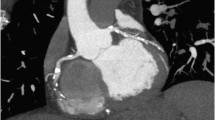
Abstract
In patients with stable symptoms suggestive of coronary artery disease (CAD), coronary CT angiography (CTA) allows for assessing several aspects of coronary atherosclerosis. Coronary artery stenoses are reliably detected, plaque formation can be quantified and characterized as calcified or non-calcified, and markers of potential instability such as expansive vascular remodeling, spotty calcification, and atheroma size can be described. As opposed to invasive coronary angiography, CTA visualizes the vessel lumen and wall. Being a purely anatomic test, even small plaques are detected with excellent sensitivity. At the other end of the spectrum, the hemodynamic significance of large plaque burden is sometimes overestimated. This may in part be corrected using mathematical modeling. Computational fluid dynamics of vascular anatomy and subtended myocardial mass provide for measures of CT-based fractional flow reserve (FFRCT). Large prospective trials have demonstrated the diagnostic utility of CTA in particular for ruling out obstructive CAD. The ability to detect non-obstructive plaque allows for improved risk prediction in comparison with functional testing, because even patients with sub-clinical atherosclerosis can be identified and selected for preventive medical treatment. This has led to incorporating CTA as one of several possible diagnostic tests for the evaluation of stable CAD in the actual European guidelines. Recently, it has even been forwarded as the first-line diagnostic test in the United Kingdom. The clinical implications of novel quantitative CTA-derived parameters such as FFRCT and non-calcified plaque volume are being examined in ongoing studies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Over the past years, the diagnostic value of coronary CT angiography (CTA) in patients with stable coronary artery disease (CAD) has been firmly established. The current guidelines of the European Society of Cardiology (ESC) consider it an indication with a IIa class of recommendation (“should be considered”) in the presence of a low to intermediate pre-test probability of CAD [1]. The English National Institute for Health and Care Excellence (NICE) in its 2016 update recommends CTA as the first-line investigation in patients with atypical or typical angina symptoms or in the presence of EKG-changes in asymptomatic patients [2]. These recommendations are based on published data on the diagnostic accuracy of CTA and the prognostic implications.
Compared with all other non-invasive approaches to investigating CAD in stable patients, CTA as discussed here is based on anatomic information and not on functional-hemodynamic testing. Strengths and limitations of CTA can be derived from this anatomical approach. As opposed to invasive coronary angiography (ICA), CTA not only provides for a luminogram, i.e., not only visualizes the inner contour of the coronary vessels, but also allows for interrogating the vessel wall, including plaque formation, non-calcified plaque volume, extent of calcification, vascular remodeling patterns, and further potential markers of unstable or progressive plaque development. Accordingly, three aspects of CAD are visualized: (1) coronary artery stenosis; (2) coronary artery plaque formation (presence—calcification—volume); and (3) markers of instability such as expansive vascular remodeling, spotty calcification, and atheroma size (Table 1). Finally, the anatomic image information lends itself to mathematical modeling using computational fluid dynamics, generating CTA-derived fractional flow reserve (FFRCT), so that even functional information, albeit purely derived from anatomy, can be made available. The clinical implications remain to be fully established, but certainly CTA is bound for increasing use in patients with stable CAD.
Technique
The advances in CTA would not be possible without astonishing technical developments over the past years. In particular, image quality and robustness have greatly increased. Currently, the rotation of the spiral CT X-ray source around the patient is possible in ≤ 250 ms. Sometimes, even two X-ray sources are installed (“dual-source CT”), complementing each other and thus halving the image acquisition time. This is coupled with an increasing number of detector rows, allowing for covering larger volumes per time interval. Modern algorithms employ high-pitch scanning modes, meaning increased table speed during rotation of X-ray source/gantry in relation to slice thickness. Overall, image acquisition times clearly below 100 ms can be obtained (down to appr. 65 ms), and the complete coronary system can be covered within 166 ms. The tube current (mAs) has been steadily increased, in turn allowing for decreased tube voltage (kV). The combination of this setting together with prospective ECG triggering or even “flash imaging” within one cardiac cycle, high-pitch image protocols, and up to 400 detector rows, accounts for lowering radiation dose down to < 1 mSv. In patients weighing < 100 kg who are accordingly prepared, a mean radiation dose of 0.3 mSv can be achieved using third generation dual-source CT technology [3]. In the German Cardiac CT registry, from 2009 to 2014, the median value of radiation dose associated with CTA declined from 5.6 to 2.5 mSv [4].
Patients with stable angina and low risk
Two large trials have analyzed the ability of CTA to diagnose stable CAD in a prospective, randomized manner. The Scottish COmputed Tomography of the HEART (SCOT-HEART) trial examined 4146 patients in 12 centers in the United Kingdom [5]. Patients with suspected angina due to CAD, mean age 57 years, were randomly assigned to either undergo CTA in addition to usual care (CTA group) or usual care alone (control group). The primary endpoint was the increase in certainty with which after 6 weeks the patients’ angina could really be attributed to CAD. This endpoint was met in that compared with the control group, the certainty of the diagnosis of angina due to CAD increased significantly (RR 1.79, CI 1.62–1.96, p < 0.0001). ICA was more appropriately used in the CTA group in that it was more likely to show CAD compared with the control group [6]. Preventive medical therapy was significantly altered, and after a median follow-up of 20 months, the rate of fatal and non-fatal myocardial infarction was halved in the CTA group [6].
The PROspective Multicenter Imaging Study for Evaluation of chest pain (PROMISE) recruited 10,003 patients with symptoms suggestive of CAD in 193 centers in the USA and Canada [7]. Their mean age was 61 years. They were randomly assigned to either undergo CTA (CTA group) or a functional test, i.e., exercise electrocardiography, nuclear stress testing, or stress echocardiography (control group). Most patients in the control group, appr. 67%, underwent nuclear stress testing. The primary endpoint was a combination of death, myocardial infarction, hospitalization for unstable angina, or major procedural complication. Secondary endpoints included invasive cardiac catheterization not showing significant CAD. Overall, median follow-up was 25 months. The rate of the primary endpoint was lower than expected, reaching only 3.2% in the total study group, and there was no overall difference between the two study groups. Although patients in the CTA group underwent ICA more frequently (12 vs. 8%), catheterization less frequently showed no significant CAD (28 vs. 53%), arguing for a more appropriate use of ICA. Accordingly, the rate of revascularization was higher in the CTA group (6 vs. 3%) [7]. On the other hand, the ability to identify truly normal anatomy and thus a truly good prognosis was improved in the CTA group [8]. The rate of normal test results was much lower (33 vs. 78%), and among these patients, the event rate was only 0.9% compared with 2.1% in patients with a normal test result in the control group. CTA identified a substantial number of patients who had no significant CAD but still non-obstructive coronary plaques. Indeed, these patients were the majority (n = 2461, 55%). They could be discriminated from those with no coronary plaques (n = 1505, 33%) and it could be demonstrated that more events occurred in this group compared with the latter (3.0 vs. 0.9%). A similar risk discrimination of strictly normal vs. mildly abnormal test results was not feasible on the basis of functional testing in the control group. In summary, risk stratification was improved using CTA [8].
Registry data corroborate the above data from the randomized trials. A Danish multicenter cohort study confirmed the prognostic implications of detecting non-obstructive coronary plaque formation using CTA [9]. Nearly 17,000 stable patients with symptoms suggestive of CAD underwent CTA scanning in the years from 2008 to 2012. The CT-scanners had as minimum technical requirement 64 slices. Patients’ mean age was 57 years, 57% were females. They were followed over a median of 3.6 years. The main endpoint was a combination of late revascularization > 90 days after CTA, myocardial infarction, and all-cause death. The composite endpoint occurred in 486 patients. Risk of an event was 1.5% in patients without CAD. In comparison, even after adjustment for risk factors, increased relative risks were observed both for non-obstructive CAD [hazard ratio (HR) 1.28] as well as obstructive one-vessel CAD (HR 1.83), two-vessel CAD (HR 2.97), and three-vessel/left main CAD (HR 4.41). Accordingly, CTA provided for risk discrimination even within the patient group without obstructive CAD.
Another registry in Denmark compared the outcome of stable patients being diagnosed using CTA or functional tests [10]. Out of a total of 86,705 patients with a mean age of 57 years, 32,961 underwent CTA. Over a median follow-up of 3.6 years, the risk of all-cause mortality and myocardial infarction was analyzed. Compared with functional testing, there was a significantly higher use of medications (statins 15.9 vs. 9.1%, aspirin 12.7 vs. 8.5%) in the CTA group. There was also a significant increase in catheterization (14.7 vs. 10.1%) and percutaneous revascularization (3.8 vs. 2.1%). After adjustment for risk factors, CTA was associated with comparable all-cause mortality [hazard ratio (HR) 0.96] and with a decreased risk of myocardial infarction (HR 0.71).
Taken together, these data suggest that the ability of CTA to visualize coronary anatomy and identify obstructive CAD in stable symptomatic patients allows for effective risk stratification. As opposed to functional testing, patients without obstructive CAD can be further categorized into those with truly normal coronary arteries who do not need further treatment and those with non-obstructive plaque formation (Figs. 1, 2, 3). The latter appear to benefit from medical therapy with statins and aspirin, explaining lower myocardial infarction rates compared with functional testing.
55-year-old man. Coronary CTA shows some coronary plaques, partially calcified. Overall, plaque burden is low. There is no suggestion of an increased coronary risk. His brother had suffered an acute myocardial infarction at age 48; he had to be resuscitated. LAD left anterior descending coronary artery, LCX left circumflex coronary artery, RCA right coronary artery
45-year-old woman without known cardiovascular risk factors. In the exercise stress test done in the setting of a ‘check-up examination’, electrocardiographic changes suggestive of ischemia had been observed. CTA shows heavily calcified plaques in the left anterior descending coronary artery, but no obstructive stenosis. The other major coronary vessels appear completely normal. Additional risk stratification showed normal values for lipoprotein(a), homocysteine, and potential markers of a thrombophilic disorder. Abbreviations as in Fig. 1
79-year-old man with persistent atrial fibrillation and progressive exertional dyspnea. Despite the arrhythmia (see electrocardiogram at the top), CTA provides for diagnostic image quality. There is a calcified plaque in the proximal left anterior descending coronary artery and distal to it a non-calcified plaque with non-obstructive stenosis. Obstructive coronary artery disease can be ruled out. The cross-sectional images of the non-calcified lesion illustrate the amount of remodeling. Abbreviations as in Fig. 1
Identification of high coronary risk
The ability of CTA to identify obstructive CAD is undisputed. Obviously, when using ICA as reference standard, comparing anatomy with anatomy, the agreement between the two methods is high (Figs. 4, 5, 6) [11]. However, because also the vessel wall is shown and not just the lumen, there is a tendency for overestimating the degree of stenosis compared with ICA. Whereas the sensitivity for detecting obstructive CAD has been reported to be 91–100%, specificity ranged between 37–99% [11]. With ICA as the reference, the area under the curve (AUC) of CTA for detecting obstructive CAD was 0.97–0.98, whereas AUC was 0.62 for exercise stress testing and 0.64 for nuclear stress testing [11]. A different picture emerged when comparing CTA to invasive fractional flow reserve [12]. On a vessel-by-vessel level, sensitivity and specificity of CTA were 91 and 58%, and the AUC was 0.85 [12]. The decrease in specificity is explained by visual overestimation of the hemodynamic effects of large plaques, sometimes perhaps also by imaging artifacts related to severe calcification, i.e., ‘blooming’ and beam hardening. It can be partially compensated for using computational fluid dynamics and generating FFRCT (see below).
63-year-old man with atypical chest pain. CTA shows voluminous plaque formation in the left anterior descending coronary artery (LAD), causing obstructive stenosis. The curved CTA image reconstruction shown here only displays a small portion of the overall image volume, and the ostium of the left circumflex coronary artery is leaving the reconstruction plane; it is not stenotic. In the proximal LAD, expansive remodeling is apparent. Invasive catheterization confirms the findings, but only provides for a luminogram. Abbreviations as in Fig. 1
51-year-old man with previous stent implantation in the left anterior descending coronary artery and atypical angina. Just proximal to the stent, CTA visualizes an obstructive de novo stenosis, confirmed by invasive angiography. The previously implanted stent has a diameter of 3 mm. Its lumen is clearly visible, and restenosis can be excluded on the basis of CTA
69-year-old man with diffuse coronary atherosclerosis. The Agatston calcium score measures 967. A previously implanted stent is visualized in the left anterior descending coronary artery (LAD). Despite the extensive calcification, obstructive stenoses can be ruled in the LAD, including the stented vessel segment. In the right coronary artery, however, a non-calcified plaque is causing a stenosis, confirmed by the invasive angiogram (arrows). Abbreviations as in Fig. 1
For over a decade, characteristics of high-risk plaques have been analyzed on the basis of CTA [13, 14]. One should not forget that the most important predictor of events is obstructive stenosis [14]. However, further independent risk predictors have been identified such as expansive vascular remodeling, low-attenuation plaque, and spotty (rather than ‘chunky’) calcification [14, 15]. Of note, the immediate precursor plaque of an acute coronary syndrome usually displays a diameter stenosis > 70%. However, this is due to recent accelerated plaque growth, a hallmark of instability. Plaques being interrogated months before the acute event tend to be non-obstructive [15]. A large plaque burden attests to previous plaque growth and inherent instability. Accordingly, it is interesting to quantify coronary atherosclerosis, which is increasingly becoming feasible using CTA and semi-automated algorithms [16]. Indeed, non-calcified plaque volume per se and CT-features of high-risk plaques can be used as predictors of myocardial ischemia, independent of stenosis severity [17, 18].
The recently published Progression of AtheRosclerotic PlAque DetermIned by Computed TomoGraphic Angiography Imaging (PARADIGM) study was a prospective, multinational registry of patients without history of CAD who underwent serial CTA with a minimum inter-scan interval of 2 years [19]. At 13 sites in 7 countries, 1255 patients were included between 2003 and 2015. Plaque progression was compared between statin-naïve (n = 474) and statin-taking (n = 781) patients. Plaques in statin-taking patients displayed a slower rate of overall progression but more rapid progression of calcification. The annual incidence of new high-risk plaque features was lower. In summary, statins appeared to be associated with stabilization of cholesterol-rich plaques toward more calcified lesions, probably representing a healing process. Of note, an increase in coronary calcification under statin treatment has also been reported in serial non contrast-enhanced CT-studies of coronary artery calcium progression [20].
Can CT angiography yield functional test results?
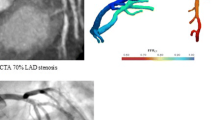
Despite its characteristic of being an anatomic test, CTA can be used to generate FFR values, FFRCT, on the basis of computational fluid dynamics (Fig. 7). In the largest trial so far, invasively measured FFR was used as the reference standard [21]. Ischemia was defined by an FFR-threshold value of 0.8. When using FFRCT instead of CTA alone, sensitivity slightly decreased from 94 to 86%, whereas specificity increased from 34 to 79%. The AUC significantly increased from 0.81 to 0.90 [21]. A substudy further analyzed the ability to predict lesion-specific ischemia, demonstrating that apart from stenosis assessment and FFRCT, the amount of low density non-calcified plaque volume also was an independent predictor of ischema [18]. In Prospective longitudinal trial of FFRCT: outcome and resource impacts (PLATFORM), consecutive stable patients with an intermediate pre-test probability of CAD either underwent usual testing (n = 287) or CTA (n = 297) with selective FFRCT [22]. Most of the patients were planned for ICA (n = 380). In this study arm, among those undergoing CTA and FFRCT, 60% did not need to undergo ICA because non-invasive testing could rule out obstructive CAD. Accordingly, there was a marked reduction in the finding of no obstructive CAD at ICA, and the majority of invasive procedures could be avoided [22].
62-year-old man with extensive calcification of mid left anterior descending coronary artery (LAD). On the basis of anatomy, CTA suggests obstructive stenosis but may over-exaggerate the degree of stenosis. CTA-based fractional flow reserve gives a value of 0.57 in the distal, arguing in favor of hemodynamic significance of the lesion. A stress magnetic resonance imaging study showed a reversible perfusion defect of the apical left ventricular myocardium, confirming hemodynamic significance of the LAD lesion (images not shown). Invasive catheterization is shown at right. There is an intermediate mid-LAD stenosis and obstructive stenosis of the ostium of the second diagonal branch (arrow). Bifurcation percutaneous intervention was performed (far right side, arrow)
In the setting of the SYNTAX II trial, CTA was used to compute a non-invasive functional SYNTAX score [23]. In 77 patients with 3-vessel CAD, CTA measures were first used to generate a CTA SYNTAX score simply on the basis of the anatomic information provided for by CTA and compared to the invasive angiographic SYNTAX score. CTA-lesions producing ischemia, i.e., displaying an FFRCT ≤ 0.8, were included in the CTA-based functional SYNTAX score and compared to invasive FFR (or instantaneous wave-free ratio, iFR) as the reference standard. The anatomic CTA-score could be obtained in 86% of the patients, the functional non-invasive score in 80. The CTA-score overestimated the invasive SYNTAX score, whereas the non-invasive functional score on the basis of CTA and FFRCT yielded similar results as the invasively determined functional SYNTAX score incorporating FFR and iFR. Overall, the combination of CTA and FFRCT performed well in patients with 3-vessel CAD [23].
In summary, FFRCT can help to improve the specificity of CTA in the presence of indeterminate lesions. Obviously, the results depend on image quality and source data, and even with the most elaborated mathematical algorithms, the basis of the test still is anatomy. Interestingly, CTA-studies combining various measures of coronary atherosclerosis have demonstrated that independent of stenosis severity and FFRCT, plaque volume also contributes to predicting ischemia [17, 18]. Nevertheless, in the presence of extensive coronary atherosclerosis, CTA sometimes yields low specificity with regard to hemodynamically significant lesions. This is perceived as the method’s Achilles’ heel, and FFRCT may help to overcome it.
References
Montalescot G, Sechtem U, Achenbach S et al (2013) 2013 ESC guidelines on the management of stable coronary artery disease: the Task Force on the management of stable coronary artery disease of the European Society of Cardiology. Eur Heart J 34:2949–3003 (Erratum in: Eur Heart J 2014;35:2260–2261)
National Institute for Health and Clinical Excellence (2016) Chest pain of recent onset: assessment and diagnosis of recent onset chest pain or discomfort of suspected cardiac origin (update). CG95. National Institute for Health and Clinical Excellence, London
Hell MM, Bittner D, Schuhbaeck A, Muschiol G, Brand M, Lell M, Uder M, Achenbach S, Marwan M (2014) Prospectively ECG-triggered high-pitch coronary angiography with third-generation dual-source CT at 70 kVp tube voltage: feasibility, image quality, radiation dose, and effect of iterative reconstruction. J Cardiovasc Comput Tomogr 8:418–425
Schmermund A, Marwan M, Hausleiter J et al (2017) Declining radiation dose of coronary computed tomography angiography: German cardiac CT registry experience 2009–2014. Clin Res Cardiol 106:905–912
SCOT-HEART Investigators (2015) CT coronary angiography in patients with suspected angina due to coronary heart disease (SCOT-HEART): an open-label, parallel-group, multicentre trial. Lancet 385:2383–2391 (Erratum in: Lancet 2015;385:2354)
Williams MC, Hunter A, Shah ASV et al, SCOT-HEART Investigators (2016) Use of coronary computed tomographic angiography to guide management of patients with coronary disease. J Am Coll Cardiol 67:1759–1768
Douglas PS, Hoffmann U, Patel MR et al (2015) Outcomes of anatomical versus functional testing for coronary artery disease. N Engl J Med 372:1291–1300
Hoffmann U, Ferencik M, Udelson JE et al (2017) Prognostic value of noninvasive cardiovascular testing in patients with stable chest pain: insights from the PROMISE trial (Prospective Multicenter Imaging Study for Evaluation of Chest Pain). Circulation 135:2320–2332
Nielsen LH, Bøtker HE, Sørensen HT et al (2017) Prognostic assessment of stable coronary artery disease as determined by coronary computed tomography angiography: a Danish multicentre cohort study. Eur Heart J 38:413–421
Jørgensen ME, Andersson C, Nørgaard BL et al (2017) Functional testing or coronary computed tomography angiography in patients with stable coronary artery disease. J Am Coll Cardiol 69:1761–1770
Nielsen LH, Ortner N, Nørgaard BL, Achenbach S, Leipsic J, Abdulla J (2014) The diagnostic accuracy and outcomes after coronary computed tomography angiography vs. conventional functional testing in patients with stable angina pectoris: a systematic review and meta-analysis. Eur Heart J Cardiovasc Imaging 15:961–971
Danad I, Szymonifka J, Twisk JWR, Nørgaard BL, Zarins CK, Knaapen P, Min JK (2017) Diagnostic performance of cardiac imaging methods to diagnose ischaemia-causing coronary artery disease when directly compared with fractional flow reserve as a reference standard: a meta-analysis. Eur Heart J 38:991–998
Motoyama S, Sarai M, Harigaya H et al (2009) Computed tomographic angiography characteristics of atherosclerotic plaques subsequently resulting in acute coronary syndrome. J Am Coll Cardiol 54:49–57
Motoyama S, Ito H, Sarai M, Kondo T et al (2015) plaque characterization by coronary computed tomography angiography and the likelihood of acute coronary events in mid-term follow-up. J Am Coll Cardiol 66:337–346
Chang HJ, Lin FY, Lee SE et al (2018) Coronary atherosclerotic precursors of acute coronary syndromes. J Am Coll Cardiol 71:2511–2522
Kishi S, Magalhães TA, Cerci RJ et al (2016) Total coronary atherosclerotic plaque burden assessment by CT angiography for detecting obstructive coronary artery disease associated with myocardial perfusion abnormalities. J Cardiovasc Comput Tomogr 10:121–127
Driessen RS, Stuijfzand WJ, Raijmakers PG et al (2018) Effect of plaque burden and morphology on myocardial blood flow and fractional flow reserve. J Am Coll Cardiol 71:499–509
Gaur S, Øvrehus KA, Dey D et al (2016) Coronary plaque quantification and fractional flow reserve by coronary computed tomography angiography identify ischaemia-causing lesions. Eur Heart J 37:1220–1227
Lee S-E, Chang H-J, Sung JM et al (2018) Effects of statins on coronary atherosclerotic plaques: the progression of atherosclerotic plaque determined by computed tomographic angiography imaging (PARADIGM) study. J Am Coll Cardiol Img. https://doi.org/10.1016/j.jcmg.2018.04.015
Dykun I, Lehmann N, Kälsch H et al (2016) Statin medication enhances progression of coronary artery calcification: the Heinz Nixdorf recall study. J Am Coll Cardiol 68:2123–2125
Nørgaard BL, Leipsic J, Gaur S et al (2014) Diagnostic performance of noninvasive fractional flow reserve derived from coronary computed tomography angiography in suspected coronary artery disease: the NXT trial (analysis of coronary blood flow using CT angiography: next steps). J Am Coll Cardiol 63:1145–1155
Douglas PS, De Bruyne B, Pontone G et al (2016) 1-year outcomes of FFRCT-guided care in patients with suspected coronary disease: the PLATFORM study. J Am Coll Cardiol 68:435–445
Collet C, Miyazaki Y, Ryan N et al (2018) Fractional flow reserve derived from computed tomographic angiography in patients with multivessel CAD. J Am Coll Cardiol 71:2756–2769
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest, neither scientific nor fincancial.
Rights and permissions
About this article
Cite this article
Schmermund, A., Eckert, J., Schmidt, M. et al. Coronary computed tomography angiography: a method coming of age. Clin Res Cardiol 107 (Suppl 2), 40–48 (2018). https://doi.org/10.1007/s00392-018-1320-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00392-018-1320-5