Abstract
Purpose
Diaphragm ultrasonography is used to identify causes of diaphragm dysfunction. However, its correlation with pulmonary function tests, including maximal inspiratory (MIP) and expiratory pressures (MEP), remains unclear. This study investigated this relationship by measuring diaphragm thickness, thickening fraction (TF), and excursion (DE) using ultrasonography, and their relationship to MIP and MEP. It also examined the influence of age, sex, height, and BMI on these measures.
Methods
We recruited healthy Japanese volunteers and conducted pulmonary function tests and diaphragm ultrasonography in a seated position. Diaphragm ultrasonography was performed during quiet breathing (QB) and deep breathing (DB) to measure the diaphragm thickness, TF, and DE. A multivariate analysis was conducted, adjusting for age, sex, height, and BMI.
Results
Between March 2022 and January 2023, 109 individuals (56 males) were included from three facilities. The mean (standard deviation) MIP and MEP [cmH2O] were 72.2 (24.6) and 96.9 (35.8), respectively. Thickness [mm] at the end of expiration was 1.7 (0.4), TF [%] was 50.0 (25.9) during QB and 110.7 (44.3) during DB, and DE [cm] was 1.7 (0.6) during QB and 4.4 (1.4) during DB. Multivariate analysis revealed that only DE (DB) had a statistically significant relationship with MIP and MEP (p = 0.021, p = 0.008). Sex, age, and BMI had a statistically significant influence on relationships between DE (DB) and MIP (p = 0.008, 0.048, and < 0.001, respectively).
Conclusion
In healthy adults, DE (DB) has a relationship with MIP and MEP. Sex, age, and BMI, but not height, are influencing factors on this relationship.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The diaphragm is the primary muscle involved in respiration. The diaphragm dysfunction is associated with exertional dyspnea, orthopneic breathing, reduced cough strength leading to aspiration risk, and difficulty in weaning from mechanical ventilation [1, 2]. Diaphragm ultrasonography is a non-invasive and convenient method to assess diaphragm function and is useful in diagnosing diaphragmatic paralysis and predicting successful weaning from mechanical ventilation [1, 2]. There are two measurement techniques: one measures the diaphragm excursion (DE) and the other evaluates diaphragm thickness and the change in thickness during respiration (thickening fraction: TF). Common diagnostic criteria for diaphragmatic paralysis include a thickness < 2 mm or a TF < 20% during deep breathing (DB) [1, 3, 4]. Furthermore, DE < 2 cm during quiet breathing (QB) has been proposed as a criterion for diaphragm dysfunction [5].
Diaphragm ultrasonography is becoming increasingly prevalent in clinical practice. Although diaphragm function might be closely related to respiratory function, the specific relationship between respiratory function tests and diaphragm ultrasonography has not been well established [6,7,8]. For instance, some studies suggested a relationship between thickness at functional residual capacity (FRC) and maximal inspiratory pressure (MIP), whereas others did not [6, 9,10,11]. Similarly, results are mixed regarding the relationship between DE and maximal expiratory pressure (MEP) or MIP [6, 12,13,14,15,16]. However, these studies had a small sample size. The populations vary between healthy individuals and patients with underlying conditions such as chronic obstructive pulmonary disease or head trauma, making it challenging to integrate the results. A recent systematic review of the relationship between diaphragm ultrasonography and respiratory function tests noted high heterogeneity in terms of study design [7]. Therefore, there is a need for standardized large-scale studies focusing on healthy individuals.
In this study, we conducted pulmonary function tests and diaphragm ultrasonography on 109 healthy Japanese volunteers to investigate the relationship between diaphragm ultrasonography parameters, including thickness, TF, and DE, with the MIP and MEP. Additionally, we explored factors that influenced these relationships.
Methods
Study Population and Setting
This study was a secondary analysis of a cross-sectional study of diaphragmatic ultrasonography on healthy Japanese [17]. Healthy adult Japanese volunteers, age 18-year-old or older, were recruited from three facilities in Tokyo, Fukuoka and Kanagawa prefecture. The recruitment period was from March 2022 to January 2023. On the examination day, volunteers provided information about their age, sex, height, weight, smoking history, and medical history. Subsequently, pulmonary function tests were conducted using a Spirometer (AutoSpiro507, Minato Medical Science Co. Ltd.) to measure the percent vital capacity (%VC), forced expiratory volume in one second (FEV1), and forced vital capacity (FVC). Only asymptomatic individuals with %VC ≥ 80% and FEV1/FVC ≥ 70% were included. MIP and MEP were measured twice each, and the better of the two results was used. All diaphragm ultrasonography measurements were performed by physicians certified as instructors in the Point of Care Ultrasound Simulation Course and by trained ultrasonography technicians.
Ultrasound Measurements of the Diaphragm
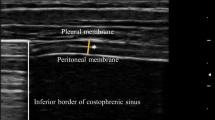
The right hemidiaphragm was measured by ultrasonography in a seated position. The DE of the right hemidiaphragm was assessed at the area around the eighth to ninth intercostal space along the anterior to mid-axillary line, where the diaphragm dome is visualized. A phased-array transducer (2.5 MHz) was placed longitudinally and perpendicularly to the chest wall and adjusted to avoid the ribs. The M-mode interrogation line was adjusted to be as perpendicular to the diaphragm as possible, and the difference in the dome’s movement during inspiration and expiration was measured. For the thickness of the right hemidiaphragm measurement, a linear transducer (7.0 MHz) was positioned longitudinally and perpendicularly at the zone of apposition of the diaphragm, near the eighth to ninth intercostal space along the anterior to mid-axillary line. It was then adjusted to avoid the ribs and positioned so that the lung was partially visible at the edge of the screen during inspiration. At this site, the diaphragm thickness during inspiration and expiration was measured. The thickness measurements were conducted in B-mode, with measurement markers placed from the center of the white line on the thoracic side of the diaphragm to the center of the white line on the peritoneal side. The TF was calculated as follows: (thickness at the end of inspiration − thickness at the end of expiration)/thickness at the end of expiration × 100. The TF and DE were measured during QB and DB.
Statistical Analysis
The patients’ characteristics, diaphragm thickness, TF, and DE are presented as the mean and standard deviation (SD) for continuous data and as counts and proportion for categorical data. Simple and multiple linear regression analyses were used to evaluate the associations between thickness, TF, and DE, and MIP and MEP. In the multiple linear regression analyses, patients’ characteristics including age, sex, height, and BMI were adjusted. We considered p < 0.05 to indicate statistical significance. All data analyses were performed using STATA version 17.0 (StataCorp LLC, College Station, TX, USA).
Results
Participants
A total of 111 Japanese volunteers were recruited for this study. Of these, two individuals were excluded because their %VC was below 80%, resulting in 109 participants being included. The proportion of male volunteers was 51%, with an average age of 31.8 years, ranging from 19 to 60 years. The average BMI was 22.5 for males and 21.7 for females, which was closely aligned with the Japanese national averages of 23.6 for males and 21.8 for females [18]. Two participants had a history of bronchial asthma but were asymptomatic on the day of measurement and had %VC and FEV1/FVC within the normal range. Patient characteristics are presented in Table 1.
Diaphragm Thickness, TF, and DE
The mean values of measurements for the diaphragm thickness (FRC), TF (QB), TF (DB), DE (QB), and DE (DB) were 1.7 mm (SD 0.4), 50.0% (SD 25.9), 110.7% (SD 44.3), 1.7 cm (SD 0.6), and 4.4 cm (SD 1.4), respectively. The thickness and TF were measured in all participants during QB and DB. DE could not be measured in one individual during QB and in 10 individuals during DB because the lung overlaid the diaphragm during inspiration. The measurement results are presented in Table 2.
Simple and Multiple Regression Analyses
Initially, a linear univariate regression analysis was conducted with MIP or MEP as the outcome variable to examine their relationship with diaphragm thickness, TF, and DE. A statistically significant relationship was observed between the MIP and DE during DB (p < 0.001), between the MEP and TF during QB (p = 0.048), and the DE during DB (p < 0.001). No relationship was found between the MIP or MEP, and thickness or TF during DB or DE during QB (Table 3).
In the multivariate linear regression analysis adjusted for age, sex, height, and BMI, a statistically significant relationship was only observed for DE during DB for the MIP (p = 0.008) and MEP (p = 0.021). Sex and BMI were statistically significant influencing factors for all parameters in relation to the MIP and MEP. Age had a significant influence on all parameters in relation to the MIP and MEP except for thickness and DE during DB for MEP. Height had no influence on any of the parameters (Table 4 and 5).
Discussion
This study investigated the relationship between ultrasonographic findings (diaphragm thickness, TF, and DE) and pulmonary function test findings (MIP and MEP) in healthy volunteers. Additionally, it explored which factors influenced these relationships. This represents the largest-scale study to date conducted on a healthy population. [6, 7, 9,10,11] Results from the multiple regression analysis, adjusted for age, sex, height, and BMI, demonstrated a statistically significant relationship between the DE (DB), and the MIP and MEP. Thickness, TF (QB), and TF (DB) had no relationship with MIP and MEP. Although previous studies reported a relationship between the thickness, TF, and DE with MIP and MEP in healthy individuals, all were univariate analyses, and none had been adjusted for variables such as age, sex, or BMI [6, 11, 19]. However, previous studies reported relationships between the diaphragm thickness, TF, and DE with age, sex, and BMI [4, 6, 20,21,22,23]. To accurately evaluate the relationship between various diaphragm ultrasonography parameters and the MIP or MEP, it is essential to adjust for these factors. In this study, the univariate analysis indicated a relationship between the TF (QB) and MEP, although it was not observed in the multivariate analysis. Sex and BMI were statistically significant influencing factors in the relationship between thickness, TF, and DE, and MIP and MEP, with female sex having a negative influence and BMI a positive influence. Therefore, future studies that compare results between groups with significantly different average BMIs, such as Asians and Western populations, must consider the background BMI in their assessments.
Ultrasonographic Findings and MIP
In the current study, no relationship was found between the thickness (FRC) and MIP. This finding is consistent with previous studies involving 13 healthy individuals [11] and 64 healthy individuals [6]. Although these studies had small sample sizes, our analysis with 109 subjects also did not demonstrate a relationship between the thickness (FRC) and MIP. Two other studies that reported there was a relationship between the thickness (FRC) and MIP had sample sizes of 36 and 24 participants, respectively. Thus, our study sample size of 109 subjects can be considered sufficiently large in comparison [9, 10]. Therefore, it is unlikely that the reason our study did not show a statistically significant relationship is due to an insufficient sample size. The first study included 36 participants, of whom 15 were weight-lifters and 3 were children [9] and the second study focused exclusively on 24 individuals aged 65 years and over [10]. Thus, the participant profiles in these studies were not representative of a general healthy population. In our findings, age had a negative influence and BMI had a positive influence on the variables (p = 0.033, p < 0.001). Considering these facts, the two previous studies did not adjust for BMI when assessing the thickness of the diaphragm in weightlifters, who are presumed to have a thicker diaphragm than the general population, nor did they adjust for age when considering the elderly, who are presumed to have a thinner diaphragm, potentially affecting the outcomes [9, 10]. Although thickness (FRC) might be an indirect indicator of muscle mass, this might not directly reflect muscle strength. Therefore, diaphragm thickness may not be directly applicable for predicting respiratory muscle strength [6, 24].
TF, either in QB or DB, had no relationship with MIP. Few studies have examined the relationship between TF and MIP in healthy individuals. A study of 10 children with an average age of 11 years by Ho et al. reported a Spearman’s rank relationship coefficient of 0.64 between the TF and MIP [19]. In our multiple regression analysis, age had a negative influence on the relationship between TF and MIP. Because the results of the study by Ho et al. were not adjusted for age, it is possible that age had a significant impact on the study findings. However, it is unclear whether children and adults can be discussed in a similar fashion. Additionally, a study by Cardenas et al. [6], involving 64 healthy individuals, reported a positive relationship between the TF (DB) and MIP. The average BMI was 26.1 for males and 25.5 for females, which is higher than our study (22.5 for males and 21.7 for females). BMI had a positive effect on the relationship between TF and MIP in our result. However, the study by Cardenas did not adjust for BMI when performing the analysis, which might have influenced their results because the higher BMI in their study might have suggested a relationship between TF and MIP.
DE (DB) had a significant relationship with the MIP (p = 0.008), whereas DE (QB) did not (p = 0.408). These results are consistent with past studies [6, 12, 15]. However, a study by Dos Santos Yamaguti et al. reported the DE (DB) did not correlate with MIP [14]. That study targeted chronic obstructive pulmonary disease patients, who, compared with healthy individuals, had a markedly reduced DE, which might have influenced the results [14]. DE is a critical factor involved in altering thoracic content volume. A larger DE results in a greater change in thoracic content volume, thereby increasing the negative pressure on the lungs. Consequently, this can lead to an increase in MIP. The lack of a relationship between DE (QB) and MIP and the observed relationship with DE (DB) can be understood from this pathophysiological perspective.
Ultrasonographic Findings and MEP
No relationships were found between the thickness, TF (QB), TF (DB), DE (QB), and MEP. Given that the diaphragm is primarily involved in inspiration, it is plausible that it does not have a relationship with MEP. However, a significant relationship was observed between the DE (DB) and MEP. The reason for this may be the influence of the MIP and MEP being related. Simple regression analysis and multiple regression analysis adjusted for age, sex, height, and BMI showed a statistically significant positive relationship between the MIP and MEP (both p < 0.001) (Supplementary Information 1). These findings might have impacted the observed relationship between the DE (DB) and MEP.
In light of these findings, diaphragmatic ultrasonography could be applied to predict successful weaning from mechanical ventilation. In fact, diaphragm ultrasonography has been used increasingly in intensive care units to predict successful weaning from mechanical ventilation [2, 25]. This involves diaphragmatic ultrasonography during a spontaneous breathing trial (SBT) to assess the TF or DE, and to predict successful weaning. The combined sensitivity and specificity for DE are 0.85 and 0.75 whereas these values are 0.80 and 0.80 for TF during QB [2]. MIP and MEP are also important indicators of successful weaning [26,27,28], but measuring these in mechanically ventilated patients during routine clinical SBT is challenging. For instance, encouraging patients who are able to communicate to take deep breaths during SBT and evaluating the DE during DB may further increase the success rate of weaning. However, the threshold values for this are unknown, necessitating further research.
This study had some limitations. Because the participants in this study had an average BMI, it is unclear whether these results are applicable to patients with severe obesity. In cases of extremely high BMI, MIP may actually decrease because of factors such as reduced thoracic compliance. Additionally, since the subjects of this study are young, healthy individuals, it is unclear whether the findings can be applied to patients with underlying diseases or to the elderly. Further research is needed in the future.
In summary, this study investigated the relationship between ultrasonographic findings (diaphragm thickness, TF, and DE) and pulmonary function test findings (MIP and MEP) in healthy volunteers. The influence of age, sex, height, and BMI on these factors were also investigated. The DE (DB) was related to the MIP and MEP, whereas thickness (FRC), TF (QB), and TF (DB) had no relationship. Female sex and older age negatively influence the relationship. Higher BMI positively influences, whereas height does not have an influence.
Abbreviations
- BMI:
-
Body mass index
- DB:
-
Deep breathing
- DE:
-
Diaphragm excursion
- FEV1:
-
Forced expiratory volume in one second
- FRC:
-
Functional residual capacity
- FVC:
-
Forced vital capacity
- MEP:
-
Maximal expiratory pressure
- MIP:
-
Maximal inspiratory pressure
- QB:
-
Quiet breathing
- SBT:
-
Spontaneous breathing trial
- SD:
-
Standard deviation
- TF:
-
Thickening fraction
- TLC:
-
Total lung capacity
- %VC:
-
Percent vital capacity
References
McCool FD, Tzelepis GE (2012) Dysfunction of the diaphragm. N Engl J Med 366(10):932–942
Parada-Gereda HM, Tibaduiza AL, Rico-Mendoza A et al (2023) Effectiveness of diaphragmatic ultrasound as a predictor of successful weaning from mechanical ventilation: a systematic review and meta-analysis. Crit Care 27(10):174
Gottesman E, McCool FD (1997) Ultrasound evaluation of the paralyzed diaphragm. Am J Respir Crit Care Med 155(5):1570–1574
Boussuges A, Rives S, Finance J, Brégeon F (2020) Assessment of diaphragmatic function by ultrasonography: current approach and perspectives. World J Clin Cases 8(12):2408–2424
Haaksma ME, Smit JM, Boussuges A et al (2022) EXpert consensus On diaphragm UltraSonography in the critically ill (EXODUS): a Delphi consensus statement on the measurement of diaphragm ultrasound-derived parameters in a critical care setting. Crit Care 26(1):99
Cardenas LZ, Santana PV, Caruso P, de Carvalho CRR, de Albuquerque ALP (2018) Diaphragmatic ultrasound correlates with inspiratory muscle strength and pulmonary function in healthy subjects. Ultrasound Med Biol 44(4):786–793
Pałac M, Rutka M, Wolny T, Podgórski M, Linek P (2022) Ultrasonography in assessment of respiratory muscles function: a systematic review. Respiration 101(9):878–892
Liu X, Yang Y, Jia J (2023) Respiratory muscle ultrasonography evaluation and its clinical application in stroke patients: a review. Front Neurosci 17:1132335
McCool FD, Conomos P, Benditt JO, Cohn D, Sherman CB, Hoppin FG Jr (1997) Maximal inspiratory pressures and dimensions of the diaphragm. Am J Respir Crit Care Med 155(4):1329–1334
Summerhill EM, Angov N, Garber C, McCool FD (2007) Respiratory muscle strength in the physically active elderly. Lung 185(6):315–320
Ueki J, De Bruin PF, Pride NB (1995) In vivo assessment of diaphragm contraction by ultrasound in normal subjects. Thorax 50(11):1157–1161
Yoon SY, Moon HI, Kim JS, Yi TI, Park YG (2020) Comparison between M-Mode ultrasonography and fluoroscopy for diaphragm excursion measurement in patients with acquired brain injury. J Ultrasound Med 39(3):535–542
Spiesshoefer J, Herkenrath S, Henke C et al (2020) Evaluation of respiratory muscle strength and diaphragm ultrasound: normative values, theoretical considerations, and practical recommendations. Respiration 99(5):369–381
Dos Santos Yamaguti WP, Paulin E, Shibao S, Chammas MC et al (2008) Air trapping: the major factor limiting diaphragm mobility in chronic obstructive pulmonary disease patients. Respirology 13(1):138–144
Ruggeri P, Lo Monaco L, Musumeci O et al (2020) Ultrasound assessment of diaphragm function in patients with late-onset Pompe disease. Neurol Sci 41(8):2175–2184
Qaiser M, Khan N, Jain A (2020) Ultrasonographic assessment of diaphragmatic excursion and its correlation with spirometry in chronic obstructive pulmonary disease patients. Int J Appl Basic Med Res 10(4):256–259
Yamada T, Minami T, Yoshino S et al (2023) Diaphragm ultrasonography: reference values and influencing factors for thickness, thickening fraction, and excursion in the seated position. Lung. https://doi.org/10.1007/s00408-023-00662-2
Ministry of Health Law, Government of Japan. Obesity status (BMI), age group and sex. Accessed 19 July 2023. https://www.mhlw.go.jp/toukei/youran/indexyk_2_1.html. Accessed 19 July 2023
Ho S, Rock K, Addison O, Marchese V (2022) Relationships between diaphragm ultrasound, spirometry, and respiratory mouth pressures in children. Respir Physiol Neurobiol 305:103950
Boussuges A, Rives S, Finance J et al (2021) Ultrasound assessment of diaphragm thickness and thickening: reference values and limits of normality when in a seated position. Front Med 8:742703
Boon AJ, Harper CJ, Ghahfarokhi LS, Strommen JA, Watson JC, Sorenson EJ (2013) Two-dimensional ultrasound imaging of the diaphragm: quantitative values in normal subjects. Muscle Nerve 47(6):884–889
Boussuges A, Finance J, Chaumet G, Brégeon F (2021) Diaphragmatic motion recorded by M-mode ultrasonography: limits of normality. ERJ Open Res 7(1):00714–02020
van Doorn JLM, Wijntjes J, Saris CGJ, Ottenheijm CAC, van Alfen N, Doorduin J (2022) Association of diaphragm thickness and echogenicity with age, sex, and body mass index in healthy subjects. Muscle Nerve 66(2):197–202
Goodpaster BH, Park SW, Harris TB et al (2006) The loss of skeletal muscle strength, mass, and quality in older adults: the health, aging and body composition study. J Gerontol A Biol Sci Med Sci 61(10):1059–1064
Summerhill EM, El-Sameed YA, Glidden TJ, McCool FD (2008) Monitoring recovery from diaphragm paralysis with ultrasound. Chest 133(3):737–743
Combret Y, Prieur G, Hilfiker R et al (2021) The relationship between maximal expiratory pressure values and critical outcomes in mechanically ventilated patients: a post hoc analysis of an observational study. Ann Intensive Care 11(1):8
Zeggwagh AA, Abouqal R, Madani N, Zekraoui A, Kerkeb O (1999) Weaning from mechanical ventilation: a model for extubation. Intensive Care Med 25(10):1077–1083
Vitacca M, Paneroni M, Bianchi L et al (2006) Maximal inspiratory and expiratory pressure measurement in tracheotomised patients. Eur Respir J 27(2):343–349
Acknowledgements
The authors are grateful to Heita Nakayama, Hiroshi Imura, Hiroyuki Ide, Megumi Honda, Namie Hattori, Riko Oda, Sachio Muramatsu, Takuya Tobari, and Yosuke Matsuzaki for their efforts in data collection and management of the research environment. This work was supported by JSPS KAKENHI Grant (Number JP20K16543). The authors thank J. Ludovic Croxford, PhD, from Edanz (https://jp.edanz.com/ac) for editing a draft of this manuscript.
Funding
This work was supported by JSPS KAKENHI Grant (Number JP20K16543).
Author information
Authors and Affiliations
Contributions
Toru Yamada, Taro Minami, Syumpei Yoshino, Ken Emoto, Suguru Mabuchi, and Masayoshi Hashimoto contributed to the design of the study. Toru Yamada, Syumpei Yoshino, Ken Emoto, and Suguru Mabuchi contributed to the data collection. The data were analyzed and interpreted by Toru Yamada, Ryoichi Hanazawa, and Akihiro Hirakawa. Toru Yamada, Taro Minami, and Masayoshi Hashimoto supervised the study. Toru Yamada wrote the first draft with input from Taro Minami. All authors contributed to the writing and review of the main manuscript, had full access to all the data in the study, and had final responsibility for the decision to submit for publication.
Corresponding author
Ethics declarations
Conflicts of interest
Taro Minami is a consultant for FUJIFILM and AA Health Dynamics in relation to a project funded by the Ministry of Economy, Trade and Industry, Japan.
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Tokyo Medical and Dental University (protocol code M2020-112, date of approval: December 21th, 2021).
Consent to Participate
Written informed consent was obtained from all participants involved in the study.
Consent to Publish
The authors affirm that human research participants provided informed consent for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yamada, T., Minami, T., Yoshino, S. et al. Relationship Between Diaphragm Thickness, Thickening Fraction, Dome Excursion, and Respiratory Pressures in Healthy Subjects: An Ultrasound Study. Lung 202, 171–178 (2024). https://doi.org/10.1007/s00408-024-00686-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00408-024-00686-2