Abstract
The objective of this study is to evaluate the relationship between social cognition (SC) and cognitive impairment in persons with multiple sclerosis (PwMS). A prospective study was conducted in 60 PwMS, 30 with relapsing-remitting MS (RRMS), 15 with secondary progressive MS (SPMS) and 15 with primary progressive MS (PPMS), and in healthy subjects (HS). All subjects were assessed by the Bordeaux Social Cognition Evaluation Protocol (PECS-B) (facial emotion recognition, theory of mind, emotional awareness and cognitive and affective alexithymia), by a large neuropsychological battery and by questionnaires (depression and anxiety). 43.3% of PwMS were impaired for at least one SC test. The proportion of PwMS with at least two impaired SC tests was similar in all three phenotypes (20%). Mean scores differed significantly between PwMS and HS only for the Reading the Mind in the Eyes Test, a test of Theory of Mind (ToM). ANOVA analyses showed an effect of phenotype on emotional awareness scores with lower scores in PPMS as compared to RRMS. ToM performance was significantly correlated (r 2 = 0.56) with executive functions, working memory and episodic memory scores. SC impairment was found in all phenotypes and was more prominent in cognitively impaired MS patients. Executive functions, and working and episodic memory performance accounts for approximately 50% of ToM performance. Emotional awareness is more impaired in progressive MS.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Social cognition (SC) refers to the cognitive processes necessary for interpersonal relationships [1]. It relies on the capacity to recognize and interpret interpersonal cues that guide social behaviour [2]. SC has been separated in various domains, such as theory of mind (ToM), empathy, emotion processing, social knowledge and social perception, with overlapping among those terms. Emotional processing refers to perceiving and using emotions, including identifying, understanding and managing emotions [3]. It can be assessed by face emotion recognition, affective prosody recognition, emotional awareness and alexithymia scales. ToM, also called mental state attribution, mentalizing or mind reading, could be separated into cognitive ToM and affective ToM [2]. Cognitive ToM refers to the ability to make inferences about mental states (intentions, dispositions, and beliefs) of other people. Affective ToM is the ability to make affective inferences about what another person is thinking or feeling and requires empathy [1].
SC has been assessed in several studies in multiple sclerosis (MS) investigating facial emotion recognition, affective prosody, ToM (cognitive ToM and affective ToM), empathy, decision making and alexithymia suggesting that in addition to the classical domains of cognitive dysfunction, individuals with MS also have impaired social cognition [4–6].
However, these studies yielded contradictory results concerning the relationships between cognitive impairment (CI) and SC in MS. It remains unclear if a specific SC impairment exists in MS or if the observed deficits are mainly secondary to cognitive impairment (CI) [4]. Indeed, data obtained in different pathologies suggested that SC impairment could be, at least in part, associated with CI, such as in executive functions (EF) [4–6]. Moreover, most of the studies were performed only in relapsing-remitting MS (RRMS) and little is known about SC performances of persons with progressive MS.
The main objective of this prospective study was to evaluate the relationship between several dimensions of SC and CI in people with MS (PwMS). The secondary objective was to describe the frequency and the characteristics of SC impairment in PwMS compared to healthy subjects (HS) matched for age, education and gender and to study the effect of clinical phenotype of three groups of PwMS, one with RRMS, one with primary progressive MS (PPMS) and the last one with secondary progressive MS (SPMS).
Subjects and methods
The study was conducted at the MS clinic of the Bordeaux University Hospital between March 2013 and June 2014 according to the declaration of Helsinki. Bordeaux ethical committee (Comité de protection des personnes) has approved the use of human subjects for this study and all patients and subjects gave informed written consent.
Subjects
Consecutive PwMS, fulfilling McDonald criteria [7], aged 18–60, native French speakers and diagnosed with RRMS, PPMS or SPMS could be included. Exclusion criteria included psychiatric comorbidity or history, current or past alcohol or drug abuse, Mini Mental State (MMS) <24; Expanded Disability Status Score (EDSS) >6, neurological impairment precluding performing NP or SC testing and relapse in the previous month.
HS were recruited specifically by the Psychology Laboratory of Bordeaux University and four groups were matched for age, gender and education to PwMS. Exclusion criteria for HS included psychiatric or neurologic comorbidity or history, current or past alcohol or drug abuse. Subjects did not receive compensation.
Measures
SC measures
The Bordeaux Social Cognition Assessment Protocol (PECS-B), validated for French speaking subjects, was used [8]. This battery addresses the different dimensions of SC.
-
ToM was assessed by three tests, the Faux Pas test, [9] the Attribution of intentions test [10] and the Reading the Mind in the Eyes test [11].
-
Facial emotion recognition was assessed by the Faces test adapted from Baron-Cohen [12] (forced or free choice) [8].
-
Emotional awareness was assessed using the Levels of Emotional Awareness Scale (LEAS) [13]. Three scores were derived: self awareness, other awareness (awareness of emotions of other people) and total emotional awareness score.
-
Emotional fluency was assessed with the emotional fluency task of the PECS-B [8].
-
Alexithymia was assessed using the Bermond–Vorst Alexithymia Questionnaire [14].
Neuropsychological (NP) testing
Cognitive functioning was assessed using the following NP tests and scores
-
Episodic memory: French adapation of the Grober and Buschke Free and Cued Selective Reminding Test [15] called “Épreuve de rappel libre/rappel indicé à 16 items” (RL-RI 16) and forward digit span [16].
-
Working memory: backward and forward digit span [16] and PASAT 3 s [17].
-
Information processing speed (IPS): digit symbol [16], Computerized Speed Cognitive Test© (CSCT) [18], D2 test, a letter cancellation test (numbers of items analysed) [19], Stroop 45- colour denomination; Stroop 45-reading [20].
-
Attention: D2 test, a crossing test measuring selective and sustained attention and visual scanning speed (error percentage) [19].
-
Executive functions: verbal fluency [21], Wisconsin card sorting test [22], and Stroop 45 inhibition score [20].
Psychological evaluation
Depression was assessed using the Beck Depression Inventory (BDI) [23] and anxiety using State form and Trait form of the State-Trait Anxiety Inventory (STAI-S, and STAI-T) [24].
Procedure
After signature of the consent form, patients had the following assessments: clinical neurological examination, NP testing and SC testing. Patient reported outcomes (BDI, STAI) were filled after the clinical examination. All assessments were performed by the same neurologist.
Analysis and statistics
Statistical analyses were performed using Statview for Windows. A p level of 0.05 represented statistical significance.
Baseline characteristics between groups were compared using Student’s t test or the Mann–Whitney U test when appropriate.
A SC and NP test was considered to be impaired if one of the calculated scores for this test was <5th percentiles of the matched HS scores (−1.64 SD). Patients were considered cognitively impaired for a given SC or NP domain if they were impaired for at least one score of this domain, except for IPS, where considering the greater number of tests used for this domain, two impaired tests were required.
Z scores were calculated for each NP and SC test and domain using the HS data as a reference sample. Values of SC and NP scores of PwMS and HS group were compared using Student’s t test or the Mann–Whitney U test when appropriate according to normality assessed by the Kolmogorov–Smirnov test. Bonferroni correction for multiple comparisons of mean SC scores was applied and a p value <0.0045 was retained as significant. Considering the limited sample size in subgroups the comparison was only carried out for the whole PwMS group to HS. To study the effect of clinical phenotype (RR, PP and SP) on SC scores, ANOVA analysis was carried out. For significant results a post hoc, Least Significant Difference (PLSD) of Fisher was carried out to compare phenotypes.
Pearson’s correlation analyses were performed between SC domains z scores and neurocognitive domains and psychological z scores and some clinical parameters (EDSS, disease duration). Multivariate linear analyses were performed with SC domains as dependent variables and cognitive domains z scores as independent variables. Only variables with a p value <0.25 in correlation analyses were entered into the models. EDSS score was forced in all models. Additionally, multivariate linear analyses were performed with SC tests z scores belonging to the SC domain significantly correlated to NP domains in the first multivariate analysis as dependent variables and NP domains z scores as independent variables.
Results
Demographic and clinical characteristics
63 MS patients were screened but three were not included, one for past alcohol abuse, two for neurological disability precluding performing NP or SC testing. Thirty PwRRMS, 15 PwPPMS and 15 PwSPMS patients were included. The HS group included 65 subjects matched for age, gender and education.
Table 1 summarizes the demographic and clinical characteristics of these subjects. Age was greater for PPMS or SPMs as compared to RRMS (p < 0.01 for both comparisons). Disease duration was longer, as expected, in SPMS as compared to RRMS and PPMS (p < 0.01 for both comparisons). The proportion of women was greater in RRMS than in SPMS and PPMS (p < 0.01 for both comparisons). Median EDSS was lower in RRMS than in PPMS or SPMS (p < 0.001 for both comparisons).
Anova analyses showed a positive effect of phenotype on BDI (F = 3.39; p < 0.05) with higher scores in progressive phenotypes than in RRMS (p < 0.05 at PLSD Fisher’s tests for PPMS and SPMS as compared to RRMS). Only two patients were diagnosed with major depression according to a BDI score >27 (3%). Six patients out of 60 used antidepressant drugs (10%).
SC results
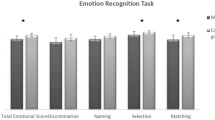
Among PwMS, 43.3% were impaired for at least one SC test and 20% for at least two tests, but only 10% were impaired for at least three tests. The proportions of PwMS with at least two SC tests impaired were similar in all three phenotypes (20%). Figure 1 shows the proportion of PwMS impaired for the different SC domains in the whole group and in the RRMS, PPMS and SPMS groups. This figure shows that the proportion of PwMS impaired for ToM ranged from 20 to 36.7% according to phenotypes and from 6.7 to 20% for facial emotion recognition. The Reading the Mind in the Eyes test was impaired in 18.3%, the Attribution of intention test in 13.3% and the Faux Pas test in only 5% of PwMS. Emotional awareness impairment is present in 7–27% according to phenotypes and cognitive alexithymia from 17 to 20%. Affective alexithymia deficits were uncommon (7%).
Table 2 summarizes scores for SC tests for each domain in the whole group of PwMS and for each phenotype.
PwMS had significantly lower performances than HS for the Reading the Mind in the Eyes test (ToM) (p = 0.0026). The other scores were not different between PwMS and HS after correction for multiple comparisons.
Anova analyses showed a positive effect of phenotype for emotional awareness, for LEAS total score (F = 3.18; p < 0.05), and other awareness score (F = 5.51; p < 0.01). Fisher’s PLSD test between PP and RRMS showed a significant difference (p < 0.05 for LEAS total score and 0.01 for other awareness score) showing lower emotional awareness scores in PPMS than in RRMS. A trend was observed (p = 0.08) for the faux pas test with lower scores in PPMS than RRMS (PLSD Fisher’s test p < 0.01).
NP assessment
The proportions of PwMS in the whole group and for each phenotype with CI for each cognitive domain are presented in Fig. 2.
Correlation analyses
Table 3 presents the univariate linear analyses between SC performances in the different domains and clinical and cognitive performances. Multivariate analyses showed a significant correlation (R 2 = 0.56; p < 0.0001) between ToM z score with executive functions, working memory and episodic memory z scores. No clinically relevant correlation was observed between other SC domains and NP domains z scores. R 2 values were, respectively, 0.086 for the correlation between facial emotion recognition z score and episodic memory z score (p < 0.05), and 0.103 for the correlation between emotional awareness z score and IPS z score (p < 0.05). Alexithymia did not correlate with any NP domain. Clinical scores (BDI, STAI S and T, EDSS and disease duration) did not correlate significantly with any SC domain in multivariate analyses.
For a better understanding of the correlation we performed additional multivariate linear analyses with individual SC test z scores of the main domain showing significant correlation in the first analysis (ToM) as dependant variables and NP domain z scores as independent variables. Table 4 presents the univariate linear analyses between the 3 ToM tests z scores and NP domains z scores. Working memory (p < 0.001) and attention (p < 0.05) accounts for approximately 34% of Reading the Mind in the Eyes test z scores (R 2 = 0.337). Working memory accounts for 27% of Attribution of intentions test z scores (R 2 = 0.269; p < 0.001). Finally, Executive functions and episodic memory accounts for 21% of the Faux pas test scores (R 2 = 0.215; p < 0.05).
Discussion
This prospective study showed that whatever the clinical phenotype, a significant number of PwMS have impairment in tasks assessing SC. Impairment could occur for any of the SC domains that we studied, ToM, facial emotion recognition, emotional awareness and alexithymia, but was more frequent for ToM. Although some differences exist between the three phenotypes, each type of impairment can occur in the three groups. ANOVA analyses showed, however, that emotional awareness scores were lower in PPMS as compared to RRMS. The proportions of PwMS with at least two SC tests impaired were 20% in all three phenotypes, which is relatively low. Finally, the main result of this study was that ToM performance correlated significantly with cognitive performance in particular executive functions, episodic memory and working memory. Other SC domains correlated poorly or did not correlate with cognitive performance.
In this study, ToM impairment was observed in 28% of PwMS, with the proportion of impaired subjects reaching approximately 35% in RRMS and SPMS. When comparing ToM test scores between PwMS and HS, only the scores for the Reading the Mind in the Eyes Test were significantly lower and this test was the most frequently SC impaired test in this sample. Several studies explored ToM impairment in MS [4–6]. Among SC domains, ToM shows the highest frequency of impairment [6]. These studies suggested that MS patients may have difficulties in cognitive ToM tests or in the attribution of intentions or thoughts, such as false belief tasks [25, 26] or in the reading the mind in the eyes test [27, 28]. Some studies used tests that assess both cognitive and affective ToM, such as the Faux Pas test, and found lower scores in PwMS than in controls [25, 26, 28–30]. We did not find frequent impairment at the Faux Pas test. However, impairment at the Faux Pas test was significant only in patients with CI in one study [29], in patients with high disability in another [28], and in one study, the impairment concerned only the cognitive ToM part of the Faux Pas test [30]. Interestingly, the Faux Pas test scores were lower in PPMS in our population. No other effect of phenotype was observed on ToM tests. Two studies using the Movie for the Assessment of Social Cognition found that identification of thoughts and intentions and emotion identification were both significantly impaired in MS [26, 31]. A recent study used a dynamic task, the Awareness of Social Inference Test (TASIT), testing the ability of individuals with MS to understand and interpret lies and sarcasm [32].
The frequency of impairment in other SC domains (facial expression recognition, emotional awareness and alexithymia) varied between 10 and 15% in our sample, and significantly lower scores for emotional awareness were observed than in HS. These impairments occurred in all phenotypes, but 27% of PPMS patients had impaired facial expression recognition and alexithymia. Emotional recognition was investigated in several studies in MS using various methodologies [4]. Several studies [25, 28, 33–36] consistently found significant differences between PwMS and HS for facial emotions recognition. To our knowledge, emotional awareness was not studied in MS previously. Interestingly, we observed that impairment at the LEAS was not rare in MS and an effect of phenotype was confirmed by ANOVA for the total score and more specifically for the score measuring emotional awareness about other persons showing poorer performances at the test in PPMS as compared to RRMS. In the PPMS and SPMS groups, 13.3–20% of subjects were impaired in this test. This suggests that this domain must be taken into account in SC assessment in MS in particular in progressive MS. Alexithymia has been widely studied in MS [36, 37]. It has been defined as difficulties in expressing one’s emotions. The frequency of alexithymia in MS varies according to country and methodology, but all these studies used the Toronto Alexithymia Scale. Previous studies in French PwMS had prevalence from 23.2 to 30% [37, 38]. Using the BVAQ, which is probably less sensitive, we observed a frequency of 12% in the whole group, but 27% in PPMS. The BVAQ allows differentiating a cognitive dimension, known to be similar to the TAS, and a psychological dimension. We observed that PwMs were essentially impaired for the cognitive dimension.
In the present study, using a large cognitive battery, we found a significant correlation between executive functioning, episodic and working memory and ToM scores. The strength of the correlation was quite high because scores in these domains accounts for more than 50% of ToM impairment. These correlations were observed for the three ToM tests used in this study: the Reading the Mind in the Eyes test, the Attribution of Intention test and the Faux Pas test, but with interesting differences. The Reading the Mind in the Eyes test can be considered as a task that measures cognitive ToM because it includes only complex mental states that involve the attribution of a belief or intention, a cognitive mental state [11]. The performance in the Eyes test and the Attribution of intentions test, two tasks measuring cognitive ToM, correlate significantly with working memory and with attention. The Faux Pas test, which studies both cognitive and affective ToM, correlates significantly with executive functioning and episodic memory.
These results are in agreement with several studies showing some correlations between ToM tasks and cognitive performance although recent meta-analyses and review showed that contradictory results have been reported [4–6]. One study found that only patients with CI have difficulties in Strange Stories tasks, the Faux Pas task and the Conversations and Insinuations video task [29]. In one study, the correlation between ToM impairment and performance in processing speed, memory and executive function tasks was observed, but ToM impairment remained significant in PwMS without CI compared to HS [31]. A recent study found correlations between TASIT scores and IPS, working memory, learning and memory, and premorbid IQ [32]. However, other studies reported negative results. One study testing 25 PwMS found a correlation between ToM scores with the Stroop test, but not with working memory and set shifting tests [26], and other studies did not find any correlations [28, 30, 36], including the only study in pediatric-onset MS [39]. In this small sample, IPS performances measured by the Symbol Digit Modalities test did not account for the differences in ToM performance [39]. These discrepancies between studies are probably due to methodological differences (samples, cognitive tests used). The association of SC with CI is supported by other works outside MS. A review of ToM and executive functions [40] suggested an association between the two, but not for a specific elementary executive process.
In our study, emotion recognition and alexithymia did not correlate with cognitive performance, in spite of a quite large NP investigation. Some authors found correlations between emotional recognition and IPS [25, 36], working memory [34, 36], and sustained attention [36], although other authors did not find such a correlation [28]. In another study [27], in which, although no overall group differences in facial affect recognition was identified, specific difficulties were observed in decoding two facial emotions: anger and fear and were related to cognitive domains, especially IPS.
SC impairment occurs in all clinical phenotypes of MS, with a higher frequency for ToM impairment, which appears to be linked to cognitive performance, mainly working memory, executive functions and episodic memory. This association between ToM, working and episodic memory and executive functioning may have important consequences for the management of PwMS. Indeed, ToM evaluation should be performed when neuropsychological testing discloses impairment in one of these domains. Emotional awareness appears also to be an important aspect of SC dysfunction, especially in progressive MS and concerning emotional awareness about other persons. This can have consequences in daily lives of PwMS and deserves further studies.
The study has some limitations. The sample size of the two groups according to MS phenotypes is small limiting the power of the comparison between them. However, we were able to show effect of phenotypes using ANOVA. Another limitation is the cross-sectional design. A longitudinal design is necessary for definitely demonstrating a causal relationship between CI and SC performances.
The consequences of these deficits in the daily life of patients, especially in their interpersonal relationships, warrant specific studies in MS to evaluate the usefulness of specific management programs.
Author contribution statement
Dr Dulau: Study concept and design, acquisition of data, analysis and interpretation, critical revision of the manuscript for important intellectual content. Dr Deloire: Study concept and design, analysis and interpretation, critical revision of the manuscript for important intellectual content, study supervision. Ms Diaz: acquisition of data, analysis and interpretation. Ms Saubusse: acquisition of data, analysis and interpretation. Ms Charré-Morin: acquisition of data, analysis and interpretation. Dr Prouteau: Study concept and design, analysis and interpretation, critical revision of the manuscript for important intellectual content, study supervision. Pr Brochet: Study concept and design, acquisition of data, analysis and interpretation, critical revision of the manuscript for important intellectual content, study supervision.
References
Dulau C (2015) Introduction to social cognition. In: Brochet B (ed) Neuropsychiatric symptoms of inflammatory demyelinating diseases. Springer, Cham, pp 181–194
Frith C, Frith U (2005) Theory of mind. Curr Biol 15:R644–R645
Green MF, Penn DL, Bentall R et al (2008) Social cognition in schizophrenia: an NIMH workshop on definitions, assessment, and research opportunities. Schizophr Bull 34:1211–1220
Dulau C (2015) Social cognition and multiple sclerosis. In: Brochet B (ed) Neuropsychiatric symptoms of inflammatory demyelinating diseases. Springer, Cham, pp 213–226
Bora E, Özakbaş S, Velakoulis D, Walterfang M (2016) Social cognition in multiple sclerosis: a meta-analysis. Neuropsychol Rev 26:160–172
Cotter J, Firth J, Enzinger C et al (2016) Social cognition in multiple sclerosis: a systematic review and meta-analysis. Neurology 87:1727–1736
Polman CH, Reingold SC, Banwell B et al (2011) Diagnostic criteria for multiple sclerosis: 2010 revisions to the McDonald criteria. Ann Neurol 69:292–302
Etchepare A, Merceron K, Amieva H et al (2014) Évaluer la cognition sociale chez l’adulte: validation préliminaire du Protocole d’Evaluation de la Cognition Sociale de Bordeaux (PECS-B). Rev Neuropsychol 6:138–149
Stone VE, Baron-Cohen S, Knight RT (1998) Frontal lobe contributions to theory of mind. J Cogn Neurosci 10:640–656
Brunet E, Sarfati Y, Hardy-Baylé MC (2003) Reasoning about physical causality and other’s intentions in schizophrenia. Cognit Neuropsychiatry 8:129–139
Baron-Cohen S, Wheelwright S, Hill J et al (2001) The “Reading the Mind in the Eyes” test revised version: a study with normal adults, and adults with Asperger syndrome or high-functioning autism. J Child Psychol Psychiatry 42:241–251
Baron-Cohen S, Wheelwright S, Jolliffe T (1997) Is there a “language of the eyes”? Evidence from normal adults, and adults with autism or Asperger syndrome. Vis Cogn 4:311–331
Lane RD, Quinlan DM, Schwartz GE et al (1990) The Levels of Emotional Awareness Scale: a cognitive-developmental measure of emotion. J Pers Assess 55:124–134
Vorst HCM, Bermond B (2001) Validity and reliability of the Bermond–Vorst Alexithymia Questionnaire. Pers Individ Diff 30:413–434
Amieva H, Carcaillon L, Rouze L’Alzit-Schuermans PR et al (2007) Cued and uncued memory tests: norms in elderly adults from the 3 Cities epidemiological study. Rev Neurol (Paris) 163:205–221
Wechsler D (1997) Wechsler Adult Intelligence Scale-III, administration and scoring manual. The Psychological Corporation, San Antonio
Rao SM and the Cognitive Function Study Group of the National Multiple Sclerosis Society (1990) A manual for the Brief Repeatable Battery of Neuropsychological Tests in multiple sclerosis. National Multiple Sclerosis Society, Milwaukee
Ruet A, Deloire MS, Charre-Morin J et al (2013) A new computerised cognitive test for the detection of information processing speed impairment in multiple sclerosis. Mult Scler 19:1665–1672
Anonymous (2005) The D2 Test of Attention: an examination of age, gender, and cross-cultural indices. Argosy University
Golden CJ (1975) A group version of the Stroop color and word test. J Pers Assess 39:386–388
Godefroy O, Azouvi P, Robert P et al (2010) Dysexecutive syndrome: diagnostic criteria and validation study. Ann Neurol 68:855–864
Vayalakara J, Devaraju-Backhaus S, Bradley J et al (2000) Abbreviated form of the Wisconsin card sort test. Int J Neurosci 103:131–137
Beck AT, Brown G, Steer RA (1996) Beck depression Inventory II manual. The Psychological Corporation, San Antonio
Spielberger CD (1983) Manual for the State-Trait Anxiety Inventory. Consulting Psychologists Press, Palo Alto
Henry A, Tourbah A, Chaunu MP et al (2011) Social cognition impairments in relapsing-remitting multiple sclerosis. J Int Neuropsychol Soc 17:1122–1131
Kraemer M, Herold M, Uekermann J et al (2012) Theory of mind and empathy in patients at an early stage of relapsing remitting multiple sclerosis. Clin Neurol Neurosurg 115:1016–1022
Henry JD, Phillips LH, Beatty WW et al (2009) Evidence for deficits in facial affect recognition and theory of mind in multiple sclerosis. J Int Neuropsychol Soc 15:277–285
Banati M, Sandor J, Mike A et al (2010) Social cognition and theory of mind in patients with relapsing-remitting multiple sclerosis. Eur J Neurol 17:426–433
Ouellet J, Scherzer PB, Rouleau I et al (2010) Assessment of social cognition in patients with multiple sclerosis. J Int Neuropsychol Soc 16:287–296
Roca M, Manes F, Gleichgerrcht E et al (2014) Cognitive but not affective theory of mind deficits in mild relapsing-remitting multiple sclerosis. Cogn Behav Neurol 27:25–30
Pottgen J, Dziobek I, Reh S et al (2013) Impaired social cognition in multiple sclerosis. J Neurol Neurosurg Psychiatry 84:523–528
Genova HM, Cagna CJ, Chiaravalloti ND, DeLuca J, Lengenfelder J (2016) Dynamic assessment of social cognition in individuals with multiple sclerosis: a pilot study. J Int Neuropsychol Soc 22:83–88
Beatty WW, Goodkin DE, Weir WS et al (1989) Affective judgments by patients with Parkinson’s disease or chronic progressive multiple sclerosis. Bull Psychon Soc 27:361–364
Berneiser J, Wendt J, Grothe M et al (2014) Impaired recognition of emotional facial expressions in patients with multiple sclerosis. Mult Scler Relat Disord 3:482–488
Phillips LH, Henry JD, Scott C et al (2011) Specific impairments of emotion perception in multiple sclerosis. Neuropsychology 25:131–136
Prochnow D, Donell J, Schäfer R et al (2011) Alexithymia and impaired facial affect recognition in multiple sclerosis. J Neurol 258:1683–1688
Chahraoui K, Duchene C, Rollot F et al (2014) Longitudinal study of alexithymia and multiple sclerosis. Brain Behav 4:75–82
Gay M-C, Vrignaud P, Garitte C et al (2010) Predictors of depression in multiple sclerosis patients. Acta Neurol Scand 121:161–170
Charvet LE, Cleary RE, Vazquez K, Belman AL, Krupp LB, US Networkfor Pediatric MS (2014) Social cognition in pediatric-onset multiple sclerosis (MS). Mult Scler 20:1478–1484
Aboulafia-Brakha T, Christe B, Martory MD et al (2011) Theory of mind tasks and executive functions: a systematic review of group studies in neurology. J Neuropsychol 5:39–55
Acknowledgements
Our research group is part of TRAIL, cluster of excellence (ANR-10-LABX-57). This project was supported by a grant from Ligue Française de la Sclérose en Plaques.
Conflicts of interest
The authors did not report disclosures relevant to the present publication. Pr Brochet or its institution received research grants and/or consulting fees from Biogen, Bayer-Healthcare, Novartis, Genzyme, Roche, Medday, Merck-Serono, Actelion and Teva. C Dulau received a travel grant from Teva. M Deloire, H Diaz, A Saubusse, J Charre-Morin, and A Prouteau have nothing to disclose.
Ethical standards
The study was conducted at the MS clinic of the Bordeaux University Hospital between March 2013 and June 2014 according to the declaration of Helsinki. Bordeaux ethical committee (Comité de protection des personnes) has approved the use of human subjects for this study.
Informed consent
All patients and subjects gave informed written consent.
Author information
Authors and Affiliations
Corresponding author
Additional information
A. Prouteau and B. Brochet share co-seniorship of this study.
Rights and permissions
About this article
Cite this article
Dulau, C., Deloire, M., Diaz, H. et al. Social cognition according to cognitive impairment in different clinical phenotypes of multiple sclerosis. J Neurol 264, 740–748 (2017). https://doi.org/10.1007/s00415-017-8417-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-017-8417-z