Abstract
Purpose
Proximal and distal colon cancers differ in terms of epidemiology, clinical presentation, and pathologic features. The aim of our study was to evaluate the impact of right-sided (RC), transverse (TC), and left-sided (LC) colon cancer on morbidity rates and oncological outcomes.
Methods
A retrospective analysis of patients with resected colon cancer between 2004 and 2014 was conducted. Cox proportional hazard models were used to assess predictors of overall (OS), and disease-specific survival (DSS), as well as disease-free survival (DFS).
Results
A total of 1189 patients were included. RC patients (n = 618) were older, predominantly women, and had a higher comorbidity rate. LC (n = 454) was associated with symptomatic presentation and increased rates of laparoscopic surgery. Multivisceral resections were more frequently performed in TC tumors (n = 117). This group was admitted 1 day longer and had a higher complication rate (RC 35.6% vs. TC 43.6% vs. LC 31.1%, P0.032). Although the incidence of abscess/leak was similar between the groups, the necessity of readmission and subsequent reoperation for a leak was significantly higher in LC patients. Pathology revealed more poorly differentiated tumors and microsatellite instability in RC. Kaplan-Meier curves demonstrated worse 5-year OS for right-sided tumors (RC 73.0%; TC 76.2%. LC 80.8%, P0.023). However, after adjustment, no differences were found in OS, DSS, and DFS between tumor location. Only pathological features were independently correlated with prognosis, as were baseline characteristics for OS.
Conclusion
Tumor location in colon cancer was not associated with survival or disease recurrence. Pathological differences beyond tumor stage were significantly more important.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Colorectal cancer is one of the most commonly diagnosed cancers in both men and women worldwide. The decline in incidence and mortality rates over the last decades reflects the impact of the reduction of risk factors, the introduction of screening programs, and altered treatment patterns [1, 2]. Previous studies reported a shift in the distribution of colon cancer toward the proximal side of the colon [3, 4]. Already in the early 1990s, a distinction in clinical outcomes and pathological features within different segments of the colon relative to colon cancer was suggested [5]. Subsequent research elaborated on this topic and a discussion as to whether to consider proximal and distal colon cancer as two different diseases was raised [6]. However, up until now, these results have had no consequences on screening or treatment patterns.
The proximal and distal parts of the large intestine are physiologically separate, due to different embryological origins. The right colon arises from the midgut, as does the proximal two-thirds of the transverse colon. The left colon, including the distal one-third of the transverse colon, derives from the hindgut. Consequently, there is not only a difference in blood supply but also potentially in gene expression and clinical presentation [6, 7]. In addition, the genetic carcinogenetic pathways may be different [8,9,10]. Three major pathways for sporadic colorectal cancer have been described: chromosomal instability (CIN), microsatellite instability (MSI), and more recently the serrated pathway classified as the CpG island methylator phenotype (CIMP). While CIN is observed in the vast majority of colorectal cancers, it is associated with distal cancers, whereas the latter two pathways have been linked to more proximal colon cancers [8, 11].
Although distinct differences exist, the influence on prognosis remains unclear. Some studies suggested higher mortality for right-sided tumors [7, 12], while others found no differences [13]. Unfortunately, most of the conducted studies were limited in their ability to adjust for a wide range of potential confounders. Furthermore, to our knowledge, none of the studies compared transverse colon cancer as a separate entity. Therefore, the aim of our study was to determine clinicopathological differences and the prognostic impact of primary tumor location in colon cancer.
Materials and methods
Study design and population
A cohort study was designed from a prospectively maintained and IRB-approved database that included all primary colon cancer patients who underwent surgical treatment at Massachusetts General Hospital between 2004 and 2014. Patients who underwent an emergency procedure (n = 152), had a total colectomy (n = 88), or received neoadjuvant therapy (n = 62) were excluded. Patients were divided into three groups: right-sided, transverse, and left-sided colon cancer. TC was defined as the resection of the transverse colon only, while RC included the resection of the cecum, ascending colon, and hepatic flexure, and LC the resection of the splenic flexure, descending colon, and sigmoid.
The main outcome measures for this study were overall survival (OS), disease-specific survival (DSS), and disease-free survival (DFS). Data on long-term outcomes was updated periodically by reviewing patient follow-up records and the Massachusetts General Hospital’s cancer registry. Secondary outcomes included patient characteristics, pathological features, and perioperative outcomes. All time to events was calculated from date of surgery.
Statistical analyses
Continuous variables were analyzed using a Kruskal-Wallis H test for the differences between all three groups, while group-specific differences compared with the remainder of the population were performed through a Mann-Whitney U test. Continuous variables are presented as the mean with a standard deviation (SD) or the median with an interquartile range (IQR) according to the distribution (Kolmogorov-Smirnov and Shapiro-Wilk test). Categorical variables are presented as the percentages of patients. Differences in dichotomous variables were assessed using a chi-square (χ2) test or Fisher’s exact test where appropriate. Long-term outcomes were performed for stage I–III colon cancer only. Kaplan-Meier curves for overall (OS), disease-specific survival (DSS), and disease-free survival (DFS) were assessed within each stage and all stages combined using log-rank testing. Multivariate analyses were performed using a Cox proportional hazard model to determine risk factors for overall, disease-specific, and disease-free survival. Variables significant in univariate analysis were entered into the model. Results are reported as hazard ratios (HR) with a 95% confidence interval (CI). All statistical analyses were performed using SPSS (IBM Corp. Released 2013. IBM SPSS Statistics for Macintosh, Version 24.0. Armonk, NY: IBM Corp.). The threshold for statistical significance was set at a two-sided P value of 0.05 or less.
Results
Of the 1189 patients who underwent an elective single-segment colectomy for colon cancer at our tertiary center between 2004 and 2014, 52.0% (n = 618) had right-sided colon cancer, 9.8% (n = 117) a transverse colon cancer, and 38.2% (n = 454) a left-sided colon cancer. Patient characteristics are summarized in Table 1. Median age (RC 71.4 vs. TC 70.3 vs. LC 62.6 years, P < 0.001) and mean ASA score (RC: 2.43 vs. TC 2.34 vs. LC 2.25, P < 0.001) were significantly different between the three groups. In terms of group-specific differences compared with the remainder of the population, patients with RC were older, had a higher ASA score (P < 0.001), and were more likely to be female (P0.003) and Caucasian (P0.014). LC patients were younger, had a lower ASA score (P < 0.001), and were more often male (P0.003) and Asian (P0.004). When considering comorbidities, RC patients had more frequently a history of diverticulitis (P0.045), chronic pulmonary disease (P0.029), prior abdominal surgery (P0.011), and anemia (P < 0.001). The incidence of anemia (P < 0.001), congestive heart failure (P0.033), chronic pulmonary disease (P0.013), or prior abdominal surgery (P0.001) was significantly lower in LC patients. TC patients had a higher incidence of alcohol abuse (P0.041). A symptomatic presentation was more often seen in patients with left-sided tumors, including complaints of changes in stool habits (P0.002), constipation (P0.014), and hematochezia (P < 0.001).
Pathology features
Statistically significant variations existed between the three groups in AJCC stage, nodal disease, tumor grade, microsatellite instability, tumor size, colonic specimen length, and lymph-node harvest (Table 2). Poorly differentiated tumors were seen more often in right-sided colon cancer (RC 22.5% vs. TC 14.5% vs. LC 11.2%, P < 0.001), as was high-microsatellite instability, and presence of both HPMS2 and HMLH1 loss (P < 0.001). Lymph-node-positive disease was correlated with left-sided cancer (RC 37.4% vs. TC 25.6% vs. LC 43.0%, P0.010). Furthermore, LC was associated with smaller tumor size, fewer lymph nodes harvested (P < 0.001), and more perineural invasion (P0.022). TC was associated with a significantly longer colonic specimen length (median RC 20 vs. TC 30 vs. LC 23 cm, P < 0.001), and less lymph-node positive disease (P0.003). R0 resections were achieved in comparable numbers (RC 92.1% vs. TC 96.6% vs. LC 93.6%, P0.182).
Peri-operative outcomes
Analysis of peri-operative outcomes is demonstrated in Table 3. Left-sided colon cancer resections were completed more often laparoscopically (RC 31.9% vs. TC 30.8% vs. LC 39.0%, P0.010) with a subsequent significantly longer median time of surgery (RC 104 vs. TC 135 vs. LC 135 min, P < 0.001). The overall laparoscopic conversion rate was 4.0%, with no difference between the groups (RC 3.1% vs. TC 6.8% vs. LC 4.6%, P0.119). The rate of adhesions was remarkably higher in RC patients (P < 0.001). Although not significantly different, multivisceral resections were more frequently performed in TC resections (RC 10.4% vs. TC 15.4% vs. LC 8.8%, P0.054). Of all patients with transverse colon cancer, 44.4% underwent a transverse colectomy and 55.6% an extended right or left colectomy.
Median length of stay and rate of complications within 30 days of surgery was significantly different between the groups. TC patients were admitted 1 day longer and developed more complications after surgery (RC 35.6% vs. TC 43.6% vs. LC 31.1%, P0.032). However, when including the requirement for blood transfusion in the complication rate, postoperative morbidity was comparable with RC but still significantly higher than the LC group (RC 46.1% vs. TC 51.3% vs. LC 35.9%, P0.049). On the contrary, although incidence of intra-abdominal abscess and/or anastomotic leakage was comparable between the groups, the rate of readmission was significantly higher in LC patients for abscess/leak (P0.038). This was reflected in higher reoperation rates after LC resection (P0.024), with anastomotic leakage (42.9%), colonic perforation (14.3%), bowel obstruction (14.3%), and fascial dehiscence (14.3%) as the main indications for reoperation. No differences were found regarding 30-day readmission and mortality rates.
Oncological outcomes
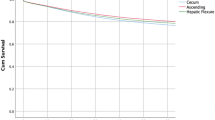
Within the full study cohort, 999 patients were diagnosed with stage I–III disease and included in the long-term analysis (Table 4). Median follow-up duration was 48.6 months and comparable between the groups. During the study period, more patients with right-sided tumors died with a significantly worse 5-year overall survival (RC 73.0%; TC 76.2%; LC 80.8%, P0.023). Nevertheless, colon cancer-specific survival was neither different between the groups (RC 91.7%; TC 94.2%; LC 91.8%, P0.372) nor was disease recurrence (RC 85.3%; TC 89.4%; LC 81.2%, P0.125). When analyzing stage-by-stage, no differences were found in either OS or DSS between the three groups. Despite a higher administration of adjuvant therapy in patients with left-sided tumors (P < 0.001), the estimated 5-year disease-free survival tended to be worse for this group compared to the remainder of the population (LC 81.2% vs. RC/TC 85.9%, P0.052). In addition, we found worse DFS for stage II left-sided colon cancer (LC 80.3% vs. RC/TC 90.2%, P0.019).
To assess risk factors for both survival and disease recurrence, a multivariate analysis was performed. Relationships between patient characteristics, clinicopathological features, and long-term outcomes in all patients undergoing curative resection for colon cancer are demonstrated in Table 5. On univariate analysis, left-sided colon cancer was associated with better overall survival compared to right-sided tumors (HR 0.73, P0.025). However, after adjustment tumor location was no longer associated with worse outcomes. Factors independently related to overall mortality included patient-related (older age, higher ASA score, BMI < 25 kg/m2, alcohol abuse), procedure-related (open surgery), and tumor-related characteristics (T3–T4 tumors, lymph-node positivity, high-grade disease, perineural invasion, R1 resection, less than 12 lymph nodes harvested) and the administration of adjuvant therapy. Regarding disease-specific survival, tumor location was not contributory in the univariate analysis. Pathological features including lymph-node disease, lymphovascular invasion, perineural invasion, and high grade disease as well as alcohol abuse were associated with worse colon cancer-specific survival. When analyzing risk factors for disease-free survival, anastomotic type appeared to be a risk factor for poorer outcomes. Compared to ileo-colonic anastomoses, patients with a colo-colonic anastomosis had worse disease-free survival (HR 1.63, P0.048). Risk was non-significantly higher when compared to colorectal anastomoses (HR 1.37, P0.238).
Discussion
Over the last several years, there has been increased interest in identifying the differences between proximal and distal colon cancer. Differences in epidemiology, patient demographics, and histological features relative to tumor site in colon cancer are observed, yet the location of the tumor is often not considered as a separate entity when outcomes are discussed. In addition, transverse colon cancers are often excluded altogether or included in either the right- or left-sided group for analysis. Therefore, the aim of our study was to assess the differences in clinicopathological characteristics as well as long-term outcomes in patients who were diagnosed with either a right-sided, left-sided, or transverse colon cancer.
In our study, patients with right-sided colon cancer were older, more likely to be female and had poorer histopathological features including more T3–T4 tumors and poor differentiation. This is fully consistent with previous data [7, 13, 14]. On the other hand, lymph-node-positive disease was more frequent in LC tumors. Transverse cancer was correlated with stage II disease, but the requirement for a multivisceral resection was higher in this group due to contiguous involvement of adjacent organs. Regarding short-term outcomes, most studies have investigated the rate of complications during admission and these tend to be higher for right-sided procedures [7, 15, 16]. Benedix et al. demonstrated a higher rate of general postoperative complications for RC patients, including pulmonary and cardiovascular complications, while surgery-related complications were almost equally distributed. Other studies concluded that risk of major complications was comparable between the two locations [15,16,17,18]. However, knowledge of differences in short-term outcomes after surgery for transverse colon cancer is scarce. Since transverse colon cancer requires either an extended colectomy or a transverse colectomy with the need for a colo-colonic anastomosis, we hypothesized that this might lead to a higher comorbidity rate and differences in short-term outcomes exist depending on the type of resection. Our study demonstrated a longer length of stay and a higher complication rate when the requirement for blood transfusion was excluded. However, when blood transfusion was incorporated in the morbidity rate, outcomes were similar with RC patients but still worse than after LC surgery. Our relatively high blood transfusion rate could be explained by the fact that we incorporated all patients who received blood whether or not this was pre-operative, intra-operative, or post-operative. This was especially true in right-sided colon cancer, since these patients often presented with anemia. LC patients had a better postoperative course with shorter admission duration and less complications, including less general postoperative complications as cardiac and renal events. Although the incidence of intra-abdominal abscesses and/or leaks was equally distributed, the necessity of readmission and subsequent reoperation for a leak was significantly higher in LC patients. This is in line with the overall belief that LC procedures are technically more challenging and due to differences in vascularization prone to develop anastomotic leakage. However, outcomes are contradictory with more recent studies suggesting no difference in incidence of anastomotic leak between right-sided and left-sided colectomies [15, 16].
The main finding in our study was the worse overall but comparable disease-specific survival for right-sided colon cancer. However, when analyzing stage-by-stage, the prognostic impact of tumor location was no longer observed. After adjusting for multiple variables, only patient characteristics and pathological features were independently related to overall and disease-specific survival. This is in contrast with previous studies including a recent systematic review and meta-analysis that reported worse overall survival for right-sided colon cancer [7, 12, 19]. Most of the studies included in the systematic review and meta-analysis evaluated only overall survival. The higher comorbidity rate and older age in the proximal colon cancer group are a reasonable explanation for the worse outcomes, especially since colon cancer-specific survival was found to be similar. Although most studies adjusted for baseline characteristics as age and ASA score, information about adjuvant therapy was often lacking. Furthermore, we excluded patients who underwent neoadjuvant therapy and patients with distant metastasis, and we were able to adjust for known histopathological risk factors besides TNM stage. The latter proved to be an important factor, since pathologic features such as poor differentiation, perineural invasion, and lymphovascular invasion were independent predictors for worse oncological outcomes.
The influence of tumor location and disease recurrence is poorly investigated. Lim et al. found worse DFS for stage III right-sided tumors, but this effect disappeared after adjusting for patient and tumor characteristics [20]. In our study, left-sided colon cancer patients tended to have a worse 5-year disease-free survival, with a significant difference in stage II disease. No differences in admission of adjuvant therapy between LC and the remainder of the population were found. After adjusting for multiple variables, risk factors for worse DFS included advanced TNM stage, extramural vascular invasion, and perineural invasion as well as type of anastomosis. Compared to ileo-colonic anastomoses, patients with a colo-colonic anastomosis had worse disease-free survival (HR 1.63, P0.048). Outcomes were comparable between colo-colonic and colorectal anastomoses (HR 1.37, P0.238) as well as colorectal and ileo-colonic anastomoses (HR 1.19, P0.419). Previous studies demonstrated that anastomoses close to the anal verge were at risk for developing anastomotic leakage [21, 22]. In rectal cancer, anastomotic leakage is associated with an increased risk of local recurrence, whereas the impact of distant recurrence remains debatable [23,24,25]. Knowledge about the impact of anastomotic leakage and recurrent disease in colon cancer is limited. Although previous studies demonstrated conflicting results, there is some evidence that anastomotic leakage is associated with reduced disease-free survival [26, 27].
Our study has several strengths and limitations. To our knowledge, this is the first study that assessed differences in clinicopathological and long-term outcomes between different segments in colon cancer and included transverse colon cancer as a separate entity. Moreover, only one study reported disease-free survival concerning this topic. Due to a prospectively maintained single-center database, another major strength of our study is the ability to adjust for multiple confounders. However, selection bias is inherent to the retrospective design, and although we adjusted for important clinicopathological factors, the determination of MSI status was not yet routine management during our study period. Consequently, the prognostic impact of MSI, associated with right-sided colon cancer, might be underestimated in our analysis.
Conclusion
Although distinct differences were found between right-sided, transverse, and left-sided colon cancer in terms of patient characteristics, histopathological features, and 30-day morbidity, tumor location in colon cancer was not independently associated with survival and disease recurrence. Nonetheless, pathological differences beyond tumor stage were significantly more important. Future research should elaborate on differences in disease characteristics leading to potential different optimal treatments in colon cancer.
Grant support
LL received a grant from the Prins Bernhard Cultuurfonds and Hendrik Muller’s Fonds.
References
Edwards BK, Ward E, Kohler BA, Eheman C, Zauber AG, Anderson RN, Jemal A, Schymura MJ, Lansdorp-Vogelaar I, Seeff LC, van Ballegooijen M, Goede SL, Ries LA (2010) Annual report to the nation on the status of cancer, 1975-2006, featuring colorectal cancer trends and impact of interventions (risk factors, screening, and treatment) to reduce future rates. Cancer 116:544–573
Vogelaar I, van Ballegooijen M, Schrag D, Boer R, Winawer SJ, Habbema JD, AGl Z (2006) How much can current interventions reduce colorectal cancer mortality in the US? Mortality projections for scenarios of risk-factor modification, screening, and treatment. Cancer 107:1624–1633
Cucino C, Buchner AM, Sonnenberg A (2002) Continued rightward shift of colorectal cancer. Dis Colon Rectum 45:1035–1040
Saltzstein SL, Behling CA (2007) Age and time as factors in the left-to-right shift of the subsite of colorectal adenocarcinoma: a study of 213,383 cases from the California Cancer Registry. J Clin Gastroenterol 41:173–177
Bufill JA (1990) Colorectal cancer: evidence for distinct genetic categories based on proximal or distal tumor location. Ann Intern Med 113:779–788
Gervaz P, Bucher P, Morel P (2004) Two colons-two cancers: paradigm shift and clinical implications. J Surg Oncol 88:261–266
Benedix F, Kube R, Meyer F, Schmidt U, Gastinger I, Lippert H (2010) Colon/rectum carcinomas (primary tumor) study group. Comparison of 17,641 patients with right- and left-sided colon cancer: differences in epidemiology, perioperative course, histology, and survival. Dis Colon Rectum 53:57–64
Azzoni C, Bottarelli L, Campanini N, Di Cola G, Bader G, Mazzeo A, Salvemini C, Morari S, Di Mauro D, Donadei E, Roncoroni L, Bordi C, Sarli L (2007) Distinct molecular patterns based on proximal and distal sporadic colorectal cancer: arguments for different mechanisms in the tumorigenesis. Int J Color Dis 22:115–126
Nawa T, Kato J, Kawamoto H, Okada H, Yamamoto H, Kohno H, Endo H, Shiratori Y (2008) Differences between right- and left- sided colon cancer in patient characteristics, cancer morphology and histology. J Gastroenterol Hepatol 23:418–423
Ponz de Leon M, Sacchetti C, Sassatelli R, Zanghieri G, Roncucci L, Scalmati A (1990) Evidence for the existence of different types of large bowel tumor: suggestions from the clinical data of a population-based registry. J Surg Oncol 44:35–43
Ogino S, Odze RD, Kawasaki T, Brahmandam M, Kirkner GJ, Laird PW, Loda M, Fucks CS (2006) Correlation of pathologic features with CpG island methylator phenotype (CIMP) by quantitative DNA methylation analysis in colorectal carcinoma. Am J Surg Pathol 30:1175–1183
Meguid RA, Slidell MB, Wolfgang CL, Chang DC, Ahuja N (2008) Is there a difference in survival between right- versus left- sided colon cancers? Ann Surg Oncol 15:2388–2394
Weiss JM, Pfau PR, O’Connor ES, King J, LoConte N, Kennedy G, Smith MA (2011) Mortality by stage for right- versus left-sided colon cancer: analysis of surveillance, epidemiology, and end results–medicare data. J Clin Oncol 29:4401–4409
Powell AG, Wallace R, McKee RF, Anderson JH, Going JJ, Edwards J, Horgan PG (2012) The relationship between tumour site, clinicopathological characteristics and cancer-specific survival in patients undergoing surgery for colorectal cancer. Color Dis 14:1493–1499
Campana JP, Pellegrini PA, Rossi GL, Ojea Quintana G, Mentz RE, Vaccaro CA (2017) Right versus left laparoscopic colectomy for colon cancer: does side make any difference? Int J Color Dis 32:907–912
Masoomi H, Buchberg B, Dang P, Carmichael JC, Mills S, Stamos MJ (2011) Outcomes of right vs. left colectomy for colon cancer. J Gastrointest Surg 15:2023–2028
Petrelli F, Tomasello G, Borgonovo K, Ghidini M, Turati L, Dallera P, Passalacqua R, Sgroi G, Barni S (2017) Prognostic survival associated with left-sided vs right-sided colon cancer: a systematic review and meta-analysis. JAMA Oncol 3:211–219
Kwaan MR, Al-Refaie WB, Parsons HM, Chow CJ, Rothenberger DA, Habermann EB (2013) Are right-sided colectomy outcomes different from left-sided colectomy outcomes?: study of patients with colon cancer in the ACS NSQIP database. JAMA Surg 148:504–510
Yahagi M, Okabayashi K, Hasegawa H, Tsuruta M, Kitagawa Y (2016) The worse prognosis of right-sided compared with left-sided colon cancers: a systematic review and meta-analysis. J Gastrointest Surg 20:648–655
Lim DR, Kuk JK, Kim T, Shin EJ (2017) Comparison of oncological outcomes of right-sided colon cancer versus left-sided colon cancer after curative resection. Medicine (Baltimore) 96:e8241
Trencheva K, Morrissey KP, Wells M, Mancuso CA, Lee SW, Sonoda T, Michelassi F, Charlson ME, Milsom JW (2013) Identifying important predictors for anastomotic leak after colon and rectal resection: prospective study on 616 patients. Ann Surg 257:108–113
Veyrie N, Ata T, Muscari F, Couchard AC, Msika S, Hay JM, Fingerhut A, Dziri C, French Associations for Surgical Research (2007) Anastomotic leakage after elective right versus left colectomy for cancer: prevalence and independent risk factors. J Am Coll Surg 205:785–793
Eberhardt JM, Kiran RP, Lavery IC (2009) The impact of anastomotic leak and intra-abdominal abscess on cancer-related outcomes after resection for colorectal cancer: a case control study. Dis Colon Rectum 52:380–386
Mirnezami A, Mirnezami R, Chandrakumaran K, Sasapu K, Sagar P, Finan P (2011) Increased local recurrence and reduced survival from colorectal cancer following anastomotic leak: systematic review and meta-analysis. Ann Surg 253:890–899
Bertelsen CA, Andreasen AH, Jørgensen T, Harling H, Danish Colorectal Cancer Group (2010) Anastomotic leakage after curative anterior resection for rectal cancer: short and long-term outcome. Color Dis 12:e76–e81
Kube R, Mroczkowski P, Granowski D, Benedix F, Sahm M, Schmidt U, Gastinger I, Lippert H (2010) Anastomotic leakage after colon cancer surgery: a predictor of significant morbidity and hospital mortality, and diminished tumour-free survival. Eur J Surg Oncol 36:120–124
Krarup PM, Nordholm-Carstensen A, Jorgensen LN, Harling H (2014) Anastomotic leak increases distant recurrence and long-term mortality after curative resection for colonic cancer: a nationwide cohort study. Ann Surg 259:930–938
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Leijssen, L.G.J., Dinaux, A.M., Kunitake, H. et al. Pathologic factors are more important than tumor location in long-term survival in colon cancer. Int J Colorectal Dis 33, 709–717 (2018). https://doi.org/10.1007/s00384-018-3027-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-018-3027-5