Abstract
Background
In patients with incident heart failure, the risk of venous thromboembolism (VTE), defined as pulmonary embolism (PE) and/or deep venous thrombosis (DVT), is sparsely described, especially potential sex differences. We conducted an observational study to evaluate risk of VTE among male and female heart failure patients.
Methods
Population-based cohort study of patients diagnosed with incident heart failure during 2000–2015, identified by record linkage between nationwide registries in Denmark. Using a pseudo-value approach, we calculated relative risks [RR] of VTE at 1 and 3 years of follow-up. Crude VTE risk for males and females are reported and contrasted after adjustment for established clinical risk factors for VTE.
Results
A total of 32,330 heart failure patients were included, of which 15,238 (47%) were females. For the combined endpoint of VTE, female sex was associated with a higher risk (1-year adjusted RR: 1.30, 95% confidence interval [CI]: 0.97–1.73; 3-year adjusted RR: 1.34, 95% CI: 1.07–1.67) compared to male patients. For the individual endpoints of PE and DVT after 1-year of follow-up, female sex was only associated with a higher risk of PE and not DVT, compared to male patients. However, female sex was associated with a higher risk of both PE and DVT after 3 years of follow-up.
Conclusions
Among incident heart failure patients, female sex is associated with a higher risk of VTE, mainly driven by an excess risk of PE. This finding may help improve clinical decision-making regarding VTE prophylaxis in patients with heart failure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Heart failure (HF) is associated with a higher risk of thromboembolic events [1]. Although many studies have focused on arterial endothelial dysfunction, venous dysfunction is also present in HF, and contribute to a prothrombotic state in HF patients [2]. Venous thromboembolism (VTE), defined as a diagnosis of pulmonary embolism (PE) or deep vein thrombosis (DVT) [3], often occur secondary to clinical conditions such as HF [4, 5]. Several guidelines recommend that all hospitalized patients with HF receive VTE prophylaxis with anticoagulant medication if the risk–benefit ratio is favorable and the patient is not already anticoagulated [6, 7]; however, there is evidence that thromboprophylaxis is underused in the HF patient group, possibly due to the difficulties in accurately assessing the individual patient’s risk [8].
Several studies have investigated the association between VTE and HF, but no study has focused on the association between sex and the risk of VTE in HF patients [9, 10]. Previous studies assessing the association between sex and risk of VTE, not specifically in HF patients, have demonstrated inconsistent findings, but most studies found an excess risk among males [11,12,13]. In the HF population, potential sex differences in risk of VTE has not been investigated, but two studies demonstrated that HF appears to be a stronger risk factor for both PE and DVT in females than in males [9, 10]. However, one of these studies focused on the association between ejection fraction and thromboembolic risk; the study did not include DVT as an endpoint and had a low proportion of females [9]. The other study only examined VTE risk in HF patients during hospitalization [10].
Establishing potential sex-related differences in risk of VTE could improve the possibility of tailoring thromboprophylactic strategies in the HF population. Therefore, we performed an observational cohort study to assess the risk of VTE in HF patients according to sex, using Danish nationwide administrative registries.
Methods
Data sources
This observational cohort study used data from three nationwide registries: (1) The Danish Civil Registration System, which holds information on sex, date of birth, vital and emigration status of all persons living in Denmark [14]; (2) the Danish National Patient Registry [15], which has registered dates of hospital admission and discharge, and discharge diagnoses classified according to the 10th revision of the International Classification of Diseases (ICD) for more than 99% of hospital admissions in Denmark since 1994; (3) the National Prescription Registry [16], which contains data on all prescriptions dispensed from Danish pharmacies since 1994, coded according to the anatomical therapeutic chemical (ATC) classification system. Data were linked via a unique personal identification number used across all Danish national registries. Information from the three registries was retrieved until December 31, 2015. These registries have previously been validated [14,15,16], and the diagnoses of HF and VTE were found to have sufficiently high validity to conduct epidemiological investigations in cohorts consisting of patients with these records [17,18,19,20,21].
Study population
We used the Danish National Patient Registry to identify all patients diagnosed with a primary discharge diagnosis of incident (first-time hospital diagnosis) HF in the period January 1, 2000–December 31, 2015 (see e-Table 1 in the supporting materials). Patients who had previously experienced a VTE were excluded to examine the risk of first-time VTE. Since cancer patients represent a subgroup with high VTE risk [22] and specialized thromboprophylactic treatment regimens, we excluded patients with a diagnosis of cancer within 3 years before HF diagnosis. Using the Danish National Prescription Registry, we identified and excluded patients with a claimed prescription of anticoagulant therapy within 12 months prior to the HF diagnosis to avoid potential effect modification by anticoagulation.
Additional comorbidities were assessed at the date of discharge from the incident HF diagnosis and identified using the Danish National Patient Registry and the Danish National Prescription Registry. Ascertainment of baseline medication status (other than oral anticoagulants) was based on prescriptions claimed between 60 days before and 10 days after hospital discharge from incident HF to express the status in the follow-up period. The 10-day period after discharge was included to give a more accurate estimate of the baseline medication status as many patients may redeem a prescription within the first days after hospital discharge. ICD-codes and ATC-codes used to define comorbidities and medical therapy are provided in the supporting materials (see e-Table 1 in the supporting materials). As diagnoses from the emergency room commonly represent working diagnoses with a documented low validity, these diagnoses were not included in our study [23].
Outcomes
The main endpoint was a primary diagnosis of VTE, defined as pulmonary embolism (PE) and/or deep venous thrombosis (DVT) (for ICD-codes see e-Table 1 in the supporting materials). Besides the main combined endpoint of VTE, PE and DVT were analyzed as separate endpoints. For all study outcomes, only primary in-hospital codes were obtained, excluding emergency room and ambulatory diagnoses, likewise to ensure higher validity of the outcomes [23].
Statistical methods
Patient baseline characteristics were presented separately for men and women using means and standard deviation for continuous measures and proportions for categorical measures. We applied time-to-event survival analyses to describe the association between sex and risk of endpoints. Follow-up began at the date of discharge from the HF diagnosis. Patients were followed for 1 and 3 years until the outcome of interest, date of death, emigration, or end of study (December 31, 2015), whichever came first.
Absolute risks of all endpoints according to sex were estimated based on Aalen-Johansen [24] estimator for the cumulative incidence function assuming death as a competing risk and adjusted for well-established risk factors of VTE and potential confounders [age, atrial fibrillation, hypertension, vascular disease, diabetes, and chronic obstructive pulmonary disease [25, 26], as well as statin and aspirin therapy (as these therapies may influence the association between sex and VTE risk)]. Adjustment was obtained applying an inverse probability of treatment weights approach [27]. For the main analysis, a pseudo-value approach was used to compare relative risk (RR) of the endpoints at 1 and 3 years follow-up according to sex, using male patients as reference, to account for competing risk of death [28, 29]. The pseudo-values were analyzed by generalized linear regression analysis with log-link function and with sex as exposure and adjusted by the pre-specified potential confounders [25, 26].
In a secondary analysis, the role of age on the association between sex and risk of VTE was examined. The study population was stratified into three age groups (< 60 years, 60–80 years, and > 80 years) and the adjusted RR calculations were repeated. The youngest age group (< 60 years) was eliminated from the secondary analysis due to few events. To maintain robust estimates of the adjusted RR in this stratified analysis, we fitted a logistic regression model to calculate a propensity score based on the previously described risk factors for VTE. This approach was selected due to a potential mismatch between the number of identified risk factors for VTE, and the limited number of events within strata [30].
Analyses were performed using Stata version 14 (Stata Corporation, College Station, TX, USA). Results are reported with 95% confidence intervals (CI).
Ethical considerations
No ethical approval is required for studies based on data from administrative Danish registries. The study was approved by the Danish Data Protection Agency (J. No. File No. 2015-57-0001).
Results
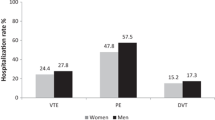
We identified 32,330 incident HF patients, of which 15,238 (47.1%) were females [Fig. 1]. The median follow-up time for the primary endpoint of VTE was 4.0 years (inter-quartile range 0.9–6.0). Baseline patient characteristics are summarized in Table 1. Mean age was higher among female patients than among male patients, 78.9 (SD 12.4) and 72.3 years (SD 13.3), respectively. The prevalence of vascular disease (including prior myocardial infarction) and diabetes was lower in female patients compared to male patients, but the prevalence of hypertension was higher in female patients. Male patients were more often using angiotensin-converting enzyme inhibitors, beta-blockers, aldosterone antagonists, statins, and aspirin compared to the female patients.
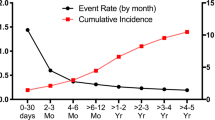
The number of events and absolute risks of VTE, DVT, and PE in each patient group at 1 and 3 years of follow-up are shown in Table 2. After 3 years of follow-up, the absolute risk of VTE was 2.0% for the female patients and 1.5% for the male patients, corresponding to incidence rates per 1000 person-years of 7.4 and 4.9, respectively. Especially, female patients had a high risk of PE (1.1%) compared to the male patients (0.8%), whereas the risk of DVT was similar between the female and male patients at 1-year follow-up, but higher among females after 3 years of follow-up. Adjusted cumulative incidence curves for VTE, DVT, and PE demonstrate a steady increase during follow-up, as seen in Figs. 2 and 3.
Adjusted cumulative incidence of deep vein thrombosis and pulmonary embolism according to sex. HF heart failure. Adjusted for age (continuous), hypertension (binary), vascular disease (binary), diabetes (binary), chronic obstructive pulmonary disease (binary), statin therapy (binary), and aspirin therapy (binary)
Multivariable analysis
For the combined endpoint of VTE, female sex was associated with a higher risk [1-year adjusted RR: 1.30, 95% confidence interval (CI): 0.97–1.73; 3-year adjusted RR: 1.34, 95% CI: 1.07–1.67] compared to male patients [Fig. 4]. After 1-year of follow-up, for the individual endpoints of PE and DVT, female sex was only associated with a higher risk of PE (adjusted RR: 1.62, 95% CI: 1.10–2.38) and not DVT (adjusted RR: 1.05, 95% CI: 0.61–1.83) compared to male patients. However, female sex was associated with a higher risk of both PE and DVT after 3 years of follow-up. Results were similar also when censoring patients who initiated oral anticoagulation during follow-up (data not shown).
Relative risks of all endpoints after 1 and 3 years of follow-up, according to sex (reference group: male patients). FUP follow-up, 95% CI 95% confidence interval. Analysis adjusted for age (continuous), hypertension (binary), vascular disease (binary), diabetes (binary), chronic obstructive pulmonary disease (binary), statin therapy (binary), and aspirin therapy (binary)
In the age-stratified analysis, female sex was associated with a higher risk of VTE (specifically a higher risk of PE), in the age groups “60–80 years” and “>80 years”; though, statistical significance was not reached (Fig. 5).
Relative risks of all endpoints after 1 and 3 years of follow-up, according to sex (reference group: male patients), and stratified into two age groups. FUP follow-up, 95% CI 95% confidence interval. Analysis adjusted for a propensity score based on age (continuous), hypertension (binary), vascular disease (binary), diabetes (binary), chronic obstructive pulmonary disease (binary), statin therapy (binary), and aspirin therapy (binary)
In the age-stratified analyses, female sex was associated with an overall higher risk of VTE in comparison to males: among patients at age 60–80 years, females had a 15% higher risk and 43% higher risk among patients aged > 80 years (Fig. 5). Importantly, the higher risk of VTE in females was mainly driven by higher risk of PE in both strata.
Discussion
In this nationwide cohort study of HF patients, we examined the difference in risk of VTE between male and female patients. We found a higher risk of VTE in female patients compared to male patients. This association was mainly driven by a higher risk of PE in female patients.
The mechanism by which HF is associated with a higher risk for VTE is not precisely determined [31]. In the general Danish population, VTE incidence is much lower than in the present heart failure population, but in contrast with our findings more frequent among men than women [32]. Multiple mechanisms lead to a high risk of VTE in this patient group, such as increased pulmonary pressure and decreased ejection fraction leading to increased venous stasis in the lower extremities [2, 33]. As ejection fraction decreases, exercise tolerance decreases resulting in poor mobility, which leads to a significantly higher risk for VTE [34]. HF is also associated with endothelial dysfunction, decreased microcirculatory blood flow, and increased blood levels of prothrombotic molecules, which all lead to a hypercoagulable state [2, 33].
Whether male and female patients have different physiological mechanisms that may explain the higher risk of PE among female patients has not been determined. In our study, female HF patients had a higher mean age, but a lower prevalence of vascular disease (such as prior myocardial infarction) and diabetes. Older age is a well-known risk factor of PE [35, 36] and could be part of the explanation for the difference in PE risk between the male and female patients. We performed a secondary analysis, stratifying the study population into two age groups (60–80 years and > 80 years), and found a clear trend for a higher risk of PE among female patients in both age groups. Another possible explanation could be that males and females do not share the same risk factors for VTE and especially for PE. For example, we did not have information about risk factors such as smoking, body mass index, and recent immobilization, which could be related to the risk of PE, but we do not expect that this missing information will completely explain our findings. In addition, females are uniquely exposed to well-known risk factors of VTE, such as hormone replacement therapy, estrogen containing oral contraceptives, pregnancy and the postpartum period [37, 38] as opposed to males. However, some of these unique risk factors might not be of much relevance in our older HF population or at least of perceived very little bearing. We had no access to information about ejection fraction, New York Heart Association (NYHA) class, or the specific aetiology of heart failure in our study population and could, therefore, not explore how such clinical characteristics would have influenced the observed sex disparities. In the general HF population, females more often have HF with preserved ejection fraction, whereas males more often have HF with reduced ejection fraction [39, 40], just as men often are more symptomatic as measured by NYHA class [41]. However, few studies have found an association between reduced ejection fraction and higher risk of VTE [9, 34], so this does not provide an explanation for the higher risk of VTE observed among women compared with men. In addition, although the baseline prevalence of chronic obstructive pulmonary disease was similar between sexes, we had no information on severity on this disease, which is known to be specifically associated with PE [42]. More males than females were using statins and antiplatelets at baseline in our study; probably reflecting the difference in vascular disease and diabetes history. This difference could potentially influence the outcome estimates; however, we included these medications in the adjusted analysis. Thus, future studies are needed to explain the etiological relation between female sex and PE risk, and whether the prognostic value of sex is consistent across different aetiologies of heart failure as well as within categories of ejection fraction.
Clinical importance
Several guidelines recommend that all hospitalized patients with HF receive VTE prophylaxis with anticoagulant medication if the risk–benefit ratio is favourable and the patient is not already anticoagulated [6, 7]. Recent evidence shows a large gap between these guidelines and clinical practice [43], and there is evidence that thromboprophylaxis is underused in the HF patient group, possibly due to the difficulties in accurately assessing the individual patient’s risk [8]. However, current guidelines only recommend anticoagulation to HF patients for long-term thromboprophylaxis if the patient has concomitant atrial fibrillation or another indication for anticoagulation [44, 45]. Given the bleeding risk associated with thromboprophylactic therapy, identification of high-risk subjects with an assumed benefit from treatment is essential. We found a higher risk of PE among female HF patients compared to male HF patients; thus, female HF patients may be a subgroup that deserves more attention in clinical practice. More focus on modifiable PE risk factors in the female HF population is warranted and specific thromboprophylactic strategies in women may be necessary to possibly reduce the burden of PE in these patients.
It should be noted that there are no completed randomized trials investigating the effect of long-term anticoagulation on VTE prevention specifically in patients with heart failure. However, previous trials of extended duration of anticoagulation in acute medically ill patients have yielded conflicting results, with some demonstrating a benefit of prolonged treatment [46, 47], while in others the antithrombotic effect was offset by bleeding risk [48]. Another trial is still ongoing [49].
Strengths and limitations
The large sample size uniquely possible with this type of cohort study minimized the risk of random error. Selection into the study was not an issue, since we investigated a nationwide population cohort of incident HF patients using administrative data, which also implies virtually no loss to follow-up.
The diagnosis of HF has previously been validated with a sensitivity of 29%, a specificity of 99%, and a positive predictive value of 81–100% [18, 50]; thus, we may have underestimated the true population HF prevalence, and also cannot be certain that all included patients had definite HF. Nonetheless, this most likely applies equally to both men and women and the risk of observational bias due to misclassification is, therefore, not a likely explanation for the observed associations. In addition, we included only patients with a primary discharge diagnosis of HF to optimize the probability of including only correctly identified patients with HF.
In Denmark, almost every patient presenting with VTE symptoms is admitted to and examined in a hospital setting. Hence, the accuracy of our findings depends on proper ICD-10 coding. Recent Danish validation studies comparing VTE diagnoses obtained from the Danish National Patient Registry with medical records have revealed positive predictive values of 75–88% for first-time VTE [19, 23]. Additionally, the positive predictive value for first-time VTE is similar between sexes, 78–86 and 71–88% for men and women, respectively [19, 23].
Generalizability of the study results
We were unable to distinguish between HF with preserved and reduced ejection fraction or estimate the functional classification, since we did not have access to echocardiograms. Furthermore, the study was carried out as a nationwide study in the Danish population, which is ethnically fairly homogeneous; thus, future studies are needed to evaluate if our findings hold in more ethnically diverse HF populations.
In conclusion, we examined the difference in risk of VTE between male and female HF patients.
Among incident HF patients, female sex is associated with a higher risk of VTE, mainly driven by an excess risk of PE. These findings may help improve clinical decision-making regarding VTE prophylaxis in patients with HF.
Abbreviations
- CI :
-
Confidence interval
- DVT :
-
Deep vein thrombosis
- HF :
-
Heart failure
- PE :
-
Pulmonary embolism
- RR :
-
Relative risk
- VTE :
-
Venous thromboembolism
References
Haeusler KG, Laufs U, Endres M (2011) Chronic heart failure and ischemic stroke. Stroke 42:2977–2982
Shantsila E, Lip GYH (2014) Thrombotic complications in heart failure: an underappreciated challenge. Circulation 130:387–389
Patel K, Fasanya A, Yadam S, Joshi AA, Singh AC, DuMont T (2017) Pathogenesis and epidemiology of venous thromboembolic disease. Crit Care Nurs Q 40:191–200
Edelsberg J, Hagiwara M, Taneja C, Oster G (2006) Risk of venous thromboembolism among hospitalized medically ill patients. Am J Heal Pharm 63:S16–S22
Spyropoulos AC, Hussein M, Lin J, Battleman D (2009) Rates of venous thromboembolism occurrence in medical patients among the insured population. Thromb Haemost 102:951–957
Geerts WH, Bergqvist D, Pineo GF, Heit JA, Samama CM, Lassen MR et al. (2008) Prevention of venous thromboembolism: American College of chest physicians evidence-based clinical practice guidelines (8th edn). Chest 133:381S–453S
Lindenfeld J, Albert NM, Boehmer JP, Collins SP, Ezekowitz JA, Givertz MM et al (2010) HFSA 2010 comprehensive heart failure practice guideline. J Card Fail 16:e1–e194
Mebazaa A, Spiro TE, Büller HR, Haskell L, Hu D, Hull R et al (2014) Predicting the risk of venous thromboembolism in patients hospitalized with heart failure. Circulation 130:410–418
Dries D, Rosenberg Y, Waclawiw M, Domanski M (1997) Ejection fraction and risk of thromboembolic events in patients with systolic dysfunction and sinus rhythm: evidence for gender differences in the studies of left ventricular dysfunction trials. J Am Coll Cardiol 29:1074–1080
Beemath A, Stein PD, Skaf E, Al Sibae MR, Alesh I (2006) Risk of venous thromboembolism in patients hospitalized with heart failure. Am J Cardiol 98:793–795
Silverstein MD, Heit JA, Mohr DN, Petterson TM, O’Fallon WM, Melton LJ (1998) Trends in the incidence of deep vein thrombosis and pulmonary embolism: a 25-year population-based study. Arch Intern Med 158:585–593
Bauersachs RM, Riess H, Hach-Wunderle V, Gerlach H, Carnarius H, Eberle S et al (2010) Impact of gender on the clinical presentation and diagnosis of deep-vein thrombosis. Thromb Haemost 103:710–717
Roseann Andreou E, Koru-Sengul T, Linkins L, Bates SM, Ginsberg JS, Kearon C (2008) Differences in clinical presentation of deep vein thrombosis in men and women. J Thromb Haemost 6:1713–1719
Pedersen CB (2011) The danish civil registration system. Scand J Public Health 39(7 Suppl):22–25
Lynge E, Sandegaard JL, Rebolj M (2011) The Danish National Patient Register. Scand J Public Health 39(7 Suppl):30–33
Kildemoes HW, Sørensen HT, Hallas J (2011) The Danish National Prescription Registry. Scand J Public Health 39(7 Suppl):38–41
Kümler T, Gislason GH, Kirk V, Bay M, Nielsen OW, Køber L et al (2008) Accuracy of a heart failure diagnosis in administrative registers. Eur J Heart Fail 10:658–660
Thygesen SK, Christiansen CF, Christensen S, Lash TL, Sørensen HT (2011) The predictive value of ICD-10 diagnostic coding used to assess Charlson comorbidity index conditions in the population-based Danish National Registry of Patients. BMC Med Res Methodol 11:83
Sundbøll J, Adelborg K, Munch T, Frøslev T, Toft Sørensen H, Erik Bøtker H et al. (2016) Positive predictive value of cardiovascular diagnoses in the Danish National Patient Registry: a validation study. BMJ Open 6:e012832
Schmidt M, Cannegieter SC, Johannesdottir SA, Dekkers OM, Horváth-Puhó E, Sørensen HT (2014) Statin use and venous thromboembolism recurrence: a combined nationwide cohort and nested case-control study. J Thromb Haemost 12:1207–1215
Delekta J, Hansen SM, AlZuhairi KS, Bork CS, Joensen AM (2018) The validity of the diagnosis of heart failure (I50.0–I50.9) in the Danish National Patient Register. Dan Med J 65:2–6
Zöller B, Ji J, Sundquist J, Sundquist K (2012) Risk of haemorrhagic and ischaemic stroke in patients with cancer: a nationwide follow-up study from Sweden. Eur J Cancer 48:1875–1883
Severinsen MT, Kristensen SR, Overvad K, Dethlefsen C, Tjønneland A, Johnsen SP (2010) Venous thromboembolism discharge diagnoses in the Danish National Patient Registry should be used with caution. J Clin Epidemiol 63:223–228
Gooley T, Leisenring W, Crowley J, Storer B (1999) Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med 18:695–706
Konstantinides SV, Torbicki A, Agnelli G, Danchin N, Fitzmaurice D, Galiè N et al (2014) 2014 ESC Guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J 35:3033–3080
Mahmoodi BK, Cushman M, Næss IA, Allison MA, Bos WJ, Brækkan SK et al (2017) Association of traditional cardiovascular risk factors with venous thromboembolism. Circulation 135:7–16
Cole SR, Hernán MA (2004) Adjusted survival curves with inverse probability weights. Comput Methods Programs Biomed 75:45–49
Klein J, Andersen P (2005) Regression modeling of competing risks data based on pseudovalues of the cumulative incidence function. Biometrics 61:223–229
Klein J, Logan B, Harhoff M, Andersen P (2007) Analyzing survival curves at a fixed point in time. Stat Med 26:4505–4519
Glynn RJ, Schneeweiss S, Stürmer T (2006) Indications for propensity scores and review of their use in pharmacoepidemiology. Basic Clin Pharmacol Toxicol 98:253–259
Haskins IN, Amdur R, Sarani B, Vaziri K (2015) Congestive heart failure is a risk factor for venous thromboembolism in bariatric surgery. Surg Obes Relat Dis 11:1140–1145
Severinsen MT, Johnsen SP, Tjønneland A, Overvad K, Dethlefsen C, Kristensen SR (2010) Body height and sex-related differences in incidence of venous thromboembolism: a Danish follow-up study. Eur J Intern Med 21:268–272
Dean SM, Abraham W (2010) Venous thromboembolic disease in congestive heart failure. Congest Hear Fail 16:164–169
Howell MD, Geraci JM, Knowlton AA (2001) Congestive heart failure and outpatient risk of venous thromboembolism: a retrospective, case-control study. J Clin Epidemiol 54:810–816
Rothberg MB, Lindenauer PK, Lahti M, Pekow PS, Selker HP (2011) Risk factor model to predict venous thromboembolism in hospitalized medical patients. J Hosp Med 6:202–209
Stein PD, Hull RD, Kayali F, Ghali WA, Alshab AK, Olson RE (2004) Venous thromboembolism according to age: the impact of an aging population. Arch Intern Med 164:2260–2265
Heit JA, Kobbervig CE, James AH, Petterson TM, Bailey KR, Melton LJ (2005) Trends in the incidence of venous thromboembolism during pregnancy or postpartum: a 30-year population-based study. Ann Intern Med 143:697–706
Gomes MPV, Deitcher SR (2004) Risk of venous thromboembolic disease associated with hormonal contraceptives and hormone replacement therapy: a clinical review. Arch Intern Med 164:1965–1976
van Heerebeek L, Paulus WJ (2016) Understanding heart failure with preserved ejection fraction: where are we today? Netherlands Hear J 24:227–236
Andersson C, Vasan RS (2014) Epidemiology of heart failure with preserved ejection fraction. Hear Fail Clin 10:377–388
Meyer S, Teerlink JR, Metra M, Ponikowski P, Cotter G, Davison BA et al (2017) Sex differences in early dyspnea relief between men and women hospitalized for acute heart failure: insights from the RELAX-AHF study. Clin Res Cardiol 106:280–292
Rizkallah J, Man SFP, Sin DD (2009) Prevalence of pulmonary embolism in acute exacerbations of COPD: a systematic review and metaanalysis. Chest 135:786–793
Jois-Bilowich P, Michota F, Bartholomew JR, Glauser J, Diercks D, Weber J et al (2008) Venous thromboembolism prophylaxis in hospitalized heart failure patients. J Card Fail 14:127–132
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, Drazner MH et al (2013) 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 62:e147–e239
Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS et al (2016) 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur Heart J 37:2129–2200
Cohen AT, Harrington RA, Goldhaber SZ, Hull RD, Wiens BL, Gold A et al (2016) Extended thromboprophylaxis with betrixaban in acutely Ill medical patients. N Engl J Med 375:534–544
Hull RD, Schellong SM, Tapson VF, Monreal M, Samama MM, Nicol P et al (2010) Extended-duration venous thromboembolism prophylaxis in acutely ill medical patients with recently reduced mobility: a randomized trial. Ann Intern Med 153:8–18
Albertsen IE, Larsen TB, Rasmussen LH, Overvad TF, Lip GY (2012) Prevention of venous thromboembolism with new oral anticoagulants versus standard pharmacological treatment in acute medically ill patients. A systematic review and meta-analysis. Drugs 72:1755–1764
Raskob GE, Spyropoulos AC, Zrubek J, Ageno W, Albers G, Elliott G et al (2016) The MARINER trial of rivaroxaban after hospital discharge for medical patients at high risk of VTE. Design, rationale, and clinical implications. Thromb Haemost 115:1240–1248
Mard S, Nielsen FE (2010) Positive predictive value and impact of misdiagnosis of a heart failure diagnosis in administrative registers among patients admitted to a University Hospital cardiac care unit. Clin Epidemiol 2:235–239
Acknowledgements
Line Melgaard, Peter Brønnum Nielsen, and Flemming Skjøth had full access to all of the data in this study and take responsibility for the integrity of the data and the accuracy of the data analysis. All authors contributed to the design, analysis, interpretation of data, drafting the article, or revising it critically for important intellectual content and approved the final version to be published. Statistics Denmark provided the data for this study.
Funding
The Obel Family Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Professor Lip: Consultant for Bayer/Janssen, BMS/Pfizer, Biotronik, Medtronic, Boehringer Ingelheim, Novartis, Verseon and Daiichi-Sankyo. Speaker for Bayer, BMS/Pfizer, Medtronic, Boehringer Ingelheim, and Daiichi-Sankyo. No fees are directly received personally. Associate Professor Larsen: An investigator for Janssen Scientific Affairs, LLC, and Boehringer Ingelheim. Speaker for Bayer, BMS/Pfizer, Janssen Pharmaceuticals, Takeda, Roche Diagnostics, and Boehringer Ingelheim. Peter Brønnum Nielsen: Speaker for Boehringer Ingelheim and Bayer Pharma AG; research Grant from BMS/Pfizer. Flemming Skjøth: Consultancy fees from Bayer. Other authors—none declared. Profs Lip and Larsen are guarantors of this paper.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Melgaard, L., Nielsen, P.B., Overvad, T.F. et al. Sex differences in risk of incident venous thromboembolism in heart failure patients. Clin Res Cardiol 108, 101–109 (2019). https://doi.org/10.1007/s00392-018-1329-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-018-1329-9