Abstract
Background
Amyotrophic lateral sclerosis (ALS) is a global disease, which adversely affects the life quality of patients and significantly increases the burden of families and society. We aimed to assess the changing incidence, prevalence of ALS around the world.
Methods
We searched Medline, Embase, Web of Science, and Cochrane library to identify articles published until September 9, 2018. Each included study was independently reviewed for methodological quality by two reviewers. We used a random-effects model to summarize individual studies and assessed heterogeneity (I2) with the χ2 test on Cochrane’s Q statistic.
Results
We identified 124 studies that were eligible for final inclusion, including 110 studies of incidence and 58 studies of prevalence. The overall crude worldwide ALS prevalence and incidence were 4.42 (95% CI 3.92–4.96) per 1,00,000 population and 1.59 (95% CI 1.39–1.81) per 1,00,000 person-years, respectively. ALS prevalence and incidence increased by age until the age of 70–79. Since 1957, incidence has been significantly rising year by year, and this upward trend was weakened after standardization. The longest survival time were in Asia (ranging from 3.74 years in South Asia to 9.23 years in West Asia).
Conclusions
With the aggravation of population aging and the rapid growth of economy, developing regions following the development pattern of the developed regions may suffer rising ALS prevalence and incidence which may increase their disease burden as well. These data highlight the need for research into underlying mechanism and innovations in health-care systems.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Amyotrophic lateral sclerosis (ALS) is a rare neurodegenerative disease. ALS starts in a single part and then spreads to most muscles including the diaphragm of body, rendering the majority of patients dead from respiratory failure eventually [1]. Patients with ALS may suffer from cognitive impairments, such as frontotemporal dementia (ALS–FTD), executive cognitive impairment (ALS–ECI), non-executive cognitive impairment (ALS-NECI), etc. [2, 3]. As a public challenge, 3,30,918 people suffered from motor neuron disease, causing 9,26,090 disability-adjusted life years (DALYs) and 34,325 deaths in 2016 [4]. In Google and Bing/Yahoo, ALS was one of the most frequently searched neurological diseases [5].
The existing epidemiological studies of ALS indicated variation in ALS prevalence and incidence across different populations, especially in terms of age and sex. Furthermore, a number of studies revealed regional and temporal variations as well. Taking Norway as an example, the ALS prevalence climbed from 3.67 per 1,00,000 population in 1988 [6] to 4.10 per 1,00,000 population in 2015 [7]. Meanwhile, the incidence rose from 1.60 per 1,00,000 person-years between 1978 and 1988 [6] to 2.10 person-years between 2000 and 2015 [7]. While the ALS prevalence and incidence of Taiwan in 2008 were 2.07 per 100 000 population and 0.53 per 1,00,000 person-years, respectively [8].
Considering the particularity of rare diseases, most related studies only collected data within a certain region or limited years, making it difficult to assess the geographical variation and temporal trend in a single study. However, systematic review and meta-analysis, as powerful statistical methods, could provide quantitative approaches for pooling the results of different studies on the same topic and estimating the diversity among different regions and years [9].To date, a couple of systematic reviews have summarized the worldwide prevalence and incidence of ALS. The results of the sex and age variation of ALS were consistent with single studies: prevalence and incidence were higher in males than females, and ascending by age [4, 10, 11]. Two systematic reviews have explored the ALS geographical variation [4, 10], and found that the incidence was higher in Europe and America than in Africa and Asia. One of those meta-analysis published in 2017 [10], which included all articles published before June 2015, failed to detect temporal trend of the standardized ALS incidence. However, since 2016 a great many high-quality studies with sample size larger than 1,00,000 [12,13,14,15] have added into this field. For instance, after 2015, two studies based on larger sample size in West Asia [16, 17] suggested the incidence was 1.3–1.8 per 1,00,000 person-years, which was nearly three times the result from a meta-analysis published in 2017 [18].
Given that all above systematic reviews were based on articles before 2016, we proposed to update the geographical characteristics and temporal trend of ALS incidence and prevalence. Therefore, we did a systematic review of population-based studies reporting the incidence and prevalence of ALS across the world from inception to September 2018 to update the geographical variation and temporal trends of ALS prevalence and incidence. This study is registered in the PROSPERO, number CRD42019119168, and followed the Meta-analysis Of Observational Studies in Epidemiology (MOOSE) guidelines (Online Resource 1).
Methods
Search strategy
In this systematic review and meta-analysis, we searched Medline, Embase, Web of Science, and Cochrane library to identify all relevant studies focusing on the global population with ALS, published up to September 9, 2018. The search strategy was made by consulting the librarians in Peking University Health Science Center (Online Resource 2). To supplement these database searches, references of all relevant studies were also screened to identify additional potential data sources. This study is registered with PROSPERO, number CRD42019119168.
Inclusion and exclusion criteria
Inclusion criteria were: (1) population-based studies, reporting the prevalence or incidence of ALS or providing the data to compute these estimates; (2) The diagnosis of ALS was based on neurological experts, self-reports, disease codes or neurological examinations according to pre-defined criteria. Exclusion criteria were: (1) duplicates, reviews, conference proceedings, case reports, experiments and clinical trials; (2) Studies reporting prevalence or incidence for specific population not for general population; (3) studies from ALS foci [10, 19]including Papua, Guam ALS-PDC focus and Wakayama prefecture; (4) non-English studies; (5) no full text.
Data extraction and quality evaluation
Two reviewers (TXL and YXY) assessed all studies independent from each other and filled the following items in a data extraction form: (1) publication year, author, study region, study period, (2) case definition criteria, number of patients, size of study population, (3) prevalence, incidence and corresponding 95% CIs, and (4) the year-specific, age-specific, region-specific, and sex-specific results.
A quality assessment tool was applied (Online Resource 3) consistent with published guidelines [20, 21] and previous relevant meta-analysis [22], which involved three aspects including sample representativeness, condition assessment, and statistical methods.
In the event of disagreement on extracted data, the article was assessed by 2 other authors (LX and SFW) independently from each other and discussed between the four authors until agreement was achieved.
Statistical analysis
To minimize the influence from studies with extreme estimates on the combined values, we first stabilized the variance of study-specific rate using the Freeman-Tukey double arcsine transformation before pooling analysis [23, 24]. Unadjusted pooled rate was calculated on the basis of the information of crude rates and corresponding standard errors provided by individual studies or estimated from statistics in individual studies. We only used crude rates, not adjusted for age or sex in the quantitative synthesis [25].
First, we did subgroup meta-analysis by sex and age to assess the variations in different populations. Age was classified into seven groups: 0–29, 30–39, 40–49, 50–59, 60–69, 70–79, and over 80 years. Second, the incidence and prevalence data were grouped by geographical region and economic level using the United Nations classification of economic regions (https://unstats.un.org/unsd/methodology/m49/, retrieved April 1, 2019), which is based on geographical proximity and economic similarities. If an author reported multiple time periods for a region, we used the most recent one. When data were reported for only one region within a subcontinent, the region was used to represent the subcontinent. Third, the temporal trends in prevalence and incidence were calculated by fitting a linear regression to the square root of the annual rates with the year as the predictor variable. Age-adjusted pooled rates were also utilized to analyze temporal trends. Age-adjusted pooled rates were calculated by direct standardization according to WHO 2000–2025 populations. We used the median year if the time points reported were longer than one year but no longer than 10 years [26] and the prevalence or incidence with time points longer than 10 years were excluded. The P values of coefficients from these regressions less than 0.05 were considered statistically significant. Furthermore, we also tried to estimate the survival time by formula: Survival time (years) = Prevalence/Incidence [27]. Meta-regression was performed to explore the sources of heterogeneity by including the proportion of males, subcontinent, proportion of population over 60 years old, study period and study quality. Heterogeneity between studies was assessed with Cochran’s Q, I2. The I2 statistic estimates the percentage of total variation across studies due to true between-study differences rather than chance. Generally, I2 values greater than 60–70% indicate the presence of substantial heterogeneity [28]. Funnel plots and Egger’s test were used to detect publication bias. Asymmetric funnel plots or P value less than 0.10 on Egger’s test implicates the existence of statistically significant publication bias [29]. This meta-analysis was done with metan, metareg, metafunnel and metabias packages in Stata 14.0.
Result
Characteristics of included studies
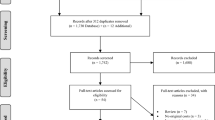
After removal of duplications and initial screening, 522 articles were eligible for full-text review (Fig. 1). On full-text review of 522 articles, 398 were excluded. Overall, 124 studies were eligible for final inclusion in the systematic review, including 110 studies of incidence and 58 studies of prevalence. Characteristics of the 110 incidence studies and 58 prevalence studies, including quality assessment, are available in Online Resource 4. Prevalence was reported for North Africa (one study), North America (13 studies), South America (three studies), Australia and New Zealand (one study), East Asia (five studies), South Asia (one study), West Asia (two studies), North Europe (13 studies), South Europe (17 studies), and West Europe (two studies). Of the 110 incidence studies, one study was in North Africa, 16 in North America, three in South America, one in Australia and New Zealand, eight in East Asia, one in South Asia, four in West Asia, 25 in North Europe, 43 in South Europe, six in West Europe, one in Caribbean, and one in Hawaii.
Sex and age distribution of prevalence and incidence
The crude worldwide ALS prevalence and incidence were 4.42 (95% CI 3.92–4.96) per 1,00,000 population and 1.59 (95% CI 1.39–1.81) per 1,00,000 person-years, respectively. Both ALS incidence and prevalence of males (incidence: 1.91, 95% CI 1.65–2.19; prevalence: 5.96, 95% CI 5.14–6.85) were higher than the corresponding values of females (incidence: 1.36, 95% CI 1.14–1.59; prevalence: 3.90, 95% CI 3.30–4.56). Figure 2 shows that prevalence and incidence increased by age before the age of 70–79, followed by a decrease.
Global variation of prevalence and incidence
The results by subcontinent and economic level are displayed in Fig. 3. Both prevalence and incidence were lowest in South Asia (prevalence: 1.57, 95% CI 0.17–4.39; incidence: 0.42, 95% CI 0.06–1.11) and highest in West Europe (prevalence: 9.62, 95% CI 4.80–16.10; incidence: 2.76, 95% CI 2.00–3.64). Divided by economic levels, developed regions were higher than developing regions in both prevalence (4.94, 95% CI 4.46–5.45 vs 1.79, 95% CI 1.58–2.02) and incidence 2.15, 95% CI 1.34–3.16 vs 0.63, 95% CI 0.44–0.85).
Temporal trend for prevalence and incidence
No significant temporal trend was detected for crude prevalence since 1959 (coefficient = − 0.000, t = − 0.00, P = 0.997) (Fig. 4). However, incidence was significantly rising by 0.00013 per year (t = 7.21, P < 0.001), and this upward trend was weakened after standardization with WHO 2000–2025 populations.
Survival time of ALS
The survival time of West Asia (9.23 years) and East Asia (4.32 years) was longer than other subcontinents. The European survival time ranged from 2.00 years in North Europe to 3.70 years in South Europe (Fig. 5).
Heterogeneity and publication bias
Heterogeneity was identified for proportion of population aged 60 or over (Table 1) and study period in univariate meta-regression. Using multivariate meta-regression, the heterogeneity for proportion of population aged 60 or over was still statistically significant. The funnel plot and the Egger test (Online Resource 5) suggested that no publication bias existed in the studies on prevalence, but might exist in studies on incidence.
Discussion
This meta-analysis summarizes available evidence on the worldwide ALS prevalence and incidence. There were several key findings: ALS prevalence varied from 1.57 per 1,00,000 population to 9.62 per 1,00,000 population and ALS incidence varied from 0.42 per 1,00,000 person-years to 2.76 per 1,00,000 person-years by subcontinent; both ALS prevalence and incidence were higher in developed regions and a slight positive temporal trend was found in crude incidence.
Compared with the meta-analysis of ALS incidence in 2017 [10], we included 66 more original studies by combining the rates using only the rates and corresponding standard errors. The sex and age variations in ALS prevalence and incidence were consistent with previous results [4, 10]. It is interesting to note that, consistent with previous studies, this study also observed a decline in ALS incidence in the older age group. This phenomenon was possibly due to the under-diagnosis of the oldest old, since the oldest old usually suffer from comorbidity, have difficulties in seeking for medical service and may die from other diseases before the diagnosis of ALS [30].
The worldwide ALS incidence in our study was similar to the result of the aforementioned meta-analysis [10], in which the incidence in European regions was higher and that in Asian regions was lower. Our study adopted the same regional classification standard as a previous meta-analysis [10], however, we first found that the prevalence in West Asia was much higher, only second to that of West Europe. This may be due to the fact that West Asia is adjacent to Europe and there may be an ancestry association between them [31]. The modern humans who replaced Neanderthals in Europe probably originated in West Asia, and the genetic distances between West Asia and Europe was also shorter than other Asian regions [32, 33]. Actually, a hexanucleotide GGGGCC repeat expansion in the C9orf72 gene (9p21.2 region) has been identified as the most frequent genetic cause of ALS in Caucasian population [34,35,36,37,38,39,40], which happens relatively rare in Asia and might be a potential explanation [40].Therefore, the prevalence of West Asia was higher than other Asian regions, similar to European regions. Furthermore, the overall incidence in our study was higher than that of the Global Burden of Disease (GBD 2016), possibly because of the inclusion of articles after 2015 in our study, which might reflect the rise of ALS incidence during recent years in another perspective.
In addition, we found that the developing regions enjoyed lower ALS prevalence and incidence in comparison to developed regions. This may be due to the fact that population aging was more severe in developed regions [41] and the ALS prevalence and incidence of elder population were higher.
Notably, crude incidence was found a statistically significant upward temporal trend, but the annual growth was small and the trend weakened by applying standardized incidence, which is consistent with the previous meta-analysis [10] and similar to the observation by Chio et al. [42], in which age effect was attenuated by standardization as well. In that study, the standardized incidence of women patients with ALS witnessed a statistically significant increase from 1995 to 2014 and the researchers explained the increase may be due to a birth cohort effect in women. We didn’t find similar results due to the limited number of studies reporting sex-age-specific incidence. But taken together, it implied that the temporal trend of crude incidence in our study might be caused by population aging [43]. As aforementioned, several studies including our study discovered that ALS incidence grew by age with a peak in 60–70 years old [4, 30]. The severity of population aging is increasing year by year, which might cause the rise of crude incidence. And we did find that age was significantly associated with the combined results in heterogeneity analysis for both prevalence and incidence. The upward temporal trend of incidence weakened after age standardization, and the P value even turned to be not significant. But we should note that the number of studies used to calculate the standardized incidence was reduced to 41 and the time range was reduced to 1970–2014 instead of the 1957–2014 in crude pooled incidence. It still cannot be excluded for the possibility of rising trend for crude incidence.
Surprisingly, the survival time in Asia regions were consistently longer than other regions. This finding was in accordance with Shahrizaila’s study published in 2016, [44] which has also reported that the median survival time in Japan, Taiwan and India was longer than that in western countries.
It is certain that the younger median age of onset in these population contributed to the longer survival in Asian and other non-European population, with age as the strongest factor influencing prognosis in ALS [45]. However, there are still other possible explanations for those differences. First, the disease progression of European patients with ALS might be faster, which might result in worse prognosis [46]. Dorst J. et al. [46] discovered that the proportion of patients with bulbar onset (patients with bulbar onset have a worse prognosis) was smaller in China than that in German, therefore the different proportions of disease subtypes might be responsible for the phenomenon as well. Second, the use of tracheostomy positive pressure ventilation (TPPV) might play a role in this phenomenon. More than 20% of ALS patients used TPPV in Japan and Taiwan [47, 48], however, the utility in West countries was fewer than 10% [48]. In addition, it implies that perhaps Asia residents may possess an inherent protective factor to ensure longer survival [44]. And the aforementioned C9orf72 gene (9p21.2 region) which is rare in Asian population was just statistically associated with inferior survival [40, 49, 50].
To the best of our knowledge, this paper is the latest meta-analysis that focuses on the ALS prevalence and incidence with 124 population-based studies included [26, 51]. Nevertheless, the findings of this study should be interpreted with caution. First, as in previous meta-analysis exploring the geographical variation [10, 30], the prevalence or incidence in certain subcontinents were represented by only few countries or regions due to lack of relevant original studies, possibly making the estimation biased. Second, the subgroups considered in this study may be not enough, limited by the number of original studies. Subgroups including occupations [52, 53], ethnicity [54] may also be meaningful to be explored. Third, publication bias in studies on incidence owing to small-sampled studies could affect the precision of our estimates [24]. This may be due to the fact that we only included the published studies.
Conclusion
This systematic review provides a comprehensive global overview of the incidence, prevalence, and survival time of ALS. The findings suggest that the ancestral origin of population in West Asia is awaited to be explored, because of its characteristics of low incidence but long survival time. Furthermore, developing regions may face increasing ALS prevalence and incidence in decades to come, since developing countries, like developed countries decades ago, are accelerating their economic development. The rising global burden of ALS requires more studies to identify plausible determinants for ALS and more innovations in health-care system to manage and control this costly disease.
References
Brown RH, Al-Chalabi A (2017) Amyotrophic lateral sclerosis. N Engl J Med 377:162–172. https://doi.org/10.1056/NEJMra1603471
Montuschi A, Iazzolino B, Calvo A, Moglia C, Lopiano L, Restagno G, Brunetti M, Ossola I, Lo Presti A, Cammarosano S, Canosa A, Chio A (2015) Cognitive correlates in amyotrophic lateral sclerosis: a population-based study in Italy. J Neurol Neurosurg Psychiatry 86:168–173. https://doi.org/10.1136/jnnp-2013-307223
Phukan J, Elamin M, Bede P, Jordan N, Gallagher L, Byrne S, Lynch C, Pender N, Hardiman O (2012) The syndrome of cognitive impairment in amyotrophic lateral sclerosis: a population-based study. J Neurol Neurosurg Psychiatry 83:102–108. https://doi.org/10.1136/jnnp-2011-300188
Logroscino G, Piccininni M, Marin B, Nichols E, Abd-Allah F, Abdelalim A, Alahdab F, Asgedom SW, Awasthi A, Chaiah Y, Daryani A, Do HP, Dubey M, Elbaz A, Eskandarieh S, Farhadi F, Farzadfar F, Fereshtehnejad S-M, Fernandes E, Filip I, Foreman KJ, Gebre AK, Gnedovskaya EV, Hamidi S, Hay SI, Irvani SSN, Ji JS, Kasaeian A, Kim YJ, Mantovani LG, Mashamba-Thompson TP, Mehndiratta MM, Mokdad AH, Nagel G, Nguyen TH, Nixon MR, Olagunju AT, Owolabi MO, Piradov MA, Qorbani M, Radfar A, Reiner RC, Sahraian MA, Sarvi S, Sharif M, Temsah O, Tran BX, Truong NT, Venketasubramanian N, Winkler AS, Yimer EM, Feigin VL, Vos T, Murray CJL (2018) Global, regional, and national burden of motor neuron diseases 1990–2016: a systematic analysis for the global burden of disease study 2016. Lancet Neurol 17:1083–1097. https://doi.org/10.1016/s1474-4422(18)30404-6
Baeza-Yates R, Sangal PM, Villoslada P (2017) Burden of neurological diseases in the US revealed by web searches. PLoS ONE 12:e0178019. https://doi.org/10.1371/journal.pone.0178019
Tysnes OB, Vollset SE, Aarli JA (1991) Epidemiology of amyotrophic lateral sclerosis in Hordaland county, western Norway. Acta Neurol Scand 83:280–285
Benjaminsen E, Alstadhaug KB, Baloch F, Gulsvik M, Odeh F (2017) Amyotrophic lateral sclerosis in Nordland County, Norway 2000–2015. Eur J Neurol 24:28
Tsai C-P, Wang K-C, Hwang C-S, Lee IT, Lee CT-C (2015) Incidence, prevalence, and medical expenditures of classical amyotrophic lateral sclerosis in Taiwan, 1999–2008. J Formos Med Assoc 114:612–619. https://doi.org/10.1016/j.jfma.2013.01.008
McKenzie JE, Beller EM, Forbes AB (2016) Introduction to systematic reviews and meta-analysis. Respirology 21:626–637. https://doi.org/10.1111/resp.12783
Marin B, Boumediene F, Logroscino G, Couratier P, Babron MC, Leutenegger AL, Copetti M, Preux PM, Beghi E (2017) Variation in worldwide incidence of amyotrophic lateral sclerosis: a meta-analysis. Int J Epidemiol 46:57–74. https://doi.org/10.1093/ije/dyw061
Chio A, Logroscino G, Traynor BJ, Collins J, Simeone JC, Goldstein LA, White LA (2013) Global epidemiology of amyotrophic lateral sclerosis: a systematic review of the published literature. Neuroepidemiology 41:118–130. https://doi.org/10.1159/000351153
D'Ovidio F, d'Errico A, Farina E, Calvo A, Costa G, Chio A (2016) Amyotrophic lateral sclerosis incidence and previous prescriptions of drugs for the nervous system. Neuroepidemiology 47:59–66. https://doi.org/10.1159/000448618
Moura MC, Casulari LA, Carvalho Garbi Novaes MR (2016) Ethnic and demographic incidence of amyotrophic lateral sclerosis (ALS) in Brazil: a population based study. Amyotroph Lateral Scler Frontotemporal Degener 17:275–281. https://doi.org/10.3109/21678421.2016.1140210
Scialo C, Novi G, Bandettini di Poggio M, Canosa A, Sormani MP, Mandich P, Origone P, Truffelli R, Mancardi GL, Caponnetto C (2016) Clinical epidemiology of amyotrophic lateral sclerosis in Liguria, Italy: an update of LIGALS register. Amyotroph Lateral Scler Frontotemporal Degener 17:535–542. https://doi.org/10.1080/21678421.2016.1197942
Tesauro M, Consonni M, Filippini T, Mazzini L, Pisano F, Chio A, Esposito A, Vinceti M (2017) Incidence of amyotrophic lateral sclerosis in the province of Novara, Italy, and possible role of environmental pollution. Amyotroph Lateral Scler Frontotemporal Degener 18:284–290. https://doi.org/10.1080/21678421.2017.1281961
Weil C, Zach N, Rishoni S, Shalev V, Chodick G (2016) Epidemiology of amyotrophic lateral sclerosis: a population-based study in Israel. Neuroepidemiology 47:76–81. https://doi.org/10.1159/000448921
Demetriou CA, Hadjivasiliou PM, Kleopa KA, Christou YP, Leonidou E, Kyriakides T, Zamba-Papanicolaou E (2017) Epidemiology of amyotrophic lateral sclerosis in the Republic of Cyprus: a 25-year retrospective study. Neuroepidemiology 48:79–85. https://doi.org/10.1159/000477126
Kahana E, Alter M, Feldman S (1976) Amyotrophic lateral sclerosis: a population study. J Neurol 212:205–213
Okumiya K, Wada T, Fujisawa M, Ishine M, Garcia Del Saz E, Hirata Y, Kuzuhara S, Kokubo Y, Seguchi H, Sakamoto R, Manuaba I, Watofa P, Rantetampang AL, Matsubayashi K (2014) Amyotrophic lateral sclerosis and parkinsonism in Papua, indonesia: 2001–2012 survey results. BMJ open 4:e004353. https://doi.org/10.1136/bmjopen-2013-004353
Boyle MH (1998) Guidelines for evaluating prevalence studies. Evid Based Ment Health 1:37–39
Loney PL, Chambers LW, Bennett KJ, Roberts JG, Stratford PW (1998) Critical appraisal of the health research literature: prevalence or incidence of a health problem. Chronic Dis Can 19:170–176
Fiest KM, Sauro KM, Wiebe S, Patten SB, Kwon CS, Dykeman J, Pringsheim T, Lorenzetti DL, Jette N (2017) Prevalence and incidence of epilepsy: a systematic review and meta-analysis of international studies. Neurology 88:296–303. https://doi.org/10.1212/wnl.0000000000003509
Noubiap JJ, Bigna JJ, Nansseu JR, Nyaga UF, Balti EV, Echouffo-Tcheugui JB, Kengne AP (2018) Prevalence of dyslipidaemia among adults in Africa: a systematic review and meta-analysis. Lancet Glob Health 6:e998–e1007. https://doi.org/10.1016/s2214-109x(18)30275-4
Noubiap JJ, Nansseu JR, Nyaga UF, Nkeck JR, Endomba FT, Kaze AD, Agbor VN, Bigna JJ (2019) Global prevalence of diabetes in active tuberculosis: a systematic review and meta-analysis of data from 2.3 million patients with tuberculosis. Lancet Glob Health 7:e448–e460. https://doi.org/10.1016/s2214-109x(18)30487-x
Etminan N, Chang HS, Hackenberg K, de Rooij NK, Vergouwen MDI, Rinkel GJE, Algra A (2019) Worldwide incidence of aneurysmal subarachnoid hemorrhage according to region, time period, blood pressure, and smoking prevalence in the population: a systematic review and meta-analysis. JAMA neurol 76:588–597. https://doi.org/10.1001/jamaneurol.2019.0006
Ng SC, Shi HY, Hamidi N, Underwood FE, Tang W, Benchimol EI, Panaccione R, Ghosh S, Wu JCY, Chan FKL, Sung JJY, Kaplan GG (2018) Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet 390:2769–2778. https://doi.org/10.1016/s0140-6736(17)32448-0
Freeman J, Hutchison GB (1980) Prevalence, incidence and duration. Am J Epidemiol 112:707–723. https://doi.org/10.1093/oxfordjournals.aje.a113043
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558. https://doi.org/10.1002/sim.1186
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315:629–634
Marin B, Fontana A, Arcuti S, Copetti M, Boumediene F, Couratier P, Beghi E, Preux PM, Logroscino G (2018) Age-specific ALS incidence: a dose-response meta-analysis. Eur J Epidemiol 33:621–634. https://doi.org/10.1007/s10654-018-0392-x
Taskent RO, Gokcumen O (2017) The Multiple histories of Western Asia: perspectives from ancient and modern genomes. Hum Biol 89:107–117
Anne MB, Judith RK, Joanna LM, Joan MH, Luciano C, Kenneth KK, Luca LC (1991) Drift, admixture, and selection in human evolution: a study with DNA polymorphisms. Proc Nati Acad Sci 88:839–843
Luca LCS (1997) Genes, peoples, and languages. Proc Natl Acad Sci 94:7719–7724
van Es MA, Veldink JH, Saris CG, Blauw HM, van Vught PW, Birve A, Lemmens R, Schelhaas HJ, Groen EJ, Huisman MH, van der Kooi AJ, de Visser M, Dahlberg C, Estrada K, Rivadeneira F, Hofman A, Zwarts MJ, van Doormaal PT, Rujescu D, Strengman E, Giegling I, Muglia P, Tomik B, Slowik A, Uitterlinden AG, Hendrich C, Waibel S, Meyer T, Ludolph AC, Glass JD, Purcell S, Cichon S, Nothen MM, Wichmann HE, Schreiber S, Vermeulen SH, Kiemeney LA, Wokke JH, Cronin S, McLaughlin RL, Hardiman O, Fumoto K, Pasterkamp RJ, Meininger V, Melki J, Leigh PN, Shaw CE, Landers JE, Al-Chalabi A, Brown RH Jr, Robberecht W, Andersen PM, Ophoff RA, van den Berg LH (2009) Genome-wide association study identifies 19p13.3 (UNC13A) and 9p21.2 as susceptibility loci for sporadic amyotrophic lateral sclerosis. Nat Genet 41:1083–1087. https://doi.org/10.1038/ng.442
Diekstra FP, Saris CG, van Rheenen W, Franke L, Jansen RC, van Es MA, van Vught PW, Blauw HM, Groen EJ, Horvath S, Estrada K, Rivadeneira F, Hofman A, Uitterlinden AG, Robberecht W, Andersen PM, Melki J, Meininger V, Hardiman O, Landers JE, Brown RH Jr, Shatunov A, Shaw CE, Leigh PN, Al-Chalabi A, Ophoff RA, van den Berg LH, Veldink JH (2012) Mapping of gene expression reveals CYP27A1 as a susceptibility gene for sporadic ALS. PLoS ONE 7:e35333. https://doi.org/10.1371/journal.pone.0035333
Uyan O, Omur O, Agim ZS, Ozoguz A, Li H, Parman Y, Deymeer F, Oflazer P, Koc F, Tan E, Ozcelik H, Basak AN (2013) Genome-wide copy number variation in sporadic amyotrophic lateral sclerosis in the Turkish population: deletion of EPHA3 is a possible protective factor. PLoS ONE 8:e72381. https://doi.org/10.1371/journal.pone.0072381
Fogh I, Ratti A, Gellera C, Lin K, Tiloca C, Moskvina V, Corrado L, Soraru G, Cereda C, Corti S, Gentilini D, Calini D, Castellotti B, Mazzini L, Querin G, Gagliardi S, Del Bo R, Conforti FL, Siciliano G, Inghilleri M, Sacca F, Bongioanni P, Penco S, Corbo M, Sorbi S, Filosto M, Ferlini A, Di Blasio AM, Signorini S, Shatunov A, Jones A, Shaw PJ, Morrison KE, Farmer AE, Van Damme P, Robberecht W, Chio A, Traynor BJ, Sendtner M, Melki J, Meininger V, Hardiman O, Andersen PM, Leigh NP, Glass JD, Overste D, Diekstra FP, Veldink JH, van Es MA, Shaw CE, Weale ME, Lewis CM, Williams J, Brown RH, Landers JE, Ticozzi N, Ceroni M, Pegoraro E, Comi GP, D'Alfonso S, van den Berg LH, Taroni F, Al-Chalabi A, Powell J, Silani V (2014) A genome-wide association meta-analysis identifies a novel locus at 17q11.2 associated with sporadic amyotrophic lateral sclerosis. Hum Mol Genet 23:2220–2231. https://doi.org/10.1093/hmg/ddt587
Du Y, Wen Y, Guo X, Hao J, Wang W, He A, Fan Q, Li P, Liu L, Liang X, Zhang F (2018) A Genome-wide expression association analysis identifies genes and pathways associated with amyotrophic lateral sclerosis. Cell Mol Neurobiol 38:635–639. https://doi.org/10.1007/s10571-017-0512-2
Shatunov A, Mok K, Newhouse S, Weale ME, Smith B, Vance C, Johnson L, Veldink JH, van Es MA, van den Berg LH, Robberecht W, Van Damme P, Hardiman O, Farmer AE, Lewis CM, Butler AW, Abel O, Andersen PM, Fogh I, Silani V, Chio A, Traynor BJ, Melki J, Meininger V, Landers JE, McGuffin P, Glass JD, Pall H, Leigh PN, Hardy J, Brown RH Jr, Powell JF, Orrell RW, Morrison KE, Shaw PJ, Shaw CE, Al-Chalabi A (2010) Chromosome 9p21 in sporadic amyotrophic lateral sclerosis in the UK and seven other countries: a genome-wide association study. Lancet Neurol 9:986–994. https://doi.org/10.1016/s1474-4422(10)70197-6
Balendra R, Isaacs AM (2018) C9orf72-mediated ALS and FTD: multiple pathways to disease. Nat Rev Neurol 14:544–558. https://doi.org/10.1038/s41582-018-0047-2
Lloyd-Sherlock P (1982) (2000) Population ageing in developed and developing regions: implications for health policy. Soc Sci Med 51:887–895
A Chio G Mora C Moglia U Manera A Canosa S Cammarosano A Ilardi D Bertuzzo E Bersano P Cugnasco M Grassano F Pisano L Mazzini A Calvo VD Piemonte Register for ALS 2017 Secular trends of amyotrophic lateral sclerosis: the piemonte and valle d'aosta register, JAMA Neurol 74: 1097–1104 10.1001/jamaneurol.2017.1387
Arthur KC, Calvo A, Price TR, Geiger JT, Chio A, Traynor BJ (2016) Projected increase in amyotrophic lateral sclerosis from 2015 to 2040. Nat Commun 7:12408. https://doi.org/10.1038/ncomms12408
Shahrizaila N, Sobue G, Kuwabara S, Kim SH, Birks C, Fan DS, Bae JS, Hu CJ, Gourie-Devi M, Noto Y, Shibuya K, Goh KJ, Kaji R, Tsai CP, Cui L, Talman P, Henderson RD, Vucic S, Kiernan MC (2016) Amyotrophic lateral sclerosis and motor neuron syndromes in Asia. J Neurol Neurosurg Psychiatry 87:821–830. https://doi.org/10.1136/jnnp-2015-312751
Chen L, Zhang B, Chen R, Tang L, Liu R, Yang Y, Yang Y, Liu X, Ye S, Zhan S, Fan D (2015) Natural history and clinical features of sporadic amyotrophic lateral sclerosis in China. J Neurol Neurosurg Psychiatry 86:1075–1081. https://doi.org/10.1136/jnnp-2015-310471
Dorst J, Chen L, Rosenbohm A, Dreyhaupt J, Hubers A, Schuster J, Weishaupt JH, Kassubek J, Gess B, Meyer T, Weyen U, Hermann A, Winkler J, Grehl T, Hagenacker T, Lingor P, Koch JC, Sperfeld A, Petri S, Grosskreutz J, Metelmann M, Wolf J, Winkler AS, Klopstock T, Boentert M, Johannesen S, Storch A, Schrank B, Zeller D, Liu XL, Tang L, Fan DS, Ludolph AC (2019) Prognostic factors in ALS: a comparison between Germany and China. J Neurol 266:1516–1525. https://doi.org/10.1007/s00415-019-09290-4
Lee CT, Chiu YW, Wang KC, Hwang CS, Lin KH, Lee IT, Tsai CP (2013) Riluzole and prognostic factors in amyotrophic lateral sclerosis long-term and short-term survival: a population-based study of 1149 cases in Taiwan. J Epidemiol 23:35–40. https://doi.org/10.2188/jea.je20120119
Rabkin J, Ogino M, Goetz R, McElhiney M, Marziliano A, Imai T, Atsuta N, Morita M, Tateishi T, Matsumura T, Mitsumoto H (2013) Tracheostomy with invasive ventilation for ALS patients: neurologists' roles in the US and Japan. Amyotroph Lateral Scler Frontotemporal Degener 14:116–123. https://doi.org/10.3109/17482968.2012.726226
Floeter MK, Traynor BJ, Farren J, Braun LE, Tierney M, Wiggs EA, Wu T (2017) Disease progression in C9orf72 mutation carriers. Neurology 89:234–241. https://doi.org/10.1212/wnl.0000000000004115
Trojsi F, Siciliano M, Femiano C, Santangelo G, Lunetta C, Calvo A, Moglia C, Marinou K, Ticozzi N, Ferro C, Scialo C, Soraru G, Conte A, Falzone YM, Tortelli R, Russo M, Sansone VA, Chio A, Mora G, Silani V, Volanti P, Caponnetto C, Querin G, Sabatelli M, Riva N, Logroscino G, Messina S, Fasano A, Monsurro MR, Tedeschi G, Mandrioli J (2019) Comparative analysis of C9orf72 and sporadic disease in a large multicenter als population: the effect of male sex on survival of C9orf72 positive patients. Front Neurosci 13:485. https://doi.org/10.3389/fnins.2019.00485
Al-Chalabi A, Hardiman O (2013) The epidemiology of ALS: a conspiracy of genes, environment and time. Nat Rev Neurol 9:617–628. https://doi.org/10.1038/nrneurol.2013.203
Korner S, Kammeyer J, Zapf A, Kuzma-Kozakiewicz M, Piotrkiewicz M, Kuraszkiewicz B, Goszczynska H, Gromicho M, Grosskreutz J, Andersen PM, de Carvalho M, Petri S (2019) Influence of environment and lifestyle on incidence and progress of amyotrophic lateral sclerosis in A German ALS population. Aging Dis 10:205–216. https://doi.org/10.14336/ad.2018.0327
Beard JD, Steege AL, Ju J, Lu J, Luckhaupt SE, Schubauer-Berigan MK (2017) Mortality from amyotrophic lateral sclerosis and parkinson's disease among different occupation groups—United States, 1985–2011. MMWR Morb Mortal Wkly Rep 66:718–722. https://doi.org/10.15585/mmwr.mm6627a2
Mehta P, Kaye W, Raymond J, Punjani R, Larson T, Cohen J, Muravov O, Horton K (2018) Prevalence of amyotrophic lateral sclerosis—United States, 2015. MMWR Morb Mortal Wkly Rep 67:1285–1289. https://doi.org/10.15585/mmwr.mm6746a1
Acknowledgements
The authors would like to thank Xuefeng Lai, Jinlang Lv, Junyou Yu, Wei Zhang, and Huan Yu for retrieving and managing articles as well as extracting data.
Funding
This study was funded by the National Natural Science Foundation (grant number 91646107).
The manuscript does not contain clinical studies or patient data.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xu, L., Liu, T., Liu, L. et al. Global variation in prevalence and incidence of amyotrophic lateral sclerosis: a systematic review and meta-analysis. J Neurol 267, 944–953 (2020). https://doi.org/10.1007/s00415-019-09652-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-019-09652-y