Abstract
Objectives
To assess the detectability of vasculitic changes of the superficial cranial arteries with contrast-enhanced 7 T MRI in three patients with GCA and intraindividual comparison with 3 T MRI.
Methods
Three patients (two female, one male) with suspected GCA underwent contrast-enhanced T1-weighted high-resolution MRI at 3 T and 7 T magnetic field strength. The clinical diagnosis, based on criteria of the American College of Rheumatology, was confirmed by biopsy of the superficial temporal artery after MRI. MR images were visually assessed for detection of vasculitic mural contrast enhancement and vessel wall thickening of the superficial cranial arteries.
Results
Both 3 T and 7 T MRI allows for visualisation of mural inflammatory changes and assessment of the vasculitic involvement pattern. Image quality of 7 T MRI was rated superior to image quality of 3 T MRI scans.
Conclusions
7 T MR imaging of vasculitic changes in patients with GCA is possible, and suggests superior image quality. Larger patient studies are necessary to quantify the diagnostic advantages of 7 T MRI.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Giant cell arteritis (GCA) is a granulomatous vasculitis of large- and medium-sized vessels that mostly affects elderly patients. Because of its unspecific symptoms it is still a diagnostic challenge [1, 2]. The diagnostic gold standard is biopsy and histological examination of the superficial temporal arteries, an invasive method regularly applied to confirm the diagnosis in terms of the essential systemic long-term medication with corticosteroids and possible adverse reactions. While a positive histology result can be considered as diagnostic proof, a negative biopsy [3] does, due to skip lesions, not rule out GCA [4–6]. The rate of false-negative histology results is estimated to be 10 % to 25 % [4, 7]. Importantly, however, an early diagnosis and immediate therapy is imperative to avoid devastating ischemic complications, such as loss of vision [1] or cerebrovascular events [8]. MRI promises observer-independent imaging approaches for a fast and accurate GCA detection. Previously, a high-resolution contrast-enhanced T1-weighted MR imaging protocol was proposed for the assessment of vascular inflammation in GCA via the identification of vessel wall thickness and mural contrast agent enhancement [9, 10]. Follow-up studies demonstrated high-resolution MRI to be a valuable imaging approach in the diagnosis of GCA, with good agreement with histological results and clinical diagnosis based on ACR criteria [4, 11]. Recently, a multi-centre trial including 98 patients with suspected GCA and histological evaluation confirmed previous single-centre results with a sensitivity of 88.7 % and a specificity of 75.0 % and a good interobserver agreement [12].
As previous MRI studies in GCA underlined the superiority of 3 T over 1.5 T imaging [4], the advent of 7 T imaging promises a further increase in the diagnostic potential. Here, we assessed the detectability of vasculitic changes of the superficial cranial arteries with contrast-enhanced 7 T MRI in three patients, and with an intraindividual comparison with 3 T MRI.
Methods
Patients
This study was approved by the Ethics Committee of the Otto-von-Guericke University Magdeburg, and followed the tenets of the Declaration of Helsinki. All participants gave their informed written consent.
Two women and one man treated for suspected GCA in the Department of Ophthalmology were included in this comparative pilot study (Table 1). They had palpable abnormalities of the superficial temporal artery and underwent temporal artery biopsy within 1, 2, and 3 days after the MRI measurements, which confirmed the diagnosis GCA in all three cases. Systemic treatment with an initial dose of 500 mg (patient #2 and #3) and 100 mg (patient #1) of corticosteroids was started on the day of admission. The longest duration of corticosteroid therapy prior to MRI was 3 days.
Criteria for patient exclusion from this study were renal dysfunction (glomerular filtration rate <60 ml/min), allergy to contrast agent, implants/stents, treatment with corticosteroids > 5 days, epilepsy, an instable general condition, participation in other studies, or former temporal artery biopsies. Between November 2010 and August 2013, fifteen patients were excluded (four with renal dysfunction, four with implants/stents, one with both attributes, three with an allergy to contrast agents, one with claustrophobia, one with a significant stenosis of the vertebral artery, and a lack of capacity of the MRI in one case). Cases in which the patient declined the participation were not registered. Because of this low inclusion rate, we decided to present this initial case series of three investigated patients with biopsy-proven GCA.
MRI measurements
Contrast-enhanced MR imaging was first performed on a whole body 7 T system (Siemens Healthcare, Germany) using a 32-channel head coil (Nova Medical, Wilmington, DE, USA). After the intravenous application of single-dose contrast media [Multihance, Bracco (patients #1 & #2), Gadovist, Bayer (patient #3)] T1-weighted spin echo images were acquired in axial orientation aligned along the anterior and posterior commissure (TR 500–600 ms (depending on SAR restrictions), TE 17 ms, matrix 1024 × 840, FoV 210 × 180 mm2, slice thickness 2 mm, gap 1 mm, bandwidth 132 Hz/px, no partial Fourier). Thirty axial slices covered a large central part of the brain. The pulse duration of the refocusing pulse was extended to stay within SAR limits. This had the positive side-effect that due to the different bandwidths of the excitation and refocusing pulses, only water signals contributed to the signal, and fat signals were not acquired. This reduced SAR further, as no separate fat saturation pulse was required. Within 1 hour after the 7 T examination, patients underwent 3 T MR imaging (MAGNEOM Verio or MAGNETOM Trio, Siemens Healthcare, Germany) also employing 32-channel head coils without further injection of contrast media. Fat-saturated T1-weighted spin echo images were acquired (TR 500 ms, TE 15 ms, matrix 1024 × 768, FoV 200 × 200 mm, slice thickness 3 mm, gap 3 mm, bandwidth 76 Hz/px, partial Fourier 6/8). MR images were visually assessed, with particular emphasis on the detection of mural contrast agent enhancement and wall thickening of the superficial cranial arteries. These signs have been recognized as typical vasculitic MRI findings in patients with GCA [9]. A 3D rendering of the arteries is not supported by the resolution applied here. For this purpose, future research applying the very high resolution angiography capability of 7 T may add further insight.
Results
Case 1
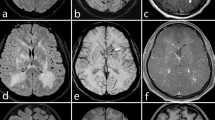
Figure 1 illustrates the results of the cranial 3 T and 7 T MRI in an 84-year-old male patient with suspected GCA due to an anterior ischemic optic neuropathy (left eye) and an elevated ESR of 78 mm in the first hour. Biopsy of the right superficial temporal artery that was performed 2 days after the MRI scans confirmed the diagnosis. Contrast-enhanced T1-weighted MR imaging visualizes the entire cranial circumference for the examination of the superficial cranial arteries and their branches. At 3 T (Fig. 1a), only slight vasculitic signs are evident. There is wall thickening and slight contrast enhancement of the wall of the frontal branch of the left (◄) and the right (→) superficial temporal arteries, which are imaged horizontally. In Fig. 1b, the corresponding 7 T images are depicted, i.e., of the cranial circumference of the same patient at nearly the same level. Mural contrast enhancement and wall thickening of the frontal branches of the right (→) and left superficial temporal artery (◄) appear more pronounced at 7 T. It should be noted that the 7 T images were acquired immediately after the intravenous administration of the contrast agent, while the patient did not receive an additional contrast injection for the 3 T scans acquired approximately 1 hour after the 7 T scan. For a more dorsal 7 T image of the same patient (Fig. 1c), the right frontal branch of the superficial temporal artery does not display any inflammatory signs (*), while inflammatory contrast enhancement and wall thickening of the parietal branch of the superficial right temporal artery is evident (→). In contrast, the left superficial cranial arteries appear to be not affected (◄ marks the parietal branch of the left superficial temporal artery).
Contrast-enhanced, T1-weighted SE image of patient #1 at 3 T (a: VERIO) and 7 T (b, c) at the corresponding location (a, b) and one more cranial slice (c) to assess the involvement of the superficial cranial arteries in the inflammatory processes. a Slight inflammatory vessel changes at 3 T, with a thickened vessel wall of the frontal temporal artery on both sides (→, ◄) and slight contrast enhancement of the frontal branch of the left superficial temporal artery (◄). b 7 T MRI of the cranial circumference of the same patient at nearly the same level. On both sides, the frontal branches of the superficial temporal artery are horizontally imaged and display wall thickening. The frontal branch of the right superficial temporal artery (→) demonstrates a more conspicuous contrast enhancement in comparison to the left side (◄). c 7 T images at a higher level demonstrating contrast enhancement and mural thickening of the right parietal branch (→). There are no inflammatory signs of the right frontal (*) and left parietal branches (◄)
Case 2
Figure 2 illustrates the results of the cranial 3 T and 7 T MRI of a 75-year-old female patient with suspected GCA due to a new onset of headache, and an elevated ESR of 76 mm in the first hour. The diagnosis was confirmed histologically by biopsy of the left superficial temporal artery 3 days after the MRI. Contrast-enhanced T1-weighted MR imaging illustrates mural inflammatory changes of the frontal branch of the left (not shown) and the right superficial temporal artery (‘→’ in Fig. 2) at both magnetic field strengths. Mural contrast enhancement with accumulation of contrast medium in the perivascular space appears stronger at 7 T than at 3 T. It should be noted that the 7 T images were acquired immediately after the application of contrast medium, while the 3 T images were acquired 30 minutes later. The signal intensity of skin and subcutaneous fat at 7 T MR image appears more inhomogeneous than at 3 T, and also shows signs of patient motion. No mural inflammatory changes of the left superficial temporal or occipital arteries are depicted at this level for either field strength.
Case 3
Figure 3 illustrates the results of the cranial 3 T and 7 T MRI of the cranial circumference of a 69-year-old female patient with suspected GCA referred by a rheumatologist due to lassitude, palpable abnormalities of the superficial temporal artery, and an elevated ESR of 98 mm in the first hour. Biopsy of the left superficial temporal artery performed 1 day after the MRI scans confirmed the diagnosis. The 3 T MRI depicts mural inflammatory changes of the parietal branch of the right superficial temporal artery (→), while there is only slight contrast enhancement on the left side (◄). Prominent inflammatory mural enhancement of the right and left parietal branches of the superficial temporal artery in nearly identical location is evident at 7 T. Solitary single-dose contrast agent was delivered directly prior to acquisition at 7 T and about 60 minutes prior to acquisition at 3 T.
Contrast-enhanced, T1-weighted images of patient #3 at 3 T (a; Verio) and 7 T (b) at nearly equivalent levels. a Inflammatory vessel changes of the parietal branch of the right (→) and, less conspicuous, of the parietal branch of the left superficial temporal artery (◄). b 7 T MRI readily reveals bright inflammatory contrast enhancement of the parietal branches on both sides
Discussion
We present the first report of high-resolution 7 T MRI of the superficial temporal arteries in three patients with biopsy-proven GCA and the intraindividual comparison to 3 T MRI. The detection of mural inflammatory changes in a bilateral assessment of the frontal and parietal branches of the superficial temporal and the superficial occipital artery within a single acquisition was possible at 7 T. The higher field strength provided strong contrast enhancement with subjectively superior image quality that may result in greater sensitivity to detect inflammatory signs. Thus, the use of 7 T imaging suggests an increase in sensitivity with the known benefits of MR-based assessment in GCA, i.e., assessment of the whole cranial circumference to evaluate the cranial involvement pattern, to localize inflamed vessel segments, and to distinguish between involved and uninvolved segments. This is important, as GCA is marked by a discontinuous affection [5]. The detection of such local inflammatory lesions may assist the optimal preparation of temporal artery biopsy, with the potential to decrease the rate of false-negative sample extraction [10]. While the three cases of the present study suggest a good agreement of MRI findings at 7 T with histological results and the diagnosis according to the ACR criteria, only follow-up studies on a greater patient cohort will be able to provide the data basis to assess the sensitivity and specificity of MR-based GCA diagnostics at 7 T.
One obstacle of 7 T MR imaging is the inhomogeneous signal intensity of the skin and fat which was observed in the present case series. It should be noted, however, that the results suggest a superior imaging quality of the superficial cranial arteries at 7 T compared to 3 T, i.e., a gain of spatial resolution and contrast to noise. This is probably due to the higher intrinsic signal-to-noise ratio at 7 T and the longer native T1 that leads to stronger contrast-agent effects. Contrast agents were delivered immediately prior to 7 T imaging, but 30 minutes (patient # 2) to 60 minutes (patient # 1 and patient # 3) prior to 3 T imaging. The elimination half-life time is 1.2 to 1.7 h for Multihance and 1.3 to 2.1 h for Gadovist. This suggests a minor, but possible effect of the contrast-agent elimination. Follow-up investigations are needed to address this issue and to quantify the resolution or contrast-to-noise advantage at higher magnetic field strength, and to evaluate possible diagnostic advantages. In this study, only three out of 15 patients could be included due to a number of contraindications for contrast-enhanced MRI at 7 T. With the introduction of 7 T to clinical diagnosis, we expect more implants to be tested and certified as MR-safe for 7 T in the future.
To avoid severe ischaemic complications of GCA a rapid initiation of high-dose corticosteroids is essential. Clinical symptoms such as headache or jaw claudication usually disappear soon after initiation of steroid therapy, but histological signs of GCA in biopsy are still detectable after 2 weeks of steroid treatment [13]. Previous studies have shown that vasculitic MRI signs in GCA disappear soon under successful therapy, and diagnostic imaging should be performed as soon as possible, preferentially before or early after the beginning of medication to avoid false-negative results [12, 14–16]. This phenomenon was taken into account in the present case series, with the longest period interval of steroid treatment before MRI comprising 3 days. Further studies are necessary to analyse how long vasculitic signs seen in MRI persist under steroid treatment, and if 7 T provides a diagnostic advantage to estimate diagnostic success and detect relapsing vasculitis.
Vasculitic mural inflammatory changes typically present as circumferential mural thickening and contrast enhancement in MRI as compared to the rather eccentric appearance of an atherosclerotic plaque. For that reason, imaging planes should be chosen in an orthogonal orientation to the vessel’s track. It should be noted, however, that the specific cause of vasculitic changes cannot be differentiated by MRI signs alone [17, 18]. Various systemic diseases such as sarcoid, systemic lupus erythematosus (SLE), gaint cell arteritis (GCA), or others may lead to mural vasculitic inflammatory changes. The specific clinical presentations of those diseases differ substantially among each other and need to be taken into account for the differential diagnosis.
High-resolution imaging at 7 T without contrast agents might be of value for the identification of vascular changes [19, 20]. For example, irregularities of the artery lumen and especially stenoses might be detected with non-contrast 7 T angiography. This imaging approach permits the assessment of the vessel’s course without the need for contrast agent. This would increase the diagnostic capabilities related to patients with allergies to contrast agents or renal failure, and would help to avoid complications. The presented case series is too small to contribute to this issue. Further studies should address the possibility to perform MR imaging without contrast medium in patients with allergies and impaired renal function. In a 3 T MRI trial including 43 patients with suspected GCA, Geiger et al. [21] found an indication for the possibility to detect vasculitic signs of the superficial cranial arteries by radial T2-weighted MRI without administration of i.v. contrast agents. A limitation of the MRI protocol used in that study is the lower spatial resolution of T2 compared with the post-contrast T1-weighted method, which makes the detection of mild and moderate GCA cases difficult. Higher resolution of T2-weighted imaging might be achieved using a higher magnetic field strength.
The present case series of three patients demonstrates that contrast-enhanced 7 T MRI allows the detailed visualization of the superficial cranial arteries and the non-invasive assessment of the cranial involvement pattern. There is an indication that the higher magnetic field strength promises further improvement of the vasculitis diagnosis. Further studies with larger patient trials are necessary to detail the diagnostic advantages of ultra-high field MR imaging.
References
Salvarani C, Cantini F, Hunder GG (2008) Polymyalgia rheumatica and giant-cell arteritis. Lancet 372:234–245
Hunder GG, Bloch DA, Michel BA et al (1990) The American College of Rheumatology 1990 criteria for the classification of giant cell arteritis. Arthritis Rheum 33:1122–1128
Hayreh SS, Podhajsky PA, Raman R, Zimmerman B (1997) Giant cell arteritis: validity and reliability of various diagnostic criteria. Am J Ophthalmol 123:285–296
Bley TA, Uhl M, Carew J et al (2007) Diagnostic value of high-resolution MR imaging in giant cell arteritis. AJNR Am J Neuroradiol 28:1722–1727
Klein RG, Campbell RJ, Hunder GG, Carney JA (1976) Skip lesions in temporal arteritis. Mayo Clin Proc 51:504–510
Taylor-Gjevre R, Vo M, Shukla D, Resch L (2005) Temporal artery biopsy for giant cell arteritis. J Rheumatol 32:1279–1282
Ness T, Bley TA, Schmidt WA, Lamprecht P (2013) The diagnosis and treatment of giant cell arteritis. Dtsch Arztebl Int 110:376–385, quiz 386
Zenone T, Puget M (2013) Characteristics of cerebrovascular accidents at time of diagnosis in a series of 98 patients with giant cell arteritis. Rheumatol Int 33:3017–3023
Bley TA, Wieben O, Uhl M, Thiel J, Schmidt D, Langer M (2005) High-resolution MRI in giant cell arteritis: imaging of the wall of the superficial temporal artery. AJR Am J Roentgenol 184:283–287
Bley TA, Weiben O, Uhl M et al (2005) Assessment of the cranial involvement pattern of giant cell arteritis with 3T magnetic resonance imaging. Arthritis Rheum 52:2470–2477
Bley TA, Wieben O, Uhl M et al (2005) Integrated head-thoracic vascular MRI at 3 T: assessment of cranial, cervical and thoracic involvement of giant cell arteritis. MAGMA 18:193–200
Klink T, Geiger J, Both M et al (2014) Giant cell arteritis: diagnostic accuracy of MR imaging of superficial cranial arteries in initial diagnosis-results from a multicenter trial. Radiology 273:844–852
Achkar AA, Lie JT, Hunder GG, O’Fallon WM, Gabriel SE (1994) How does previous corticosteroid treatment affect the biopsy findings in giant cell (temporal) arteritis? Ann Intern Med 120:987–992
Bley TA, Ness T, Warnatz K et al (2007) Influence of corticosteroid treatment on MRI findings in giant cell arteritis. Clin Rheumatol 26:1541–1543
Bley TA, Markl M, Schelp M et al (2008) Mural inflammatory hyperenhancement in MRI of giant cell (temporal) arteritis resolves under corticosteroid treatment. Rheumatology (Oxford) 47:65–67
Hauenstein C, Reinhard M, Geiger J et al (2012) Effects of early corticosteroid treatment on magnetic resonance imaging and ultrasonography findings in giant cell arteritis. Rheumatology 51:1999–2003
Choi YJ, Jung SC, Lee DH (2012) Vessel wall imaging of the intracranial and cervical carotid arteries. J Stroke 17:238–255
Küker W (2007) Cerebral vasculitis: imaging signs revisited. Neuroradiology 49:471–479
Fischer A, Maderwald S, Johst S et al (2014) Initial evaluation of non-contrast-enhanced magnetic resonance angiography in patients with peripheral arterial occlusive disease at 7 T. Investig Radiol 49:331–338
Wrede KH, Dammann P, Monninghoff C et al (2014) Non-Enhanced MR imaging of cerebral aneurysms: 7 Tesla versus 1.5 Tesla. PLoS One 9(1):e84562
Geiger J, Bley T, Uhl M, Frydrychowicz A, Langer M, Markl M (2010) Diagnostic value of T2-weighted imaging for the detection of superficial cranial artery inflammation in giant cell arteritis. J Magn Reson Imaging 31:470–474
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Dr. Bley receives ongoing research support from the “Deutsche Forschungsgemeinschaft, DFG” for MRI research.
Conflict of interest
Dr. Bley had past consulting activities for HeartFlow, GSK, and MSD, and received past payments for lectures from Bayer, Bracco, HeartFlow, Guerbet, and Siemens. Beyond this, all authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge, or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Goll, C., Thormann, M., Hofmüller, W. et al. Feasibility study: 7 T MRI in giant cell arteritis. Graefes Arch Clin Exp Ophthalmol 254, 1111–1116 (2016). https://doi.org/10.1007/s00417-016-3337-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-016-3337-7