Abstract
Purpose
There has been a lot of controversies about the correlation between vitamin D and colorectal cancer (CRC). In this meta-analysis, we purposed to explore the relationship between vitamin D and the incidence of CRC/the prognosis of CRC.
Methods
A systematic search for articles in databases (Pubmed, Web of Science, EBSCO and Cochrane Library) was terminated in April 2020. The primary outcomes were the incidence rate of CRC and the long-term survival of patients with CRC.
Results
According to the estimated pooled OR from 21 eligible studies, covering 904,152 people, the use of vitamin D was inversely associated with the incidence of CRC [OR = 0.87, (0.82–0.92)]. Among the four studies included in this meta-analysis, covering 7486 patients, compared the overall survival (OS) of CRC between the vitamin D users and the non-users. Based on the estimated pooled HR, vitamin D potentially improved the long-term survival of CRC patients [HR = 0.91, (0.83–0.98)].
Conclusion
This meta-analysis demonstrates that vitamin D not only has a positive impact on the incidence of CRC from either the dietary or supplemental sources but also benefits clinical outcomes and improves the long-term survival of CRC patients. However, further studies are recommended to clarify the above phenomena.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal cancer (CRC) is among the common malignancies, with over 1.8 million incident cases and 860,000 deaths annually [1]. Though the international incidence rates vary, migration studies have documented increasing rates among groups moving from low incidence to high incidence areas [2]. As a result, the role for an environmental factor (s) such as diet in CRC is depicted [3, 4]. Vitamin D is the collective name for cholecalciferol (vitamin D3) and ergocalciferol (vitamin D2) [5]. There exists controversy on how vitamin D deficiency is associated with the aetiology of some common cancers. In a study, the receiver operating characteristic (ROC) curve for serum 25(OH)D level suggested that once the level fell below 30 ng/mL, the risk for advanced colorectal adenoma was not progressive but stepwise as levels fell below this serum level. It is worth evaluating a minimum period of vitamin D deficiency [6]. Observational evidence immensely supports that vitamin D is related to the incidence of CRC. Previous studies had revealed the intestine as an organ with the highest expression of vitamin D receptor (VDR) [7, 8]. Moreover, intestines harbors 1α, 25-dihydroxy vitamin D3 or calcitriol, the most active vitamin D metabolite, that mediates vital homeostatic functions [9]. Some vitro studies have demonstrated vitamin D-induced growth arrest and apoptosis of CRC cells, modulation of the Wnt signalling pathway, DNA repair, and immunomodulation [10, 11], thus supporting a causal relationship of vitamin D with CRC [12].
Moreover, various studies using animals, including some that analyze the effect of diets with vitamin D and carcinogen content, and others with xenotransplanted mice, indeed agreed with the beneficial effect of vitamin D, calcitriol, or its analogues on colorectal tumorigenesis [13]. Collectively, these findings demonstrate a protective function of vitamin D against CRC by reducing risk and attenuating the tumorigenic process. Several studies have also investigated the correlation between vitamin D and the incidence of CRC/the prognosis of CRC. However, individual studies have yield inconsistent results, potentially caused by limitations associated with an individual study. For instance, based on prospective studies, the use of vitamin D was inversely associated with the incidence of CRC in the Nurses’ Health Study [14], but not in the Japan Public Health Centre [15]. Although several studies [16,17,18,19] have explored the correlation between vitamin D and the prognosis of CRC, a majority of them had small series with conflicting results. The association of vitamin D with the incidence of CRC and the prognosis of CRC remains elusive. To provide insight on these controversies, we conducted a comprehensive meta-analysis pooling the available data to examine the relationship between vitamin D and the incidence of CRC/the prognosis of CRC to guide its clinical application.
Methods
Search strategy
In this meta-analysis, we conducted a systematic search of databases (Pubmed, Web of Science, EBSCO and Cochrane Library) which was terminated in April 2020. The subject search was combined with free text terms and medical subject heading, including (‘CRC’ OR ‘colorectal carcinomas’ OR ‘colorectal neoplasms’ OR ‘colon cancer’ OR ‘colon neoplasms’ OR ‘colonic neoplasms’ OR ‘rectum cancer’ OR ‘rectum neoplasms’ OR ‘rectal neoplasms’ OR ‘rectal cancer’) AND (‘vitamin D’). Moreover, the references in the literature were manually searched to avoid omitting relevant studies.
Selection criteria
Each study retrieved for inclusion was assessed by two authors independently. The inclusion criteria included the following: (1) Randomized controlled trials (RCTs) or observational studies including cohort and case-control studies. (2) Reports on the correlation between vitamin D and the incidence of CRC or the correlation between vitamin D and prognosis of CRC. (3) Result (including odds ratio (OR)) of the correlation between vitamin D and the incidence of CRC as well as the result (including hazard ratio (HR)) of the correlation between vitamin D and prognosis of CRC. Exclusion criteria included the following: (1) Articles that were not original. (2) Articles without relevant outcome or detailed data. (3) Duplicate reports. However, on an encounter with duplicate articles, the literature that had the newest or comprehensive data were included. (4) The disease was not CRC, like polyp, adenoma, and precancerous lesions.
Data extraction and study quality assessment
Data extraction and study quality assessment were independently conducted by two investigators. Uncertain inclusions of a study between the two investigators were resolved by consensus of two investigators and the third reviewer. The database was used to record the available information, including author, year of publication, country, cohort characteristics, sample size, time to recruit, follow-up time, vitamin D type, cancer type, total vitamin D dose, study design, and adjustment factors. This meta-analysis was conducted per the guidelines of the Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) 2015 statement [20]. The quality of RCTs and observational studies was determined by the Cochrane Collaboration’s tool and the Newcastle-Ottawa Quality Assessment Scale (NOS) checklist, respectively, by two independent authors.
Statistical analysis
Statistical analyses were conducted using STATA 12.0 Software (Stata, College Station). OR, 95% confidence interval (CI) were utilized to analyze the correlation between vitamin D and the incidence of CRC. HR, 95% CI were utilized to analyze the correlation between vitamin D and prognosis of CRC. Cochran’s Q statistic and I2 statistics were applied to evaluate the heterogeneity in the studies. Considering the variability of vitamin D type and dose, a random-effects model was used for meta-analysis. The sources of heterogeneity were investigated through the subgroup analyses. Publication bias was assessed using Begg’s and Egger’s regression asymmetry test. Furthermore, the sensitivity analysis was conducted to test the stability of the results by excluding each study successively.
Results
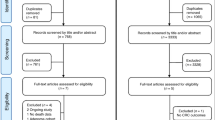
A total of 2245 studies were screened after the database search. Additionally, 4 studies were identified through other sources. Following the deletion of 1470 duplicates, 731 studies were excluded by screening their title and abstract. Additionally, 48 studies were evaluated via reading full-text articles. Finally, 25 eligible articles [14,15,16,17,18,19, 21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39] were included in quantitative synthesis (Fig. 1), 21 studies [14, 15, 21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39] for the correlation between vitamin D and the incidence of CRC and 4 studies [16,17,18,19] for the correlation between vitamin D and prognosis of CRC. The quality of 25 studies was assessed as shown in Supplementary Table 1.
The characteristics of 21 studies demonstrating the correlation between vitamin D and the incidence of CRC are shown in Table 1. Geographic locations were categorized as Asia, Europe, and North America. Total vitamin D intake was from dietary and vitamin D supplement. Cancer types were CRC classified as colon cancer and rectal cancer. Adjustment factors were mainly sex, age, education, energy, body mass index, and smoking. The characteristics of 4 studies of the correlation between vitamin D and the prognosis of CRC are shown in Table 2. Geographic location was categorized as Europe and North America. Total vitamin D intake was from dietary and vitamin D supplement. Adjustment factors were mainly sex, age, alcohol, energy, and smoking.
Vitamin D and risk of CRC incidence
A total of 21 studies reported OR in CRC among vitamin D (total intake) higher intake compared with the low intake. The directions of the estimated OR and 95% CI were not consistent among the studies. According to the estimated pooled OR, the use of vitamin D was inversely associated with the incidence of CRC [OR = 0.87, (0.82–0.92)]. Heterogeneity was prominent (I2 = 51.3%) (Fig. 2). Having found an inverse association between total vitamin D and incidence of CRC, we further probed whether dietary vitamin D and supplementary vitamin D could reduce the risk of CRC. The results of the meta-analysis of 17 studies are shown in Fig. 3. Our assessments revealed that dietary vitamin D was associated with a decrease in the incidence of CRC [OR = 0.92, (0.87–0.97)]. The results of the meta-analysis of 9 studies are displayed in Fig. 4. Similarly, supplementary vitamin D was related to a decline in the incidence of CRC [OR = 0.89, (0.81–0.96)].
Subgroup analysis
The relationship between total vitamin D usage and the incidence of CRC among male and female were examined in 5 and 11 studies, respectively. The pooled ORs for male and female were 0.90 (95%CI, 0.84–0.97) and 0.87 (95%CI, 0.81–0.92), respectively. To compare OR in colon cancer and rectal cancer, 7 and 8 studies were used, respectively. The pooled ORs for colon cancer and rectal cancer were 0.82 (95%CI, 0.73–0.90) and 0.88 (95%CI, 0.78–0.97), respectively. Concerning the subsites of colon cancer, the pooled ORs for proximal colon cancer and distal colon cancer were 0.83 (95%CI, 0.74–0.91) and 0.83 (95%CI, 0.76–0.91), respectively. Accordingly, the pooled ORs in Europe and North America were 0.84 (95%CI, 0.72–0.96) and 0.87 (95%CI, 0.82–0.91). From the subgroup analysis of the above aspects, the potential effect of vitamin D to reduce the incidence of cancer was consistent.
The relationship between dietary vitamin D usage and the incidence of CRC in Europe and North America was retrieved in 9 studies and 8 studies, respectively. With the estimated pooled OR, vitamin D was inversely associated with the incidence of CRC in North America [OR = 0.94, (0.90, 0.99)] and in Europe [OR = 0.84, (0.72, 0.96)]. However, the estimated pooled OR showed a supplementary vitamin D examined in 9 studies was inversely associated with the incidence of CRC in North America [OR = 0.86, (0.77–0.94)] (Table 3).
Vitamin D and prognosis of CRC
We included four studies in the analysis to compare the OS of CRC between vitamin D users and non-users. The estimated pooled HR revealed that the use of vitamin D was associated with longer OS [HR = 0.91, (0.83–0.98)] (Fig. 5). Only 3 studies were used to compare the OS of CRC between the supplementary vitamin D users and non-users. According to the estimated pooled HR, the use of supplementary vitamin D improved OS of CRC [HR = 0.88, (0.77–0.99)] (Fig. 6).
Publication bias and sensitivity analysis
No significant publication bias was observed on the influence of total vitamin D and CRC incidence using Egger’s plot (p = 0.278) or Begg’s test (p = 0.697) (Supplementary Fig. 1). Sensitivity analysis was performed to test the stability of the results by excluding each study successively. The detailed sensitivity analysis results are illustrated in Supplementary Fig. 2. Similarly, there was no significant publication bias on the influence of dietary vitamin D and CRC incidence (Supplementary Fig. 3). The detailed sensitivity analysis results are indicated in Supplementary Fig. 4. Moreover, there was no significant publication bias on the influence of supplemental vitamin D and CRC incidence (Supplementary Fig. 5). The detailed sensitivity analysis performed to test the stability of their results is shown in Supplementary Fig. 6.
Discussion
In the present study, we conducted a meta-analysis on 21 articles to evaluate the correlation between vitamin D and the incidence of CRC whereas the 4 articles were evaluated on the correlation between vitamin D and prognosis of CRC. Notably, vitamin D intake was shown to be related to a decline in the incidence of CRC as well as improving the long-term survival of CRC patients. The effect was consistently observed in subgroup analyses of gender, CRC sites, colonic cancer subsites, and geographical differences. However, this estimate was robust across the sensitivity analysis. Hence, our findings provide an insight into the protective action of vitamin D against CRC by reducing CRC risk and improving the prognosis of patients with CRC.
Previous studies demonstrated that vitamin D potentially reduces the risk of colorectal adenomas [40], a finding that was inconsistent with reports from other studies [41, 42]. Elsewhere, a meta-analysis [43] reported that neither dietary intake of vitamin D nor supplemental intake of vitamin D was significantly associated with the incidence of colorectal adenoma. Thus, this discrepancy might be attributed from multiple factors, such as the limited number of articles on either dietary or supplemental vitamin D intake, differences in the dose of supplemental vitamin D, the absorption rate of dietary intake of vitamin D, and metabolism of vitamin D, and other confounding factors, such as age, physical activity, and sun exposure. On the contrary, another meta-analysis [44] indicated that vitamin D intake was inversely associated with colorectal adenomas incidence and recurrent adenomas. The result further supported the role of vitamin D in preventing colorectal adenoma incidence and their recurrence. More importantly, vitamin D may display an additional protective effect other than their preventive effects against colorectal adenomas [44]. It is deductive to speculate the intake of vitamin D inhibits the progression of adenoma or precancerous polyps to CRC, thereby reducing the risk of CRC.
Some studies have found that vitamin D has slight neoplasm inhibitory impacts, for example, promoting bile acid catabolism and modulating > 200 genes involved in cell cycle regulation, DNA repair, immunomodulation, and protection against oxidative stress and inflammation [45,46,47,48]. Similarly, vitamin D may control the development of colorectal cells via the attachment of vitamin D identifying receptors [46]. In Smad3-/- mice with inactivated TGF-β signalling pathways increased dietary vitamin D intake significantly suppressed p-P38 MAPK activity and lowered the accumulation of colonic inflammatory cells by increased 25(OH) D circulation, which ultimately reduced the incidence of colon carcinomas [49]. Besides, vitamin D facilitates the differentiation of colon carcinoma cells via the promotion of the expression of adhesion proteins, such as E-cadherin, and the inhibition of the Wnt/β-catenin signalling pathway [50].
Although controversies exist on the correlation between vitamin D and the incidence of CRC several studies [15, 22, 24] had revealed that there was no protective effect on CRC risk. The non-significant results may be attributed to inadequate measures of vitamin D intake; natural dietary sources of vitamin D are limited. Conversely, an American cohort study [21] reported a significant protective effect of vitamin D on CRC hazard. An inverse association between the use of vitamin D, the prognosis of CRC by Jeffreys et al. [17] concurs with our finding, though not consistent with a report by Antunac et al. [16]. Also, a recent meta-analysis [51] demonstrated a clinically beneficial effect from vitamin D supplementation on survival outcomes in CRC patients.
Interestingly, both dietary and supplemental sources of vitamin D showed significant effects. However, there are food products that are rich in vitamin D3 [52] such as fish oils, fish, and fortified-products (dairy products). For instance, fish consumption was revealed to have an inverse correlation with CRC risk in humans according to a previous meta-analysis [53]. Therefore, we advise that individuals vulnerable to vitamin D deficiency should have a well-mixed diet with modest fish. Considering that dietary sources of vitamin D were limited, supplemental vitamin D was also necessary. Following numerous reports from previous studies [16, 33], vitamin D supplementation potentially prevents or slows the progression of CRC probably due to inadequate duration of the trial since the development of CRC is associated with long latency. Therefore, in our work, vitamin D supplementary used may be insufficient to demonstrate a protective effect. Additionally, published meta-analyses of the overall risk of colon cancer also correlate to serum level of 25(OH)D at or around 30 ng/mL [6]. Of interest, the Endocrine Society recommends children under one year of age require 400–1000 IU daily, children one year and older 600–1000 IU daily, and all adults 1500–2000 IU daily to sustain a circulating serum level of 25(OH)D > 30 ng/mL [54], which may have a protective effect on the incidence of CRC. The subgroup analysis of gender indicated the total vitamin D intake was significantly associated with a lower risk of CRC both in male and female. Several cohort studies [26, 27] in male have shown no association or only weak inverse association of total vitamin D intake with CRC risk probably due to higher misclassification of vitamin D intake in male than in the female. On the other hand, a cohort study [27] in female found no association between total vitamin D intake and CRC risk. Different physiological mechanisms may be operational; these mechanisms may involve gender-specific gut motility or other hormonal influences [55]. Sex hormone metabolism potentially modifies or mask the associations between vitamin D status and CRC risk [26], since the vitamin D receptor has been linked with the estrogen receptor system [56, 57]. The intriguing finding in our study was that vitamin D was inversely associated with the incidence of CRC in both male and female. This may be due to the large sample size of our study, though further studies are recommended to ascertain this.
From the studies that provided the colon subsite-specific data [15, 23, 26, 28, 31, 33], total vitamin D was inversely associated with distal and proximal colon cancers. The two sides of the colorectum differ in respect to the embryological origin and bile acid metabolism, and in some cases, tumors in these sites may develop along different carcinogenic pathways [58]. Though there were differences across colonic subsites in molecular characteristics and associations with genetic and environmental factors[58], our study indicated that there were no differences via the subgroup analysis of colonic subsites (distal and proximal colon cancers). For instance, our study showed that total vitamin D has a slightly stronger association with colon cancer than rectal cancer, [OR = 0.82, (0.73–0.90)] versus [OR = 0.88, (0.78–0.97)], despite the limited evidence from epidemiologic studies and clinical trials. In the previous studies, there was no inconsistency on whether the use of vitamin D could affect the incidence of CRC by colorectal sites. Some cohort studies found total vitamin D intake has an inverse association with colon cancer risk only [26, 31]. In another cohort study, total vitamin D intake was observed to have a negative relationship with rectal cancer risk merely [28]. Furthermore, other studies have indicated that vitamin D is not associated with the incidence of colon cancer or rectal cancer [24, 33]. Thus, further investigation of vitamin D with risk of colorectal sites neoplasms is of paramount importance.
Herein, we reported an inverse relationship between total vitamin D usage and the incidence of CRC both in Europe and in North America. Nonetheless, pooling data from European studies resulted in a stronger association than that of North America between dietary vitamin D intake and the incidence of CRC. This provides support for the importance of differences in dietary habits, race, the relative composition of foods that make up dairy products, and other demographic differences across populations that could contribute to the variability. Notably, the sample size in Asian use lacked reliable data to evaluate the association between vitamin D and the incidence of CRC.
Previous study has indicated that a 50% lower risk of CRC was associated with a serum 25(OH)D level > 33 ng/mL, compared to < 12 ng/mL[59]. Additionally, Sy and Bautista [60] also noted the overall risk of colon polyps would be related to serum 25(OH)D level at or around 30 ng/mL. More importantly, the study found that vitamin D insufficiency is highly prevalent among patients with a family history of colon cancer [6]. Above all, we suggest that people with a positive family history could control regularly their vitamin D level to prevent the incidence of CRC. In a study [61] by Sellers et. al., total and supplemental vitamin D intakes were associated inversely with risk among family history positive women. The cellular effect of vitamin D metabolites is influenced by the biology of the vitamin D receptor complex [6]. Interestingly, Peters et al. [62] did not find attributable patterns of vitamin D receptor variants in families with an increased occurrence of colon cancer. So, it still requires to be further explored whether people with a positive family history need to take regularly vitamin D. Our result demonstrated that vitamin D intake can improve the prognosis of CRC patients. Previous study has shown that vitamin D insufficiency is highly common among patients with CRC [63,64,65]. Therefore, CRC patients of vitamin D deficiency should be advised to take the vitamin D to a normal level. Additionally, we also advise CRC patients with a normal level to take vitamin D supplementation because vitamin D toxicity is extremely uncommon. Studies have shown in adults that 10000 IUs daily for at least 5 months did not cause any toxicity [66]. More importantly, it still needs more research to further guide clinical application.
The use of vitamin D contributed to a significant reduction in CRC incidence, which is inconsistent with a previous meta-analysis [67]. However, there were several strengths to our study. To our knowledge, this is the first comprehensive meta-analysis that assessed the relationship between vitamin D and the incidence of CRC as well as between vitamin D and the prognosis of CRC. In our study, relatively larger sample size was used, thereby updating and extending earlier results. Moreover, based on our study, the pooled estimates were stable following the exclusion of any trial at a time with sensitivity analysis. On the contrary, there were some associated limitations to our meta-analysis. For instance, we found that heterogeneity existed in our meta-analysis as indicated by the I2. It is predictable because of the presence of inter-studies differences in study design (prospective observational studies, retrospective observational studies and RCTs), enrolled populations, duration of follow-up, and the cohort characteristics. The studies included did not evaluate the serum 25(OH)D baseline status of CRC patients, which is hard to assess whether the CRC patients had a prior deficiency. Less consistency also existed on the dose-response relation; therefore, the optimal dose of vitamin D still needs to be further evaluated via large-scale prospective studies to lower the risk of CRC and improve the long-term survival of CRC patients.
Conclusion
This meta-analysis demonstrates a meaningful beneficial effect between the incidence of CRC and vitamin D from either dietary or supplement sources. The effect was concordantly observed via subgroup analyses of gender, CRC sites, colonic cancer subsites, and geographical differences. Although vitamin D could benefit clinical outcomes and improve the long-term survival of CRC patients, their certainty requires further studies. Therefore, there is a need to fully evaluate the optimal dosing of vitamin D using adequately powered RCTs and to augment the duration of follow-up.
Data availability
The datasets supporting the conclusions of this article are included within the article.
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424
Boyle P, Zaridze D, Smans M (1985) Descriptive epidemiology of colorectal cancer. Int J Cancer 36(1):9–18
Haenszel W, Berg J, Segi M, Kurihara M, Locke FB (1973) Large-bowel cancer in Hawaiian Japanese. J Natl Cancer Inst 51(6):1765–1779
Lipkin M, Reddy B, Newmark H, Lamprecht SA (1999) Dietary factors in human colorectal cancer. Annu Rev Nutr 19:545–586
Kulda V (2012) Vitamin D metabolism. Vnitr Lek 58(5):400–404
Ahmad I, Trikudanathan G, Feinn R, Anderson J, Nicholson M, Lowe S, Levine JB (2016) Low serum vitamin D: A surrogate marker for advanced colon adenoma? J Clin Gastroenterol 50(8):644–648
Arai H, Miyamoto KI, Yoshida M, Yamamoto H, Taketani Y, Morita K, Kubota M, Yoshida S, Ikeda M, Watabe F, Kanemasa Y, Takeda E (2001) The polymorphism in the caudal-related homeodomain protein Cdx-2 binding element in the human vitamin D receptor gene. J Bone Miner Res 16(7):1256–1264
Jeffery LE, Henley P, Marium N, Filer A, Sansom DM, Hewison M, Raza K (2018) Decreased sensitivity to 1,25-dihydroxyvitamin D3 in T cells from the rheumatoid joint. J Autoimmun 88:50–60
Barbáchano A, Fernández-Barral A, Ferrer-Mayorga G, Costales-Carrera A, Larriba MJ, Muñoz A (2017) Endocrinology C: The endocrine vitamin D system in the gut. Mol Cell Endocrinol 453:79–87
Theodoratou E, Tzoulaki I, Zgaga L, Ioannidis JP (2014) Vitamin D and multiple health outcomes: umbrella review of systematic reviews and meta-analyses of observational studies and randomised trials. BMJ 348:g2035
Autier P, Boniol M, Pizot C, Mullie P (2014) Vitamin D status and ill health: a systematic review. Lancet Diabetes Endocrinol 2(1):76–89
Fleet JC, DeSmet M, Johnson R, Li Y (2012) Vitamin D and cancer: a review of molecular mechanisms. Biochem J 441(1):61–76
Barbáchano A, Larriba MJ, Ferrer-Mayorga G, González-Sancho JM, Muñoz A (2018) Vitamin D and colon cancer. Health Disease and Therapeutics 2:837–862
Martínez M, Giovannucci E, Colditz G, Stampfer M, Hunter D, Speizer F, Wing A, Willett WC (1996) Calcium, vitamin D, and the occurrence of colorectal cancer among women. J Natl Cancer Inst 88(19):1375–1382
Pritchard R, Baron J, Gerhardsson de Verdier M (1996) Dietary calcium, vitamin D, and the risk of colorectal cancer in Stockholm, Sweden. Cancer Epidemiol Biomarkers Prev 5(11):897–900
Antunac Golubic Z, Barsic I, Librenjak N, Plestina S (2018) Vitamin D supplementation and survival in metastatic colorectal cancer. Nutr Cancer 70(3):413–417
Jeffreys M, Redaniel MT, Martin RM (2015) The effect of pre-diagnostic vitamin D supplementation on cancer survival in women: a cohort study within the UK Clinical Practice Research Datalink. BMC Cancer 15:670
Lewis C, Xun P, He K (2016) Vitamin D supplementation and quality of life following diagnosis in stage II colorectal cancer patients: a 24-month prospective study. Support Care Cancer 24(4):1655–1661
Yang B, McCullough ML, Gapstur SM, Jacobs EJ, Bostick RM, Fedirko V, Flanders WD, Campbell PT (2014) Calcium, vitamin D, dairy products, and mortality among colorectal cancer survivors: the Cancer Prevention Study-II Nutrition Cohort. J Clin Oncol 32(22):2335–2343
Gu L, Khadaroo PA, Chen L, Li X, Zhu H, Zhong X, Pan J, Chen M (2019) Comparison of long-term outcomes of endoscopic submucosal dissection and surgery for early gastric cancer: A systematic review and meta-analysis. J Gastrointest Surg 23(7):1493–1501
Kearney J, Giovannucci E, Rimm E, Ascherio A, Stampfer M, Colditz G, Wing A, Kampman E, Willett WC (1996) Calcium, vitamin D, and dairy foods and the occurrence of colon cancer in men. Am J Epidemiol 143(9):907–917
Boutron M, Faivre J, Marteau P, Couillault C, Senesse P, Quipourt V (1996) Calcium, phosphorus, vitamin D, dairy products and colorectal carcinogenesis: a French case-control study. Br J Cancer 74(1):145–151
Marcus PM, Newcomb PA (1998) The association of calcium and vitamin D, and colon and rectal cancer in Wisconsin women. Int J Epidemiol 27(5):788–793
Järvinen R, Knekt P, Hakulinen T, Aromaa A (2001) Prospective study on milk products, calcium and cancers of the colon and rectum. Eur J Clin Nutr 55(11):1000–1007
Kampman E, Slattery M, Caan B, Potter JD (2000) Calcium, vitamin D, sunshine exposure, dairy products and colon cancer risk (United States). Cancer Causes Control 11(5):459–466
McCullough M, Robertson A, Rodriguez C, Jacobs E, Chao A, Carolyn J, Calle E, Willett W, Thun MJ (2003) Calcium, vitamin D, dairy products, and risk of colorectal cancer in the Cancer Prevention Study II Nutrition Cohort (United States). J Clin Oncol 14(1):1–12
Slattery M, Neuhausen S, Hoffman M, Caan B, Curtin K, Ma K, Samowitz W (2004) Dietary calcium, vitamin D, VDR genotypes and colorectal cancer. Int J Cancer 111(5):750–756
Terry P, Baron J, Bergkvist L, Holmberg L, Wolk A (2002) Cancer: Dietary calcium and vitamin D intake and risk of colorectal cancer: a prospective cohort study in women. Nutr Cancer 43(1):39–46
Kesse E, Boutron-Ruault M, Norat T, Riboli E, Clavel-Chapelon F (2005) Dietary calcium, phosphorus, vitamin D, dairy products and the risk of colorectal adenoma and cancer among French women of the E3N-EPIC prospective study. Int J Cancer 117(1):137–144
Lin J, Zhang S, Cook N, Manson J, Lee I, Buring JE (2005) Intakes of calcium and vitamin D and risk of colorectal cancer in women. Am J Epidemiol 161(8):755–764
Lipworth L, Bender T, Rossi M, Bosetti C, Negri E, Talamini R, Giacosa A, Franceschi S, McLaughlin J, La Vecchia CJN et al (2009) Dietary vitamin D intake and cancers of the colon and rectum: a case-control study in Italy. Nutr Cancer 61(1):70–75
Park S, Murphy S, Wilkens L, Nomura A, Henderson B, Kolonel LN (2007) Calcium and vitamin D intake and risk of colorectal cancer: the Multiethnic Cohort Study. Am J Epidemiol 165(7):784–793
Wactawski-Wende J, Kotchen J, Anderson G, Assaf A, Brunner R, O'Sullivan M, Margolis K, Ockene J, Phillips L, Pottern L et al (2006) Calcium plus vitamin D supplementation and the risk of colorectal cancer. N Engl J Med 354(7):684–696
Banqué M, Raidó B, Masuet C, Ramon JM (2012) Food groups and nutrient intake and risk of colorectal cancer: a hospital-based case-control study in Spain. Nutr Cancer 64(3):386–392
Hosseinzadeh P, Javanbakht M, Alemrajabi M, Gholami A, Amirkalali B, Sohrabi M, Zamani F (2019) The association of dietary intake of calcium and vitamin D to colorectal cancer risk among Iranian population. Asian Pac J Cancer Prev 20(9):2825–2830
Manson J, Cook N, Lee I, Christen W, Bassuk S, Mora S, Gibson H, Gordon D, Copeland T, D'Agostino D et al (2019) Vitamin D supplements and prevention of cancer and cardiovascular disease. N Engl J Med 380(1):33–44
Sun Z, Wang P, Roebothan B, Cotterchio M, Green R, Buehler S, Zhao J, Squires J, Zhao J, Zhu Y et al (2011) Calcium and vitamin D and risk of colorectal cancer: results from a large population-based case-control study in Newfoundland and Labrador and Ontario. Can J Public Health 102(5):382–389
Um C, Prizment A, Hong C, Lazovich D, Bostick RM (2019) Associations of calcium, vitamin D, and dairy product intakes with colorectal cancer risk among older women: the Iowa women's health study. Nutr Cancer 71(5):739–748
Vallès X, Alonso M, López-Caleya J, Díez-Obrero V, Dierssen-Sotos T, Lope V, Molina-Barceló A, Chirlaque M, Jiménez-Moleón J, Fernández Tardón G et al (2018) Colorectal cancer, sun exposure and dietary vitamin D and calcium intake in the MCC-Spain study. Environ Int 121:428–434
Hartman TJ, Albert PS, Snyder K, Slattery ML, Caan B, Paskett E, Iber F, Kikendall JW, Marshall J, Shike M, Weissfeld J, Brewer B, Schatzkin A, Lanza E, Polyp Prevention Study Group (2005) The association of calcium and vitamin D with risk of colorectal adenomas. J Nutr 135(2):252–259
Baron JA, Barry EL, Mott LA, Rees JR, Sandler RS, Snover DC, Bostick RM, Ivanova A, Cole BF, Ahnen DJ, Beck GJ, Bresalier RS, Burke CA, Church TR, Cruz-Correa M, Figueiredo JC, Goodman M, Kim AS, Robertson DJ, Rothstein R, Shaukat A, Seabrook ME, Summers RW (2015) A trial of calcium and vitamin D for the prevention of colorectal adenomas. N Engl J Med 373(16):1519–1530
Crockett SD, Barry EL, Mott LA, Ahnen DJ, Robertson DJ, Anderson JC, Wallace K, Burke CA, Bresalier RS, Figueiredo JC, Snover DC, Baron JA (2019) Calcium and vitamin D supplementation and increased risk of serrated polyps: results from a randomised clinical trial. Gut 68(3):475–486
Huang D, Lei S, Wu Y, Weng M, Zhou Y, Xu J, Xia D, Xu E, Lai M, Zhang H (2020) Additively protective effects of vitamin D and calcium against colorectal adenoma incidence, malignant transformation and progression: a systematic review and meta-analysis. Clin Nutr 39(8):2525–2538
Wei MY, Garland CF, Gorham ED, Mohr SB, Giovannucci E (2008) Vitamin D and prevention of colorectal adenoma: a meta-analysis. Cancer Epidemiol Biomark Prev 17(11):2958–2969
Newmark HL, Lipkin M (1992) Calcium, vitamin D, and colon cancer. Cancer Res 52(7 Suppl):2067s–2070s
Lamprecht SA, Lipkin M (2003) Chemoprevention of colon cancer by calcium, vitamin D and folate: molecular mechanisms. Nat Rev Cancer 3(8):601–614
Dusso AS, Brown AJ, Slatopolsky E (2005) Vitamin D. Am J Physiol Ren Physiol 289(1):F8–F28
Cross HS, Nittke T, Kallay E (2011) Colonic vitamin D metabolism: implications for the pathogenesis of inflammatory bowel disease and colorectal cancer. Mol Cell Endocrinol 347(1-2):70–79
Meeker S, Seamons A, Paik J, Treuting P, Brabb T, Grady W, Maggio-Price LJ (2014) Increased dietary vitamin D suppresses MAPK signaling, colitis, and colon cancer. Cancer Res 74(16):4398–4408
Ricca C, Aillon A, Viano M, Bergandi L, Aldieri E, Silvagno F (2019) Vitamin D inhibits the epithelial-mesenchymal transition by a negative feedback regulation of TGF-β activity. J Steroid Biochem Mol Biol 187:97–105
Vaughan-Shaw PG, Buijs LF, Blackmur JP, Theodoratou E, Zgaga L, Din FVN, Farrington SM, Dunlop MG (2020) The effect of vitamin D supplementation on survival in patients with colorectal cancer: systematic review and meta-analysis of randomised controlled trials. Br J Cancer 123:1705–1712
Wranicz J, Szostak-Wegierek D (2014) Health outcomes of vitamin D. Part II. Role in prevention of diseases. Rocz Panstw Zakl Hig 65(4):273–279
Wu S, Feng B, Li K, Zhu X, Liang S, Liu X, Han S, Wang B, Wu K, Miao D, Liang J, Fan D (2012) Fish consumption and colorectal cancer risk in humans: a systematic review and meta-analysis. Am J Med 125(6):551–559 e555
Holick M, Binkley N, Bischoff-Ferrari H, Gordon C, Hanley D, Heaney R, Murad M, Weaver C (2011) Evaluation, treatment, and prevention of vitamin D deficiency: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab 96(7):1911–1930
Lampe JW, Fredstrom SB, Slavin JL, Potter JD (1993) Sex differences in colonic function: a randomised trial. Gut 34(4):531–536
Speer G, Cseh K, Winkler G, Takacs I, Barna I, Nagy Z, Lakatos P (2001) Oestrogen and vitamin D receptor (VDR) genotypes and the expression of ErbB-2 and EGF receptor in human rectal cancers. Eur J Cancer 37(12):1463–1468
Gilad LA, Bresler T, Gnainsky J, Smirnoff P, Schwartz B (2005) Regulation of vitamin D receptor expression via estrogen-induced activation of the ERK 1/2 signaling pathway in colon and breast cancer cells. J Endocrinol 185(3):577–592
Iacopetta B (2002) Are there two sides to colorectal cancer? Int J Cancer 101(5):403–408
Gorham E, Garland C, Garland F, Grant W, Mohr S, Lipkin M, Newmark H, Giovannucci E, Wei M, Holick MF (2007) Optimal vitamin D status for colorectal cancer prevention: a quantitative meta analysis. Am J Prev Med 32(3):210–216
Sy A, JEK B (2013) Association between serum vitamin D levels and colonic carcinomatous polyps. J Gastrointest Cancer 44(4):481–485
Sellers TA, Bazyk AE, Bostick RM, Kushi LH, Olson JE, Anderson KE, Lazovich D, Folsom AR (1998) Diet and risk of colon cancer in a large prospective study of older women: an analysis stratified on family history (Iowa, United States). Cancer Causes Control 9(4):357–367
Peters U, Hayes RB, Chatterjee N, Shao W, Schoen RE, Pinsky P, Hollis BW, McGlynn KA, Prostate LC (2004) Ovarian Cancer Screening Project T: Circulating vitamin D metabolites, polymorphism in vitamin D receptor, and colorectal adenoma risk. Cancer Epidemiol Biomark Prev 13(4):546–552
Fakih M, Trump D, Johnson C, Tian L, Muindi J, Sunga AY (2009) Chemotherapy is linked to severe vitamin D deficiency in patients with colorectal cancer. Int J Colorectal Dis 24(2):219–224
Moan J, Porojnicu A, Lagunova Z, Berg J, Dahlback A (2007) Colon cancer: prognosis for different latitudes, age groups and seasons in Norway. J Photochem Photobiol B 89:148–155
Mezawa H, Sugiura T, Watanabe M, Norizoe C, Takahashi D, Shimojima A, Tamez S, Tsutsumi Y, Yanaga K, Urashima M (2010) Serum vitamin D levels and survival of patients with colorectal cancer: post-hoc analysis of a prospective cohort study. BMC Cancer 10:347
Heaney R, Davies K, Chen T, Holick M, Barger-Lux MJ (2003) Human serum 25-hydroxycholecalciferol response to extended oral dosing with cholecalciferol. Am J Clin Nutr 77(1):204–210
Huncharek M, Muscat J, Kupelnick B (2009) Colorectal cancer risk and dietary intake of calcium, vitamin D, and dairy products: a meta-analysis of 26,335 cases from 60 observational studies. Nutr Cancer 61(1):47–69
Code availability
Not applicable.
Funding
This study was funded by the Key Laboratory of Diagnosis and Treatment of Digestive System Tumors of Zhejiang Province (Grant No. 2019E10020), Ningbo Clinical Research Center for Digestive System Tumors (Grant No. 2019A21003).
Author information
Authors and Affiliations
Contributions
XY designed the research process. YBX searched the database for corresponding articles. MXQ and JZH extracted useful information from the articles above. DMG and TY used statistical software for analysis. LHX and YBX drafted the meta-analysis. XY polished this article. All authors had read and approved the manuscript and ensured that this was the case.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Fig. 1.
Publication bias of the correlation of total vitamin D and colorectal cancer incidence (Egger’s, p=0.278; Begg’s, p=0.597). (PNG 208 kb)
Supplementary Fig. 2.
Sensitivity analysis of the correlation of total vitamin D and colorectal cancer incidence. (PNG 488 kb)
Supplementary Fig. 3.
Publication bias of the correlation of dietary vitamin D and colorectal cancer incidence (Egger’s, p=0.617; Begg’s, p=0.923). (PNG 211 kb)
Supplementary Fig. 4.
Sensitivity analysis of the correlation of dietary vitamin D and colorectal cancer incidence. (PNG 476 kb)
Supplementary Fig. 5.
Publication bias of the correlation of supplementary vitamin D and colorectal cancer incidence (Egger’s, p=0.337; Begg’s, p=0.374). (PNG 185 kb)
Supplementary Fig. 6.
Sensitivity analysis of the correlation of supplementary vitamin D and colorectal cancer incidence. (PNG 332 kb)
Supplementary Table 1.
Quality assessment of the enrolled studies. (DOCX 21 kb)
ESM 1
(DOC 64 kb)
Rights and permissions
About this article
Cite this article
Xu, Y., Qian, M., Hong, J. et al. The effect of vitamin D on the occurrence and development of colorectal cancer: a systematic review and meta-analysis. Int J Colorectal Dis 36, 1329–1344 (2021). https://doi.org/10.1007/s00384-021-03879-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-021-03879-w