bergh 1980; Faulkner et al. 1990; De Ruiter and De Haan 2000; Allen et al. 2008; Racinais and Oksa 2010; Cahill et al. 2011). Conversely, the mild effect of hypoxia on mechanical function (fatigue) has been widely attributed to increases in energetic metabolite interference with Ca2+ handling and the contractile proteins (Edwards 1981; Fulco et al. 1996; Fitts 1994; Haseler et al. 1999; Hogan et al. 1999; Amann and Calbet 2007; Perrey and Rupp 2009; Christian et al. 2014a).
In this study, it was hypothesised that during combined hypoxic-cold stress, co-activation (Oksa et al. 2002) and muscle blood flow reductions (Yanagisawa et al. 2004; Gregson et al. 2011) in cold would accentuate the low oxygen delivery (Amann and Calbet 2007) during hypoxia, resulting in an interaction on net fatigue (MVC) due to altered function at the mechanical level (FI and ∆EMG/MVC). However, contrary to this hypothesis the effect on mechanical fatigue was additive showing no interactions; 3.1 ± 0.3 μV per kg (FI increased by 62 % of N) (Figs. 4, 5). One explanation for this is that the inhibitory influence of increased energetic metabolites during hypoxia (Fitts 1994; Haseler et al. 1999; Hogan et al. 1999; Perrey and Rupp 2009), was not influenced by the direct slowing effect of cooling on ATP hydrolysis, Ca2+ handling and the contractile proteins during cold exposure (Kossler et al. 1987; Sweitzer and Moss 1990; Ferretti 1992; Oksa et al. 2002). It suggests that despite each stressor hindering mechanical function, cold and hypoxia may influence fatigue through sufficiently independent cellular mechanisms, so as not to interact with one another during low-resistance exercise.
The observed additive effect may also result from multiple inter-mechanism interactions and/or an interaction cancelation. A synergistic and antagonistic interaction of similar magnitude would result in net additive effects, and thus to investigate this further, interactive studies examining the individual mechanisms that contribute to hypoxic-cold fatigue are required.
Electrophysiological factors during fatigue in hypoxic-cold
In the present study, MVC sequentially decreased in peak force output and EMGrms amplitude over time, across all conditions (Fig. 3). This suggests changes in corticospinal drive (Gandevia 2001) and/or action potential propagation (Bigland-Ritchie et al. 1981) were partially responsible for fatigue observed during the exercise protocol. However, the decline in combined forearm EMGrms was not generally affected by condition, thus it is unlikely that electrophysiological factors are primarily responsible for the environmental influences on fatigue in the present study. Moreover, despite changes in cognitive function as a result of immediate exposure to environmental stress (Gaoua et al. 2011), we observed no influence on MVC or EMGrms at the start of the exposure, suggesting central drive and muscle fibre recruitment remained largely unaffected.
Perceptual responses to fatigue in hypoxic-cold
Despite no significant condition effect on motor unit recruitment (EMGrms) during MVC (Fig. 3), the rise in the relative work rate (recruitment/voluntary drive) during submaximal repetitive exercise over time and across conditions did appear to mildly increase RPE (Table 1). RPE reflected the temporal decline in overall fatigue, showing a trend for decrease early in hypoxia (MVC1) and a small effect on effort during the latter stages of the protocol with cooling (MVC3 and MVC4). Since participants received no specific detailing on the interpretation of RPE, the increase is likely in response to both a greater mental effort (Marcora and Staiano 2010) and a higher peripheral discomfort (Christian et al. 2014b) with fatigue.
The effect on mechanical and perceived fatigue, yet not MVC recruitment could be because of the inherent limitations on conscious regulation during closed loop protocols. In this study, the only option was to (a) stop exercise, or (b) attenuate motor output during a brief MVC; each providing little or no relief from fatigue. As such, neither perceptual tolerance to fatigue, nor the volitional regulation of neural drive in response to high or maximal levels of peripheral fatigue were investigated. In fact, even given a greater scope for regulation, recovery between bouts may have resulted in maintained neural drive during brief maximal contractions, since afferent feedback is most relevant during a prolonged mental effort with no immediate recovery (Cahill et al. 2011; Amann et al. 2013; Christian et al. 2014a; Lloyd et al. 2014).
Additional perspectives and limitations
Previous studies have shown that hypoxia can cause vasodilation in non-acral skin during cold exposure (e.g. Cipriano and Goldman 1975; Johnston et al. 1996; Simmons et al. 2010). However, the vasoconstrictor response of acral skin (finger pad) during cold exposure was not significantly affected by hypoxia in the present study. A possible explanation is that the large core-to-skin temperature gradient (~−11 °C) at the non-exercised finger was sufficient to abolish the hypoxic vasodilation effect (Simmons et al. 2011), contrary to observations of more proximal skin at milder temperatures for shorter durations (Cipriano and Goldman 1975; Simmons et al. 2010). Furthermore, to our knowledge no direct relationship has previously been shown between perfusion of skin microvasculature and local muscle blood flow. As such, substantiation of the link between skin, local muscle blood flow, and fatigue during both local cold, and combined hypoxic-cold stress, would be an interesting avenue for future studies.
In the present study, aural temperature was used to illustrate changes in core temperature. The results showed a small but significant shift of −0.46 °C during cold exposure, despite a probable increase in metabolic rate during exercise. It should be noted that some of this drop in aural temperature may have been caused by local tissue cooling of the aural canal in this study. However, previous studies using a similar duration and severity of hypoxic-cold have reported similar changes after 75-min rest measured by rectal temperature (−0.4 °C; Robinson and Haymes 1990) and a follow-up study in our lab under similar conditions as the present study (using rectal temperature assessment) has also produced drops in core temperature, although smaller (−0.2 °C after 40-min rest) and after an initial rise (0.15 °C).
Surface EMG reflects not only descending drive to the muscle but also the electrode/muscle interface. As such the effect of changes in local tissue temperature around electrode site cannot be ruled out as a contributing factor to the present observations (Racinais 2013). Also, because flexor and extensor EMG was not measured during the submaximal exercise bouts, co-activation and the temporal rise in mechanical fatigue cannot be concluded from the present results. Additionally, due to the use of small forearm muscles in this study, inter-muscle cross talk is a potential limitation. Finally, it should be recognised that the absence of a separate familiarisation for MVC trials is also a limitation. While this may be minimised by allowing practise attempts prior to the prediction of workloads, and by using young, regular exercisers who are well accustomed to physical activity, the importance of this on reproducibility, validity and reliability should be acknowledged in the context of this study.
Conclusion
In conclusion, the decrease in MVC force and increase in electromechanical ratio and FI (Figs. 2, 4, 5, respectively) support previous findings, suggesting that independent exposure to cold and hypoxia can significantly increase muscle fatigue compared to control conditions. The main finding of this study is that when moderate hypoxia and cold exposure are combined, the decline in MVC force and rise in electromechanical ratio suggest the level of fatigue increases additively, with no interactions; however, further research is warranted using alternative stressor severities and exercise modalities, as this may lead to different results.
Abbreviations
- ANOVA:
-
Analysis of variance
- AU:
-
Arbitrary units
- C:
-
Condition normoxic-cold
- EMG:
-
Electromyography
- ED:
-
Extensor digitorum
- FCR:
-
Flexor carpi radialis
- FDS:
-
Flexor digitorum superficialis
- FFT:
-
Fast Fourier transform
- FI:
-
Fatigue index
- FIO2 :
-
Fraction of inspired oxygen
- H:
-
Condition hypoxic-thermoneutrality
- HC:
-
Conditions hypoxic-cold
- HR:
-
Heart rate
- LDF:
-
Laser Doppler flowmetry
- MVC:
-
Maximal voluntary contraction
- N:
-
Condition normoxic-thermoneutrality
- RMS:
-
Root mean square
- RPE:
-
Rate of perceived exertion
- SpO2 :
-
Peripheral arterial oxygen saturation
- Tco :
-
Core temperature
- Ta :
-
Ambient temperature
- Tsk :
-
Skin temperature
References
Allen DG, Lamb GD, Westerblad H (2008) Skeletal muscle fatigue: cellular mechanisms. Physiol Rev 88:287–332
Amann M, Calbet JAL (2007) Convective oxygen transport and fatigue. J Appl Physiol 104:861–870
Amann M, Dempsey JA (2007) Peripheral muscle fatigue from hyperoxia to moderate hypoxia—a carefully regulated variable? Physiology News 66:28–29
Amann M, Eldridge MW, Lovering AT, Stickland MK, Pegelow DF, Dempsey JA (2006a) Arterial oxygenation influences central motor output and exercise performance via effects on peripheral locomotor muscle fatigue. J Physiol 575:937–952
Amann M, Romer LM, Pegelow DF, Jacques AJ, Hess CJ, Dempsey JA (2006b) Effects of arterial oxygen content on peripheral locomotor muscle fatigue. J Appl Physiol 101:119–127
Amann M, Pegelow DF, Jacques AJ, Dempsey JA (2007a) Inspiratory muscle work in acute hypoxia influences locomotor muscle fatigue and exercise performance of healthy humans. Am J Physiol Regul Integr Comp Physiol 293:R2036–R2045
Amann M, Romer LM, Subudhi AW, Pegelow DF, Dempsey JA (2007b) Severity of arterial hypoxaemia affects the relative contributions of peripheral muscle fatigue to exercise performance in healthy humans. J Physiol 581:389–403
Amann M, Venturelli M, Ives SJ, McDaniel J, Layec G, Rossman MJ, Richardson RS (2013) Peripheral fatigue limits endurance exercise via a sensory feedback-mediated reduction in spinal motoneuronal output. J Appl Physiol 115:355–364
Bergh U (1980) Human power at subnormal body temperatures. Acta Physiol Scand 478:1–39
Bergh U, Ekblom B (1979a) Influence of muscle temperature on maximal muscle strength and power output in human skeletal muscles. Acta Physiol Scand 107:33–37
Bergh U, Ekblom B (1979b) Physical performance and peak aerobic power at different body temperatures. J Appl Physiol 46:885–889
Bigland-Ritchie B, Donovan EF, Roussos CS (1981) Conduction velocity and EMG power spectrum changes in fatigue of sustained maximal efforts. J Appl Physiol 51:1300–1305
Blomstrand E, Bergh U, Essen-Gustavsson B, Ekblom B (1984) Influence of low muscle temperature on muscle metabolism during intense dynamic exercise. Acta Physiol Scand 120:229–236
Cahill F, Kalmar JM, Pretorius T, Gardiner PF, Giesbrecht GG (2011) Whole-body hypothermia has central and peripheral influences on elbow flexor performance. Exp Physiol 96:528–538
Cè E, Rampichini S, Agnello L, Limonta E, Veicsteinas A, Esposito F (2012) Combined effects of fatigue and temperature manipulation on skeletal muscle electrical and mechanical characteristics during isometric contraction. J Electromyogr Kinesiol 22(3):348–355
Christian RJ, Bishop DJ, Billaut F, Girard O (2014a) Peripheral fatigue is not critically regulated during maximal, intermittent, dynamic leg extensions. J Appl Physiol 117:1063–1073
Christian RJ, Bishop DJ, Billaut F, Girard O (2014b) The role of sense of effort on self-selected power output. Front Physiol 5:115
Cipriano LF, Goldman RF (1975) Thermal responses of unclothed men exposed to both cold temperatures and high altitudes. J Appl Physiol 39:796–800
De Ruiter CJ, De Haan A (2000) Temperature effect on the force/velocity relationship of the fresh and fatigued human adductor pollicus muscle. Pflügers Arch 440:163–170
Edwards R (1981) Human muscle function and fatigue. In: Porter R, Whelan J (eds) Human Muscle Fatigue Physiological Mechanisms. Pitman, London, pp 1–8
Enoka RM, Stuart DG (1992) Neurobiology of muscle fatigue. J Appl Physiol 72:1631–1648
Faulkner JA, Zerba E, Brooks SV (1990) Muscle temperature of mammals: cooling impairs most functional properties. Am J Physiol Regul Integr Comp Physiol 28:R259–R265
Ferretti G (1992) Cold and muscle performance. Int J Sports Med 13(Suppl 1):185–187
Fitts RH (1994) Cellular mechanisms of muscle fatigue. Physiol Rev 74:49–94
Folt CL, Chen CY, Moore MV, Burnaford J (1999) Synergism and antagonism among multiple stressors. Limnol Oceanogr 44:864–877
Fulco CS, Cymerman A, Muza SR, Rock PB, Pandolf KB, Lewis SF (1994) Adductor pollicis muscle fatigue during acute and chronic altitude exposure and return to sea level. J Appl Physiol 77:179–183
Fulco CS, Lewis SF, Frykman PN, Boushel R, Smith S, Harman EA, Cymerman A, Pandolf KB (1996) Muscle fatigue and exhaustion during dynamic leg exercise in normoxia and hypobaric hypoxia. J Appl Physiol 81:1891–1900
Gandevia SC (2001) Spinal and supraspinal factors in human muscle fatigue. Physiol Rev 81:1725–1789
Gaoua N, Grantham J, El Massioui F, Girard O, Racinais S (2011) Cognitive decrements do not follow neuromuscular alterations during passive heat exposure. Int J Hyperthermia 27:10–19
Gautier H, Bonora M, Schultz SA, Remmers JE (1987) Hypoxia-induced changes in shivering and body temperature. J Appl Physiol 62:2477–2484
Goodall S, Ross EZ, Romer LM (2010) Effect of graded hypoxia on supraspinal contributions to fatigue with unilateral knee-extensor contractions. J Appl Physiol 109:1842–1851
Gregson W, Black MA, Jones H, Milson J, Morton J, Dawson B, Atkinson G, Green D (2011) Influence of cold water immersion on limb and cutaneous blood flow at rest. Am J Sports Med 39(6):1316–1323
Haseler LJ, Hogan MC, Richardson RS (1999) Skeletal muscle phosphocreatine recovery in exercise-trained humans is dependent on O2 availability. J Appl Physiol 86:2013–2018
Hermens HJ, Freriks B, Disselhorst-Klug C, Rau G (2000) Development of recommendations for SEMG sensors and sensor placement procedures. J Electromyogr Kinesiol 10:361–374
Hogan MC, Richardson RS, Haseler LJ (1999) Human muscle performance and PCr hydrolysis with varied inspired oxygen fractions: a 31P-MRS study. J Appl Physiol 86:1367–1373
Johnston CE, White MD, Wu M, Bristow GK, Giesbrecht GG (1996) Eucapnic hypoxia lowers human cold thermoregulatory response thresholds and accelerates core cooling. J Appl Physiol 80:422–429
Katayama K, Amann M, Pegelow DF, Jacques AJ, Dempsey JA (2007) Effect of arterial oxygenation on quadriceps fatigability during isolated muscle exercise. Am J Physiol Regul Integr Comp Physiol 292:R1279–R1286
Kossler F, Lange F, Kuchler G (1987) Isometric twitch and tetanic contraction of frog skeletal muscles at temperatures between 0 to 30°C. Biomed Biochim Acta 46:809–814
Lloyd A, Hodder S, Faulkner S, Fry A, Havenith G (2014) Muscle temperature limits isometric endurance via sensory feedback-mediated central fatigue. Med Sci Sports Exerc 46(5S):180–187
Marcora SM, Staiano W (2010) The limit to exercise tolerance in humans: mind over muscle? Eur J Appl Physiol 109(4):763–770
McArdle WD, Magel JR, Lesmes GR, Pechar GS (1976) Metabolic and cardiovascular adjustment to work in air and water at 18, 25, and 33°C. J Appl Physiol 40(l):85–90
Millet GY, Aubert D, Favier FB, Busso T, Benoît H (2008) Effect of acute hypoxia on central fatigue during repeated isometric leg contractions. Scand J Med Sci Sports 19:695–702
Millet GY, Muthalib M, Jubeau M, Laursen PB, Nosaka K (2012) Severe hypoxia affects exercise performance independently of afferent feedback and peripheral fatigue. J Appl Physiol 112:1335–1344
Moor Instruments (2004) MoorLab laser Doppler blood flow user manual
Mucke R, Heuer D (1989) Behaviour of EMG parameters and conduction velocity in contractions with different muscle temperatures. Biomed Bichim Acta 5(6):459–464
Oksa J, Rintamäki H, Rissanen S (1997) Muscle performance and electromyographic activity of the lower leg muscles with different levels of cold exposure. Eur J Appl Physiol 75:484–490
Oksa J, Ducharme M, Rintamäki H (2002) Combined effect of repetitive work and cold on muscle function and fatigue. J Appl Physiol 92:354–361
Perrey S, Rupp T (2009) Altitude-induced changes in muscle contractile properties. High Alt Med Biol 10:175–182
Racinais S (2013) Hot ambient conditions shift the Force/EMG relationship. Springerplus 2:317
Racinais S, Oksa J (2010) Temperature and neuromuscular function. Scand J Med Sci Sports 20:1–18
Ray CA, Hume KM, Gracey KH, Mahoney ET (1997) Muscle cooling delays activation of the muscle metaboreflex in humans. Am J Physiol Heart Circ Physiol 273:2436–2441
Robinson KA, Haymes EM (1990) Metabolic effects of exposure to hypoxia plus cold at rest and during exercise in humans. J Appl Physiol 68:720–725
Segal SS, Faulkner JA, White TP (1986) Skeletal muscle fatigue in vitro is temperature dependent. J Appl Physiol 61:660–665
Simmons GH, Fieger SM, Minson CT, Halliwill JR (2010) Hypoxic cutaneous vasodilation is sustained during brief cold stress and is not affected by changes in CO2. J Appl Physiol 108:788–792
Simmons GH, Barrett-O’Keefe Z, Minson CT, Halliwill JR (2011) Cutaneous vascular and core temperature responses to sustained cold exposure in hypoxia. Exp Physiol 96:1062–1071
Sweitzer N, Moss R (1990) The effect of altered temperature on Ca2 + -sensitive force in permeabilized myocardium and skeletal muscles. J Gen Physiol 96:1221–1245
Thompson CS, Holowatz LA, Kenney WL (2005) Cutaneous vasoconstrictor responses to norepinephrine are attenuated in older humans. Am J Physiol Regul Integr Comp Physiol 288:R1108–R1113
Tipton M (2012) A case for combined environmental stressor studies. Extrem Physiol Med 1:7–8
Todd G, Butler JE, Taylor JL, Gandevia SC (2005) Hyperthermia: a failure of the motor cortex and the muscle. J Physiol 563:621–631
West W, Hicks A, Clements L, Downing J (1995) The relationship between voluntary electromyogram, endurance time and intensity of effort in isometric handgrip exercise. Eur J Appl Physiol 71(301):30
Wood SC (1991) Interactions between hypoxia and hypothermia. Annu Rev Physiol 53:71–85
Yanagisawa O, Kudo H, Takahashi N, Yoshioka H (2004) Magnetic resonance imaging evaluation of cooling on blood flow and oedema in skeletal muscles after exercise. Eur J Appl Physiol 91:737–740
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Nicolas Place.
Rights and permissions
About this article
Cite this article
Lloyd, A., Hodder, S. & Havenith, G. The interactive effect of cooling and hypoxia on forearm fatigue development. Eur J Appl Physiol 115, 2007–2018 (2015). https://doi.org/10.1007/s00421-015-3181-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-015-3181-1