Abstract
Purpose
Fish oil (FO) elicits diverse beneficial effects. Reduction in or prevention of body mass (BM) gain in animal models may be associated with modulation of brown adipose tissue (BAT). We aimed to evaluate the effects of different high-fat diets with FO on BAT metabolism and thermogenic markers.
Methods
C57BL/6 male mice (3-month-old) were fed different diets during 8 weeks: standard-chow diet (SC 10 % fat), high-fat lard diet (HF-L 50 % fat), high-fat lard plus FO diet (HF-L+FO 50 % fat), and high-fat FO diet (HF-FO 50 % fat). We evaluated BM and performed an oral glucose tolerance test. At euthanasia, plasma was collected for leptin, and triacylglycerol measurement and interscapular BAT was dissected and stored for molecular analyses.
Results
HF-L group showed elevated BM; glucose intolerance associated with diminished TC10 and GLUT4 expressions; hypertriglyceridemia associated with increased CD36 and diminished CPT1 expression; elevated expression of pro-inflammatory cytokines; and reduced PPAR expression. Furthermore, these animals showed hyperleptinemia with increased expression of thermogenic markers (beta3-AR, PGC1alpha, and UCP1). Conversely, HF-L+FO and HF-FO groups showed reduced BM gain with regularization of glucose tolerance and triglyceridemia, GLUT4, TC10, CD36, CPT1, and cytokines expressions. Both groups exhibited elevated PPAR and thermogenic markers expression in a dose-dependent way.
Conclusions
FO improves metabolic profile and upregulates thermogenic markers, suggesting an elevated thermogenesis that leads to reduced BM gain.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Fish oil (FO) is rich in n-3 polyunsaturated fatty acids (PUFA), especially eicosapentaenoic (EPA) and docosahexaenoic (DHA) acids. First described to target hypertriglyceridemia, FO is now recognized with beneficial effects on cardiovascular and inflammatory diseases, among others. One of the mechanisms by which FO exerts its positive impact is through activation of the peroxisome proliferator-activated receptors (PPAR), transcription factors involved in gene expression regulation [1]. Several animal studies proved that the FO (or its components) can prevent or reduce weight gain [2–4]. Moreover, some studies have already confirmed that FO (i.e., n-3 PUFA) stimulates thermogenesis in brown adipose tissue (BAT), although the mechanisms are still to be elucidated [5, 6].
BAT can generate heat through uncoupling mitochondrial fuel oxidation from ATP production, characterizing non-shivering thermogenesis [7]. This ability of BAT is conferred by the brown fat-specific uncoupling protein 1 (UCP1) and is controlled by the beta3 adrenergic receptor (beta3-AR) pathway [8]. BAT activation can be achieved by cold or overfeeding (diet-induced thermogenesis). In relation to diet-induced thermogenesis, diet composition, more specifically fat composition, can chronically modulate BAT thermogenesis [9].
Recent evidences, which human adult BAT can be activated and can have a role in the metabolism rekindle studies in this area [10]. Therefore, we have the aim of studying the effects of different high-fat diets containing FO on BAT metabolism and thermogenesis.
Materials and methods
Animals and diet
All procedures were approved by the local ethics committee for animal experimentation (protocol number CEUA/018/2013) in accordance with the conventional guidelines for animal experimentation (NIH publication number 85-23, revised 1996). The animals were maintained under controlled conditions (20 ± 2 °C and 12 h/12 h dark/light cycle) with free access to food and water.
Forty C57Bl/6 male mice with 3 months of age were randomly assigned into four groups (n = 10 each group):
-
(a)
standard-chow group (SC; 40 g soybean oil/Kg diet, 10 % of the total energy content from lipids);
-
(b)
high-fat lard group (HF-L; 40 g soybean oil and 238 g lard/Kg diet, 50 % of the total energy content from lipids);
-
(c)
high-fat lard plus FO group (HF-L+FO; 40 g soybean oil, 119 g lard and 119 g FO/Kg diet, 50 % of the total energy content from lipids); and
-
(d)
high-fat FO group (HF-FO; 40 g soybean oil and 238 g FO/Kg diet, 50 % of total energy content from lipids).
The diets were elaborated with purified nutrients by PragSolucoes (Jau, SP, Brazil) based on American Institute of Nutrition’s recommendations (AIN 93M) [11]. Table 1 shows the diet composition (FO from menhaden, approximately 25 % of EPA and DHA). Diets were offered over an 8-week period.
Body mass and food intake
The food intake was checked daily, and body mass (BM) weekly. Energy intake was the product of food intake by the energy content of the diet.
Indirect calorimetry and food quotient
The respiratory quotient (RQ) and the energy expenditure were measured (Apparatus Oxylet system, Panlab/Harvard, Barcelona, Spain) at the end of the experiment. The monitoring system evaluated the respiratory metabolism by measuring the oxygen consumption (VO2) and the carbon dioxide production (VCO2). Data were collected during 48 h and averaged over the entire testing period.
The food quotient (FQ) was calculated using the following equation: (0.835* %protein) + (1.000* %carbohydrate) + (0.710* %fat) [12].
Blood analyses
The oral glucose tolerance test (OGTT) was achieved at the conclusion of the experiment using a 25 % glucose solution (1.0 g/kg) administered by orogastric gavage after a 6-h fasting period. Blood was obtained from the tail vein. Glycemia was measured before glucose administration (0 min) and 15, 30, 60, and 120 min after glucose administration (glucometer Accu-Check, Roche, SP, Brazil). The results are presented as the ‘area under the curve’ (AUC; arbitrary units, a.u.), calculated using the trapezoidal rule (GraphPad Prism version 6.05, La Jolla, CA, USA).
At killing, animals were deprived of food for 6 h and were intraperitoneally anesthetized with 150 mg/kg sodium pentobarbital. Blood samples were obtained, and the serum was separated by centrifugation (120 g, 15 min) and stored individually at −80 °C. Leptin was measured using the SinglePlex kit EZML-82K (Millipore, Billerica, MA, USA). The triacylglycerol was performed using an automated spectrophotometer (Bioclin System II, Quibasa, MG, Brazil).
Adiposity index
We dissected and weighed the following fat pads: the inguinal fat pad (subcutaneous fat located between the lower part of the rib cage and the mid-thigh), the retroperitoneal fat pad (connected to the posterior abdominal wall near the kidneys), and the epididymal fat pad (located in the lower part of the abdomen and connected to the epididymis). The adiposity index was determined as the ratio between the sum of the fat masses (epididymal, retroperitoneal, and inguinal) divided by the total BM, as a percentage [13].
Brown adipose tissue
The interscapular brown adipose tissue (iBAT) was dissected, and samples were rapidly frozen and stored at −80 °C. Alternatively, the samples were kept in freshly prepared fixative solution (4 % formaldehyde w/v, 0.1 M phosphate buffer, pH 7.2).
RT-qPCR
Total RNA of iBAT was extracted using Trizol reagent (Invitrogen, CA, USA). Nanovue spectroscopy (GE Life Sciences) was used to determine RNA amount. Then, 1 µg RNA was treated with DNAse I (Invitrogen, CA, USA). Afterward, oligo (dT) primers for mRNA and Superscript III reverse transcriptase (both from Invitrogen, CA, USA) were applied to the synthesis of first-strand cDNA. Quantitative real-time PCR used a Biorad CFX96 cycler and the SYBR Green mix (Invitrogen, CA, USA). The primers used are described in Table S1: beta3-AR; PPARgamma coactivator 1 alpha, PGC1alpha; UCP1; PPARalpha; PPARbeta/delta; PPARgamma; cluster of differentiation 36, CD36; carnitine palmitoyltransferase-1, CPT-1; interleukin-6 (IL-6); and tumor necrosis factor alpha (TNFalpha). The endogenous beta-actin was used to normalize the expression of the selected genes. After the pre-denaturation and polymerase activation program (4 min at 95 °C), 44 cycles of 95 °C for 10 s and 60 °C for 15 s were followed by a melting curve program (60 °C to 95 °C with a heating rate of 0.1 °C/s). Negative controls consisted of wells in which the cDNA was substituted for deionized water. The relative expression ratio of the mRNA was calculated using the Eq. 2−ΔΔCt, in which −ΔCT represents the ratio between the quantity of cycles (CT) of the target genes with the endogenous control.
Western blotting
Total proteins of iBAT were extracted in homogenizing buffer containing protease inhibitors. Equivalent quantities of total protein resuspended in SDS-containing sample buffer were heated for 5 min at 100 °C and separated by SDS-PAGE. After electrophoresis, the proteins were electroblotted onto polyvinyl difluoride transfer membranes (Amersham Biosciences, Piscataway, N.J., USA). The blockade of the membrane was made with nonfat dry milk. Homogenates were incubated with the primary antibodies: beta3-AR (44 kDa; SC-1473; Santa Cruz Biotechnology); PGC1alpha (91 kDa; SC-5816; Santa Cruz Biotechnology); UCP1 (33 kDa; SC-6529; Santa Cruz Biotechnology); PPARalpha (52 kDa; SC-9000; Santa Cruz Biotechnology); PPARgamma (67 kDa; SC-7273; Santa Cruz Biotechnology); TC10 (24 kDa; T 8950; Sigma); and glucose transporter type 4 (GLUT-4, 55 kDa; 07-1404; Millipore). Beta-actin (SC81178; Santa Cruz Biotechnology) served as a loading control. We used ECL for protein expression detection system and the Bio-Rad Molecular Imaging ChemiDoc XRS Systems (Bio-Rad, Hercules, CA, USA). The chemiluminescence intensity of the bands was quantified using the ImageJ software, version 1.48 (NIH, imagej.nih.gov/ij, USA). The integral absorbance values were measured, and the results are shown as percentages of the SC group.
Immunofluorescence
For UCP-1 immunofluorescence, tissue sections (5 μm thick) were submitted to citrate buffer, pH 6.0, at 60 °C for 20 min for antigen retrieval, glycine 2 %, and blocking buffer (PBS/5 % BSA). The iBAT sections were incubated overnight at 4 °C with anti-UCP1 antibody (SC-6529; Santa Cruz Biotechnology), diluted 1:50 in PBS/1 % BSA, followed by incubation for 1 h at room temperature with fluorochrome-conjugated secondary antibody anti-goat IgG-Alexa 488 (Invitrogen, Molecular Probes, Carlsbad, CA, USA), diluted 1:50 in PBS/1 % BSA. After rinsing in PBS, the slides were mounted with SlowFade Antifade (Invitrogen, Molecular Probes, Carlsbad, CA, USA). Digital immunofluorescence images were captured using confocal microscopy (Nikon Confocal Laser Scanning Microscopy—Model C2; Nikon Instruments, Inc., New York, USA).
Data analysis
Values are shown as the mean ± standard deviation (SD). After confirmation of the homogeneity of variances, the differences among the groups were tested using a one-way ANOVA followed by the Holm–Sidak post hoc test. In all cases, a P value ≤0.05 was considered statistically significant (GraphPad Prism version 6.05, La Jolla, CA, USA).
Results
BM and food intake
All animals started the experiment with similar BM. After 2 weeks of the diet, the HF-L group was heavier than the other groups (+12 %, P = 0.006, compared to the SC group). The difference remained until the end of the experiment (+19 %, P < 0.0001, compared to the SC group). The HF-L+FO group had higher BM than both the SC group (+12 %, P = 0.0003) and the HF-FO group (+8 %, P = 0.008), but lower BM than the HF-L group (−6 %, P = 0.02). The HF-FO group stayed similar to the SC group (Fig. 1).
Body mass evolution. Values are the mean ± SD, n = 5 per group. Significant differences are indicated (P < 0.05), as determined by one-way ANOVA and Holm–Sidak post hoc test. Groups are standard-chow group (SC), high-fat lard group (HF-L), high-fat lard plus FO group (HF-L+FO), and high-fat FO group (HF-FO)
The four groups showed similar food intake (Table 2). However, because the HF groups ingested a hyperenergetic diet, these animals had an elevated energy intake when compared with the SC group (P < 0.05). There were no differences between the HF groups.
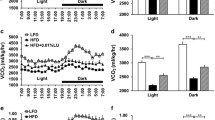
Indirect calorimetry and food quotient (Table 2)
The HF groups showed reduced RQ, when compared with the SC group: −9 %, P = 0.03 for the HF-L group; −10 %, P = 0.02, for the HF-L+FO group; and −12 %, P = 0.01, for the HF-FO group. There were no differences between these three groups.
The calculated FQ was 0.95 for the SC diet and 0.83 for the HF diets.
In relation to the energy expenditure, animals from the HF-L group showed reduced values in comparison with the SC group (−10 %, P = 0.0005). On the opposite way, the HF-L+FO group showed similar values to the SC group, and the HF-FO group demonstrated elevated values in comparison with the SC group (+6 %, P = 0.02). The HF-L+FO and the HF-FO groups showed elevated values when compared to the HF group (+12 %, P = 0.0002; +18 %, P < 0.0001; respectively). Lastly, the HF-FO group was elevated in comparison with the HF-L+FO group (+5 %, P = 0.02).
Blood analyses (Table 2)
The HF-L group was the only one to present an increase in the AUC from the OGTT test, being 30 % higher than the SC group (P < 0.0001). Both HF-L+FO and HF-FO groups had similar AUC to the SC group and diminished AUC compared to the HF-L group (−24 %, P < 0.0001; and −22 %, P < 0.0001; respectively).
Plasma leptin was higher in the HF-L group than in the SC group (P < 0.0001), but standard in both FO groups (similar to the SC group). It is remarkable that the plasma leptin was diminished in FO groups in comparison with the HF-L group (−87 %, P < 0.0001; −86 %, P < 0.0001; respectively).
The triacylglycerol was found higher in the HF-L group (+11 %, P = 0.0163) and regularized in the HF-L+FO and the HF-FO groups, compared to the SC group. The HF-L+FO group was 9 % lower (P = 0.01), and the HF-FO group was 14 % lower (P < 0.0001) than the HF-L group.
Adiposity index
The HF-L group had higher adiposity index compared to the SC group (+73 %, P < 0.0001). As observed in BM, the adiposity index in the HF-L+FO group continued elevated in comparison with the SC group (+39 %, P = 0.0186), but diminished in comparison with the HF-L group (−19 %, P = 0.0276). The HF-FO group had similar adiposity index to the SC group, as shown in Table 2.
RT-qPCR
Regarding beta3-AR, PGC1alpha, and UCP1 mRNA expressions, the HF-L group (+19 %, P = 0.007; +50 %, P = 0.04; +49 %, P = 0.002; respectively), the HF-L+FO group (+33 %, P < 0.0001; +111 %, P = 0.001; +129 %, P < 0.0001; respectively), and the HF-FO group (+73 %, P < 0.0001; +359 %, P < 0.0001; +239 %, P < 0.0001; respectively) were elevated in comparison with the SC group. Besides that, the HF-FO+L was elevated in comparison with the HF-L group (+12 %, P = 0.02; +41 %, P = 0.04; +53 %, P < 0.0001; respectively), and the HF-FO group was also elevated in comparison with the HF-L (+45 %, P < 0.0001; +206 %, P < 0.0001; +127 %, P < 0.0001) and the HF-L+FO group (+30 %, P < 0.0001; +118 %, P < 0.0001; +48 %, P < 0.0001) for all the three analyses (Fig. 2).
Gene and protein expressions of beta3-AR, PGC1alpha, and UCP1 in iBAT. Values are the mean ± SD, n = 5 per group. Significant differences are indicated (P < 0.05), one-way ANOVA and Holm–Sidak post hoc test: a ≠ SC, b ≠ HF-L, c ≠ HF-L+FO. Groups are standard-chow group (SC), high-fat lard group (HF-L), high-fat lard plus FO group (HF-L+FO), and high-fat FO group (HF-FO)
PPARbeta/delta gene expression was reduced in the HF-L group, in comparison with the SC group (−76 %, P = 0.04) (Fig. 3). In the groups fed FO, the PPARbeta/delta gene expression was dose dependent: HF-L+FO group was 66 % higher for PPARalpha (P = 0.04), 82 % higher for PPARbeta/delta (P = 0.04), and 109 % higher for PPARgamma (P = 0.01), and the HF-FO was 413 % higher for PPARalpha (P < 0.0001), 227 % higher for PPARbeta/delta (P < 0.0001), and 284 % higher for PPARgamma (P < 0.0001), in contrast to the SC group.
Gene and protein expressions of PPAR in iBAT. Values are the mean ± SD, n = 5 per group. Significant differences are indicated (P < 0.05), one-way ANOVA and Holm–Sidak post hoc test: a ≠ SC, b ≠ HF-L, c ≠ HF-L+FO. Groups are standard-chow group (SC), high-fat lard group (HF-L), high-fat lard plus FO group (HF-L+FO), and high-fat FO group (HF-FO)
Gene expression of CD36 was elevated in the HF groups compared to the SC group (more than 70 % high; P = 0.03 for all analyses, but no difference was observed among the HF groups). In addition, the gene expression of CPT-1 was reduced in the HF-L group (−28 %, P = 0.002) and elevated in both the HF-L+FO group (+28 %, P = 0.002) and the HF-FO group (+22 %, P = 0.006), compared to the SC group (Fig. 4).
Gene and protein expressions of CD36, CPT1, TC10, and GLUT-4 in iBAT. Values are the mean ± SD, n = 5 per group. Significant differences are indicated (P < 0.05), one-way ANOVA and Holm–Sidak post hoc test: a ≠ SC, b ≠ HF-L, c ≠ HF-L+FO. Groups are standard-chow group (SC), high-fat lard group (HF-L), high-fat lard plus FO group (HF-L+FO), and high-fat FO group (HF-FO)
IL-6 and TNFalpha gene expressions were increased in the HF-L group (P < 0.0001, for both genes) as observed in Fig. 5. The HF-L+FO and HF-FO groups had similar gene expressions to the SC group, but reduced gene expressions than the HF-L group (IL-6 −65 % and TNFalpha −93 % for the HF-L+FO group; and IL-6 −60 % and TNFalpha −95 %, for the HF-FO group; P < 0.0001, for all analyses).
Gene expression of IL-6 and TNFalpha in iBAT. Values are the mean ± SD, n = 5 per group. Significant differences are indicated (P < 0.05), one-way ANOVA and Holm–Sidak post hoc test: a ≠ SC, b ≠ HF-L, c ≠ HF-L+FO. Groups are standard-chow group (SC), high-fat lard group (HF-L), high-fat lard plus FO group (HF-L+FO), and high-fat FO group (HF-FO)
Western blotting
Beta3-AR, PGC1alpha, and UCP-1 protein expressions were slightly elevated in the HF-L group (+30 %, P = 0.04; +8 %, P = 0.03; +27 %, P = 0.008; respectively), compared to the SC group. In contrast, they were elevated in the HF-L+FO group (+76 %, P = 0.0003; +15 %, P = 0.0008; +59 %, P < 0.0001; respectively) and highly elevated in the HF-FO group (+118 %, P < 0.0001; +26 %, P < 0.0001; +83 %, P < 0.0001; respectively), compared to the SC group. Thermogenic protein responses to FO were dose dependent (Fig. 2).
Both the PPARalpha and PPARgamma showed reduced protein expressions in the HF-L group (−21 %, P = 0.04; −16 %, P = 0.04; respectively). These protein expressions were elevated in the other HF groups (Fig. 3). Interestingly, the response of PPARalpha and PPARgamma protein expressions in FO groups was dose dependent. Consequently, the HF-L+FO group showed a 24 % higher PPARalpha expression (P = 0.04) and 29 % higher PPARgamma expression (P = 0.002) than the SC group. In addition, the HF-FO group showed a 47 % higher PPARalpha expression (P = 0.0009) and 100 % higher PPARgamma expression (P < 0.0001) than the SC group.
The HF-L group showed reduced TC10 and GLUT-4 protein expressions than the SC group (−26 %, P = 0.04; −47 %, P = 0.006; respectively). Both the HF-L+FO and HF-FO groups showed higher protein expression of TC10 (+38 %, P = 0.005; +44 %, P = 0.003; respectively) than the HF-L group. The GLUT-4 protein expression was similar comparing both the FO groups and the SC group (Fig. 4).
Immunofluorescence
The HF-L group showed a more intense UCP1 staining than the SC group. In addition, the FO groups showed more intense UCP1 staining than both the SC and HF-L groups (Fig. 6).
iBAT hematoxylin and eosin staining and immunofluorescence. SC group showed typical multilocular brown adipocytes (a), whereas HF-L (b) and HF-L+FO (c) groups showed larger lipid droplets within iBAT. FO restored brown adipocyte cytoarchitecture in HF-FO (d). Likewise, tissues were labeled for UCP1 and showed different intensity of positive immunoreactions. Less intensity was found in SC group (a′), followed by HF-L group (b′). The FO in the diet complied with higher intensity of UCP1 labeling in a dose-dependent manner: in HF-L+FO group (c′), intermediate intense labeling, and HF-FO group (d′), the most intense labeling
Discussion
We observed that the HF-FO diets could reduce/prevent weight gain and adiposity without affecting food intake through stimulation of energy expenditure. The effects of FO on iBAT were associated with improvement in glucose intolerance and triacylglycerol levels, reduced gene expression of pro-inflammatory cytokines, and increased gene and protein expressions related to fatty acid/glucose uptake and oxidation in iBAT coupled with elevated protein and gene expressions of PPAR and thermogenic markers.
As expected, the HF-L group showed the greatest BM [13]. Interestingly, we observed that the ability of FO to prevent BM gain was dose dependent in agreement with previous studies. Rats fed a diet with 83 g/kg of n-3 PUFA had intermediate BM between high saturated fatty acids group and control group [4]. However, when 218 g/kg of FO was given to animals, there was a more reduced BM gain [2]. As illustrated by the adiposity index, this BM gain seen in the HF-L and the HF-L+FO groups was accounted for by increases in fat mass.
All the three HF groups showed similar food intake but elevated energy intake in comparison with the SC group due to high energy density of the HF diets [13]. N-3 PUFA does not alter the food intake as shown previously [14]. Therefore, the prevention of BM gain seen in the FO groups cannot be attributed to food intake changes rather than metabolic alterations.
RQ represents the fuel utilization and indicates unbalanced diets. In this way, the three HF diets were able to reduce it, representing increased fat oxidation. FQ, in comparison with RQ, is useful to predict energy homeostasis. A higher RQ than FQ means that the energy balance is positive. Therefore, energy intake is higher than energy expenditure leading to lipid accumulation and increased BM [15] as seen in the HF-L and the HF-L+FO. Mice fed a HF diet with lard showed slightly elevated RQ in comparison with FQ, indicating positive energy balance as seen in the present study [16].
When energy intake exceeds, energy expenditure (positive energy balance) obesity is established. In the present study, energy intake was similar among the three HF groups, but energy expenditure differed between the groups. HF-L group showed diminished energy expenditure compared to the SC group, justifying the positive energy balance and BM gain. On the other hand, the HF-FO group showed elevated energy expenditure compared with the SC group, which is indicative that the adaptive thermogenesis may be activated in these animals. Energy expenditure comprises three main elements: obligatory energy expenditure (required for cells and organs normal functioning), physical activity, and adaptive thermogenesis. The thermogenesis is known to be induced by either cold or diet and, in rodents, is mostly developed by BAT [17].
Leptin is produced and secreted by adipocytes, being responsible for energy homeostasis. The more the adipose tissue increases; the more the leptin is secreted, leading to elevated plasma leptin levels in obese animals (as in the HF-L group) [13, 18]. Central actions of leptin include the activation of sympathetic nervous system (SNS), and although obese mice are leptin resistant in relation to appetite control, the same does not happen regarding thermogenic control. Obese mice with hyperleptinemia show increased stimulus to iBAT [18]. Elevated expression of thermogenic genes and proteins was also found in the present study (i.e., beta3-AR, PGC1alpha, and UCP1). Overfeeding is responsible for stimulating the SNS, which releases catecholamines that act via beta3-AR. This fact leads to a variety of intracellular changes resulting in the transcription of PGC1alpha, which is highly expressed in BAT and functions as a coactivator of a range of nuclear receptors that upregulate UCP1 gene expression [7, 19]. As reported in our study, HF-obese mice and rats fed a cafeteria diet had elevated mRNA expression of beta3-AR and UCP1, compared to lean mice [20, 21].
On the contrary, although high-PUFA diets also yield augmentation of BAT adaptive thermogenesis, they are thought to modulate this tissue in a leptin-independent way [22]. Mice fed with a high-PUFA diet had elevated capacity for non-shivering thermogenesis and upregulated UCP1 [5]. Regardless some reports that every PUFA is capable of inducing thermogenesis to the same extent [22, 23], other studies affirm that different types of fatty acids modulate different responses in UCP1. For instance, mice fed a beef tallow diet (saturated and monounsaturated fatty acids) had lower stimulus to thermogenesis than corn oil-fed mice (PUFA) [24]. A high-FO diet given to rats elevated UCP1 mRNA expression to a bigger extent than a high-safflower diet (rich in n-6 PUFA) [6]. The gene and protein expression pattern observed in the present HF groups is in agreement with the previous studies.
One of the potential regulators of UCP1 and adaptive thermogenesis in this alternative HF diets (i.e., PUFA diets) is the PPAR [21]. PPARalpha and PPARgamma are well-known targets for FO [1]. PPARalpha is highly expressed in BAT and regulates gene expression of PGC1alpha and UCP1 [25]. Besides that, PPARalpha is also responsible for the transcription of genes involved in fatty acid oxidation, a process that is essential for thermogenesis [26]. PPARgamma presents the same roles as PPARalpha in BAT: gene expression regulation of PGC1alpha and UCP1 [27]. The activation of PPAR can lead to activated thermogenesis, which could explain our findings (HF-L+FO and HF-FO groups). The PPARbeta/delta was proven to increase fatty acid oxidation and thermogenesis (UCP1 expression) [28]. However, the PPARbeta/delta upregulation may not be associated with n-3 PUFA intake but rather with endogenous fatty acid ligands generated in BAT thermogenesis [29].
Although elevated UCP1 mRNA expression could represent recruitment of BAT, when only verifying this gene expression we cannot conclude that BAT function, i.e., thermogenic capacity, is activated. Recent publication considered that to estimate heat production in BAT, it is necessary to measure total UCP1 protein level per mouse [30]. In the present study, we did not evaluate the total UCP1 protein level per mouse. However, not only the gene expression was measured, but the protein expression of UCP1, which reinforces the results. Thus, it is prudent to conclude that thermogenic markers were upregulated in iBAT, without any inferences in the thermogenic capacity of the tissue.
Adaptive thermogenesis requires the fuel utilization from fatty acids or glucose. More importantly, this use may account for a whole-body metabolic improvement [22, 31]. Mice lack CD36 when exposed to cold for 12 h had a drastic reduction in body temperature. When exposed to cold for 24 h, the mice died, demonstrating that CD36 performs a crucial role in fatty acid uptake for thermogenesis in BAT [32]. In the same way, mice submitted to cold displayed an augmentation of CPT1 gene expression, representing an enhancement of fatty acid degradation [31]. The increased fatty acid substrate availability to thermogenesis was seen in the HF-L+FO and the HF-FO groups, with improvement of plasma triacylglycerol. Of interest, FO was already proven to elevate gene expression of CD36 and CPT1 in the liver [33, 34]. Our findings in the HF-L group, despite the growth in fatty acid uptake (represented by CD36), indicate that the mitochondria entrance for thermogenesis was compromised (diminished CPT1). Therefore, thermogenesis was probably affected.
An augmentation of glucose uptake for oxidation can be explained by elevated GLUT-4 [32] as we saw in the HF-L-FO and the HF-FO groups, leading to an improvement in glucose tolerance. Cold exposure in rats promoted augmentation of GLUT-4 expression and glucose uptake in BAT [35]. In this regard, TC10 is crucial for GLUT4 translocation in the cell membrane and glucose transportation within the cell [36]. FO fed mice had increased GLUT-4 gene expression in skeletal muscle associated with reduced plasma glucose [37]. On the contrary, a reduction in GLUT4 expression, as observed in the HF-L group, can represent diminished glucose transport within the cell and decreased substrate for thermogenesis. Female rats fed a high-fat diet showed reduced GLUT-4 protein expression, leading to insulin resistance [38].
Diabetic obese mice have elevated inflammation in BAT with increased immunostaining for IL-6 and TNFalpha [39]. In agreement with this report, the HF-L group showed iBAT inflammation, as indicated by elevated gene expressions of IL-6 and TNFalpha. Unlike the HF-L group, the groups receiving FO showed a pro-inflammatory pattern similar to the SC group, in which the dose was not decisive for the result. The n-3 PUFA are well-known anti-inflammatory compounds, acting, at least partially, via PPAR activation [1], and diminishing inflammation in the liver and white adipose tissue in mice models of obesity [3, 40]. Interestingly, BAT inflammation is associated with insulin resistance [41]. Therefore, it is possible to hypothesize that the reduced inflammation in the FO groups contributed to the improvement of their glucose tolerance and nutrient utilization.
In addition, the present study used a high fish oil dose in order to compare the effects of equivalent HF diets (same amount of energy and lipids) that only differ in fatty acid composition. The authors know that such dose (that is a non-physiological dose) cannot be translated to humans, and this is a limitation of the present study. However, the study may give an actual indication of the beneficial effects of fish oil.
In conclusion, HF diets rich in FO can prevent/reduce BM gain and adiposity and improve metabolic profile (glucose tolerance and triacylglycerol levels). The changes are related to elevated expression of proteins and genes associated with glucose (TC10 and GLUT4) and fatty acid uptake and oxidation (CD36 and CPT1), diminished gene expression of cytokines (IL-6 and TNFalpha), upregulation of PPAR (the three isoforms), and elevated expression of thermogenic markers (beta3-AR, PGC1alpha and UCP1). The results indicate that the FO has beneficial effects on mice iBAT.
References
Calder PC (2012) Mechanisms of action of (n-3) fatty acids. J Nutr 142(3):592S–599S. doi:10.3945/jn.111.155259
Lionetti L, Mollica MP, Sica R, Donizzetti I, Gifuni G, Pignalosa A, Cavaliere G, Putti R (2014) Differential effects of high-fish oil and high-lard diets on cells and cytokines involved in the inflammatory process in rat insulin-sensitive tissues. Int J Mol Sci 15(2):3040–3063. doi:10.3390/ijms15023040
Liu X, Xue Y, Liu C, Lou Q, Wang J, Yanagita T, Xue C, Wang Y (2013) Eicosapentaenoic acid-enriched phospholipid ameliorates insulin resistance and lipid metabolism in diet-induced-obese mice. Lipids Health Dis 12:109. doi:10.1186/1476-511X-12-109
Liu HQ, Qiu Y, Mu Y, Zhang XJ, Liu L, Hou XH, Zhang L, Xu XN, Ji AL, Cao R, Yang RH, Wang F (2013) A high ratio of dietary n-3/n-6 polyunsaturated fatty acids improves obesity-linked inflammation and insulin resistance through suppressing activation of TLR4 in SD rats. Nutr Res 33(10):849–858. doi:10.1016/j.nutres.2013.07.004
Sadurskis A, Dicker A, Cannon B, Nedergaard J (1995) Polyunsaturated fatty acids recruit brown adipose tissue: increased UCP content and NST capacity. Am J Physiol 269(2 Pt 1):E351–E360
Takahashi Y, Ide T (2000) Dietary n-3 fatty acids affect mRNA level of brown adipose tissue uncoupling protein 1, and white adipose tissue leptin and glucose transporter 4 in the rat. Br J Nutr 84(2):175–184
Richard D, Carpentier AC, Dore G, Ouellet V, Picard F (2010) Determinants of brown adipocyte development and thermogenesis. Int J Obes (Lond) 34(Suppl 2):S59–S66. doi:10.1038/ijo.2010.241
Kajimura S, Saito M (2014) A new era in brown adipose tissue biology: molecular control of brown fat development and energy homeostasis. Annu Rev Physiol 76:225–249. doi:10.1146/annurev-physiol-021113-170252
Morrison SF, Madden CJ, Tupone D (2014) Central neural regulation of brown adipose tissue thermogenesis and energy expenditure. Cell Metab 19(5):741–756. doi:10.1016/j.cmet.2014.02.007
Cypess AM, Lehman S, Williams G, Tal I, Rodman D, Goldfine AB, Kuo FC, Palmer EL, Tseng YH, Doria A, Kolodny GM, Kahn CR (2009) Identification and importance of brown adipose tissue in adult humans. N Engl J Med 360(15):1509–1517. doi:10.1056/NEJMoa0810780
Reeves PG, Nielsen FH, Fahey GC Jr (1993) AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J Nutr 123(11):1939–1951
Livesey G, Elia M (1988) Estimation of energy expenditure, net carbohydrate utilization, and net fat oxidation and synthesis by indirect calorimetry: evaluation of errors with special reference to the detailed composition of fuels. Am J Clin Nutr 47(4):608–628
Catta-Preta M, Martins MA, Cunha Brunini TM, Mendes-Ribeiro AC, Mandarim-de-Lacerda CA, Aguila MB (2012) Modulation of cytokines, resistin, and distribution of adipose tissue in C57BL/6 mice by different high-fat diets. Nutrition 28(2):212–219. doi:10.1016/j.nut.2011.05.011
Flachs P, Horakova O, Brauner P, Rossmeisl M, Pecina P, Franssen-van Hal N, Ruzickova J, Sponarova J, Drahota Z, Vlcek C, Keijer J, Houstek J, Kopecky J (2005) Polyunsaturated fatty acids of marine origin upregulate mitochondrial biogenesis and induce beta-oxidation in white fat. Diabetologia 48(11):2365–2375. doi:10.1007/s00125-005-1944-7
Longo KA, Charoenthongtrakul S, Giuliana DJ, Govek EK, McDonagh T, Distefano PS, Geddes BJ (2010) The 24-hour respiratory quotient predicts energy intake and changes in body mass. Am J Physiol Regul Integr Comp Physiol 298(3):R747–R754. doi:10.1152/ajpregu.00476.2009
Foucault AS, Even P, Lafont R, Dioh W, Veillet S, Tome D, Huneau JF, Hermier D, Quignard-Boulange A (2014) Quinoa extract enriched in 20-hydroxyecdysone affects energy homeostasis and intestinal fat absorption in mice fed a high-fat diet. Physiol Behav 128:226–231. doi:10.1016/j.physbeh.2014.02.002
Lowell BB, Spiegelman BM (2000) Towards a molecular understanding of adaptive thermogenesis. Nature 404(6778):652–660. doi:10.1038/35007527
Enriori PJ, Sinnayah P, Simonds SE, Garcia Rudaz C, Cowley MA (2011) Leptin action in the dorsomedial hypothalamus increases sympathetic tone to brown adipose tissue in spite of systemic leptin resistance. J Neurosci 31(34):12189–12197. doi:10.1523/JNEUROSCI.2336-11.2011
Sugimoto S, Nakajima H, Kodo K, Mori J, Matsuo K, Kosaka K, Aoi W, Yoshimoto K, Ikegaya H, Hosoi H (2014) Miglitol increases energy expenditure by upregulating uncoupling protein 1 of brown adipose tissue and reduces obesity in dietary-induced obese mice. Nutr Metab (Lond) 11(1):14. doi:10.1186/1743-7075-11-14
Shi H, Akunuru S, Bierman JC, Hodge KM, Mitchell MC, Foster MT, Seeley RJ, Reizes O (2009) Diet-induced obese mice are leptin insufficient after weight reduction. Obesity (Silver Spring) 17(9):1702–1709. doi:10.1038/oby.2009.106
Margareto J, Marti A, Martinez JA (2001) Changes in UCP mRNA expression levels in brown adipose tissue and skeletal muscle after feeding a high-energy diet and relationships with leptin, glucose and PPARgamma. J Nutr Biochem 12(3):130–137
Cannon B, Nedergaard J (2004) Brown adipose tissue: function and physiological significance. Physiol Rev 84(1):277–359. doi:10.1152/physrev.00015.2003
Takeuchi H, Matsuo T, Tokuyama K, Shimomura Y, Suzuki M (1995) Diet-induced thermogenesis is lower in rats fed a lard diet than in those fed a high oleic acid safflower oil diet, a safflower oil diet or a linseed oil diet. J Nutr 125(4):920–925
Mercer SW, Trayhurn P (1987) Effect of high fat diets on energy balance and thermogenesis in brown adipose tissue of lean and genetically obese ob/ob mice. J Nutr 117(12):2147–2153
Hondares E, Rosell M, Diaz-Delfin J, Olmos Y, Monsalve M, Iglesias R, Villarroya F, Giralt M (2011) Peroxisome proliferator-activated receptor alpha (PPARalpha) induces PPARgamma coactivator 1alpha (PGC-1alpha) gene expression and contributes to thermogenic activation of brown fat: involvement of PRDM16. J Biol Chem 286(50):43112–43122. doi:10.1074/jbc.M111.252775
Iizuka K, Wu W, Horikawa Y, Saito M, Takeda J (2013) Feedback looping between ChREBP and PPARalpha in the regulation of lipid metabolism in brown adipose tissues. Endocr J 60(10):1145–1153
Festuccia WT, Blanchard PG, Richard D, Deshaies Y (2010) Basal adrenergic tone is required for maximal stimulation of rat brown adipose tissue UCP1 expression by chronic PPAR-gamma activation. Am J Physiol Regul Integr Comp Physiol 299(1):R159–R167. doi:10.1152/ajpregu.00821.2009
Wang YX, Lee CH, Tiep S, Yu RT, Ham J, Kang H, Evans RM (2003) Peroxisome-proliferator-activated receptor delta activates fat metabolism to prevent obesity. Cell 113(2):159–170
Mottillo EP, Bloch AE, Leff T, Granneman JG (2012) Lipolytic products activate peroxisome proliferator-activated receptor (PPAR) alpha and delta in brown adipocytes to match fatty acid oxidation with supply. J Biol Chem 287(30):25038–25048. doi:10.1074/jbc.M112.374041
Nedergaard J, Cannon B (1831) UCP1 mRNA does not produce heat. Biochim Biophys Acta 5:943–949. doi:10.1016/j.bbalip.2013.01.009
Komatsu M, Tong Y, Li Y, Nakajima T, Li G, Hu R, Sugiyama E, Kamijo Y, Tanaka N, Hara A, Aoyama T (2010) Multiple roles of PPARalpha in brown adipose tissue under constitutive and cold conditions. Genes Cells 15(2):91–100. doi:10.1111/j.1365-2443.2009.01368.x
Bartelt A, Bruns OT, Reimer R, Hohenberg H, Ittrich H, Peldschus K, Kaul MG, Tromsdorf UI, Weller H, Waurisch C, Eychmuller A, Gordts PL, Rinninger F, Bruegelmann K, Freund B, Nielsen P, Merkel M, Heeren J (2011) Brown adipose tissue activity controls triglyceride clearance. Nat Med 17(2):200–205. doi:10.1038/nm.2297
Bargut TC, Frantz ED, Mandarim-de-Lacerda CA, Aguila MB (2014) Effects of a diet rich in n-3 polyunsaturated fatty acids on hepatic lipogenesis and beta-oxidation in mice. Lipids 49(5):431–444. doi:10.1007/s11745-014-3892-9
Yamazaki T, Nakamori A, Sasaki E, Wada S, Ezaki O (2007) Fish oil prevents sucrose-induced fatty liver but exacerbates high-safflower oil-induced fatty liver in ddy mice. Hepatology 46(6):1779–1790. doi:10.1002/hep.21934
Shimizu Y, Nikami H, Tsukazaki K, Machado UF, Yano H, Seino Y, Saito M (1993) Increased expression of glucose transporter GLUT-4 in brown adipose tissue of fasted rats after cold exposure. Am J Physiol 264(6 Pt 1):E890–E895
Chiang SH, Baumann CA, Kanzaki M, Thurmond DC, Watson RT, Neudauer CL, Macara IG, Pessin JE, Saltiel AR (2001) Insulin-stimulated GLUT4 translocation requires the CAP-dependent activation of TC10. Nature 410(6831):944–948. doi:10.1038/35073608
Yu YH, Wu SC, Cheng WT, Mersmann HJ, Shen TL, Ding ST (2011) The function of porcine PPARgamma and dietary fish oil effect on the expression of lipid and glucose metabolism related genes. J Nutr Biochem 22(2):179–186. doi:10.1016/j.jnutbio.2010.01.006
Nadal-Casellas A, Bauza-Thorbrugge M, Proenza AM, Gianotti M, Llado I (2013) Sex-dependent differences in rat brown adipose tissue mitochondrial biogenesis and insulin signaling parameters in response to an obesogenic diet. Mol Cell Biochem 373(1–2):125–135. doi:10.1007/s11010-012-1481-x
Han L, Tang MX, Ti Y, Wang ZH, Wang J, Ding WY, Wang H, Zhang Y, Zhang W, Zhong M (2013) Overexpressing STAMP2 improves insulin resistance in diabetic ApoE(-)/(-)/LDLR(-)/(-) mice via macrophage polarization shift in adipose tissues. PLoS One 8(11):e78903. doi:10.1371/journal.pone.0078903
Oliver E, McGillicuddy FC, Harford KA, Reynolds CM, Phillips CM, Ferguson JF, Roche HM (2012) Docosahexaenoic acid attenuates macrophage-induced inflammation and improves insulin sensitivity in adipocytes-specific differential effects between LC n-3 PUFA. J Nutr Biochem 23(9):1192–1200. doi:10.1016/j.jnutbio.2011.06.014
Valverde AM, Teruel T, Navarro P, Benito M, Lorenzo M (1998) Tumor necrosis factor-alpha causes insulin receptor substrate-2-mediated insulin resistance and inhibits insulin-induced adipogenesis in fetal brown adipocytes. Endocrinology 139(3):1229–1238. doi:10.1210/endo.139.3.5854
Acknowledgments
The authors would like to thank Mrs. Thatiany Marinho, Mrs. Aline Penna, Mrs. Gezileia Lau, and Mrs. Michele Soares for their technical assistance. This study was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Fundação de Amparo a Pesquisa do Estado do Rio de Janeiro (FAPERJ), and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Brazil.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bargut, T.C.L., Silva-e-Silva, A.C.A.G., Souza-Mello, V. et al. Mice fed fish oil diet and upregulation of brown adipose tissue thermogenic markers . Eur J Nutr 55, 159–169 (2016). https://doi.org/10.1007/s00394-015-0834-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00394-015-0834-0