Abstract
Hair is a preferred material to detect exposure or use of illegal drugs in children. In the present study, we investigated a total of 387 hair samples for commonly applied illegal drugs of children up to 16 years. Analysis was by liquid chromatography/mass spectrometry with LOQs of 0.01 ng/mg hair for all analytes except tetrahydrocannabinol carboxylic acid with an LOQ of 0.1 pg/mg hair. Results were firstly compared with our in-house statics on results from adults’ hair, and secondly to literature data. We started from the assumption that drug concentrations decrease with increasing age.
Results were assigned to 4 different age groups (< 1 year, 1–< 6 years, 6–< 14 years, 14–16 years). As expected, higher results were obtained in age groups 1 and 2. The lowest concentrations were present in age group 3, whereas an increase could be observed in group 4 except heroin. In babies, positive results may be due to in utero exposure, breast milk feeding, and a close physical contact. All drugs under investigation such as cannabinoids, cocaine, amphetamines, and opiates have been detected in breast milk as well as in skin excretions such as sebum, sweat and cutaneous cells. For most drugs, average concentrations in children hair were lower than in adult hair when compared with our in-house statistics. Interestingly, the increase of cannabinoids, cocaine, and amphetamines concentrations in adolescents’ hair points to a deliberate use of these drugs possibly in addition to passive exposure. This observation shows that age groups 1 and 4 are most vulnerable if caregivers or parents are drug users, even if the sources of positive drug findings differ.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
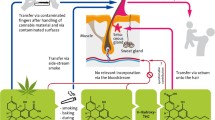
Drug exposure of a child may result in physical, cognitive, and behavioral problems. Many matrices including hair have been proposed to detect in utero exposure. Analysis of neonatal hair can reveal drug use of the mother during the last trimester [1]. Possible sources are fetal blood and amniotic fluid. In babies, positive findings may also be due to drugs being present in the mother’s breast milk or to environmental exposure. Especially in infants’ hair, drugs may be deposited onto the hair by drug containing aerosols or dust, or by bodily contact to consuming caregivers. The latter concerns transfer either through careless handling of the drug or by sebum or sweat of the drug-consuming individual. Also, accidental intake may be an issue.
Knowledge on children hair physiology is very limited. Newborns with a low birth weight generally have thinner hair than those of normal weight [2]. After delivery, the scalp hair being almost exclusively in the anagen stage passes to the catagen stage, and then to the telogen stage. After delivery, the hair is thus replaced within about 3 months. Then, the hair growth stages are similar to that of adults. A baby’s hair has an approximate diameter of 20 μm, and that of a child of about 60 μm, whereas the diameter of adult hair averages 100 μm [3]. Blonde hair is thinner than heavily pigmented hair [4]. After the onset of puberty, hair grows longer and more coarsely [5].
Due to the different hair morphology of babies, infants, children, or adolescents, and a possibly closer contact to family members in the younger age, we assumed in agreement with previous findings that drug concentrations in hair will decrease with increasing age of a child [6, 7]. To check our assumption, we compiled results from hair samples of children having been exposed to commonly used illegal drugs.
Materials and methods
Specimens
A total of 387 hair samples from children (age up to 16 years) which were collected between 2014 and 2018 were analyzed for cocaine, cannabinoids, opiates, and amphetamines by high-performance liquid chromatography/tandem mass spectrometry (LC-MS/MS) using an in-house method accredited to the norm DIN EN ISO/IEC 17025. All cases were related to concerns regarding the child’s welfare by the caregivers being identified as drug users at least in the past. Hair analysis had been ordered from the authorities to decide on either supervision or guardianship. For the present investigation all identifying information except age or sex (except one case) had been removed before data analysis.
Results were assigned to the following 4 age groups: group 1, babies, age < 1 year, n = 21; group 2, infants, age 1–< 6 years, n = 122; group 3 children, age 6– < 14 years, n = 117; group 4 adolescents, age 14–16 years, n = 127.
Method
Depending on availability, the proximal sections of 1 to 6 cm in length were washed twice with 5 mL light petroleum and 5 mL methanol through shaking. After drying, the samples were chopped into small snippets < 1 mm. Extraction was performed with 3 mL methanol through ultrasonication at 50 °C for 4 h following addition of respective deuterated standards. After cooling to room temperature, 1.5 mL of the supernatant was evaporated to dryness under nitrogen and reconstituted in 30 μL acetonitrile and 120 μL 2 mM ammonium acetate. LC-MS/MS analysis was performed by gradient elution on a Sciex 6500 QTrap mass spectrometer (Sciex, Darmstadt, Germany) coupled to an Agilent 1290 Infinity LC system (Agilent, Waldbronn, Germany) equipped with a Kinetex C18 reversed-phase column (3.0 mm ×50 mm; 2.6 μm particle size; Phenomenex, Aschaffenburg, Germany). For data processing MultiQuant 3.0 was used (Sciex, Darmstadt, Germany). Calibration standards ranged from 0.01 to 1.0 ng/mg hair.
The limit of quantification (LOQ) was 0.01 ng/mg hair for all analytes except THC-carboxylic acid which was 0.1 pg/mg hair. THC-carboxylic acid has been determined on request in a very few cases, also depending on the availability of sufficient material. Further validation data have been summarized in Table 1.
Results and discussion
Minimum, maximum, and median values of all analytes are summarized in Table 2. Subsequently, the text is arranged in decreasing frequency of findings from the following drugs or drug groups: cannabinoids, cocaine, amphetamines, and opiates (heroin).
Cannabinoids
THC being present in 112 cases was most frequently detected; its concentrations range from 0.01 ng/mg hair to 15 ng/mg hair. Interestingly, the median THC was highest in age group 4, albeit significantly lower compared with the median of 0.23 ng THC/mg hair derived from our in-house statistical evaluation of adult hair specimens [8]. Also, the median value of adolescent hair being approximately twice that compared with the other age groups suggests that some of them had not only be exposed to environmental cannabis but might have used it accidentally or deliberately. It is known that the incidence of parent drug use may affect the prevalence of teen drug use [9]. Adolescence is a developmental period being associated with increased risk-taking behaviors which promote drug use if living in an environment characterized by caregiver substance use [10].
The finding of lower drug concentrations in age groups 1–3 is in line with results in hair samples from very young children reported by [11]; authors tested 8 out of 12 hair specimens from children aged 2 to 24 months positive for THC at concentrations ranging from 0.039 to 1.89 ng/mg hair. As already mentioned in the introduction, drug residues in age group 1 may result from in utero as well as postnatal exposure. THC is excreted into breastmilk in small quantities and its duration of detection ranges from 6 days to greater than 6 weeks [12]. Also, a physical contact may lead to positive results for THC in children hair. At present, THC as well as cannabidiol and cannabinol have been determined from human skin residues such as skin particles, sebum, and sweat collected in sweat patches [13] whereas THC-carboxylic acid was not detectable at an LOD of 0.3 ng/patch. Due to the low number of cases THC-positive cases along with the additional order of determining THC-carboxylic acid (n = 4), reliable data analysis will depend on further analytical results. We are quite aware that a thorough interpretation especially for age group 4 will not be possible until tetrahydrocannabinolic acid A, THC-carboxylic acid and hydroxy-THC will be analyzed in addition [14, 15]. However, these issues require to perform further analyses which are often limited by the small amounts of hair especially from very young children.
Cocaine
The second highest occurrence was with cocaine at median values ranging from 0.078 to 0.14 ng cocaine/mg hair (n = 75). The maximum concentrations of cocaine determined in age groups 1 and 4 were higher than those reported by Pichini et al. [16] (range 0.15–3.81 ng cocaine/mg hair, n = 23) of 2- to 10-year-old children presenting to an urban pediatric emergency department without suggestive signs or symptoms of drug ingestion. In the present investigation, benzoylecgonine could be detected in all samples except one being positive for cocaine. As cocaine is known to readily arise from environmental exposure and prone to form benzoylecgonine by hydrolysis, an assessment of drug use is usually based on norcocaine and cocaethylene concentrations. Related to the number of positive results for cocaine, norcocaine and cocaethylene were detectable in 50 and 33% of specimens from age group 1, respectively. For deliberate use which appears unlikely in this age group, results may reflect exposure through related parties. According to the findings of Cone at al. [17], cocaine is the major analyte in sweat, whereas metabolites are excreted in lower amounts. Therefore, physical contact may be a likely source just like in utero exposure or ingestion via breast milk. Beside cocaine, all major metabolites had also been detected in random breast milk samples [18]. Percentages decreased to 23% for norcocaine and to 14% for cocaethylene in age group 3, probably pointing to a decreasing contact with their caregivers. Compared to our in-house statistics, percentages in age groups 2 and 3 were all lower compared with those for norcocaine and cocaethylene being 71% and 78% in adult samples [8]. However, in group 4 there was again an increase in percentages for both norcocaine and cocaethylene as already observed for THC, reaching 35% for norcocaine and 29% for cocaethylene, and suggesting active use beside environmental exposure.
Amphetamines
Amphetamine (alone), methamphetamine, and 3,4-methylenedioxymethamphetamine (MDMA) could be detected in 65, 9, and 41 specimens, respectively. The concentration range for methamphetamine was far lower than that observed in the study of Castaneto et al. [19] where methamphetamine was detected in 67 samples of children (1 month to 18 years) at concentrations of 0.1–22 ng methamphetamine/mg hair suspected to be environmentally exposed to household drug intake. This may partly be due to the low number of methamphetamine positive cases but also to a severe exposure of the children in clandestine laboratories. Maximum values of all drugs were detectable in age group 4, where a deliberate or accidental use seems most probable. This is also reflected by respective median values of group 4 for methamphetamine and MDMA, but not for amphetamine. The median values for amphetamine and methamphetamine in all groups was always lower than those being available from our in-house statistical evaluation on adults’ hair [8]. However, the median MDMA concentration in group 4 matches that from our in-house statistics of adults (0.51 versus 0.52 ng MDMA/mg hair), pointing towards a preferred use of MDMA compared with amphetamine and methamphetamine already at a younger age. This may reflect the increase of MDMA use in adolescents in the last few years [20]. Amphetamines have been detected in both skin excretions as well as in breast milk, but at high dosages may impair milk production [21, 22].
Opiates (heroin)
Proving positive for 6-acetylmorphine as the primary metabolite of heroin, exposure to the drug could be identified in 16 cases. Interestingly, there was no positive finding for heroin, 6-acetylmorphine or morphine in age group 4. This is in line with observations in the USA where young adults aged 18–25 among had used heroin far more frequently in the past year than teens aged 12–17 [23].
Heroin itself was detectable in very low concentrations in groups 1–3 in 12 cases ranging from 0.01 to 0.04 ng/mg hair. Regarding the summarized molar concentration of 6-acetylmorphine and morphine, group 1 appeared to be most exposed compared with the other groups, possibly due to in utero as well as environmental exposure after birth. Exposure may be via the placenta, breastmilk, and inhalation of smoked heroin [24].
Also, the highest median concentrations of both heroin and 6-acetylmorphine were detectable in age groups 1–2 indicating again a closer bodily contact of younger children to their consuming caregivers or parents. Heroin and metabolites may preferably be transferred via sweat or sebum to children’s hair. This assumption is supported by findings from Cone et al. [16]; the working group observed heroin being the major analyte excreted in sweat, and 6-acetylmorphine appeared rapidly after heroin administration and continued to increase in sweat samples while the content of heroin decreased suggesting hydrolysis of heroin in sweat. Comparing the present data to that of our in-house statistics on ranges in adult hair, median concentrations in adult hair were grossly more than 20 times higher (0.60 ng heroin/mg hair, 0.74 ng 6-acetylmorphine/mg hair) compared with the present result [8]. However, the median concentration of morphine, which is preferably used to assess drug consumption behavior, was about 70 times higher compared with that in our present population, which again provides evidence of drug transfer via sweat or sebum to childrens’ hair for excretion of morphine into sweat is significantly lower than for both, heroin and 6-acetylmorphine [25].
Case report
Further, to put forward our introductory assumption, subsequent specimens obtained from male and female siblings including 3 boys and 2 girls (aged 1–8 years) from a family were detailed. Results obtained at the first collection date and after a 6-month period are summarized in Table 3. At the first sampling date, in each of the 5 samples, cocaine including major metabolites, amphetamine, and THC were tested positive. THC-carboxylic acid was present in none of the samples; due to the small amount of hair available, the analyte could not be determined from the sample of the 5-year-old child at the time of first testing. The higher cocaine concentrations of the two youngest children may be explained through a closer contact to the drug-consuming mother. In addition, differences between boys and girls could be noticed; however, a conclusive explanation cannot be given.
Six months later, a second hair sample was collected for drug analysis (Table 3). In contrast to the first analysis, amphetamine was not detectable in any of the samples collected at the second date. Cocaine and benzoylecgonine were detectable in one sample only, but neither norcocaine nor cocaethylene were still present. In the specimen of this female twin, the highest cocaine and benzoylecgonine concentrations were measured in the first sample. However, concentrations of THC had increased meanwhile compared with the first time of collection. Also, traces of THC-carboxylic acid were detectable in the hair of the three younger children. Except the “cocaine” positive sample of the female twin, there was a decreasing tendency in THC concentrations with increasing age.
The changes in drug findings could be explained by the children having been taken from the cocaine-consuming parent to the cannabis-consuming parent after the first sample collection, this thesis is also supported by the findings of THC-carboxylic acid. This change in caregiver’s drug habits may support external contamination of the children hair. This is in line with the findings of Pragst et al. [26] showing that drugs being smoked or available as a powder are deposited to a higher degree onto children hair compared with those taken as a solid or fluid preparation. Also, authors showed a decreasing concentration with increasing age for nearly all drugs under investigation, which may result from a closer physical contact and a less resistant cuticle of the hair in the young age.
Conclusion
The present study provides evidence that with an increasing age of the child, the risk of being exposed to drugs from drug consuming caregivers or environmental exposure through smoke, dust, or drug residues decreases. Especially in babies, in utero exposure, breast milk or a close physical contact is a likely source of drug findings in hair. The increase of drug concentrations in adolescents’ hair, especially of cannabinoids, amphetamines, and cocaine, suggests an accidental or deliberate use of the drug, possibly in addition to a passive exposure. Results also revealed heroin being a less attractive drug for adolescents which is supported by epidemiological data.
References
Gray T, Huestis M (2007) Bioanalytical procedures for monitoring in utero drug exposure. Anal Bioanal Chem 388(7):1455–1465. https://doi.org/10.1007/s00216-007-1228-9
Berger HM, King J, Doughty S, Wharton BA (1978) Nutrition, sex, gestational age, and hair growth in babies. Arch Dis Child 53:290–294
Robbins CR (2012) Chemical and physical behavior of human hair, 5th edn. Springer, Heidelberg
Pötsch L, Skopp G (2004) Inkorporation von Fremdsubstanzen in das Haar. In: Madea B, Mußhoff F (Hrsg.) Haaranalytik. Technik und Interpretation in Medizin und Recht, Deutscher Ärzte-Verlag, Köln, pp 29-98
Neureiter V, Pietrusky F, Schütt E (1940) Handwörterbuch der gerichtlichen Medizin und naturwissenschaftlichen Kriminalistik. Springer Verlag, Berlin
Pragst F, Broecker S, Hastedt M, Herre S, Andresen-Streichert H, Sachs H, Tsokos M (2013) Methadone and illegal drugs in hair from children with parents in maintenance treatment or suspected for drug abuse in a German community. Ther Drug Monit 35:737–752
Wang X, Drummer OH (2015) Review: Interpretation of the drug presence in the hair of children. Forensic Sci Int 257:458–472
Musshoff F, Schwarz G, Sachs H, Skopp G, Franz T (2020) Concentration distribution of more than 100 drugs and metabolites in forensic hair samples. Int J Legal Med 134:99–995. https://doi.org/10.1007/s00414-020-02259-3
Delaney-Black V, Chiodo LM, Hannigan JH, Greenwald MK, Janisse J, Patterson G, Huestis MA, Ager J, Sokol RJ (2010) Just say “I don’t”: lack of concordance between teen report and biological measures of drug use. Pediatrics 126(5):887–893. https://doi.org/10.1542/peds.2009-3059
Lambert BL, Bann CM, Bauer CR, Shankaran S, Bada HS, Lester BM, Whitaker TM, LaGasse LL, Hammond J, Higgins RD (2013) Risk-taking behavior among adolescents with prenatal drug exposure and extrauterine environmental adversity. J Develop Behav Pediatrics 34(9):669–679. https://doi.org/10.1097/01.DBP.0000437726.16588.e2
Kintz P, Ameline A, Eibel A, Gheddar L, Feisthauer E, Geraut A, Berthelon L, Farrugia A, Raul JS (2017) Interpretation of cannabis findings in the hair of very young children. Mission Impossible. Curr Pharm Biotech 18(10):791–795. https://doi.org/10.2174/1389201019666171129180206
National Library of Medicine (US) (2006) Drugs and Lactation Database (LactMed). Cannabis, Bethesda (MD)
Gambelunghe C, Fucci N, Aroni K, Bacci M, Marcelli A, Rossi R (2016) Cannabis use surveillance by sweat analysis. Ther Drug Monit 38(5):634–639. https://doi.org/10.1097/FTD.0000000000000327
Franz T, Skopp G, Schwarz G, Musshoff F (2018) Proof of active cannabis use comparing 11-hydroxy-∆9-tetrahydrocannabinol with 11-nor-9-carboxy-tetrahydrocannabinol concentrations. Drug Test Anal 10(10):1573–1578. https://doi.org/10.1002/dta.2415
Moosmann B, Roth N, Auwarter V (2015) Finding cannabinoids in hair does not prove cannabis consumption. Sci Rep 5:14906. https://doi.org/10.1038/srep14906
Pichini S, Garcia-Algar O, Alvarez A, Gottardi M, Marchei E, Svaizer F, Pellegrini M, Rotolo M, Pacifici R (2014) Assessment of unsuspected exposure to drugs of abuse in children from a Mediterranean city by hair testing. Int J Environ Res Pub Health 11(2):2288–2298. https://doi.org/10.3390/ijerph110202288
Cone EJ, Hillsgrove MJ, Jenkins AJ, Keenan RM, Darwin WD (1994) Sweat testing for heroin, cocaine, and metabolites. J Anal Toxicol 18(6):298–305. https://doi.org/10.1093/jat/18.6.298
National Library of Medicine (US) (2006) Drugs and Lactation Database (LactMed). Cocaine, Bethesda (MD)
Castaneto MS, Barnes AJ, Scheidweiler KB, Schaffer M, Rogers KK, Stewart D, Huestis MA (2013) Identifying methamphetamine exposure in children. Ther Drug Monit 35(6):823–830. https://doi.org/10.1097/FTD.0b013e31829685b2
European Monitoring Centre for Drugs and Drug Addiction (2017) European drug report 2017. Trends and developments. Office for Official Publications of the European Communities, Lisbon
National Library of Medicine (US) (2006) Drugs and Lactation Database (LactMed). Amphetamine, Bethesda (MD)
(2014) Guidelines for Testing Drugs under International Control in Hair, Sweat and Oral Fluid
National Institute on Drug Abuse (NIDA) (2018) Heroin. https://www.drugabuse.gov
National Library of Medicine (US) (2006) Drugs and Lactation Database (LactMed). Heroin, Bethesda (MD)
Kintz P, Brenneisen R, Bundeli P, Mangin P (1997) Sweat testing for heroin and metabolites in a heroin maintenance program. Clin Chem 43(5):736–739
Pragst F, Krumbiegel F, Thurmann D, Westendorf L, Methling M, Niebel A, Hartwig S (2019) Hair analysis of more than 140 families with drug consuming parents. Comparison between hair results from adults and their children. Forensic Sci Int 297:161–170. https://doi.org/10.1016/j.forsciint.2019.01.039
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This is a retrospective study using data from which all identifying information except age has been removed.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Franz, T., Skopp, G. & Mußhoff, F. Findings of illicit drugs in hair of children at different ages. Int J Legal Med 135, 465–471 (2021). https://doi.org/10.1007/s00414-020-02479-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00414-020-02479-7