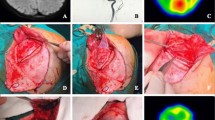
Abstract
Background
Despite the consensus on the necessity of revascularizing surgery in Moyamoya angiopathy in Asia, the indication in Caucasian Moyamoya patients is discussed controversially.
Objective
The safety of revascularizing surgery in Europe should be clarified.
Methods
This study retrospectively analyzed the rate of complications as well as clinical symptoms within the first 3 months after bypass surgery between superficial temporal artery and middle cerebral artery (STA–MCA).
Results
64 direct bypass procedures in 45 patients (95.5% Caucasians) were analyzed. The magnetic resonance imaging at day 6 showed subdural hematoma in 60.3%. The mean diameter of these hematomas on magnetic resonance imaging was 5.1 mm (SD 3.4 mm) and increased in 25% at follow-up. No difference was found between those patients with early (day 1) or late (day 7) restarts of antiplatelet therapy. Magnetic resonance imaging at day 6 revealed hyperperfusion syndrome after six of 64 procedures (9.3%). Three of these six had clinical symptoms; two-thirds were transient within seconds. Magnetic resonance imaging depicted stroke after seven procedures (10.9%). Five of these seven patients had no new symptoms. Altogether, after ten procedures (15%), patients complained about clinical symptoms. These were all transient. No new transient ischemic attacks occurred during the 3 month follow-up and no new lesions were detected in magnetic resonance imaging. Only two patients underwent surgery for asymptomatic subdural hematoma. All other subdural hematomas resolved spontaneously.
Conclusion
Revasculating surgery is a safe procedure in Caucasian patients with Moyamoya angiopathy. The observed complications have a good prognosis.
Similar content being viewed by others

Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Moyamoya angiopathy (MMA) is a non-arteriosclerotic vasculopathy characterized by progressive stenoses and occlusions of the intracranial internal carotid arteries (ICA) and proximal parts of middle (MCA) and anterior cerebral arteries (ACA) on both sides. Due to hemodynamic insufficiency, children and young adults with MMA have a high risk for transient ischemic attacks (TIA) and stroke. In addition to hemodynamic stroke, thrombotic material may also arise locally at the stenoses due to hemodynamic insufficiency and a reduced washout. Moreover, the extensive “Moyamoya” collaterals are fragile and bear a high risk for cerebral hemorrhage. The direct bypass surgery (EC/IC bypass) is mainly characterized by creating an anastomosis between the extracranial superficial temporal artery (STA) and a distal branch of the MCA (STA–MCA–bypass). The goal is to achieve a rapid restoration of cerebral perfusion in adult and pediatric patients with MMA and to reduce the risk of future ischemic stroke [1]. It is also intended to decrease the risk of cerebral hemorrhage by diminution of the fragile collateral networks. Indirect bypass techniques include the implantation of highly vascularized tissue like muscle or omentum to the brain surface. Indirect bypass surgery is mainly performed in young children when the donor artery STA is too small for direct bypass techniques.
While it is known since 1985 that extracranial–intracranial revascularizing surgery is ineffective in arteriosclerotic diseases [2,3,4], there is a common consensus among Asian experts that bypass surgery is beneficial in hemodynamic compromised stages of MMA [5,6,7,8,9].
As a first attempt, the French guidelines published just recently, aimed to clarify clinical practice of MMA in Europe based on literature analysis [10]. However, outside of Asia, study data for the indication of bypass surgery are sparse [11, 12]. Therefore, it is important to analyze the risks of direct bypass surgery. Asian studies [13] have reported hyperperfusion syndrome [14, 15], postoperative ischemia [16], and seizures [17] as the main complications after bypass surgery. Postoperative subdural hemorrhages following bypass surgery are nearly completely neglected in the scientific literature [13, 18].
In this study, we aimed to characterize the development of subdural hematomas in the postoperative time course and investigate the rates of spontaneous restitution and the percentage of patients requiring neurosurgical interventions. In addition, we attempted to elucidate if the timepoint of restarting antiplatelet therapy after bypass surgery has an influence on the risk, extent, and prognosis of postoperative subdural hematoma.
Objective
This study aimed to analyze safety of STA–MCA–bypass in Europe in consecutive patients of dominantly Caucasian family origin.
Specifically, we sought to analyze the early postoperative course after STA–MCA–bypass surgery to address the following questions: is there a difference in bleeding complications with early re-initiation of aspirin or clopidogrel? On the other hand, is the incidence of stroke increased in patients with longer intermission of antiplatelet therapy? Which patients have clinical symptoms early after surgery? How is the prognosis of complications within 3 months after surgery?
Methods
We retrospectively analyzed the rate, extent, and development of subdural hematoma, hyperperfusion syndrome, stroke in magnetic resonance imaging (MRI) and clinical symptoms early within the first 3 months after STA–MCA bypass surgery performed by one neurosurgeon (FD) in a single German institution. Between January 1st, 2015 and March 9th, 2017, all 45 consecutive patients with MMA and hemodynamic insufficiency underwent 64 STA–MCA bypass surgical interventions due to hemodynamic insufficiency. All patients routinely underwent cranial computer tomography on the first day after surgery and MRI of the brain at day 6 after surgery. The MRI protocol included axial fluid-attenuated inversion recovery (FLAIR) sequences, axial T1-weighted, axial diffusion-weighted, and susceptibility-weighted images as well as intracranial time-of-flight (TOF) MR angiography. In addition, a clinical follow-up with another MRI of the brain was performed about 3 months after bypass surgery. In cases with subdural hematoma detected on the routine computer tomography (CT) scan or MRI, the extent was measured in the position of the largest diameter. In all patients, antiplatelet therapy was discontinued 7 days before bypass surgery. Due to the assumption that early restart of antiplatelet therapy after bypass surgery influences the rate of hematoma, we changed the clinical concept between 2015 and 2016 towards longer suspension of antiplatelet therapy. Therefore, retrospectively, it was possible to divide the 64 procedures into two groups. Group 1 resumed their antiplatelet therapy between days 1–3 after surgery. Group 2 resumed their antiplatelet therapy between days 5–7.
In case of bilateral surgery, the second hemisphere was operated after a 3 month waiting period. No patient was operated bilateral simultaneously.
The study was retrospective and the decisions for all procedures and therapy were based on clinical reasoning and not influenced by the study. The study was approved by the local ethics-committee. All patients signed an informed consent. Statistical comparisons were made using Mann–Whitney U test (Wilcoxon test). Statistical analysis was performed with SPSS 20 (IBM, Armonk, NY). p values < 0.05 were considered significant.
Results
Participants
Sixty-four procedures were operated using the STA as donor vessel and a distal branch of the MCA as acceptor vessel in 45 patients. In 48 (75%) procedures, only this direct bypass surgery was performed; in 16 (25%), procedures direct surgery was combined with indirect bypass techniques. The participants were 11 males and 34 females. All participants were adults due to referring bias to our adult Moyamoya center. The age at time of surgery was 41.8 years (age range 18–64, mean, median 43 years, SD 13.68). The great majority (95.5%) of patients were of Caucasian ethnicity (42 Germans, 1 Russian). Only one patient was Arab and one patient was of Asian ethnical origin. The great majority of patients had bilateral MMA (n = 40, 88.8%), and only five patients were suffering from a unilateral variant.
Postoperative rate and extent of subdural hemorrhage
Within 24 h of surgery, CT scans revealed subdural hematoma in 29 of 64 procedures (45.3%). All subdural hematomas were located at the ipsilateral side to surgery. The mean diameter of these hematomas was 3.9 mm (median 3, SD 2.1, minimum 1 mm, and maximum 10 mm). In eight procedures, a follow-up CT scan was available between the first postoperative day and the MR scan at day 6. In those eight procedures (in 5 cases), the hematoma increased with a mean of 7.6 mm (median 7, SD 4.4, minimum 2 mm, and maximum 12 mm). The MR scan, which was performed at day 6 after surgery (mean 6.01, median six, and SD 4.19), showed subdural hematoma in 38 of 63 procedures (60.3%). The mean diameter of these hematomas in MRI was 5.1 mm (median 4.5 mm and SD 3.4 mm). In 26 of 63 (25.4%) MR scans, there was an increase of the diameter in comparison with the first CT scan at day 1 after surgery with a mean difference of 4.2 mm (median 3 mm and SD 2.9 mm). In 27 MRI scans, the diameter remained stable, and 10 MRI scans revealed an reduced diameter of hematomas with a mean difference of 2.9 mm (median 2.5 mm and SD 2.1). In two patients, an operative decompression was performed (diameter 15 mm in both cases) based on the MRI finding although patients had been asymptomatic. No patient showed epidural hemorrhage.
Antiplatelet therapy
Antiplatelet therapy (aspirin 100 mg per day or clopidogrel 75 mg per day) was prescribed in 63 of 64 procedures before surgery and was stopped 7 days before surgery. This therapy was re-initiated at the first postoperative day after 33 procedures. In 30 procedures (47.6%), the antiplatelet therapy was given at day 5.2 (mean, median 7, SD 2.2) after surgery. In those 33 procedures in which antiplatelet therapy was restarted early at day 1 after surgery, 21 (63.6%) MRI scans showed subdural hematoma. In those 22 procedures, in which antiplatelet therapy was restarted at day 4 or later after surgery (mean day 6.52, median day 7, and SD 1.08) 12 MR scans revealed subdural hematomas. This difference was not significant (p = 0.455). In the 15 patients with re-initiation at day 7 8 (53.3%) MRI scans showed subdural hematoma. The comparison between the 15 procedures who restarted antiplatelet therapy at day 7 and those 33 procedures who were restarted on antiplatelet therapy at day 1 also revealed no statistically significant difference (p = 0.45) (Fig. 1).
Early hyperperfusion syndrome in MRI at day 6 after surgery
In the MR scans performed at day 6 FLAIR hyperintensity and diffusion restriction suggesting hyperperfusion were evident after 6 of 64 procedures (9.3%). Three of six (50%) patients had clinical symptoms. Two of these were transient for hours very early after surgery and completely disappeared at day 2 (see Table 1). In all six cases, MRI performed 3 months later showed no abnormality with complete resolution of the hyperintensities.
The six patients with hyperperfusion syndrome did not differ in age (45.33 years mean, median 38 years, SD 18.01) compared to the other 39 patients (40.92 years mean, median 43, SD 13.23; p = 0.725).
Diffusion-weighted images in MRI at day 6 after surgery
MRI imaging revealed diffusion restriction with typical reduction of ADC map as a sign of ischemia after seven procedures (10.9%) (Fig. 2). Five of these seven patients had no new postoperative symptoms. As shown in Table 1, one patient had paresthesia for seconds, which completely resolved, and one patient had symptoms not related to the area of stroke. All diffusion-restricted areas appeared as little spots suggesting microembolic lesions. In four of these seven patients these diffusion-restricted spots were located in the same hemisphere as surgery, in two patients the other hemisphere was affected and in one patient spots were found in both hemispheres. Six of seven patients were older than 40 years, with a mean age of 46.85 years (median 46.85, SD 1.99). The age of those 38 patient without ischemia was 40.86 (mean, median 39.5, SD 13.91). This difference was not significant (t test, p = 0.266). Among the group of 7 patients with stroke visible on MRI at day 6, 5 had received antiplatelet therapy early at day 1, and at days 2 and 7, one each. None of the stroke patients smoked in the first 6 days after surgery or had a coagulation disorder.
Clinical symptoms
Almost all patients suffered from wound pain and headaches during the first days after surgery, which responded to analgesic medication. Therefore, pain was not analyzed as all patients received analgetic drugs on a regular basis. In addition, postoperative soft tissue hematoma and swelling was not analyzed as it was present in all patients and it is hard to quantify.
Altogether, after ten procedures (15%) patients complained of clinical symptoms other than postoperative pain and headaches.
After five procedures (8%) patients reported sensory symptoms in the form of paresthesiae of the fingers and the mouth for seconds during the early days after surgery. These symptoms and the other postoperative complaints are presented in Table 1. After those ten procedures with clinical postoperative complaints, the first CT revealed subdural bleedings in 5 (50%; mean diameter 2.4, median 0.5, SD 2.9), and MRI at day 6 revealed subdural hematoma in additional two (7 of 10, 70%) procedures (mean diameter 4.5, median 4.5, standard 4.7). After those 54 procedures in which patients presented no postoperative complaints, 24 (44.4%) early CT scans revealed postoperative hematoma (mean diameter 3.8 mm, median 3 mm, SD 2.07) and 30 (55.5%) MRI scans showed postoperative subdural hematoma (mean diameter 5 mm, median 4 mm, SD 3.17). The percentage of bleedings in CT (p = 7.46) and in MRI (p = 0.396) and the diameter of subdural hematoma (p = 0.07 for CT, p = 0.67 for MRI) did not differ between those patients with or without symptoms.
Early postoperative duplex sonography
After all 64 procedures in all 45 patients extra- and transcranial duplex sonography within the first 6 days revealed bypass patency (100%).
MRI 3 months after surgery
In 63 of 64 procedures, MRI and clinical follow-up were available at 3–4 months after surgery. No new diffusion restrictions occurred during follow-up. Neither ischemias nor bleedings were seen in MRI and all early postoperative findings like subdural hematoma and hyperperfusion syndrome resolved completely (Figs. 3, 4). This included the 36 cases, which showed a subdural hemorrhage on MRI at day 6, and which did not undergo surgical decompression. The spontaneous resolution in all these 36 non-operated cases included also 5 cases who had presented a diameter of hematoma of more than 9 mm (15, 14, 11, 10, and 9 mm, respectively).
Clinical symptoms 3 months after surgery
In 62 of 63 procedures (98.4%), patients reported no persisting clinical deficit after surgery within 3 months. One patient was lost to clinical follow-up due to living in Kuwait. After one procedure (1.5%), a persisting worsening of aphasia was reported without a new MRI lesion in comparison with the preoperative MRI. This worsening was interpreted as a postictal phenomenon due to a prior generalized seizure and residual aphasia (see Table 1). After three procedures (4.6%), patients reported wound healing complications. These included wound dehiscence and incomplete necrosis, but resolved with appropriate treatment at the 3 month follow-up.
Duplex sonography 3 months after surgery
Extra- and intracranial duplex sonography 3 months after surgery revealed bypass patency in all 44 patients after a total of 63 procedures (100%).
Discussion
This is the first study addressing systematically clinical symptoms, development of subdural hematomas and ischemias in mainly Caucasian European patients with MMA within the first 3 months after STA–MCA surgery. Yu et al. reported in 2016 that no systematic study on complications after bypass surgery is available and summarized the complications in a review article [13].
The literature about revascularisation surgery especially in European Caucasians is sparse and clinically relevant questions are still unsolved.
Recently, the Berlin group addressed perioperative risks of 37 patients with MMA and found eight ischemic complications (11%) [19]. Other studies from European neurosurgical groups focused on angiographic patency, hemodynamic outcomes or had a mainly neurosurgical focus [20,21,22,23,24,25,26,27]. Other experiences from France, Italy, and Germany are mainly based on literature review [10], case series, or small cohorts [28,29,30,31].
North American studies addressing the perioperative outcome are based on more ethnically mixed cohorts than typically found in Europe and are, therefore, difficult to compare [32, 33].
Despite the reported low risk, European neurologists are still often reluctant to indicate bypass surgery presumably out of fear of a complicated postoperative course or a low benefit.
Thus, this study was intended to address open questions from a clinical neurologist’s point of view. Important points were the analysis of the effect of antiplatelet drugs on the risk of hematoma and to elucidate if seizures and other clinical symptoms are related to hyperperfusion syndrome or bleedings.
For the first time, we demonstrated a high incidence of subdural hematoma (60.3%) early after STA–MCA bypass surgery.
Almost all of these subdural hematomas resolved spontaneously. It is remarkable that also subdural hematomas larger than 9 mm in diameter disappeared during the 3 month follow-up completely. Patients with or without symptoms did not differ in percentage and diameter of subdural hemorrhage. Even patients with extended hemorrhages remained free from symptoms. There was no significant difference concerning percentage and diameter of subdural hematoma between patients with or without early restart of aspirin or clopidogrel. However, we have to acknowledge that possibly, the size of the cohort was too small to rule out an effect of antiplatelet therapy on subdural hematoma. The review by Yu et al. only briefs comments on subdural hematoma [13]. Andoh et al. reported three patients who postoperatively developed chronic subdural hematoma. It was suspected that a preexisting brain atrophy might play a role [18]. In our experience, ipsilateral subdural hematoma is a common (60.4%) but benign complication of bypass surgery.
Based on our findings, we suggest deciding if antiplatelet therapy should be restarted based on an early CT scan performed at day 1 after surgery. The question if the risk of hematoma outweighs the benefit of preventing early embolic strokes when considering an early restart of antiplatelet therapy is not an easy one. Due to the small number of diffusion-restricted spots, the benefit of stroke prevention should be interpreted with caution. However, from a pathophysiological point of view, we believe that longer discontinuation of antiplatelet therapy could increase the risk of embolic stroke in MMA. At the same time, our data suggest that an early resumption of antiplatelet therapy does not seem to increase the risk of clinically relevant complications due to subdural hematoma. This is supported by a study from Schubert et al. with 158 patients and 168 operated hemispheres demonstrating that antiplatelet therapy was not associated with an elevated risk of hemorrhage or need for revision surgery [34]. However, the data do not allow predicting which patient will develop stroke. Age was not a significant discriminator, although six of seven patients were older than 40 years. Kazumata et al. addressed the question of postoperative ischemia in 2014. They evaluated 236 patients and 358 operations. Postoperative ischemia was detected in 4.1% of patients. The risk was higher in adult patients compared to pediatric patients (7.9% per surgery) [16].
Hyperperfusion syndrome following direct bypass surgery has frequently been described in Asian patients [14]. The sudden increase in cerebral blood flow combined with impaired cerebrovascular autoregulation may be the pathomechanism underlying this phenomenon. Incidences between 18 and 50% were described in the Asian literature [35]. It is recommended to lower blood pressure as a preventative measure [13].
Interestingly, our cases diagnosed with hyperperfusion syndrome on MRI, did not differ clinically from patients without these MRI findings.
Only few studies have described clinical symptoms as complication of bypass surgery. With regard to postoperative seizures, Jin et al. analyzed 43 patients with 53 operations, including both indirect and direct bypass procedures [17]. They found postoperative seizures in 10 of 53 hemispheres (18.9%) [17]. A Japanese study by Narisawa et al. analyzed postoperative and suspected a link between seizures and increase in CBF. Forty-four patients with 64 operated hemispheres were included in the study of whom three presented with postoperative seizures within 1–10 days after surgery [36]. The clinical question whether transient paresthesia periorally and in the hands without motor abnormalities represent focal seizures or should be summarized as a “cheiro-oral syndrome” has not been discussed in depth. Sasamori et al. found in 8 of 35 operated hemispheres (22.9%) such transient paresthesia and interpreted this as a transient “cheiro-oral syndrome”. Whether this phenomenon should be interpreted as a focal Jacksonian seizure is relevant, as this would result in a driving ban, in Germany for at least 1 year. On the other hand, we have not considered these transient sensory symptoms as an indication for antiepileptic drug treatment in most cases.
Only one patient reported persistent aphasia accentuated after surgery. However, at the 3 month clinical follow-up, 3 T MRI revealed no new structural lesions. Therefore, we interpreted the accentuation of the aphasia as a possible postictal phenomenon.
Wound healing complications occurred in 3 of 64 procedures (4.6%) compared to a frequency of 21.4% reported previously in the literature [37]. A reason for this good result may be that an one STA—instead of double-type-STA procedure was used necessitating only a small burr hole.
In conclusion, our study provides a detailed description of the early postoperative complications in dominantly Caucasian (95.5%) patients with MMA after revascularizing surgery in a single German institution. Clinically relevant complications were extremely rare with 98% wellbeing 3 months after surgery. Despite the excellent prognosis, this study elucidates that subdural hematoma is a common radiological finding after surgery (60.3%). Interestingly, subdural hematomas, hyperperfusion syndrome, and ischemias in general were subclinical and only evident in high-resolution MRI. The good spontaneous prognosis of postoperative subdural hematomas and other complications should not hide the fact that preoperative patient information should include the small but evident risk of serious complications.
Clearly, an interdisciplinary approach is advised and referral of Moyamoya patients to specialized centers is recommended as it is a rare disease in Europe [10].
To achieve a better data basis for indications to bypass surgery, future studies have to clarify the long-term prognosis regarding new strokes and hemorrhages in Caucasian Europeans.
Conclusion
This study shows excellent prognosis after 3 months after surgery. The indication for revascularization surgery should be based on hemodynamic insufficiency.
References
Kraemer M, Heienbrok W, Berlit P (2008) Moyamoya disease in Europeans. Stroke 39(12):3193–3200
EC/IC Bypass Study Group (1985) Failure of extracranial-intracranial arterial bypass to reduce the risk of ischemic stroke. Results of an international randomized trial. The EC/IC Bypass Study Group. N Engl J Med 313(19):1191–1200
Powers WJ, Clarke WR, Grubb RL Jr et al (2011) Extracranial-intracranial bypass surgery for stroke prevention in hemodynamic cerebral ischemia: the Carotid Occlusion Surgery Study randomized trial. Jama 306(18):1983–1992
Powers WJ, Grubb RL Jr, Raichle ME (1989) Clinical results of extracranial-intracranial bypass surgery in patients with hemodynamic cerebrovascular disease. J Neurosurg 70(1):61–67
Miyamoto S, Yoshimoto T, Hashimoto N et al (2014) Effects of extracranial-intracranial bypass for patients with hemorrhagic moyamoya disease: results of the Japan Adult Moyamoya Trial. Stroke 45(5):1415–1421
Research Committee on the Pathology and Treatment of Spontaneous Occlusion of the Circle of Willis; Health Labour Sciences Research Grant for Research on Measures for Infractable Diseases (2012) Guidelines for diagnosis and treatment of moyamoya disease (spontaneous occlusion of the circle of Willis). Neurol Med Chir (Tokyo) 52(5):245–266
Jeon JP, Kim JE, Cho WS, Bang JS, Son YJ, Oh CW (2018) Meta-analysis of the surgical outcomes of symptomatic moyamoya disease in adults. J Neurosurg 128(3):793–799
Yao Z, You C (2018) Effect of surgery on the long-term functional outcome of moyamoya disease: a meta-analysis. Turk Neurosurg. https://doi.org/10.5137/1019-5149.JTN.22598-18.3
Qian C, Yu X, Li J, Chen J, Wang L, Chen G (2015) The efficacy of surgical treatment for the secondary prevention of stroke in symptomatic moyamoya disease: a meta-analysis. Medicine 94(49):e2218
Herve D, Kossorotoff M, Bresson D et al (2018) French clinical practice guidelines for Moyamoya angiopathy. Rev Neurol (Paris) 174(5):292–303
Hanggi D, Steiger HJ, Vajkoczy P (2014) The role of MCA-STA bypass surgery after COSS and JET: the European point of view. Acta Neurochir Suppl 119:77–78
Hanggi D, Steiger HJ, Vajkoczy P (2012) Cerebrovascular Section of the European Association of Neurological Surgeons. EC-IC bypass for stroke: is there a future perspective? Acta Neurochir (Wien) 154(10):1943–1944
Yu J, Shi L, Guo Y, Xu B, Xu K (2016) Progress on complications of direct bypass for Moyamoya disease. Int J Med Sci 13(8):578–587
Fujimura M, Tominaga T (2012) Lessons learned from moyamoya disease: outcome of direct/indirect revascularization surgery for 150 affected hemispheres. Neurol Med Chir (Tokyo) 52(5):327–332
Fujimura M, Mugikura S, Kaneta T, Shimizu H, Tominaga T (2009) Incidence and risk factors for symptomatic cerebral hyperperfusion after superficial temporal artery-middle cerebral artery anastomosis in patients with moyamoya disease. Surg Neurol 71(4):442–447
Kazumata K, Ito M, Tokairin K et al (2014) The frequency of postoperative stroke in moyamoya disease following combined revascularization: a single-university series and systematic review. J Neurosurg 121(2):432–440
Jin SC, Oh CW, Kwon OK et al (2011) Epilepsy after bypass surgery in adult moyamoya disease. Neurosurgery 68(5):1227–1232 discussion 1232.
Andoh T, Sakai N, Yamada H et al (1992) Chronic subdural hematoma following bypass surgery—report of three cases. Neurol Med Chir (Tokyo) 32(9):684–689
Czabanka M, Boschi A, Acker G et al (2016) Grading of moyamoya disease allows stratification for postoperative ischemia in bilateral revascularization surgery. Acta Neurochir (Wien) 158(10):1895–1900
Pena-Tapia PG, Kemmling A, Czabanka M, Vajkoczy P, Schmiedek P (2008) Identification of the optimal cortical target point for extracranial-intracranial bypass surgery in patients with hemodynamic cerebrovascular insufficiency. J Neurosurg 108(4):655–661
Horn P, Scharf J, Pena-Tapia P, Vajkoczy P (2008) Risk of intraoperative ischemia due to temporary vessel occlusion during standard extracranial-intracranial arterial bypass surgery. J Neurosurg 108(3):464–469
Hori S, Acker G, Vajkoczy P (2016) Radial artery grafts as rescue strategy for patients with Moyamoya disease for whom conventional revascularization failed. World Neurosurg 85:77–84
Kuhn FP, Warnock G, Schweingruber T, Sommerauer M, Buck A, Khan N (2015) Quantitative H2[(15)O]-PET in pediatric moyamoya disease: evaluating perfusion before and after cerebral revascularization. J Stroke Cerebrovasc Dis 24(5):965–971
Khan N, Yonekawa Y (2008) Moyamoya angiopathy in Europe: the beginnings in Zurich, practical lessons learned, increasing awareness and future perspectives. Acta Neurochir Suppl 103:127–130
Khan N, Yonekawa Y (2005) Moyamoya angiopathy in Europe. Acta Neurochir Suppl 94:149–152
Khan N, Schuknecht B, Boltshauser E et al (2003) Moyamoya disease and Moyamoya syndrome: experience in Europe; choice of revascularisation procedures. Acta Neurochir (Wien) 145(12):1061–1071 discussion 1071.
Yonekawa Y, Ogata N, Kaku Y, Taub E, Imhof HG (1997) Moyamoya disease in Europe, past and present status. Clin Neurol Neurosurg 99(Suppl 2):S58–S60
Thines L, Petyt G, Aguettaz P et al (2015) Surgical management of Moyamoya disease and syndrome: current concepts and personal experience. Rev Neurol (Paris) 171(1):31–44
Hanggi D, Mehrkens JH, Schmid-Elsaesser R, Steiger HJ (2008) Results of direct and indirect revascularisation for adult European patients with Moyamoya angiopathy. Acta Neurochir Suppl 103:119–122
Ulrich PT, Januschek E (2011) Revascularisation surgery and long-term follow-up in juvenile Moyamoya syndrome: a retrospective analysis. Acta Neurochir Suppl 112:39–43
Lanterna LA, Brembilla C, Gritti P, Bernucci C (2016) Universal bypass for treatment of symptomatic moyamoya disease or Moyamoya syndrome. Analysis of a personal case series on Behalf of the Italian Moyamoya Association. Acta Neurochir Suppl 123:129–132
Guzman R, Lee M, Achrol A et al (2009) Clinical outcome after 450 revascularization procedures for moyamoya disease. Clinical article. J Neurosurg 111(5):927–935
Teo M, Johnson J, Steinberg GK (2017) Strategies for and outcome of repeat revascularization surgery for Moyamoya disease: an American Institutional Series. Neurosurgery 81(5):852–859
Schubert GA, Biermann P, Weiss C et al (2014) Risk profile in extracranial/intracranial bypass surgery—the role of antiplatelet agents, disease pathology, and surgical technique in 168 direct revascularization procedures. World Neurosurg 82(5):672–677
Uchino H, Kuroda S, Hirata K, Shiga T, Houkin K, Tamaki N (2012) Predictors and clinical features of postoperative hyperperfusion after surgical revascularization for moyamoya disease: a serial single photon emission CT/positron emission tomography study. Stroke 43(10):2610–2616
Narisawa A, Fujimura M, Shimizu H, Tominaga T (2007) Seizure following superficial temporal-middle cerebral artery anastomosis in patients with moyamoya disease: possible contribution of postoperative cerebral hyperperfusion. No Shinkei Geka 35(5):467–474
Takanari K, Araki Y, Okamoto S et al (2015) Operative wound-related complications after cranial revascularization surgeries. J Neurosurg 123(5):1145–1150
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical standards
This study was approved by local ethics-committee.
Rights and permissions
About this article
Cite this article
Kraemer, M., Sassen, J., Karakaya, R. et al. Moyamoya angiopathy: early postoperative course within 3 months after STA–MCA–bypass surgery in Europe—a retrospective analysis of 64 procedures. J Neurol 265, 2370–2378 (2018). https://doi.org/10.1007/s00415-018-8997-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-018-8997-2