Abstract
Background
Resurfacing of the glenohumeral joint for patients with glenohumeral arthritis has gained popularity since the first introduction. We report the mid-term results of the Global C.A.P. uncemented resurfacing shoulder prosthesis (DePuy Synthes).
Methods
From January 2007 to December 2009, 48 humeral cementless resurfacing prostheses in 46 patients were performed. All patients were diagnosed with primary glenohumeral osteoarthritis. Patients were contacted for review; the Constant Score, visual analog pain scale, Dutch Simple Shoulder Test, SF-12 scores and physical examination were assessed both preoperatively and yearly postoperatively. Complications and revision surgery were documented. Radiographs were evaluated for component size, offset, inclination, height, loosening and subluxation.
Results
Forty-six patients (12 males) with a mean age of 72 years old (range 59–89) were included. At a mean 6.4-year follow-up (range 5–8), the Constant Score, visual analog pain scale and the Dutch Simple Shoulder Test scores improved significantly (p < 0.05) from baseline. Three patients were lost to follow-up. One patient died and two patients were not able to attend the follow-up appointments, due to other health-related issues. Eleven patients (23%) had a revision operation.
Conclusions
The most important findings of this study of the Global C.A.P. shoulder resurfacing arthroplasty were an increase of range of motion, a reduction of pain complaints, but a concerning high rate of revision after mid-term follow-up.
Level of evidence
Therapeutic Level IV.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Shoulder pathology is a common source of pain and disability affecting patients with a prevalence of 17% [1]. Shoulder replacement can provide satisfactory results through restoration of shoulder congruity that improves range of motion and decreases pain sensation [2].
The optimal surgical treatment for glenohumeral osteoarthritis with an intact rotator cuff is still under debate [3–5]. Good outcomes of total shoulder arthroplasty have been shown to last for an average of at least 10 years. Glenoid component loosening in up to 39% remains the most frequent indication for revision surgery [6–12].
Resurfacing shoulder replacement of the proximal humerus is a viable alternative to conventional shoulder replacement to restore shoulder function in patients with osteoarthritis. The first surface replacement was designed by Copeland and was performed only in young and active patients in the mid 1980s [13, 14]. After this initial period, surface replacement has been popularized and increasingly used in elderly patients and has also been described as a viable treatment option for many indications, such as osteoarthritis, avascular necrosis, rheumatoid arthritis, rotator cuff tear and post-traumatic arthritis [6, 15–22]. Some of the advantages are the preservation of the humeral bone stock which eases the conversion to a stemmed total or reverse shoulder prosthesis if a revision becomes necessary [13, 19, 22, 23]. Other potential benefits include the shorter operation time, less risk of periprosthetic fractures and less per-operative blood loss. Studies report satisfactory results at short- and mid-term follow-up [18, 23–27]. The purpose of this study is to asses mid-term patient reported outcome measures, revision rate and radiographs of the Global Conservative Anatomic Prosthesis (C.A.P.) uncemented resurfacing shoulder prosthesis (DePuy Synthes, Warsaw, USA). This study has been performed as an extension to the ongoing follow-up study, short-term results published in 2014 [26]. The authors expect satisfactory patient reported outcome results and a revision rate lower or equal to the literature.
Patients and methods
This study was performed as an extension to the ongoing follow-up study in patients treated with uncemented Global C.A.P. resurfacing shoulder prosthesis, short-term results published in 2014 [26]. The study was approved by the Northern Dutch Review board (M1330348), and all patients had signed informed consent.
Patient population
Patients older than 18 years, with an intact and sufficient rotator cuff, adequate bone stock of the proximal humerus [>60% estimated on radiographs and magnetic resonance imaging (MRI)], with failed conservative treatment (physiotherapy, intra-articular injections with corticosteroids or arthroscopic debridement), glenoid centric type A1, A2 or B1 according of Walch classification assessed on MRI [28], and treated with a resurfacing prosthesis between January 2007 and December 2009 were included in this study. In all patients, preoperative radiographs and MRI scans were assessed. To minimize selection bias, only patients with an intact cuff and glenohumeral osteoarthritis were included.
Intervention
The senior authors performed all operations in two clinics, Alrijne Hospital (Leiderdorp, the Netherlands) and Spaarne Gasthuis (Hoofddorp, the Netherlands). All shoulders were treated with a cementless humeral resurfacing implant (Global C.A.P., DePuy Synthes, Warsaw, USA). Thirty minutes before the first incision a first-generation cephalosporin was administered intravenously. Preoperative interscalene block was used in combination with general anesthesia. Patients were placed in the beach chair position with their arm draped freely. In all shoulders, a deltopectoral approach was used. Care was taken with preservation of the tendon of the pectoralis major and the vessels of the humeral circumflex. Soft tissue releases of the tendon of the subscapularis and the anterior and posterior aspects of the capsule were performed to improve range of motion if necessary. This could also include a 360° release of the tendon of the subscapularis. The tendon of the subscapular muscle was cut close to its insertion at the minor tubercle, leaving a small part of the tendon attached. The reattachment could be done safely and strongly with multiple stitches. The construction was tested by external rotation of the arm before closure. Tenodesis or tenotomy of the long head of the biceps was only performed when tendinopathy was diagnosed intraoperatively by the senior authors. A lateral clavicle resection was performed in patients with a symptomatic AC joint diagnosed by the senior authors during physical examination prior the operation.
With respect for anatomic (retro) version and inclination the appropriate size implant was placed. Only the affected glenoid was treated with a chondropick to enhance micro fracturing of the eroded articular surface to stimulate the growth of fibrous tissue. No glenoid implants were used.
Rehabilitation
Postoperative patients used a standard sling for up to 6 weeks. Immediately, postoperative patients were stimulated to start with forward elevation and abduction and to perform front-to-back pendulum exercises. To minimize the tension in the re-attached subscapularis tendon, external rotation was allowed within the maximum degree of that obtained during surgery. Patients followed a routine rehabilitation protocol after the resurfacing shoulder arthroplasty. This protocol consisted of supervised physiotherapy for 3–6 months and self exercises.
Clinical and radiological assessment
The senior authors did the baseline assessments in all patients, including demographic details, diagnosed primary osteoarthritis, radiographs and MRI in the outpatient clinic. Two physician’s assistants (PS and MC), assessed the pain score according to the visual analog pain scale (VAS) [29, 30], the Dutch version of the Simple Shoulder Test (DSST) [31], the range of motion and strength to derive a Constant score [30, 32–37], and the patient’s activities and daily living (SF-12) [38–40]. The physician assistants did not participate in the perioperative care and did not see the postoperative X-rays.
The first day postoperative and at 3 months and annually radiographs anteroposterior and axillary were taken. Signs of loosening, such as radiolucent lines, and their evolution over time were made. Definite loosening was defined as a change in position of the implant over time. Unchanged position but progressive radiolucencies of >2 mm wide from the component were defined as probably loosening [19]. Analyses were made for luxation of the prosthesis and migration of the prosthesis outside the center of the glenoid and the length of gleno-humeral offset was assessed to measure overstuffing [41].
Statistics
For analyzing the preoperative and postoperative scores, we used Wilcoxon signed ranks test. The study data were not normally distributed, and they cannot be transformed to a normal distribution by means of a logarithmic transformation. A p value of <0.05 was considered significant. The Constant score increased and the pain (VAS) score decreased after 2 year follow-up. This can be explained by the patients with the poor scores had a revision surgery and were not included for further data analysis. To minimize selection bias, only patients with an intact cuff and glenohumeral osteoarthritis were included. Statistic software of SPSS (SPSS Inc., Chicago, Illinois, USA) version 20.0 was used.
Results
Forty-eight resurfacing humeral head surface replacement arthroplasty operations were performed in 46 patients. This cohort consists of 36 female and 12 male patients with a mean age of 72 years old (range 59–89 years). The short-term results were described in a previous publication [26].
Three patients (6%) were lost to follow-up. One patient died because of reasons not related to the prosthesis or operation. Two patients were not able to attend at the follow-up appointments due to health-related issues. The health issues were not related to the implant or operation. Eleven of 48 prosthesis (23%) had a revision operation.
Mean follow-up was 6.4 years (range 5.1–7.9). In six patients (13%), an additional lateral clavicle resection was performed. Thirty-eight patients (79%) had a biceps tenodesis and three patients (6%) had a biceps tenotomy.
The mean Constant score (corrected for gender and age [37]) improved from points 47 ± 18, preoperatively to 83 ± 22 points at follow-up (p < 0.001). The mean Dutch Simple Shoulder Test (DSST) improved from 20 ± 21 points, preoperatively to 67 ± 30 points at follow-up (p < 0.001). The pain score, according to the visual analog scale (VAS), decreased from 66 ± 19, preoperatively to 29 ± 28 points at follow-up (p < 0.001).
The SF-12, divided in a mental and a physical score, the mean SF-12 mental score improved from 49 ± 12 points preoperatively, to 51 ± 8 points at follow-up (p = 0.45). The mean SF-12 physical score improved from 35 ± 8 points preoperatively, to 39 ± 11 points at follow-up (p = 0.05). All preoperative, short-term (2 year) and mid-term follow-up data are listed in Table 1.
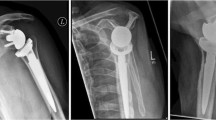
Radiology
For 36 shoulders, radiographs were available. No loosening or dislocation were seen at mid-term follow-up. Some degree of superior migration, as an indication of rotator cuff failure or insufficiency, was noted in 15 of the 36 shoulders (42%). Six (17%) patients had severe migration and nine (25%) had mild superior migration, see Table 2. Twenty-one (58%) shoulders showed no superior migration. Moderate-to-severe glenoid erosion was present in twelve (33%) of the shoulders at a mid-term follow-up, see Table 3.
Complications
The early complications were described in the 2 year follow-up. No revision surgery was performed or necessary within the short-term follow-up [26].
Revision surgery
Eleven patients (23%), five males and six females, had a revision operation to a reverse shoulder arthroplasty or total shoulder arthroplasty, see Table 4. Mean time of revision 54 months (range 34–81 months). Mean constant score prior to revision 55 (range 28–85). Patients had a mean VAS of 59 (range 15–75) prior to revision.
All revision surgeries were a complete revision of the resurfacing prosthesis and glenoid. All cultures taken during revision surgery were negative in the all mentioned patients, except the low grade infection. All revised patients had satisfactory results after revision surgery.
Discussion
The most important finding of this study of the Global C.A.P. shoulder resurfacing arthroplasty were an increase of range of motion, a reduction of pain complaints, but an increased revision rate after mid-term follow-up in contrast of 2 year follow-up [26].
Outcome assessment bias was minimized by having assessors who were not involved with the initial operation. Although we realize that TSA is the gold standard for treatment of osteoarthritis of the shoulder today [3, 42–44], we think that (short) stemmed and resurfacing hemi-shoulder prostheses are still a valid treatment option in selected patients because of the limited survival of the glenoid component in TSA after long-term follow-up. Glenoid loosening has been reported to be between 0 and 20% at medium term follow-up and 39% mid-term to long-term follow-up [6–12, 45], with more than 5% rate of revision surgery at long-term follow-up. Several factors such as rotator cuff tears, component malposition and glenoid instability can contribute to glenoid component failure [8, 45, 46].
This resurfacing prosthesis has a hydroxyapatite coating. Advantages of hydroxyapatite-coated surface replacement of the shoulder, when compared to stemmed implants, are less bone resection, primary press-fit cementless fixation with bone in-growth into a hydroxyapatite coating, easier replication of the native anatomy, reduced risk of intraoperative humeral shaft fracture and stem perforation, preservation of humeral bone stock, and easier revision surgery [19, 26, 47–49].
In line with Cofield, we think that the revision rate alone is not sensitive to a failed procedure due to the subjective assessment by the surgeon. This assessment by the surgeon should be used in combination with pain and satisfaction assessed by the patient. Especially, patients reporting pain equal or worse than their preoperative condition should also be considered as a failure [19, 50].
A resurfacing shoulder arthroplasty is less difficult to remove than a stemmed hemiarthroplasty. In contrast to Al-Hadithy and Alizadehkaiyat, our revision operations were achieved easily with the removal of the implant [24, 25]. During revision, significantly reduced bone density under the implant was observed. This observation is in line with the findings of Schmidutz et al. [5]. However, the metaphyseal bone was adequate enough to make short stem prosthesis possible. No step cut of the glenoid and bone grafting of the glenoid was necessary in all revised patients [51]. All patients had no complications and satisfactory results after revision surgery [51].
Glenoid changes after resurfacing prosthesis were assessed by measuring the joint space and determination of possible bone loss of the glenoid. This space might increase by the formation of fibrosis, because of the micro fracturing. Glenoid erosion in hemiarthroplasty is one of the major reasons for revision to total or reverse shoulder arthroplasty [52–55]. As in our study and in the literature, radiological glenoid deterioration is not correlated with pain or deterioration of clinical results [26, 41, 56, 57].
Periprosthetic fractures did not occur in our series. Possibly because of the absence of stress shielding in the midshaft with a resurfacing implant [58, 59]. A stemmed prostheses create a stress riser effect at the tip of the stem in the midshaft of the humerus [19]. Periprosthetic fractures, which have a reported prevalence of 3%, account for approximately 20% of all complications associated with total shoulder arthroplasty. This can be reduced using this prosthesis [23, 60–64].
The conclusions of this study have to be drawn in the light of some limitations. Although the patients were enrolled prospectively in a computerized database, there was no control group treated with a stemmed implant or a resurfacing prosthesis with a glenoid component as a TSP. The reported study group was small but nonetheless comparable to other published studies of shoulder resurfacing [11, 19, 22, 23]. Our revision rate (23%) was higher compared to the rate reported by Levy at al. They reported a revision rate of 14% in the resurfacing shoulder replacement after 10 years follow-up [22]. In contrast to the series reported by Streubel et al., our patients had satisfactory results after revision surgery [65].
In the literature, high rates of survival are described after mid-term and long-term follow-up. There is certainly a discrepancy in the literature with respect to revisions. Particularly recent literature from 2013 reported a significant high percentage of revisions due to glenoid erosion and pain [13, 18, 22–27, 59, 66, 67]. Relevant studies and revisions are mentioned in Table 5.
Sperling et al. reported similar revision rate in a stemmed hemiarthroplasty of 22% [55]. A more recent study from Bartelt et al. showed similar results at short-term follow-up with a high rate of revision of 30% at mid-term follow-up [68].
Nevertheless, a small sample size suggests caution in interpreting the incidence of uncommon complications. Performing a “new” type of surgery on a large scale would not be considered wise because of the recent lessons we have learned from, for example, the metal-on-metal discussion in hip surgery.
Long-term and precise follow-up is essential to determine if treatment with this cementless resurfacing implant for end-stage osteoarthritis of the shoulder is viable.
Conclusion
In conclusion: we report the clinical and radiologic outcome for the uncemented Global C.A.P. resurfacing prosthesis for the treatment of primary osteoarthritis in patients with an intact rotator cuff with more than 6 years of follow-up. The mid-term of the global C.A.P. resurfacing prosthesis are in line with other studies with a concerning revision rate of 23%.
References
Merolla G, Bianchi P, Lollino N, Rossi R, Paladini P, Porcellini G (2013) Clinical and radiographic mid-term outcomes after shoulder resurfacing in patients aged 50 years old or younger. Musculoskelet Surg 97(Suppl 1):23–29. doi:10.1007/s12306-013-0261-4
Cuomo F, Birdzell MG, Zuckerman JD (2005) The effect of degenerative arthritis and prosthetic arthroplasty on shoulder proprioception. J Shoulder Elb Surg 14(4):345–348. doi:10.1016/j.jse.2004.07.009
van den Bekerom MP, Geervliet PC, Somford MP, van den Borne MP, Boer R (2013) Total shoulder arthroplasty versus hemiarthroplasty for glenohumeral arthritis: a systematic review of the literature at long-term follow-up. Int J Shoulder Surg 7(3):110–115. doi:10.4103/0973-6042.118915
Lebon J, Delclaux S, Bonnevialle N, Rongieres M, Bonnevialle P, Mansat P (2014) Stemmed hemiarthroplasty versus resurfacing in primary shoulder osteoarthritis: a single-center retrospective series of 78 patients. Orthop Traumatol Surg Res 100(6 Suppl):S327–S332. doi:10.1016/j.otsr.2014.05.012
Schmidutz F, Sprecher CM, Milz S, Gohlke F, Hertel R, Braunstein V (2015) Resurfacing of the humeral head: an analysis of the bone stock and osseous integration under the implant. J Orthop Res 33(9):1382–1390. doi:10.1002/jor.22902
Torchia ME, Cofield RH, Settergren CR (1997) Total shoulder arthroplasty with the Neer prosthesis: long-term results. J Shoulder Elb Surg 6(6):495–505
Hawkins RJ, Greis PE, Bonutti PM (1999) Treatment of symptomatic glenoid loosening following unconstrained shoulder arthroplasty. Orthopedics 22(2):229–234
Hasan SS, Leith JM, Campbell B, Kapil R, Smith KL, Matsen FA III (2002) Characteristics of unsatisfactory shoulder arthroplasties. J Shoulder Elb Surg 11(5):431–441
Bohsali KI, Wirth MA, Rockwood CA Jr (2006) Complications of total shoulder arthroplasty. J Bone Jt Surg Am 88(10):2279–2292. doi:10.2106/JBJS.F.00125
Bishop JY, Flatow EL (2005) Humeral head replacement versus total shoulder arthroplasty: clinical outcomes–a review. J Shoulder Elb Surg 14(1 Suppl S):141S–146S. doi:10.1016/j.jse.2004.09.027
Bailie DS, Llinas PJ, Ellenbecker TS (2008) Cementless humeral resurfacing arthroplasty in active patients less than fifty-five years of age. J Bone Jt Surg Am 90(1):110–117
Antuna SA, Sperling JW, Cofield RH, Rowland CM (2001) Glenoid revision surgery after total shoulder arthroplasty. J Shoulder Elb Surg 10(3):217–224. doi:10.1067/mse.2001.113961
Levy O, Funk L, Sforza G, Copeland SA (2004) Copeland surface replacement arthroplasty of the shoulder in rheumatoid arthritis. J Bone Jt Surg Am 86-A(3):512–518
Stewart MP, Kelly IG (1997) Total shoulder replacement in rheumatoid disease: 7- to 13-year follow-up of 37 joints. J Bone Jt Surg Br 79(1):68–72
Deshmukh AV, Koris M, Zurakowski D, Thornhill TS (2005) Total shoulder arthroplasty: long-term survivorship, functional outcome, and quality of life. J Shoulder Elb Surg 14(5):471–479. doi:10.1016/j.jse.2005.02.009
Cofield RH (1984) Total shoulder arthroplasty with the Neer prosthesis. J Bone Jt Surg Am 66(6):899–906
Haines JF, Trail IA, Nuttall D, Birch A, Barrow A (2006) The results of arthroplasty in osteoarthritis of the shoulder. J Bone Jt Surg Br 88(4):496–501. doi:10.1302/0301-620X.88B4.16604
Mullett H, Levy O, Raj D, Even T, Abraham R, Copeland SA (2007) Copeland surface replacement of the shoulder. Results of an hydroxyapatite-coated cementless implant in patients over 80 years of age. J Bone Jt Surg Br 89(11):1466–1469
Levy O, Copeland SA (2001 Mar) Cementless surface replacement arthroplasty of the shoulder. 5- to 10-year results with the Copeland mark-2 prosthesis. J Bone Jt Surg Br 83(2):213–221
Boileau P, Walch G, Noel E, Liotard JP (1994) Neer’s shoulder prosthesis: results according to etiology. Rev Rhum Ed Fr 61(9):607–618
Burgess DL, McGrath MS, Bonutti PM, Marker DR, Delanois RE, Mont MA (2009) Shoulder resurfacing. J Bone Jt Surg Am 91(5):1228–1238. doi:10.2106/JBJS.H.01082
Levy O, Tsvieli O, Merchant J, Young L, Trimarchi A, Dattani R et al (2015) Surface replacement arthroplasty for glenohumeral arthropathy in patients aged younger than fifty years: results after a minimum ten-year follow-up. J Shoulder Elb Surg 24(7):1049–1060. doi:10.1016/j.jse.2014.11.035
Levy O, Copeland SA (2004) Cementless surface replacement arthroplasty (Copeland CSRA) for osteoarthritis of the shoulder. J Shoulder Elb Surg 13(3):266–271
Al-Hadithy N, Domos P, Sewell MD, Naleem A, Papanna MC, Pandit R (2012) Cementless surface replacement arthroplasty of the shoulder for osteoarthritis: results of fifty Mark III Copeland prosthesis from an independent center with four-year mean follow-up. J Shoulder Elb Surg 21(12):1776–1781. doi:10.1016/j.jse.2012.01.024
Alizadehkhaiyat O, Kyriakos A, Singer MS, Frostick SP (2013) Outcome of Copeland shoulder resurfacing arthroplasty with a 4-year mean follow-up. J Shoulder Elbow Surg 22(10):1352–1358. doi:10.1016/j.jse.2013.01.027
Geervliet P, van den Bekerom M, Spruyt P, Curvers M, Visser C, van Noort A (2014) Short-term results of the global C.A.P. uncemented resurfacing shoulder prosthesis. Orthopedics 37(1):42–47. doi:10.3928/01477447-20131219-07
Thomas SR, Wilson AJ, Chambler A, Harding I, Thomas M (2005) Outcome of Copeland surface replacement shoulder arthroplasty. J Shoulder Elb Surg 14(5):485–491
Walch G, Badet R, Boulahia A, Khoury A (1999) Morphologic study of the glenoid in primary glenohumeral osteoarthritis. J Arthroplasty 14(6):756–760
Bijur PE, Silver W, Gallagher EJ (2001) Reliability of the visual analog scale for measurement of acute pain. Acad Emerg Med 8(12):1153–1157
Richards RR, An KN, Bigliani LU, Friedman RJ, Gartsman GM, Gristina AG et al (1994) A standardized method for the assessment of shoulder function. J Shoulder Elb Surg 3(6):347–352. doi:10.1016/S1058-2746(09)80019-0
van Kampen DA, van Beers LW, Scholtes VA, Terwee CB, Willems WJ (2011) Validation of the Dutch version of the Simple Shoulder Test. J Shoulder Elb Surg. doi:10.1016/j.jse.2011.09.026
Blonna D, Scelsi M, Marini E, Bellato E, Tellini A, Rossi R et al (2012) Can we improve the reliability of the Constant-Murley score? J Shoulder Elb Surg 21(1):4–12. doi:10.1016/j.jse.2011.07.014
Conboy VB, Morris RW, Kiss J, Carr AJ (1996) An evaluation of the Constant-Murley shoulder assessment. J Bone Jt Surg Br 78(2):229–232
Constant CR, Murley AH (1987) A clinical method of functional assessment of the shoulder. Clin Orthop Relat Res 214:160–164
Constant CR, Gerber C, Emery RJ, Sojbjerg JO, Gohlke F, Boileau P (2008) A review of the Constant score: modifications and guidelines for its use. J Shoulder Elb Surg 17(2):355–361. doi:10.1016/j.jse.2007.06.022
Roy JS, MacDermid JC, Woodhouse LJ (2010) A systematic review of the psychometric properties of the Constant-Murley score. J Shoulder Elb Surg 19(1):157–164. doi:10.1016/j.jse.2009.04.008
Katolik LI, Romeo AA, Cole BJ, Verma NN, Hayden JK, Bach BR (2005) Normalization of the Constant score. J Shoulder Elb Surg 14(3):279–285. doi:10.1016/j.jse.2004.10.009
Gandek B, Ware JE, Aaronson NK, Apolone G, Bjorner JB, Brazier JE et al (1998) Cross-validation of item selection and scoring for the SF-12 Health Survey in nine countries: results from the IQOLA Project. International Quality of Life Assessment. J Clin Epidemiol 51(11):1171–1178
Jenkinson C, Layte R, Jenkinson D, Lawrence K, Petersen S, Paice C et al (1997) A shorter form health survey: can the SF-12 replicate results from the SF-36 in longitudinal studies? J Public Health Med 19(2):179–186
Ware J Jr, Kosinski M, Keller SD (1996) A 12-Item Short-Form Health Survey: construction of scales and preliminary tests of reliability and validity. Med Care 34(3):220–233
Mechlenburg I, Amstrup A, Klebe T, Jacobsen SS, Teichert G, Stilling M (2013) The Copeland resurfacing humeral head implant does not restore humeral head anatomy. A retrospective study. Arch Orthop Trauma Surg 133(5):615–619. doi:10.1007/s00402-013-1715-8
Bryant D, Litchfield R, Sandow M, Gartsman GM, Guyatt G, Kirkley A (2005) A comparison of pain, strength, range of motion, and functional outcomes after hemiarthroplasty and total shoulder arthroplasty in patients with osteoarthritis of the shoulder. A systematic review and meta-analysis. J Bone Jt Surg Am 87(9):1947–1956. doi:10.2106/JBJS.D.02854
Duan X, Zhang W, Dong X, Liu M, Gao Y, Huang F et al (2013) Total shoulder arthroplasty versus hemiarthroplasty in patients with shoulder osteoarthritis: a meta-analysis of randomized controlled trials. Semin Arthritis Rheum 43(3):297–302. doi:10.1016/j.semarthrit.2013.04.002
Jain NB, Hocker S, Pietrobon R, Guller U, Bathia N, Higgins LD (2005) Total arthroplasty versus hemiarthroplasty for glenohumeral osteoarthritis: role of provider volume. J Shoulder Elb Surg 14(4):361–367. doi:10.1016/j.jse.2004.10.007
Melis B, Bonnevialle N, Neyton L, Levigne C, Favard L, Walch G et al (2012) Glenoid loosening and failure in anatomical total shoulder arthroplasty: is revision with a reverse shoulder arthroplasty a reliable option? J Shoulder Elb Surg 21(3):342–349. doi:10.1016/j.jse.2011.05.021
Cofield RH, Edgerton BC (1990) Total shoulder arthroplasty: complications and revision surgery. Instr Course Lect 39:449–462
Boileau P, Walch G (1997) The three-dimensional geometry of the proximal humerus. Implications for surgical technique and prosthetic design. J Bone Jt Surg Br 79(5):857–865
Mansat P, Coutie AS, Bonnevialle N, Rongieres M, Mansat M, Bonnevialle P (2013) Resurfacing humeral prosthesis: do we really reconstruct the anatomy? J Shoulder Elb Surg 22(5):612–619. doi:10.1016/j.jse.2012.07.014
Roberts SN, Foley AP, Swallow HM, Wallace WA, Coughlan DP (1991) The geometry of the humeral head and the design of prostheses. J Bone Jt Surg Br 73(4):647–650
Cofield RH (1983) Unconstrained total shoulder prostheses. Clin Orthop Relat Res 173:97–108
Geervliet PC, Houtveen M, Sierevelt IN, Visser C, van Noort A (2017) Revision from Global C.A.P. resurfacing prosthesis: results, survival and group comparison (Submitted for publication)
Geervliet PC, Somford MP, Winia P, van den Bekerom MP (2015) Long-term results of shoulder hemiarthroplasty in patients with rheumatoid arthritis. Orthopedics 38(1):e38–e42. doi:10.3928/01477447-20150105-58
Groh GI, Wirth MA (2011 Jul) Results of revision from hemiarthroplasty to total shoulder arthroplasty utilizing modular component systems. J Shoulder Elb Surg 20(5):778–782. doi:10.1016/j.jse.2010.09.014
Sanchez-Sotelo J (2011) Total shoulder arthroplasty. Open Orthop J 5:106–114. doi:10.2174/1874325001105010106
Sperling JW, Cofield RH, Rowland CM (2004) Minimum fifteen-year follow-up of Neer hemiarthroplasty and total shoulder arthroplasty in patients aged fifty years or younger. J Shoulder Elb Surg 13(6):604–613. doi:10.1016/S1058274604001296
Ohl X, Nerot C, Saddiki R, Dehoux E (2010) Shoulder hemi arthroplasty radiological and clinical outcomes at more than two years follow-up. Orthop Traumatol Surg Res 96(3):208–215. doi:10.1016/j.otsr.2010.01.001
Alolabi B, Youderian AR, Napolitano L, Szerlip BW, Evans PJ, Nowinski RJ et al (2014) Radiographic assessment of prosthetic humeral head size after anatomic shoulder arthroplasty. J Shoulder Elb Surg 23(11):1740–1746. doi:10.1016/j.jse.2014.02.013
Orr TE, Carter DR (1985) Stress analyses of joint arthroplasty in the proximal humerus. J Orthop Res 3(3):360–371. doi:10.1002/jor.1100030313
Pritchett JW (2011) Long-term results and patient satisfaction after shoulder resurfacing. J Shoulder Elb Surg 20(5):771–777. doi:10.1016/j.jse.2010.08.014
Bonutti PM, Hawkins RJ (1992) Fracture of the humeral shaft associated with total replacement arthroplasty of the shoulder. A case report. J Bone Jt Surg Am 74(4):617–618
Boyd AD Jr, Thornhill TS, Barnes CL (1992) Fractures adjacent to humeral prostheses. J Bone Jt Surg Am 74(10):1498–1504
Groh GI, Heckman MM, Wirth MA, Curtis RJ, Rockwood CA Jr (2008) Treatment of fractures adjacent to humeral prostheses. J Shoulder Elb Surg 17(1):85–89. doi:10.1016/j.jse.2007.05.007
Hawkins RJ, Bell RH, Jallay B (1989) Total shoulder arthroplasty. Clin Orthop Relat Res 242:188–194
Wirth MA, Rockwood CA Jr (1996) Complications of total shoulder-replacement arthroplasty. J Bone Jt Surg Am 78(4):603–616
Streubel PN, Simone JP, Cofield RH, Sperling JW (2016) Revision of failed humeral head resurfacing arthroplasty. Int J Shoulder Surg 10(1):21–27. doi:10.4103/0973-6042.174514
Rasmussen JV, Polk A, Sorensen AK, Olsen BS, Brorson S (2014) Outcome, revision rate and indication for revision following resurfacing hemiarthroplasty for osteoarthritis of the shoulder: 837 operations reported to the Danish Shoulder Arthroplasty Registry. Bone Jt J Apr 96-B(4):519–525. doi:10.1302/0301-620X.96B4.31850
Smith T, Gettmann A, Wellmann M, Pastor F, Struck M (2013) Humeral surface replacement for osteoarthritis. Acta Orthop 84(5):468–472. doi:10.3109/17453674.2013.838658
Bartelt R, Sperling JW, Schleck CD, Cofield RH (2011) Shoulder arthroplasty in patients aged fifty-five years or younger with osteoarthritis. J Shoulder Elb Surg 20(1):123–130. doi:10.1016/j.jse.2010.05.006
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The original CAP study was funded by a grant (Spaarne Gasthuis #116347 and Alrijne Hospital #221090) from DePuy/Synthes, Warsaw, IN, USA, which participated in the study design and data management. The implant used in this study was not provided free of charge. The study sponsors had no role in the in the collection, analysis, interpretation of data, in the writing of the manuscript, and in the decision to submit the manuscript for publication.
Financial remuneration
Pieter Geervliet: The author, their immediate family, and any research foundation with which they are affiliated have not received any financial payments or other benefits from any commercial entity related to the subject of this article. Michel van den Bekerom: The author, their immediate family, and any research foundation with which they are affiliated have not received any financial payments or other benefits from any commercial entity related to the subject of this article. Paul Spruyt: The author, their immediate family, and any research foundation with which they are affiliated have not received any financial payments or other benefits from any commercial entity related to the subject of this article. Maud Curvers: The author, their immediate family, and any research foundation with which they are affiliated have not received any financial payments or other benefits from any commercial entity related to the subject of this article. Arthur van Noort: Dr Van Noort is a key opinion leader for Johnson and Johnson. Cornelis Visser: Dr Visser is a key opinion leader for Johnson and Johnson.
Additional information
IRB name and number: ACLU 2016.0054.
Ethical Committee name and number: METC M1330348.
Rights and permissions
About this article
Cite this article
Geervliet, P.C., van den Bekerom, M.P.J., Spruyt, P. et al. Outcome and revision rate of uncemented glenohumeral resurfacing (C.A.P.) after 5–8 years. Arch Orthop Trauma Surg 137, 771–778 (2017). https://doi.org/10.1007/s00402-017-2688-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00402-017-2688-9