Abstract
Cannabidiol (CBD), which is derived from hemp, is gaining recognition because of its anti-inflammatory and lipid-modulating properties that could be utilized to treat acne. We conducted experiments to quantitatively assess the effects of CBD on acne-related cellular pathways. SEB-1 sebocytes and HaCaT keratinocytes were exposed to various CBD concentrations. CBD exhibited a concentration-dependent impact on cell viability and notably reduced SEB-1 viability; furthermore, it induced apoptosis and a significant increase in the apoptotic area at higher concentrations. Additionally, CBD remarkably reduced pro-inflammatory cytokines, including CXCL8, IL-1α, and IL-1β. Additionally, it inhibited lipid synthesis by modulating the AMPK-SREBP-1 pathway and effectively reduced hyperkeratinization-related protein keratin 16. Simultaneously, CBD stimulated the synthesis of elastin, collagen 1, and collagen 3. These findings emphasize the potential of CBD for the management of acne because of its anti-inflammatory, apoptotic, and lipid-inhibitory effects. Notably, the modulation of the Akt/AMPK-SREBP-1 pathway revealed a novel and promising mechanism that could address the pathogenesis of acne.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acne is a highly prevalent disease with a peak incidence during the teenage years and the potential to persist into the third decade of life; furthermore, it ranks as the eighth most prevalent disease worldwide [1, 2]. Although the exact cause of acne remains unknown, it is believed to result from a combination of several factors rather than a single cause. The pathophysiology of acne can be broadly categorized into four factors, abnormal follicular keratinization, increased sebum production, the inflammatory response, and the presence and activity of Cutibacterium acnes bacteria, which are considered significant contributors to this condition [3]. C. acnes colonizes at the pilosebaceous units, specifically in regions densely populated with sebaceous follicles, which create a lipid-rich environment that serves as a pathological hallmark of this skin condition [4]. Various treatments have been attempted for active acne lesions and scars, but their effectiveness has been disappointing, resulting in significantly higher levels of perceived stigmatization for individuals with acne than for those with healthy skin [5].
In 2019, the Ministry of Food and Drug Safety of Korea implemented the “Amendment to the Narcotic Control Act,” which permitted the domestic importation of hemp-based drugs licensed abroad, including cannabidiol (CBD), which is the main nonpsychotropic substance found in hemp (Fig. 1). This progressive move was aimed at broadening treatment opportunities for rare and intractable diseases in Korea. CBD has demonstrated dose-dependent inhibition of excessive lipid synthesis and anti-proliferative and anti-inflammatory effects on sebaceous gland cells [6]. In another study involving human keratinocyte cell lines, the application of a 0.6% hemp seed hexane extract solution demonstrated a reduction in C. acnes-induced inflammation by suppressing pro-inflammatory cytokines, specifically IL-1β and IL-8 [7]. Additionally, CBD has been shown to inhibit keratinocyte hyperproliferation in vitro through multiple receptors [8]. In vivo experiments have also shown promising results. During a 12-week split-body trial involving patients with psoriasis, the use of topical 2.5% CBD ointment and placebo twice daily resulted in significant improvements, especially in scaling, without any major adverse effects [9]. Therefore, CBD could be an effective treatment for acne by targeting potential pathophysiological mechanisms.
To advance our understanding of the effects of CBD on cell lines, this study aimed to examine the potential of CBD to treat and prevent acne by exploring specific pathways, such as the intracellular inflammatory response and triglyceride reduction, in sebocytes. Additionally, this study sought to introduce a fresh approach to acne treatment and move beyond conventional methods. Through comprehensive analyses, this innovative approach could provide more effective treatment options, particularly for patients susceptible to inflammation and scarring.
Materials and methods
Test material and culture
CBD
CBD was prepared at concentrations of 0.1, 1, 5, 10, 50, and 100 µM (Kolmar Korea Co., Ltd, Seoul, Republic of Korea).
Cell culture and preparation
The SEB-1 immortalized human sebocyte cell line, which is a transformed cell line generated by transfection with the SV40 large T antigen, has been mentioned in many reports of acne-related studies [10, 11]. Passage 28 SEB-1 immortalized human sebocytes were used in all experiments. The standard culture medium for growing SEB-1 cells consisted of 5.5 mM glucose supplemented with Ham’s F-12 (3:1) (Invitrogen, Carlsbad, CA, USA), 2.5% fetal bovine serum (HyClone, Logan, UT, USA), adenine (180 µM) (Sigma, St. Louis, MO, USA), hydrocortisone (0.4 µg/ml), insulin (10 ng/ml), epidermal growth factor (3 ng/ml) (Austral Biologicals, San Ramon, CA, USA), and cholera toxin (0.12 nM) (Sigma). Serum-free sebocyte medium used in lipogenesis experiments consisted of DMEM containing 5.5mM glucose and antibiotics without any other additives [12].
The Detroit 551 cell (human embryonic skin fibroblast cell line, ATCC CCL-110) was maintained in Eagle’s minimum essential medium (EMEM) and Dulbecco’s modified Eagle’s medium (Sigma, St. Louis, MO, USA) plus 10% fetal bovine serum (FBS) (Invitrogen, San Diego, CA, USA), at 37 °C and in a humidified atmosphere containing 5% CO2 [13]. The HaCaT cell (immortalized human keratinocyte line) was cultured as monolayers in a standard culture medium (DMEM supplemented with 5% fetal calf serum, 2 mM glutamine, 100 IU/ml penicillin) at 37 °C in a humidified atmosphere containing 5% CO2.
Cytotoxicity analysis
SEB-1 cells were treated with different concentrations of CBD and cultured for 24 h, 48 h, and 72 h. After the respective incubation periods, 10 µl of CCK8 solution (Dojindo, Rockville, MD, USA) was added to each well. After a 1-hour reaction at 37 °C, cytotoxicity was assessed by measuring absorbance at 450 nm using a spectrophotometer. To evaluate the potential cytotoxic effects of CBD on other cell lines, HaCaT cells were subjected to similar treatments, and their respective cytotoxicity levels were determined. ApopTag peroxidase in situ apoptosis detection (cell TUNEL stain) was used to assess whether apoptosis was induced.
Inhibitory concentration of CBD on C. acnes
C. acnes (ATCC 6919), the causative microorganism of acne, was purchased from the Korea Microbial Strains Bank (Seoul, Republic of Korea). These strains were cultured in reinforced clostridium medium (Becton Dickinson, Franklin Lakes, NJ, USA) at 37 °C under anaerobic conditions. Before using the strains in the experiments, C. acnes was cultured in tryptic soy broth (BD) medium supplemented with 5% (volume/volume) defibrinated sheep blood at 37 °C until reaching an OD600 of 1.0 (logarithmic growth phase). The strains that reached the log value were filtered after centrifugation at 5,000 g and 4 °C for 15 min; then, they were heat killed at 95 °C for 20 min before use. C. acnes strains at a concentration of 2 × 107 CFU/ml were prepared in liquid medium. Different concentrations of CBD were added to 96-well plates. Then, plates were incubated with C. acnes strains for 48 to 72 h under anaerobic conditions. The suppression of C. acnes was confirmed by assessing the reduction of C. acnes using absorbance measurements.
Inflammatory cytokine analysis
To confirm the anti-inflammatory effect of CBD, an inflammatory cytokine quantitative PCR was performed after inducing inflammation in SEB-1 cells by adding C. acnes or lipopolysaccharide (LPS). The analysis was conducted using a real-time PCR and a 384-well instrument (LightCycler® 480; Roche, Indianapolis, IN, USA) with SYBR® Green quantitative PCR Master (Applied Biosystems, Foster City, CA, USA) for GAPDH, TNF-α, IL-1α, IL-1β, IL-8, and IL-6.
Lipogenesis analysis
After treating SEB-1 sebocytes with CBD at concentrations of 5 and 10 µM for 48 h, proteins and pathways involved in lipid synthesis were evaluated using a Western blot analysis. To understand the potential mediators of CBD-induced SREBP-1 inhibition, AKT and AMPK, which are both known to regulate SREBP-1, were studied [14, 15]. The antibodies used included β-actin mouse antibody (Santa Cruz Biotechnology, Santa Cruz, CA, USA), SREBP-1 rabbit antibody (Santa Cruz Biotechnology), phospho-AMPKα1/2 rabbit antibody (Abcam, Cambridge, MA, USA), and phospho-AKT Thr308 (Cell Signaling Technology, Beverly, MA, USA). HMG CoA synthase and HMG CoA reductase, which are related to the triglyceride subpathway, were analyzed by performing a real-time PCR with a 384-well instrument and SYBR® Green quantitative PCR Master as mentioned.
SEB-1 cells were seeded on four-well culture slides at 5 × 104 cells per well. After 48 h, they were washed with phosphate-buffered saline and fixed with 10% formalin for 10 min. SEB-1 cells were stained using the NovaUltra Oil red O stain kit (IHC World, Woodstock, MD, USA) and observed under a microscope at 400× magnification to verify differences in lipogenesis.
Immunocytochemistry staining intensity was quantified using a fluorescence microscope with the NIS elements BR imaging system (version 4.30; Nikon, Minato, Japan), and an analysis was performed using the TINA densitometric program (Raytest Isoto-penmebgerate, Straubenhardt, Germany).
Hyperkeratinization-related protein
The change in keratin 16 was assessed through western blot and mRNA analyses using keratin 16 rabbit antibody (Santa Cruz Biotechnology) after subjecting HaCaT cells to 0-, 5-, or 10-µM CBD treatment for 24 h. To ensure accurate protein quantification, the proportion of keratin 16 to β-actin was used for normalization, thereby maintaining consistency in protein loading across the gel.
Scar formation-related protein synthesis of fibroblasts
The changes in collagen 1, collagen 3, and elastin in fibroblasts were assessed by performing a western blot analysis, mRNA analysis, and DAPI staining. Proteins were extracted using cell lysis buffer (Cell Signaling Technology), and their contents were determined using the BCA Protein Assay (Pierce, Rockford, IL, USA). Equal protein amounts were separated on 10% SDS-PAGE gels and transferred to a membrane. Antibodies against collagen 1, collagen 3, and elastin were applied after exposing the fibroblast cells to 0-, 1-, or 5-µM CBD treatment for 48 h. To ensure accurate quantification, ratios of protein to β-actin were used for normalization. Immunocytochemistry staining was performed by using the same method as that used for the lipogenesis analysis.
Statistical analysis
The results are expressed as the mean and standard deviation of three independent experiments. A statistical significance test of the control group and experimental group was performed using SPSS (version 27.0; Armonk, NY, USA), and P < 0.05 was considered statistically significant. Data were evaluated using a one-way analysis of variance and GraphPad Prism (version 5.0) software to determine statistical significance between treatments.
Results
Assessment of the toxic effects of CBD on SEB-1 and HaCaT cells
To investigate the temporal effects of CBD on the viability of SEB-1 and HaCaT cells, a CCK8 assay was performed (Fig. 2a). When treated with CBD, both SEB-1 and HaCaT cells exhibited a significant reduction in cellular growth. When subjected to a CBD concentration of 1 µM, SEB-1 cells had an average viability rate of 98.2%; therefore, the difference was not statistically significant. Viability rates of SEB-1 cells subjected to CBD concentrations of 5 µM, 10 µM, and 20 µM were 92.4%, 63.7%, and 37.7%, respectively, at 24 h. These rates further decreased to 85.5%, 62.1%, and 10.7%, respectively, at 48 h, and 85.9%, 59.8%, and 9.5%, respectively, at 72 h. The CBD concentration appeared to have a greater inhibitory effect on proliferation. HaCaT cells also exhibited a concentration-dependent response; however, they showed a higher threshold of inhibition with a CBD concentration of 10 µM. Furthermore, after 72 h, the rates of HaCaT cell viability after the application of CBD concentrations of 5 µM, 10 µM, and 20 µM were 104.5%, 68.4%, and 22.2%, respectively.
A TUNEL (terminal deoxynucleotidyl transferase dUTP nick end labeling) assay was performed to confirm apoptosis of SEB-1 cells treated for 48 h with CBD concentrations of 5 µM or 10 µM (Fig. 2b). At a concentration of 5 µM, the area of apoptosis was 20.05%, and it was 36.65% at a concentration of 10 µM; however, it was 1% for the control group (P < 0.001 compared to baseline, respectively).
CBD induces apoptosis in SEB-1 cells and HaCaT cells. (a) SEB-1 cells and HaCaT cells were exposed to CBD at concentrations of 1, 5, 10 or 20 µM for 48 h, followed by confirmation of apoptosis using the TUNEL assay method. (b) Quantitative analysis of apoptosis staining was conducted and the results were graphed. Representative micrograph images of SEB-1 cells treated with CBD showing the induction of apoptosis (400X magnification). Data are presented from a sample size of n = 9 independent experiments. All are at the same magnification (bar = 50 μm). CBD, cannabidiol; TUNEL, terminal deoxynucleotidyl transferase dUTP nick end labeling. *, P < 0.05, †, P < 0.001
Effects of CBD on LPS-induced or C. acnes-induced inflammation of SEB-1 cells
CBD was treated in conjunction with C. acnes or LPS at concentrations of 0, 5, and 10 µM, followed by a 24-hour incubation period for the protein and mRNA analyses. Decreases observed in the mRNA levels of CXCL8, IL-1α, and IL-1β corresponded to the escalating concentrations of CBD (Fig. 3a). The relative level of CXCL8 increased to 7.12 after incubation with C. acnes. However, with the addition of 5 µM and 10 µM of CBD, the levels decreased to 5.00 and 3.81, respectively (P < 0.05 for both). Regarding IL-1α and IL-1β, the levels decreased from 3.79 to 2.44 and 1.82, 2.82 to 2.50 and 1.86 with CBD concentrations of 0, 5, and 10 µM (all P < 0.05). These reductions were consistently evident during the protein analysis and western blot analysis, in which both CXCL8 and TNF-α cytokines exhibited dose-dependent reductions with CBD treatment (Fig. 3b).
CBD modulates inflammatory cytokine expression in SEB-1 cells exposed to C. acnes. SEB-1 cells were treated with CBD at concentrations of 0 µM, 5 µM, and 10 µM in the presence of C. acnes to assess changes in inflammatory cytokines. (a) Quantitative real-time PCR validated reduced mRNA expression levels of CXCL8, IL-1α, IL-1β, and TNFα post-CBD treatment compared to the untreated control. (b) Western blot analysis quantified protein levels of CXCL8 and TNFα. Quantified data were graphically represented, showcasing decreased protein levels in response to CBD treatment. CBD, cannabidiol; C. acnes, Cutibacterium acnes. *, P < 0.05
Effects of CBD on lipogenesis via the AMPK-SREBP-1 signaling pathway in SEB-1 sebocytes
The lipid-inhibitory effect of CBD was confirmed through the regulation of AMPK-SREBP-1 signaling using western blotting and quantitative polymerase chain reaction (PCR) methods. CBD activated AMPK and reduced SREBP, PPARγ, and lipid production in SEB-1 sebocytes (Fig. 4a). These findings revealed a significant decrease in SREBP-1 mRNA as CBD concentrations increased (0.87 and 0.66, respectively) compared to the baseline values with 5 and 10 µM CBD (P < 0.05) (Fig. 4b). This decrease was accompanied by a reduction in PPARγ and increase in p-AMPKα levels (0.85 and 0.57 for PPARγ and 1.52 and 2.10 for p-AMPKα, respectively) compared to the baseline levels with 5 and 10 µM CBD (all P < 0.05) (Fig. 4b). To validate the involvement of the AMPK pathway in this effect, treatment with an AMPK inhibitor, compound C, was conducted, which reversed the effect of CBD on SREBP-1, PPARγ, and p-AMPKα (1.00 and 1.13, 1.09 and 1.08, and 1.05 and 1.09, respectively) compared to the baseline values with 5 and 10 µM CBD (Fig. 4b). This suggests that CBD may decrease SREBP-1 by inhibiting PPARγ and increasing p-AMPKα, ultimately leading to a reduction in lipid synthesis in sebocytes through the Akt/AMPK-SREBP-1 signaling pathway. In SEB-1 sebocytes exposed to C. acnes, no significant increase in the expression of genes associated with lipogenesis was observed. However, similar to SEB-1 cells not exposed to C. acnes, exposure to CBD showed a trend of inhibiting gene expression.
Effect of CBD on lipogenesis pathway in SEB-1 cells. CBD reduces SEB-1 lipogenesis by inhibiting the expression of sterol regulatory element binding protein (SREBP)-1 through the AMP-activated protein kinase (AMPK) pathway. (a) SEB-1 cells were pretreated by CBD with or without 20 µM of compound C for 24 h before western blot. Levels of phospho-/total AMPK, PPARγ, and SREBP1 were measured. (b) Western blot bands of SREBP-1/β-actin, PPARγ/β-actin, and phospho-/total AMPKα were quantified and presented in a graph. (c) Oil red staining analysis was performed and graphed (400X). All are at the same magnification (bar = 50 μm). CBD, cannabidiol; SREBP, sterol regulatory element binding protein; AMPK, AMP activated protein kinase; PPARγ, peroxisome proliferator-activated receptor gamma. *, P < 0.05; †, P < 0.001
To confirm the inhibition of lipogenesis, an Oil Red O staining analysis was performed to assess the area of red lipid droplets. The lipid droplet area decreased to 84.6% compared to the baseline values after 48 h with 5-µM CBD treatment, and it further decreased to 33.5% after 48 h with 10-µM CBD treatment, demonstrating concentration-dependent inhibition (P < 0.05 and P < 0.001, respectively) (Fig. 4c).
DAPI (4′,6-diamidino-2-phenylindole) staining has shown corresponding results, indicating that CBD increases p-AMPKα and inhibits SREBP-1, and that compound C was able to reverse this action (Fig. 5). The green fluorescence signals of p-AMPKα and SREBP-1 were quantified using ImageJ software. The signal intensity divided by the cell number per field of p-AMPKα was 0.52 when treated with 0 μM CBD, significantly increasing to 0.76 with 10 μM CBD treatment (P<0.05). However, this value decreased to 0.36 after subsequent treatment with 20 μM compound C, even lower than that of the baseline. For SREBP-1, the signal intensity divided by the cell number per field was 0.35 upon treatment with 0 μM CBD, but significantly dropped to 0.26 with 10 μM CBD treatment (P<0.05). Treatment with 20 μM compound C with CBD 10 μM showed a signal intensity of 0.61, indicating the reversal of CBD’s effects, even higher than default value.
Representative DAPI staining images of CBD treated SEB-1 cells. Representative DAPI staining images of CBD treated SEB-1 cells. SEB-1 cells were treated with CBD at concentrations of 0 µM and 10 µM for a duration of 48 h. DAPI staining was performed to assess the impact of CBD treatment. Phosphorylated AMPKα expression and SREBP-1 expression were evaluated using DAPI staining (400X). All are at the same magnification (bar = 50 μm). CBD, cannabidiol; SREBP, sterol regulatory element binding protein; AMPK, AMP activated protein kinase; DAPI, 4’,6-diamidino-2-phenylindole
Effects of CBD on hyperkeratinazation-related protein keratin 16 using HaCaT cells
HaCaT cells were treated with CBD concentrations of 0, 5, and 10 μM and underwent western blot and quantitative real-time mRNA analyses to assess alterations in hyperkeratinization-related protein keratin 16. The western blot analysis revealed that keratin 16 decreased to 0.99 and 0.73 after 5- and 10-μM CBD treatments compared to baseline values (P = 0.89 and P = 0.01, respectively) (Fig. 6a). In line with the western blot analysis results, the quantitative real-time method also indicated a decrease in the keratin 16 levels. Specifically, after 5- and 10-μM CBD treatment, the keratin 16 levels decreased to 0.63 and 0.58, respectively (P<0.05) (Fig. 6b). These results confirm the inhibitory effect of CBD on hyperkeratinization.
Inhibition of hyperkeratinization by CBD treatment in HaCaT cells. HaCaT cells were subjected to CBD treatment at concentrations of 0 µM, 5 µM, and 10 µM for a duration of 24 h. (a) Western blot analysis was employed to validate the reduction in keratin 16 levels. (b) Alterations in the mRNA expression of keratin 16 were verified through real-time PCR. CBD, cannabidiol. *, P < 0.05
Effects of CBD on scar formation-related protein synthesis of fibroblasts
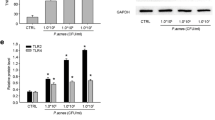
DAPI staining was performed to investigate alterations in collagen 1 and collagen 3 synthesis in fibroblasts (Fig. 7a and b). Upon exposure to CBD at a concentration of 5 µM, the synthesis of both collagen 1 and collagen 3 increased significantly, as evident from the merged images. Subsequently, fibroblasts were subjected to 0, 1, and 5 µM of CBD for further analyses using western blot and quantitative real-time mRNA assessments to evaluate changes in scar-related proteins. At a CBD concentration of 1 µM, the relative quantities of elastin, collagen 1, and collagen 3 exhibited minor changes measuring 1.02, 1.71, and 1.47, respectively, in comparison to the baseline value of 1.00; however, these changes were not statistically significant (P = 0.40, P = 0.11, and P = 0.87, respectively) (Fig. 7c and d). In contrast, when exposed to a higher CBD concentration of 5 µM, the values significantly increased to 1.88, 2.59, and 1.80 (P < 0.05, P < 0.001, and P < 0.05, respectively) (Fig. 7c and d). The quantitative real-time analysis yielded consistent findings, with 5-µM CBD treatment resulting in mRNA levels of elastin, collagen 1, and collagen 3 reaching 2.00, 1.78, and 1.95, respectively (all P < 0.05) (Fig. 7e).
Changes in the expression of proteins related to scar formation induced by CBD. (a, b) Fibroblasts were treated with CBD at concentrations of 0 µM and 5 µM for a duration of 48 h. DAPI staining was performed to assess the impact of CBD treatment. Collagen 1 (a) and collagen 3 (b) expression were evaluated using DAPI staining (400×). All are at the same magnification (bar = 50 μm). (c) Western blot analysis confirmed changes in the expression of elastin, collagen 1, collagen 3, and p-Erk at CBD concentration of 1 µM or 5 µM. (d) Quantification of western blot bands for elastin/β-actin, collagen 1/β-actin, and collagen 3/β-actin was performed. (e) Quantitative real-time analysis confirmed the mRNA expression levels of elastin, collagen 1, and collagen 3. CBD, cannabidiol; DAPI, 4’,6-diamidino-2-phenylindole; p-ERK, phosphorylated ERK. *, P < 0.05; †, P < 0.00
Discussion
Acne, a prevalent dermatological condition with multifactorial origins, remains a challenging concern in both adolescent and adult populations [16]. CBD demonstrated effects on sebum production, inflammation, apoptosis, and hyperkeratinization, which underscore its potential to address multiple pathophysiological aspects of acne. By quantitatively assessing the impact of CBD on sebocyte viability, this study establishes a dose-dependent reduction, emphasizing CBD’s anti-proliferative properties. Moreover, the induction of apoptosis suggests a potential avenue to curtail hyperproliferation. The ability of CBD to counteract the C. acnes-induced inflammatory response by downregulating cytokines like CXCL8, IL-1α, and IL-1β further positions it as an anti-inflammatory agent for acne.
One of the study’s notable findings is CBD’s modulation of lipid synthesis through the Akt/AMPK-SREBP-1 pathway. This unique lipid-modulating effect is significant, as sebum overproduction is a hallmark of acne and contributes to pore blockages. Previous study on cultured human sebocytes demonstrated that activating transient receptor potential vanilloid-4 (TRPV4) ion channels resulted in the downregulation of nuclear receptor interacting protein-1 (NRIP1), which influences glucose and lipid metabolism [6]. While there isn’t a direct and well-established connection between the Akt/AMPK-SREBP-1 pathway and the TRPV4 ion channel, both pathways can influence cellular processes related to lipid metabolism and inflammation [17, 18]. The Akt/AMPK-SREBP-1 pathway primarily regulates lipid synthesis and metabolism within cells, while the TRPV4 ion channel has been associated with various cellular functions, including calcium signaling and regulation of cellular responses to mechanical and osmotic stimuli [19]. It’s possible that these pathways could indirectly interact or influence each other through broader cellular signaling networks. For instance, TRPV4 activation might impact cellular responses that intersect with the Akt/AMPK-SREBP-1 pathway, potentially influencing lipid metabolism or inflammation. However, such interactions would likely be complex and might involve intermediary signaling molecules or pathways that have yet to be fully elucidated. In parallel investigations, CBD treatment at 10 µM within an LPS-induced acne-like context exhibited decreased levels of TNF-α, IL-1β, and IL-6 [6].
Additionally, a separate in vitro study employing hemp seed hexane extracts (HSHE) demonstrated that the application of 0.6% HSHE to a human keratinocyte cell line led to a reduction in inflammation induced by C. acnes [7]. Notably, this effect was achieved through the down-regulation of pro-inflammatory cytokines, specifically IL-1β and IL-8. In a human study evaluating the safety and efficacy of cannabis seed extracts for acne, 3% cannabis seed extracts were found to reduce inflammation-induced erythema of the skin in a 12 week study and was found to be safe, well tolerated, non-allergenic, and non-irritating in the patient volunteers [20]. Finally, in UV-irradiated keratinocytes, CBD notably augmented the efficacy of antioxidant enzymes like superoxide dismutase and thioredoxin reductase [21]. This enhancement could offer potential benefits for individuals with acne, as a majority of the lesions manifest on sun-exposed regions of the skin. Collectively, these studies, along with our own findings, offer a coherent narrative of the potential for botanical extracts, such as hemp and cannabis seed extracts, to modulate inflammatory responses associated with acne.
There are several potential reasons why keratinocytes exhibit greater resistance in CBD-mediated inhibition experiments compared to sebocytes. A study showed that CBD was able to penetrate human keratinocytes and balance the oxidative stress response resulting from UVB irradiation and hydrogen peroxide [22]. This suggests that keratinocytes may be more resistant to CBD-induced inhibition because they are better able to absorb and utilize CBD. Another possible reason may be variations in the expression and density of cannabinoid receptors, such as CB1 and CB2 receptors between sebocytes and keratinocytes. Keratinocytes form the predominant component of the epidermis, serving as a protective barrier against external stressors. This characteristic might confer inherent resilience to external agents like CBD.
Presently, the primary approaches to address acne scars involve procedural methods such as subcision, chemical peels, and laser treatments, but their effectiveness is not consistently satisfactory [23, 24]. In a recent study, the use of topical epidermal growth factor ointment was found to stimulate elastin, collagen 1, and collagen 3 production within acne scars, leading to clinical improvements. Moreover, this study noted a reduction in IL-1 alpha and keratin 16, suggesting that CBD may have potential in enhancing the clinical outcomes of acne scarring [25]. In another study investigating CBD’s anti-wrinkle properties, UV-damaged fibroblasts exposed to 4 µM CBD exhibited a significant 27.7% increase in collagen content [26]. Although this concentration may seem similar to the 5 µM concentration discussed in this paper, it’s crucial to consider that the increase in collagen content showed notable distinctions. This variance might be linked to the UV-damaged condition of the cells, which could potentially hinder collagen synthesis. In addition to its known effect in inhibiting the action of matrix metalloproteinases, preventing extracellular matrix (ECM) degradation, CBD appeared to promote ECM generation at the mRNA level, demonstrating the potential for treatment approaches rather than procedures for acne scarring [27].
It is imperative to acknowledge the limitations of this study. While the cellular level investigations provide valuable mechanistic insights, clinical trials are necessary to ascertain the real-world efficacy and safety of CBD-based treatments. Ethical and legal considerations surrounding CBD usage should also be taken into account, especially given variations in regulations across regions. Future research avenues include well designed clinical trials to validate the efficacy of CBD-based therapies, alongside investigations into potential adverse effects.
In conclusion, this study highlights CBD’s potential to address multiple facets of acne pathophysiology through its anti-inflammatory, apoptotic, lipid-inhibitory effects, and modulation of the Akt/AMPK-SREBP-1 pathway. Additionally, it suggests the potential for CBD to contribute to the improvement of acne scarring through the synthesis of collagen and elastin. These findings offer a fresh perspective on acne management, suggesting that CBD-based treatments could provide a more comprehensive approach for individuals prone to inflammation and scarring. While further research is warranted, CBD’s unique mechanism of action presents a promising avenue for advancing acne therapeutics and improving patients’ quality of life.
Data Availability
The data presented in this study are available on request from the corresponding author.
References
Lynn DD, Umari T, Dunnick CA, Dellavalle RP (2016) The epidemiology of acne vulgaris in late adolescence. Adolesc Health Med Ther 7:13–25
Hay RJ, Johns NE, Williams HC, Bolliger IW, Dellavalle RP, Margolis DJ et al (2014) The global burden of skin disease in 2010: an analysis of the prevalence and impact of skin conditions. J Invest Dermatol 134:1527–1534
Dreno B (2017) What is new in the pathophysiology of acne, an overview. J Eur Acad Dermatol Venereol 31:8–12
Fox L, Csongradi C, Aucamp M, Du Plessis J, Gerber M (2016) Treatment modalities for acne. Molecules 21:1063
Van Beugen S, Schut C, Kupfer J, Bewley AP, Finlay AY, Gieler U et al (2023) Perceived stigmatization among dermatological outpatients compared with controls: an observational Multicentre Study in 17 European countries. Acta Dermato-Venereologica 103:adv6485
Olah A, Toth BI, Borbiro I, Sugawara K, Szollosi AG, Czifra G et al (2014) Cannabidiol exerts sebostatic and antiinflammatory effects on human sebocytes. J Clin Invest 124:3713–3724
Jin S, Lee MY (2018) The ameliorative effect of hemp seed hexane extracts on the Propionibacterium acnes-induced inflammation and lipogenesis in sebocytes. PLoS ONE 13:e0202933
Wilkinson JD, Williamson EM (2007) Cannabinoids inhibit human keratinocyte proliferation through a non-CB1/CB2 mechanism and have a potential therapeutic value in the treatment of psoriasis. J Dermatol Sci 45:87–92
Puaratanaarunkon T, Sittisaksomjai S, Sivapornpan N, Pongcharoen P, Chakkavittumrong P, Ingkaninan K et al (2022) Topical cannabidiol-based treatment for psoriasis: a dual-centre randomized placebo-controlled study. J Eur Acad Dermatol Venereol 36:e718–e720
Thiboutot D, Jabara S, Mcallister JM, Sivarajah A, Gilliland K, Cong ZY et al (2003) Human skin is a steroidogenic tissue: steroidogenic enzymes and cofactors are expressed in epidermis, normal sebocytes, and an immortalized sebocyte cell line (SEB-1). J Invest Dermatology 120:905–914
Lu J, Cong T, Wen X, Li X, Du D, He G et al (2019) Salicylic acid treats acne vulgaris by suppressing AMPK/SREBP1 pathway in sebocytes. Exp Dermatol 28:786–794
Nelson AM, Gilliland KL, Cong Z, Thiboutot DM (2006) 13-cis retinoic acid induces apoptosis and cell cycle arrest in human SEB-1 sebocytes. J Invest Dermatol 126:2178–2189
Chai HJ, Li JH, Huang HN, Li TL, Chan YL, Shiau CY et al (2010) Effects of sizes and conformations of fish-scale collagen peptides on facial skin qualities and transdermal penetration efficiency. J Biomed Biotechnol 2010:757301
Yoon JY, Kwon HH, Min SU, Thiboutot DM, Suh DH (2013) Epigallocatechin-3-gallate improves acne in humans by modulating intracellular molecular targets and inhibiting. J Invest Dermatology 133:429–440
Kwon HH, Yoon JY, Park SY, Min S, Kim YI, Park JY et al (2015) Activity-guided purification identifies lupeol, a pentacyclic triterpene, as a therapeutic agent targeting multiple pathogenic factors of acne. J Invest Dermatology 135:1491–1500
Tan JK, Bhate K (2015) A global perspective on the epidemiology of acne. Br J Dermatol 172(Suppl 1):3–12
Tang S, Wu W, Tang W, Ge Z, Wang H, Hong T et al (2017) Suppression of rho-kinase 1 is responsible for insulin regulation of the AMPK/SREBP-1c pathway in skeletal muscle cells exposed to palmitate. Acta Diabetol 54:635–644
Grace MS, Bonvini SJ, Belvisi MG, Mcintyre P (2017) Modulation of the TRPV4 ion channel as a therapeutic target for disease. Pharmacol Ther 177:9–22
Alessandri-Haber N, Dina OA, Joseph EK, Reichling D, Levine JD (2006) A transient receptor potential vanilloid 4-dependent mechanism of hyperalgesia is engaged by concerted action of inflammatory mediators. J Neurosci 26:3864–3874
Ali A, Akhtar N (2015) The safety and efficacy of 3% cannabis seeds extract cream for reduction of human cheek skin sebum and erythema content. Pak J Pharm Sci 28:1389–1395
Jastrzab A, Gegotek A, Skrzydlewska E (2019) Cannabidiol regulates the expression of keratinocyte proteins involved in the inflammation process through transcriptional regulation. Cells 8:827
Baswan SM, Klosner AE, Glynn K, Rajgopal A, Malik K, Yim S et al (2020) Therapeutic potential of cannabidiol (CBD) for skin health and disorders. Clin Cosmet Investig Dermatol 13:927–942
Boen M, Jacob C (2019) A review and update of treatment options using the acne scar classification system. Dermatol Surg 45:411–422
Connolly D, Vu HL, Mariwalla K, Saedi N (2017) Acne scarring-pathogenesis, evaluation, and treatment options. J Clin Aesthet Dermatol 10:12–23
Kim DH, Yang JH, Cho SI, Yoon JY, Kim JW, Choi S et al (2022) Clinical and histological effects of topical epidermal growth factor on acne and acne scars. Dermatology 238:837–845
Chen XL, Su J, Wang RA, Hao R, Fu CG, Chen JJ et al (2023) Structural optimization of cannabidiol as multifunctional cosmetic raw materials. Antioxidants 12:314
Rawal SY, Dabbous MK, Tipton DA (2012) Effect of cannabidiol on human gingival fibroblast extracellular matrix metabolism: MMP production and activity, and production of fibronectin and transforming growth factor β. J Periodontal Res 47:320–329
Acknowledgements
Prof. Dae Hun Suh greatly appreciate the permission of use of SEB-1 cell line by Prof. Diane M. Thiboutot who made it.
Funding
Open Access funding enabled and organized by Seoul National University. This work was supported by the Promotion of Innovative Businesses for Regulation-Free Special Zones funded by the Ministry of SMEs and Startups (MSS), Republic of Korea (Grant No. P0016082).
Author information
Authors and Affiliations
Contributions
J.H.L. and D.H.K. contributed to conceptualization and data curation. J.H.L. was responsible for the original draft and also contributed to writing, review, and editing. J.Y.Y., Y.G.K., G.H.K. and B.J.P. played a role in methodology. J.Y.Y. is responsible for formal analysis. D.H.S. provided supervision, validation, and visualization, in addition to review and editing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Statement on any prior presentation
Contents of the manuscript have not been previously published and are not currently submitted elsewhere.
IRB approval status
Not applicable.
Conflict of interest
Jun Hyo Lee, Dong Hyo Kim, Ji Young Yoon and Dae Hun Suh declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results. Yoon Gyung Kwon, Geun-Hyeong Kim, and Byoung Jun Park are employees of Kolmar Korea, but they did not influence the interpretation of the experimental data.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lee, J.H., Yoon, J.Y., Kim, D.H. et al. Potential of cannabidiol as acne and acne scar treatment: novel insights into molecular pathways of pathophysiological factors. Arch Dermatol Res 316, 428 (2024). https://doi.org/10.1007/s00403-024-03131-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00403-024-03131-9