Abstract
Purpose
Behavioral evaluation of language development is an important index for the usefulness of cochlear implantation. However, it could not apply to infants and very young children. It is useful to adopt an objective measure to examine speech discrimination in this population. Thus, the current study aimed to predict the different behavioral language performance (good versus poor) in cochlear implant (CI) recipients through the auditory cortical assessment of speech discrimination with mismatch negativity (MMN).
Methods
The study comprised 40 CI children who were divided into two groups according to their behavioral language evaluation outcomes: 20 good and 20 poor CI performers. They were age, gender, and socioeconomically matched. The MMN was examined and compared between both groups with finding out the relationship between MMN and different variables.
Results
MMN existed in all good performers and 87.5% of the poor performers. There were significantly shorter latency, larger amplitude, and a larger area of MMN in the good performers. The MMN results correlated with a significant predictive effect on the behavioral measures of language evaluation.
Conclusion
The MMN is a clinically applicable objective measure of speech discrimination proficiency. Hence, it could be useful in CI programming and auditory cortical monitoring during rehabilitation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
It has been clearly shown that longstanding congenital deafness is difficult to reverse into normal hearing performance. Sensory deprivation causes a permanent effect on the development of the entire central auditory system [1]. However, early cochlear implantation represents a dramatic solution to children with severe hearing impairment. The primary goal of cochlear implantation is to permit speech perception and production [2].
Speech abilities in cochlear implant (CI) recipients are variable and questionable, with the same device and same circumstances [3, 4]. The recovery of auditory abilities after implantation shows a considerable individual variability. In particular, speech intelligibility is extremely variable across individuals [5]. Speech performance scores can reach 70–80% for sentence recognition in a quiet environment but for some CI users, speech perception may remain challenging [6].
Variance in CI outcomes could be attributed to multiple factors. Duration of hearing deprivation and residual hearing was reported to be highly predictive factors related to the variability in speech perception [3, 7]. In addition, hearing aid (HA) use, age of implantation, intelligence quotient (IQ), preoperative radiological assessment showing the survival of spiral ganglion cells, device-related factors, and surgical factors [7, 8]. However, these factors were found to represent only a limited portion of CI outcome variability. A great proportion (up to 80%) could be related to central factors beyond these personal and behavioral features [4, 7]. A comprehensive study of these central factors could provide a thorough understanding of mechanisms underlying the variability in CI outcomes.
Auditory cortical processing of the CI recipients revealed an association with auditory performance outcomes [9]. Clinically, event-related potentials can be used to assess the central mechanisms underlying speech perception in those individuals objectively. Mismatch Negativity (MMN), in particular, may provide insight into the fine discrimination abilities of the central auditory pathway [10]. The measures of MMN were found to have a correlation with psychometric test scores of language development in children [11]. It was reported that MMN amplitude in CI users with good language performance was similar to that of adults with normal hearing [12, 13].
Moreover, psychoacoustic measures may not suitable for difficult-to-test subjects such as infants and very young children especially with the tendency for early implantation. Using an objective electrophysiological method to evaluate cortical auditory processing of speech would be more appropriate to cope with this population. Restricted data are available about the MMN as an objective tool for speech evaluation in CI children with different behavioral language performance [14].
Consequently, the current study aimed to explore the results of MMN in CI children with good versus poor behavioral outcomes to find out the contributing role of auditory cortical processing to individual variability in CI performance. In addition, the study aimed to find out the relationship between both MMN and behavioral measures, yielding some declarative data for the objective evaluation of variable speech discrimination abilities to be applied for CI children who cannot administer behavioral tests.
Methods
Subjects
This comparative cross-sectional study included 40 Arabic-speaking monaural CI children of both genders with an age range of 5–10 years old and variable behavioral performance. The CI recipients were selected pre-operatively following the CI candidacy criteria of the Department of Health, Western Australia [15]. There were absent response on the tonal free field, speech free field audiometry, otoacoustic emissions, and auditory brainstem response. Aided thresholds were higher than the average normal speech spectrum with poor spoken language development despite proper amplification and adequate auditory training over at least 6 months of hearing aid fitting. In addition, high-resolution imaging was studied to determine any anatomical deformities and to examine the patency of the cochlear turns that could interfere with electrode insertion.
All CI recipients fulfilled the following criteria to be involved in the study: a pre-lingual hearing loss with no other disabilities; an average IQ as tested by Stanford-Binet Intelligence Scale; 4th edition [16]; surgery was performed within the critical period for language development [17]; an average aided CI-threshold not exceeding 25 dB HL in the frequency range 0.5–4 kHz; and an implant use duration of 36–60 months with proper CI programming and speech therapy.
Psychoacoustic speech performance scores were used as an index to divide the CI children into good and poor performers following a clinically accepted criterion [13]. Individuals with correct scores equal to or better than 65% using PBKG were considered as good performers (G1: n = 20). Poor performers (G2: n = 20) were defined as having less than 65% correct Word Recognition Score (WRS) on the Arabic Phonetically Balanced Kindergarten (PBKG) words [18] or when they gave no response except on other speech measures including the Arabic Word Intelligibility by Picture Identification (WIPI) test [19] or simple verbal command using simple orders as pointing to body parts, raising a hand, or opening the mouth.
Procedure
The study started with obtaining a detailed history; the socio-economic status (SES) of both CI subgroups using the Socioeconomic Status Scale [20]; the Arabic translated Meaningful Auditory Integration Scale (MAIS) inventory [21, 22]; the aided CI-threshold in the frequency range 0.5–4 kHz, using a calibrated audiometer (Orbiter 922 v. 2; Madsen, Taastrup, Denmark); psychoacoustic speech evaluation; and objective evaluation of speech discrimination using MMN. Other tests were performed including the Stanford-Binet Intelligence Scale (4th edition) to assess IQ and the Arabic edition of “Modified Preschool Language Scale-fourth” (PLS-4) for language evaluation [23].
The Arabic translated Meaningful Auditory Integration Scale (MAIS) questionnaire
This scale included ten questions examining the listening behavior and speech skills of the CI recipients in real-life situations. The parents answered these questions that were modified by the examiner. Each question scored from 0 to 4 where: 0 indicates never; = 1 indicates rarely; 2 indicates occasionally; 3 indicates frequently; and 4 indicates always with a 40 total score [21].
Psychoacoustic speech evaluation
Behavioral evaluation of speech discrimination proceeded according to the child performance using the WRS test, the Arabic WIPI test, and then responding to simple verbal order:
The WRS is an open set speech test of 25 PBKG monosyllabic words suitable for children vocabulary [18]. The child seated in a sound-treated booth facing a loudspeaker located one meter, zero azimuths from the child's head and conducted speech at 40 dB SL. Correct word recognition was given a score of 4% with total correct responses were added together.
The Arabic WIPI test [19] is formed of six lists of 25 monosyllabic words within the child’s vocabulary. Each word is represented graphically in one of 25 pages. Each page contained six pictures from which the child selected the picture representing the target word. A test list was presented at an average conversational level in a face-to-face condition, outside a sound-booth. The children were asked to point to the correct picture. The examiner turned the pages as the child made a selection. A practice item was presented before testing to ensure that each child understood the task. Correct picture identification was scored by 4%. Number of correct identification were added together to calculate the final score.
Responding to the simple verbal command using simple orders as pointing to body parts, raising a hand, or opening the mouth.
Mismatch negativity (MMN)
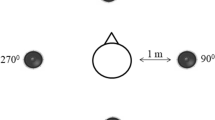
The auditory evoked system, OtoAccess (v 1.3; Eclipse 25; Assens, Denmark) was used to examine the MMN cortical response in a sound-treated room. Two stimuli were presented as an odd-ball paradigm with a frequent tone-burst of 750 Hz and a deviant tone-burst of 1000 Hz, with a probability of 80% and 20%, respectively. The sound was delivered through a loudspeaker located one meter at zero azimuth from the subject's head, at a level of 70 dB nHL. Responses were obtained with a non-inverting electrode placed in the middle of the forehead, an inverting electrode placed over the mastoid of the non-implanted ear to avoid electric artifact from the CI and a ground electrode placed on the forehead just below the non-inverting one.
Recording was carried out while the child was watching a movie on a laptop placed above the loudspeaker to distract his/her attention to the stimulus. The picked-up potentials were filtered with a band-pass filter of 0.83–33 Hz and presented in a time window of 600 ms with a pre-stimulus interval of − 50 ms. The MMN was presented as a negative trough in the difference wave obtained by subtracting the frequent from the deviant responses. Response parameters involved latency, amplitude, duration, and area of the negative trough located nearly after 250 ms [22].
The modified Preschool Language Scale fourth (PLS-4); Arabic edition
The modified PLS-4 was used to examine several aspects of the receptive and expressive language skills were evaluated, including attention, play, gesture, vocal development, social communication, vocabulary, concepts, language structure, integrative language skills, and phonological awareness. Scoring involved age-related subscales: receptive, expressive, and total language age (TLA) with an upper limit of language development of 6 years and 11 months (83 months) [23].
Statistical analysis
The data were analyzed using the SPSS 22.0 software. The normality of different variables was assessed using Shapiro–Walk test. The continuous data were presented as mean ± Standard deviation (SD) while the categorical data were presented as frequencies and percentages. The normally distributed data were compared using the parametric tests (t test and one-way ANOVA test) whereas the abnormally distributed data were compared using the non-parametric test (Mann–Whitney (U) test and Kruskall–Wallis H test). The distribution of categorical variables was compared using χ2 or Fisher’s exact tests when appropriate.
Correlation coefficient was performed with Person's correlation test for parametric testing and with Spearman’s correlation test for non-parametric testing. Linear regression analysis was applied to find out the predictor effect of different significantly correlated variables on MMN. The level of significance (p) was set at < 0.05. To ensure the validity of the statistical analysis and the strength of significance, the degrees of freedom (df) and Effect size (ES) were evaluated, respectively.
Results
Table 1 represents the personal and CI device criteria of both study groups. The good and poor CI performers were matching in age, sex, and SES. Chi-square test revealed a significantly different distribution of the causes of hearing loss, but with a small ES (0.03). The heredofamilial etiology was the commonest among the two groups. Other etiologies such as meningitis and hypoxia dominated in the poor CI performers. Both groups had a comparable duration of HA usage, age at implantation, duration of implant, and distribution of the CI devices and speech strategies.
As shown in Table 2, the two groups exhibited equivalent average aided response and IQ. Other behavioral measures including WRS, MAIS, and language age were significantly higher with df = 38 and large ES in the G1 group. In both study groups, the different behavioral measures of language evaluation and IQ revealed significant inter-correlations (Table 3).
Figure 1 represents the MMN incidence in both groups and revealed a non-significant difference (Fisher’s exact = 0.154, p > 0.05). There were significantly shorter latency, larger amplitude, and a larger area of MMN with df = 38 and large ES in G1 as compared to the G2 group, but the MMN duration did not differ significantly (Table 4). Figure 2 illustrates examples of MMN response from a good CI performer with shorter latency (panel a) versus a poor CI performer (panel b).
Example of (a; upper trace) MMN from a 7-year-old boy with right-sided CI and good language results, and (b; lower trace) MMN from a 10-year-old girl with right-sided CI and poor language results. Each trace contains three waves: the upper (f) is a response to a frequent stimulus; the middle (r) is a response to rare stimulus; the lower is a difference wave (MMN)
On studying the relationship between different factors and MMN outcomes (Table 5); personal and implantation features did not show any significant relationship in both groups; aided response had a significant correlation with all MMN measures except the duration in G1 group; and other behavioral measures (WRS, IQ, MAIS, and language age) exhibited significant correlations with most of the MMN measures in the two groups. Linear regression analysis was applied to the significant correlations and revealed an overall significant predictive effect of the MMN outcomes on the aided response (in G1) and behavioral measures (in both groups) (p < 0.001).
Discussion
Psychoacoustic language evaluation in CI children
The behavioral measures of language development are the fundamental modalities that provide a comprehensive description of linguistic skills and auditory performance. Despite the comparable age, audibility, auditory experience, intelligence, and SES of the study groups, the behavioral measures of language evaluation were significantly lower in poor than good performers. Previous behavioral studies showed similar findings in CI users [24, 25]. The significant inter-correlation among these measures reflects the parallel central auditory maturation integrated with the peripheral auditory stimulation through CIs.
Auditory cortical processing in good versus poor-performing CI children
This study aimed to investigate the cortical auditory processing of speech in CI children with different behavioral language outcomes. The inclusion criteria of the participants of both study groups established analogous personal and implantation factors. Therefore, it would be possible to estimate a confined contribution of central auditory processing to language development in CI children. In this study, MMN recorded in all good CI performers and 87.5% of poor CI performers. The literature presented the incidence of MMN with considerable variability.
Some investigators used tonal contrasts to elicit MMN in good versus poor CI performers to examine the auditory cortical processes consistent with their speech discrimination. In postlingual hearing-impaired adults with CI, the MMN existed in five out of six ears (83.3%) with good performance and in only one out of six ears (16.7%) with moderate-to-poor performance [26]. Kelly [27] reported a reduced or absent MMN in poor (n = 4) as compared to good performers (n = 8) using a two-deviant tonal paradigm in 12 adults with CI.
More complex stimulus paradigms were also used to record MMN in CI users with different behavioral speech outcomes. Kraus et al. [10] and Groenen et al. [12] presented a speech paradigm that elicited MMN in all adult CI participants with preferable speech outcomes but not in any of the poor performers. In 35 CI children (7–17 years), speech stimuli produced MMN in 80–85% of good performers and in 15–20% of poor performers [28]. Using a two-deviant speech paradigm, Turgeon et al. [13] recorded MMN from all adults with CI, either good (n = 10) or poor (n = 10) performers; however, the MMN in the poor performers had smaller amplitude and longer latency.
The different findings in these studies may be due to the variability in age, sample size, stimulus parameters, and equipment used. Despite the different outcomes, there was a general agreement that the CI users with proficient behavioral speech results showed a prevalent MMN response. Those with poor behavioral results presented a degraded or even absent MMN. Consequently, the incidence of MMN represents the underlying neurophysiologic mechanisms responsible for speech discrimination that reflect and go parallel with the outcomes of psychoacoustic speech evaluation.
Additional findings in this study were significantly shorter latency, larger amplitude, and a larger area of MMN in good versus poor CI children. These data are consistent with previous studies, which have shown a tendency of good CI performers to exhibit larger MMN amplitude, approximating that of normal-hearing individuals [15, 26, 27, 29]. The significant differences of MMN measures between good and poor performers suggest different pre-attentive speech discrimination capabilities that are impaired in the poor performers. Hence, MMN can be used as an objective measure to categorize speech discrimination dexterity in CI subjects especially in those who are difficult to test behaviorally [28, 30].
Factors determining the proficiency of language development in CI children
Despite the comparable personal and implantation criteria, CI users in the current study had variable language skills (good versus poor) as revealed with the behavioral and MMN measurements. Researchers suggested several factors that determine the proficiency of language development in CI individuals. Longstanding hearing impairment prior to cochlear implantation causes auditory deprivation with a negative impact on language development, especially in the pediatric population [31, 32]. The auditory deprivation interferes with proper auditory cortical maturation [26] and cognitive function evolution [33,34,35]. Previous studies provided evidence of cross-modal auditory cortical reorganization following a prolonged auditory deprivation where other sensations such as vision [4, 29, 36] and touch [37] replace the hearing sensation. Thus, the ability to process auditory cues provided with the CI is markedly impaired within the reduced auditory temporal cortex [38].
Eppsteiner et al. [39] suggested that the gene mutations in genetic hearing loss could affect the proficiency of language outcomes in CI recipients. They reported TMPRSS3 gene mutations in the spiral ganglion of a poor performer and LOXHD1 gene mutations in the membranous labyrinth of two good performers. Further, bacterial meningitis can impair the auditory performance in CI individuals due to different cochlear and central auditory lesions, depth of electrode insertion, and number of active electrodes [40]. It is the case in this study where the meningitis is among the causes of hearing loss in the poor CI performers.
Relationship between different variables and auditory cortical processing in the CI subgroups
Another important aim of this study was to find out the relationship between different subjective criteria and MMN. The different personal and implantation data showed no significant association with the outcomes of MMN recording. This could be attributed to the inclusion criteria of the studied CI children involving a limited range of implantation age, implantation duration, and appropriate rehabilitation. Some investigators found significant relationships between the MMN latency and subjects’ age [41], MMN latency and age at implantation [32], and MMN latency and amplitude and duration of HA use [42]. These differences in outcomes relative to that of the current study could be attributed to the recording of MMN early after implantation [41, 42] and to different stimulus parameters (e.g. smaller frequency deviance [32].
The aided response correlated with MMN parameters in good performers but not in poor ones. This indicates the presence of factors contributing to impaired performance other than the impaired cortical auditory processing in poor performers as mentioned in the previous section. The MMN results correlated with a significant predictive effect on IQ and behavioral measures of language evaluation. Similar findings were also reported in the previous studies [13, 41, 43, 44]. Thus, MMN could provide an objective indicator of speech discrimination for proper CI programming, central auditory plasticity monitoring, and follow-up during auditory rehabilitation.
Conclusion
This study showed that the assessment of cortical auditory processing could reflect the behavioral language outcomes in CI children. There was a tendency of good CI performers to have a shorter latency, larger amplitude, and a larger area of MMN than the poor ones. These results suggested that the electrophysiological MMN recording provided an objective assessment of speech discrimination. Hence, it could be an easily applied determinant of CI proficiency in difficult-to-test subjects such as infants and young children. Given the predictive effect of MMN on the behavioral tests of a language evaluation, it could be a clinically useful tool in CI programming, monitoring, and follow-up during rehabilitation programs.
References
Kral A, Sharma A (2012) Developmental neuroplasticity after cochlear implantation. Trends Neurosci 35:111–122
Kral A, O'Donoghue GM (2010) Profound deafness in childhood. N Engl J Med 363(15):1438–1450
Blamey P, Artieres F, Başkent D et al (2013) Factors affecting auditory performance of postlinguistically deaf adults using cochlear implants: an update with 2251 patients. Audiol Neurootol 18(1):36–47
Anderson CA, Lazard DS, Hartley DEH (2017) Plasticity in bilateral superior temporal cortex: effects of deafness and cochlear implantation on auditory and visual speech processing. Hear Res 343:138–149
Summerfield AQ, Marshall DH (1995) Cochlear implantation in the UK 1990–1994. Report by MRC institute of hearing research on the evaluation of the national cochlear implantation program. HMSO Publication Centre, London
Garnham C, O’Driscoll M, Ramsden R et al (2002) Speech understanding in noise with a Med-El COMBI 40+ cochlear implant using reduced channels sets. Ear Hear 23:540–552
Lazard D, Vincent C, Venail F et al (2012) Pre-, per- and postoperative factors affecting performance of postlinguistically deaf adults using cochlear implants: a new conceptual model over time. PLoS ONE 7(11):e48739
Holden LK, Finley CC, Firszt JB et al (2013) Factors affecting open-set word recognition in adults with cochlear implants. Ear Hear 34(3):342–360
Blamey PJ, Arndt P, Bergeron F (1996) Factors affecting auditory performance of postlinguistically deaf adults using cochlear implants. Audiol Neuro Otol 1:293–306
Kraus N, Micco AG, Koch DB et al (1993) The mismatch negativity cortical evoked potential elicited by speech in cochlear-implant users. Hear Res 65:118–124
Huotilainen M, Lovio R, Kujala T et al (2011) Could audiovisual training be used to improve cognition in extremely low birth weight children? Acta Paediatr 100:1489–1494
Groenen P, Snik A, van den Broek P (2011) On the clinical relevance of mismatch negativity: results from subjects with normal hearing and cochlear implant users. Audiol Neuro Otol 1(1):112–124
Turgeon C, Lazzouni L, Lepore F et al (2014) An objective auditory measure to assess speech recognition in adult cochlear implant users. Clin Neurophysiol 125:827–835
Dinces E, Chobot-Rhodd J, Sussman E (2009) Behavioral and electrophysiological measures of auditory change detection in children following late cochlear implantation: a preliminary study. Int J Pediatr Otorhinolaryngol 73:843–851
Department of Health, Western Australia (2011) Clinical guidelines for adult cochlear implantation. Health Networks Branch, Department of Health, Western Australia, Perth
Terman L, Merrill M (1972) Stanford binet intelligence scale. Highton, Mifflin Company, Boston
Sharma A, Campbell J (2011) A sensitive period for cochlear implantation in deaf children. J Matern Fetal Neonatal Med 1:51–53
Soliman S (1976) Speech discrimination audiometry, using Arabic phonetically Balanced 30 words. Ain Shams Medical J 1:24–30
Elkholi W, Soliman S, Kamal N (1991) Speech discrimination using Arabic word intelligibility by picture identification (WIPI) test in children with sensori-neural hearing loss. Master Thesis
Fahmy S, Nofal L, Shehata S et al (2015) Updating indicators for scaling the socioeconomic level of families for health research. J Egypt Public Health Assoc 90:1–7
Robbins AM, Renshaw JJ, Berry SW (1991) Evaluating meaningful auditory integration in profoundly hearing impaired children. Am J Otol 12:144–150
Zein D, Ismail N, Mobasher M et al (2018) Outcomes of pediatric cochlear implantation contributing factors. MD thesis. El Azhar University
Abo Hasseba A, El Sady S, El Shoubary A et al (2011) Standardization, translation and modification of the preschool language scale. MD thesis
Marchman VA, Fernald A (2008) Speed of word recognition and vocabulary knowledge in infancy predict cognitive and language outcomes in later childhood. Dev Sci 11:F9–F16
Cejas I, Mitchell CM, Hoffman M et al (2018) Comparisons of IQ in children with and without cochlear implants: longitudinal findings and associations with language. Ear Hear 39(6):1187–1198
Zhang F, Hammer T, Banks HL et al (2011) Mismatch negativity and adaptation measures of the late auditory evoked potential in cochlear implant users. Hear Res 275:17–29
Kelly AS (2005) Electrophysiological and speech perception measures of auditory processing in experienced adult cochlear implant users. Clin Neurophysiol 116:1235–1246
Singh S, Liasis A, Rajput K et al (2004) Event-related potentials in pediatric cochlear implant patients. Ear Hear 25(6):598–610
Ortmann M, Knief A, Deuster D et al (2013) Neural correlates of speech processing in prelingually deafened children and adolescents with cochlear implants. PLoS ONE 8:e67696
Näätänen R, Petersen B, Torppa R et al (2017) The MMN as a viable and objective marker of auditory development in CI users. Hear Res 353:57–75
Ismaail NM, Shalapy AA, Ibraheem OA et al (2019) Temporal resolution in school-age children with cochlear dead regions. J Am Acad Audiol. https://doi.org/10.3766/jaaa.18004
Hamid M, Kotait MA, Kolkaila EA (2019) Mismatch negativity in children with cochlear implant. Int J Otorhinolaryngol Head Neck Surg 5(5):1149–1155
Wass M, Ibertsson T, Lyxell B et al (2008) Cognitive and linguistic skills in swedish children with cochlear implants—measures of accuracy and latency as indicators of development. Scand J Psychol 49:559–576
Kral A, Kronenberger W, Pisoni D et al (2016) Neurocognitive factors in sensory restoration of early deafness: a connectome model. Lan Neuro 15(6):610–621
Ibraheem OA, Abdelghani M, Hassan EM et al (2020) Cognitive functions in cochlear-implanted children: a multidisciplinary approach (in press)
Ortmann M, Zwitserlood P, Knief A et al (2017) When hearing is tricky: speech processing strategies in prelingually deafened children and adolescents with cochlear implants having good and poor speech performance. PLoS ONE 12(1):e0168655
Jr A, Bernstein LE, Sungkarat W et al (2007) Vibrotactile activation of the auditory cortices in deaf versus hearing adults. NeuroReport 18(7):645–648
Sandmann P, Dillier N, Eichele T et al (2012) Visual activation of auditory cortex reflects maladaptive plasticity in cochlear implant users. Brain 135:555–568
Eppsteiner RW, Shearer AE, Hildebrand MS et al (2012) Prediction of cochlear implant performance by genetic mutation: the spiral ganglion hypothesis. Hear Res 292(1–2):51–58
de Brito R, BittencourtGomez AGMV et al (2013) Cochlear implants and bacterial meningitis: a speech recognition study in paired samples. Int Arch Otorhinolaryngol 17(1):57–61
Liang MJ, Zhang XY, Chen T et al (2014) Evaluation of auditory cortical development in the early stages of post cochlear implantation using mismatch negativity measurement. Otol Neurotol 35(1):e7–14
Li Y, Shen M, Long M (2019) A preliminary study of auditory mismatch response on the day of cochlear implant activation in children with hearing aids prior implantation. PLoS ONE 14(1):e0210457
Kileny PR, Boerst A, Zwolan T (1997) Cognitive evoked potentials to speech and tonalstimuli in children with implants. Otolaryngol Head Neck Surg 1117:161–169
Gabr T (2018) Mismatch negativity (MMN): indexing auditory discrimination in cochlear implants. Hear Balance Commun 16(1):21–28
Funding
None.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, study procedures, data collection and analysis were performed by OAI and NHG. The first draft of the manuscript was written by OAI and NHG. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declared that there was no conflict of interest.
Ethical approval
All the study procedures were performed using non-invasive testing, following the guidelines of the International Review Board of Zagazig University with an ID: 5613-29-9-2019.
Informed consents
Study took place after explaining the procedures to the participants/parents and obtaining informed consent the parents of the participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ibraheem, O.A., Kolkaila, E.A., Nada, E.H. et al. Auditory cortical processing in cochlear-implanted children with different language outcomes. Eur Arch Otorhinolaryngol 277, 1875–1883 (2020). https://doi.org/10.1007/s00405-020-05958-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00405-020-05958-0