Abstract
Purpose
Asymmetric conjoined twining (ACT) is a form of conjoined twining which is a rare malformation of monochorionic monoamniotic twin pregnancy. Most publications were single case reports. We reported a cohort of five cases with ACT from a single tertiary medical center and reviewed the case reports of ACT over the last decade to enrich the clinical research of this disease and summarized the clinical features of the disease.
Methods
We reviewed five cases of ACT admitted in Tianjin Children's Hospital from 17 March, 2008, through 7 March 2017. The cohort was analysed from general information, imaging manifestations, separation surgery, histopathological findings, outcome and follow-up. We searched the English literatures on case reports of ACT over the past decade from the PubMed database and presented details about the clinical characteristics, treatment, and prognosis of all cases.
Results
There were four males and one female in our cohort. Among the five cases, two parasites were located in epigastrium, two in rachis, and one in retroperitoneum (fetus in fetu, FIF). All of the parasites were separated successfully by operation in five cases and were confirmed to be ACT by histopathology reports. Four patients made an uneventful recovery except for one case of wound infection. All of them were doing well in follow-up. In the literature review, we found 41 cases of exoparasitic heteropagus twining (EHT) and 63 cases of FIF.
Conclusions
ACT is very rare and usually diagnosed by prenatal ultrasonography (US). Computed tomography (CT) and magnetic resonance imaging (MRI) examinations are essential imaging examinations before separation surgery to delineate the anatomical relationship between the autosite and the parasite. In general, the separation surgery of ACT is less complicated and the prognosis is better compared with the symmetric conjoined twining (SCT).
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Conjoined twining is a rare form of twin pregnancy, including symmetric conjoined twining (SCT) and asymmetric conjoined twining (ACT). ACT is extremely rare, including exoparasitic heteropagus twining (EHT) and endoparasitic heteropagus twining, which also named fetus in fetu (FIF). The earliest case was proposed by Friedrich Meckel in 1800 [1]. It is usually detected by prenatal ultrasonography (US). Computed tomography (CT) and magnetic resonance imaging (MRI) examinations should be performed before separation surgery to determine the location, extent, organ distribution and fusion of the junction between the autosite and the parasite, as well as the distribution of the blood vessels between the twins. Here we reviewed five cases of ACT. Among them, two parasites were located in epigastrium, two in rachis and one in retroperitoneum (FIF). All of them were separated by operation successfully. Besides, a review of literature was presented for the sake of a further understanding of ACT.
Patients and methods
Patients
There were five cases of ACT admitted in Tianjin Children's Hospital from 17 March 2008 to 7 March 2017.
The general information of all patients, such as age, sex, relevant family history, history of gestation, gestational weeks at birth, mode of delivery, method and timing of diagnosis, deformity type, physical examinations were collected. Written informed consents were obtained from parents of the five patients and the study was approved by the ethics committee of Tianjin Children's Hospital (Tianjin, China).
Imaging examinations
It was very important to perform the related imaging examinations before separation surgery to delineate the anatomical relationship between the autosite and the parasite. CT and MRI examinations clarified the visceral and vascular communication between the parasite and the autosite. Echocardiography (UCG) revealed cardiac malformations in autosite. US and UCG were performed in all five cases, CT and MRI in four cases (Case 1, 3, 4 and 5).
Separation surgeries
After defining the anatomical relationship between the autosite and the parasite through physical examinations and necessary imaging examinations, all patients underwent separation surgeries.
Literature review
We searched the English literatures on case reports of ACT over the last decade from the PubMed database and collected the general information, clinical characteristics, treatment, and prognosis of all cases.
Results
Patients’ details
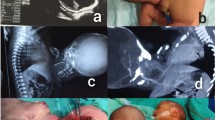
The age, sex, relevant family history, history of gestation, gestational weeks at birth, mode of delivery, method and timing of diagnosis, deformity type, physical examinations of the five cases were shown in Table 1. There were four males and one female. Three of them were admitted to the Department of Neonatal Surgery (Case 1, 2 and 5), and two were admitted to the Department of Pediatric Neurosurgery (Case 3 and 4). Among these cases, two parasites were located in epigastrium (Case 1 and 2), two in rachis (Case 3 and 4) and one in retroperitoneum (FIF) (Case 5). Only one case was diagnosed before birth (Case 5). The physical examinations of them were listed in Table 1. The malformations of cases 1, 2, 3 and 4 were shown in Figs. 1a, b, 2a, 3a, b and 4a–c, respectively. The parasites’ extremities of the three cases (Case 1, 2 and 4) could not move spontaneously or respond to external stimulation. The scrotum and penis of parasite presented in two cases, one of which showed no automatic micturition (Case 1), and the other could urinate autonomously (Case 2). In case 4, there was an anus and a perineum at the junction of the parasite’s limb and the autosite’s buttock, but neither urination nor defecation was observed. In case 3, an irregular mass on the back of the autosite could be seen (Fig. 3). The located skin on the left side of the junction was red and dry without exudation. Besides, an abnormal bony processes could be found on the right side of the basement.
Imaging details
The imaging details were shown in Table 2. US and UCG were performed in all five cases, CT and MRI in four cases (Case 1, 3, 4 and 5) (Figs. 1c, d, 3c–e, 4d, e, 5a, b). In case 5, a heterogeneous mass with clear boundary and intact capsule was seen in the right upper abdomen of the child, including multiple long bones and a probable vertebral body formation, suggesting that it might be a FIF.
Intraoperative findings
The details of surgeries, such as time of operation, surgical findings and medical operations, were shown in Table 3. All patients underwent single-stage surgery performed by a multidisciplinary team. The time of separation surgery ranged from 9th day to 6th month of life. All of the parasites were separated successfully from the autosite (Figs. 1e, 2b, c, 3f, 4f, 5c, d).
Histopathological findings
Histopathological examinations were performed in five cases and all of them were confirmed as ACT. The histopathological details were shown in Table 4.
Outcome and follow-up
Case 1 showed wound infection after operation, and wound healed gradually after regular dressing change. The other four cases made an uneventful recovery without wound dehiscence, urinary and fecal incontinence, or heart failure. The postoperative wounds of cases 1 and 3 were shown in Figs. 1f and 3g, respectively. In our five cases, the shortest follow-up lasted for 3 months and the longest lasted for 2.5 years. All of the five patients were doing well, enjoying a normal quality of life. The outcome and follow-up were shown in Table 5.
Literature review
The details about the general information, clinical characteristics, treatment, and prognosis of all cases were presented in Supplementary Table A (EHT) [2,3,4,5,6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38] and Supplementary Table B (FIF) [39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84,85,86,87,88].
In the literature review, we found 41 cases of EHT and 63 cases of FIF. In EHT, 17 were females, 24 were males. In FIF, 32 were females, 28 were males and 3 were not mentioned. In terms of parasitic sites, the most common site of EHT was abdomen, especially repigastrium, followed by rachis, while the most common site of FIF was retroperitoneum. When it came to treatment (mentioned in the literature), most cases received surgical treatment and most of them had a good prognosis, whether EHT or FIF. Specifically, in the 41 cases of EHT, 33 cases underwent separation surgery, of which 25 cases were fine after operation, 4 cases had postoperative complications such as wound infection and wound dehiscence, 2 cases left congenital heart disease and 2 cases died after operation; 7 cases did not receive surgical treatment. The treatment of 1 cases was not mentioned in EHT. In the 63 cases of FIF, 48 cases underwent separation surgery, of which 42 cases were fine after operation, 1case left epilepsy and delayed psychomotor development, 1 case left urinary retention and 4 cases died after operation; 4 cases did not receive surgical treatment. The treatments of 11 cases were not mentioned in FIF.
Discussion
ACT, also known as heteropagus conjoined twining, incomplete conjoined twining, parasitic twining, is a rare congenital developmental malformation. ACT means a relatively mature fetus parasitizing a severely developmentally deficient fetus in any portion of the body. We reviewed the case reports of EHT and FIF over the past decade (Supplementary Table A and Supplementary Table B), except for cases of termination before 20 weeks of pregnancy. The incidence is 0.5–1 in 1000000 births [89]. There is a male predominance in ACT as opposed to female predominance in SCT [17, 90]. In literature review, there were 24 males and 17 females in EHT while 28 males and 32 females in FIF. In our five cases, four cases were male and one was female. Case 3 had been published previously as an 11-day-old infant with an accessory penis and scrotum on the posterior thoracic region [91].
The mechanism of conjoined twining has not been clear yet. Two possible theories have been proposed, namely the fission theory and the fusion theory. Both theories indicate that ACT occurs around two weeks after fertilization. The fission theory claims that a single zygote cannot divide completely [92], while the fusion theory proposes that the fusion of two embryos occurs prior to implantation [93, 94]. At present, most people support the fission theory. In the literature, DNA analysis of one case showed that the ACT was dizygotic, confirming the possibility of fusion theory [95]. Another ischemic theory has also been proposed, in which the parasite results from ischemia and resorption of the autosite [92, 96].
ACT is classified into EHT and FIF. Spencer et al. suggested that FIF should have at least one of the following characteristics: (1) a completely encapsulated mass, (2) partially or completely covered by normal skin, (3) one or more clearly identifiable anatomical structures, (4) attached to the autosite by only a few relatively large blood vessels and (5) either close to the attachment site of conjoined twins, or connected with the neural tube or gastrointestinal tract [90]. In our case 5, parasite originated from the retroperitoneum of the autosite was wrapped in a complete capsule with both lower extremities and feet. Imaging examinations showed a heterogeneous mass with multiple long bones and a probable vertebral body formation. The histopathological examinations demonstrated cartilage and bone-like tissue which were suggestive of rudimentary vertebrae and long bones of lower limbs. As a result, the final diagnosis of FIF was made. The common parasitic sites of EHT include epigastrium (omphalopagus), rachis (rachipagus), thorax (thoracopagus), abdomen and ischium (ischiopagus). A few parasites can be located in extracranial region (cephalopagus) and perineum as well. However, there are also reports of atypical EHT, such as atypical ischiopagus [97]. As for FIF, the parasite can be located in anywhere of the autosite. Retroperitoneum, abdominal cavity and thoracic cavity are more usual. In the literature review, the most common site of EHT was abdomen, especially repigastrium, followed by rachis, while the most common site of FIF was retroperitoneum. In our five cases, two parasites were located in epigastrium, two in rachis and one in retroperitoneum (FIF).
Clinical features are associated with the type of ACT. The most common feature of EHT is supranumerary limbs, especially in thoracopagus and omphalopagus. The most limbs of parasites could not move spontaneously or respond to external stimulus. While two different cases have been reported in the literature. In one case, a pair of asymmetric conjoined twins had a common dual nerve supply, and stimulation of any part of the body resulted in movement of all limbs [98]. In another case, the extremities of the parasite exhibited spontaneous movement in the toes and apparent sensation [30]. In our five cases, three cases had supranumerary limbs, and none of them moved spontaneously or responded to external stimulus. Other manifestations such as abdominal wall defect and abdominal visceral fusion are usual in omphalopagus, while spinal defects are usually found in rachipagus. The clinical features of FIF are related to the location of parasite. For example, parasite located in retroperitoneum can be characterized by abdominal distension and low fever, while that in mouth causes oropharynx developmental deformities.
ACT with other malformations are less common than SCT, especially in FIF. The most common malformation is omphalocele, followed by cardiac anomalies [32]. Other malformations are rare, such as genitourinary system malformations and digestive system malformations. In our five cases, two cases had omphalocele. All of the patients underwent echocardiography and all of them found varying degrees of cardiac malformations. In addition, the soft tissue of anterior chest wall, sternum and costal cartilage defects were found in case 1. In the meantime, bilateral testicular hydrocele was also found. Case 4 showed lumbosacral spinal dysraphism, sacral dysplasia, tethered cord, myelomeningocele, expansion of central spinal canal from 9th thoracic to 4th lumbar vertebral level and lipoma of filum terminale.
ACT is usually identified by prenatal US. Sonographic findings in conjoined twins include inseparable fetal bodies and skin contours, no change in the relative positions of the fetuses and shared organs [99]. The earliest diagnosis was reported to be made at the 9th week of pregnancy [100]. But in our five cases, three mothers did not undergo US prenatally, so the malformations were not found before birth. Case 2 was an abandoned baby and the details could not be obtained. Only one patient was diagnosed by prenatal US. Prenatal three-dimensional ultrasound examination can provide a clearer image, which contributes to diagnosis and prenatal counseling. It was reported that two cases of ACT were diagnosed by prenatal three-dimensional ultrasound [13, 101]. It is very important to perform CT and MRI examinations before operation. These examinations are helpful to understand the connection site, organ distribution and fusion of twins, and to judge the blood supply of parasite. With the development of three-dimensional CT reconstruction technique, it is possible to present the structure of twins in three dimensions. It helps us to understand the twins’ position relationship more intuitively, which is crucial for the design of operation schemes and the evaluation of prognosis.
Whether early termination of pregnancy is required, as well as the timing of termination need to be determined individually, according to the types and forms of ACT. Surgery can be performed during 1st week of patient's life to the age of 1 year [30]. That’s because the patient's vital signs are relatively stable during this period. In addition, if the surgical age is too late, the growth and development of the autosite might be affected. In our five cases, the time of separation surgeries ranged from 9th day to 6th month of life. Cases 3, 4 and 5 were older than one week at admission, so the separation surgeries were performed after preoperative preparation. Case 1 was admitted to the hospital 12 h after birth. The operation was performed nine days later when the vital signs were stable and the preoperative preparation was ready. Case 2 was an abandoned infant whose timing of surgery was also influenced by several other factors. Compared with SCT, the operation procedure of ACT is less complicated because of the less extensive vascular and visceral connections. The surgical purpose of ACT is to remove the redundant parasite, while that of SCT is to separate the two surviving individuals. What’s more, some ethical issues will arise when only one fetus can be preserved during the operation of SCT [102].
The prognosis of ACT is related to the location of connection and the degree of organ fusion between the parasite and the autosite, as well as other malformations. The presence and severity of cardiac malformations are the main determinant factors of the prognosis [15]. In general, the prognosis of ACT is better than that of SCT. In our study, except one case of wound infection, the remaining four patients recovered smoothly. All of them were doing well in follow-up. In the literature review, the prognosis (mentioned in the literature) of most cases were good, except for five cases of preoperative death, one case of intraoperative death, five cases of postoperative death, one case of early termination of pregnancy and two cases of death in utero. In the EHT, one case died in utero because of severe congenital cardiac anomaly. The other two cases with multiple cardiac anomalies died of cardiac arrest and acute respiratory disorder before surgery, respectively. One case died before doing any investigations because of septicemia. Two cases died postoperatively, one of them was due to respiratory distress and cardiac failure, and the other was not able to determine the exact cause of death. In FIF, one case died in utero at 37 weeks and the two cases who died before operation were all cases of parasite located in intracalvarium. One case of FIF died for cardiac arrest during the operation. Among the three cases of FIF who died after surgery, two cases died for serious postoperative complications, one case died for complex congenital heart disease.
Conclusion
ACT is very rare and usually diagnosed by prenatal US. CT and MRI examinations are essential before separation operation to delineate the anatomical relationship between the autosite and the parasite. In most cases, the surgical operation of asymmetric twins is less complicated and the prognosis is better compared with the SCT.
References
Lewis RH (1961) Foetus in foetu and the retroperitoneal teratoma. Arch Dis Child 36:220–226. https://doi.org/10.1136/adc.36.186.220
Abubakar AM, Ahidjo A, Chinda JY et al (2010) The epigastric heteropagus conjoined twins. J Pediatr Surg 46:417–420. https://doi.org/10.1016/j.jpedsurg.2010.09.045
Ozkan-Ulu H, Yilmaz Y, Sari FN, Altug N, Uras N, Dilmen U (2011) An unusual case of heteropagus: autosite with a complex cardiac malformation. Pediatr Neonatol 52:358–360. https://doi.org/10.1016/j.pedneo.2011.08.011
Qasim M, Shaukat M (2011) Epigastric heteropagus twin. APSP J Case Rep 2:24
Zhang J, Duan H, Zhang Y, Yi Z, Bao S (2011) Parasitic rachipagus conjoined twins with spina bifida, diplomyelia, scoliosis, tethered cord syndrome, and ventricular septal defect—case report. Neurol Med Chir (Tokyo) 51:736–739. https://doi.org/10.2176/nmc.51.736
Okumura M, Liao AW, Brizot Mde L, Zugaib M, Schultz R (2011) Unusual presentation of a sacral parasitic conjoined twin. J Ultrasound Med 30:281–283. https://doi.org/10.7863/jum.2011.30.2.281
Xie JT, Zhou L, Yang ZL, Sun HY (2012) Epigastric heteropagus conjoined twins: two case studies and associated DNA analysis. Clinics (Sao Paulo) 67:527–529. https://doi.org/10.6061/clinics/2012(05)22
Solak A, Ergün S, Polat I, Sahin N, Genç B (2012) A rare form of heteropagus twinning: three-armed infant with spinal dysraphism. Case Rep Pediatr 2012:831649. https://doi.org/10.1155/2012/831649
Terata M, Kikuchi A, Kanasugi T, Oyama R, Fukushima A, Sugiyama T (2013) Prenatal diagnosis of parasitic conjoined twins with three-dimensional ultrasound. Congenit Anom (Kyoto) 53:131–133. https://doi.org/10.1111/j.1741-4520.2012.00378.x
Kesan K, Gupta A, Gupta RK et al (2013) Gluteal flap for omphalocele repair in a case of epigastric heteropagus: a novel approach for surgical management. Indian J Plast Surg 46:127–129. https://doi.org/10.4103/0970-0358.113731
Komla G, Komla A, Kpatekana S et al (2013) Experience of managing conjoined and parasitic twins from a developing country. APSP J Case Rep 4:52
Pandey A, Singh SP, Pandey J, Gupta V, Verma R (2013) Lumbosacral parasitic twin associated with lipomeningomyelocele: a rare occurrence. Pediatr Neurosurg 49:110–112. https://doi.org/10.1159/000358096
Panda SS, Das RR (2013) Incomplete conjoined twin. Indian J Pediatr 80:712. https://doi.org/10.1007/s12098-013-1026-7
Dar SH, Liaqat N, Iqbal J, Latif T, Iqbal A (2014) An epigastric heteropagus twin with ruptured giant omphalocele. J Neonatal Surg 3(2):23
Calderoni DR, Mizukami A, Nunes PH, Kharmandayan P (2014) Thoraco-omphalopagus asymmetric conjoined twins: report of a case and complete review of the literature. J Plast Reconstr Aesthet Surg 67:e18-21. https://doi.org/10.1016/j.bjps.2013.06.022
Bayri Y, Tanrıkulu B, Ekşi MS, Dağçınar A (2014) Accessory lower limb associated with spina bifida: case report. Childs Nerv Syst 30:2123–2126. https://doi.org/10.1007/s00381-014-2475-7
Kelani AB, Moumouni H, Younsa H et al (2016) A case of cephalomelia discovered in a baby born in Niger. Childs Nerv Syst 32:205–208. https://doi.org/10.1007/s00381-015-2831-2
Baskaran D, Aleem MA, Ravi R (2015) Parasitic twin with gastroschisis is one of the rarest variant of conjoined twins: a case report. Indian J Surg 77:90–91. https://doi.org/10.1007/s12262-014-1165-8
Anca FA, Negru A, Mihart AE, Grigoriu C, Bohîlțea RE (2015) Special forms in twin pregnancy—asymmetric conjoined twins. J Med Life 8:115–118
Stahr N, Guggenberger R, Kellenberger CJ, Wisser J, Subotic U (2015) In utero and postnatal imaging findings of parasitic conjoined twins (ischiopagus parasiticus tetrapus). Pediatr Radiol 45:767–770. https://doi.org/10.1007/s00247-014-3172-0
Gokcen EC, Wamisho BL (2015) Delayed presentation of a heteropagus (parasitic) twin: a case report of a 17-year-old patient. J Pediatr Orthop B 24:567–572. https://doi.org/10.1097/BPB.0000000000000140
Navaei AA, Habibi Z, Moradi E, Nejat F (2015) Parasitic rachipagus twins; report of two cases. Childs Nerv Syst 31:1001–1003. https://doi.org/10.1007/s00381-015-2664-z
Nega W, Damte M, Girma Y, Desta G, Hailemariam M (2016) Craniopagus parasiticus—a parasitic head protruding from temporal area of cranium: a case report. J Med Case Rep 10:340. https://doi.org/10.1186/s13256-016-1023-3
Sahlu A, Mesfin B, Tirsit A, Debebe T, Wester K (2016) Parasitic twin—a supernumerary limb associated with spinal malformations. A case report. Acta Neurochir (Wien) 158:611–614. https://doi.org/10.1007/s00701-016-2710-y
Rattan KN, Singh J, Dalal P, Sonika P, Rattan A (2016) Sacral rachipagus parasite: a case report. J Neonatal Surg 5:16
Raj P, Birua H (2017) Epigastric heteropagus twin. J Neonatal Surg 6:47. https://doi.org/10.2169/jns.v6i2.491
Raheja A, Mahapatra AK (2017) Rachipagus parasitic twin. Neurol India 65:1443–1444. https://doi.org/10.4103/0028-3886.217969
Malik M, Bahadur Singh U, Hedge S, Mahajan JK, Samujh R (2018) Omphalocele and epigastric heteropagus: implications and treatment. Oxf Med Case Rep. https://doi.org/10.1093/omcr/omy074
Khushdil A, Niaz H, Ahmed Z (2018) Epigastric heteropagus conjoined twins. J Coll Physicians Surg Pak 28:S42–S43. https://doi.org/10.29271/jcpsp.2018.03.S42
Jabari S, Carbon R, Besendörfer M et al (2018) Asymmetric omphalopagus in a triplet after in vitro fertilization: a rare case of conjoined twinning. Case Rep Pediatr 2018:9349606. https://doi.org/10.1155/2018/9349606
Muhelo AR, Montemezzo G, Da Dalt L et al (2018) Successful management of a parasitic ischiopagus conjoined twins in a low-income setting. Clin Case Rep 6:385–390. https://doi.org/10.1002/ccr3.1374
Kavecan I, Obrenovic M, Privrodski JJ, Privrodski B, Jeckovic M (2018) Parasitic twin presenting rudimentary upper limbs causes a unique spectrum of anomalies of autosite. Balkan Med J 35:445–446. https://doi.org/10.4274/balkanmedj.2018.0781
Khavanin N, Ruge JR, Vicari FA, Belin EJ, Kellogg RG, Steinberg JP (2018) Parasitic rachipagus conjoined twin: case report. J Neurosurg Pediatr 22:313–316. https://doi.org/10.3171/2018.3.PEDS1822
Mekonnen T (2018) Tail-like congenital duplication of lower extremity (extra leg or vestigial parasitic twin). Ethiop J Health Sci 28:103–107. https://doi.org/10.4314/ejhs.v28i1.14
Abiramalatha T, Balasubramanian R, Suman FR, Agarwal P, Balakrishnan U, Amboiram P (2019) Diagnostic dilemma of an umbilical mass in a newborn infant—a twin or a tumor. Fetal Pediatr Pathol 2019:1–5. https://doi.org/10.1080/15513815.2019.1707920
Pang H, Zang J, Qiu L (2020) Prenatal diagnosis of parasitic conjoined twins using three-dimensional ultrasound: a case report. Int J Gynaecol Obstet 148:265–266. https://doi.org/10.1002/ijgo.13017
Takrouney MH, Ibrahim IA, Abdel-Ghaffar HS et al (2020) Conjoined twins: a report of four cases. Int J Surg Case Rep 73:289–293. https://doi.org/10.1016/j.ijscr.2020.06.072
Falyoun MS, Mashlah Q, Aldiri Q, Mohamad AR, Daowd LK, Ayash M (2020) Epigastric heteropagus and omphalocele. J Surg Case Rep. https://doi.org/10.1093/jscr/rjaa437
Agrawal V, Joshi MK, Gomber S (2011) Unusual presentation of fetus-in-fetu mimicking malignant teratoma. Trop Gastroenterol 32:141–143
Bhadane S, Singh H, Kachewar S, Sasane A (2011) Foetus-in-foetu. Med J Armed Forces India 67:267–269. https://doi.org/10.1016/S0377-1237(11)60057-9
Gunaydin M, Celik FC, Tander B et al (2011) Two cases of fetus in fetu. J Pediatr Surg 46:e9–e12. https://doi.org/10.1016/j.jpedsurg.2011.05.012
Kim YJ, Sohn SH, Lee JY et al (2011) Misdiagnosis of fetus-in-fetu as meconium peritonitis. Korean J Pediatr 54:133–136. https://doi.org/10.3345/kjp.2011.54.3.133
Angoulvant F, Bonnard A (2011) Foetus-in-foetu in a 3-year-old girl. Acta Paediatr 100:e145. https://doi.org/10.1111/j.1651-2227.2011.02341.x
Ali SR, Qadir M, Ahmed S, Kumar P (2011) Foetus-in-foetu. J Pak Med Assoc 61:1132–1133
Mohta A, Khurana N (2011) Fetus-in-fetu or well-differentiated teratoma—a continued controversy. Indian J Surg 73:372–374. https://doi.org/10.1007/s12262-011-0251-4
Parashari UC, Luthra G, Khanduri S, Bhadury S, Upadhyay D (2011) Diagnostic dilemma in a neglected case of fetus-in-fetu solved with Magnetic Resonance Imaging and MDCT—a case report and review of literature. J Radiol Case Rep 5:29–37. https://doi.org/10.3941/jrcr.v5i10.833
Savelli S, Antonello M, Pasquini L, Noccioli B, Fonda C (2011) A well-documented multimodality imaging approach to fetus in fetu: pre- and postnatal imaging features. Pediatr Radiol 41:1337–1341. https://doi.org/10.1007/s00247-011-2070-y
Bajaj AK, Dave N, Garasia MB (2011) A rare case of oral fetus in fetu with mandibular cleft and absent hyoid. Paediatr Anaesth 21:706–707. https://doi.org/10.1111/j.1460-9592.2011.03577.x
Sun J, VongPhet S, Zhang Z, Mo J (2012) Fetus-in-fetu: imaging and pathologic findings. Abdom Imaging 37:147–150. https://doi.org/10.1007/s00261-011-9757-2
Gan Y, Wu JH, Zhou J et al (2012) Abdominal fetus-in-fetu in a two-year-old boy. JRSM Short Rep 3:50. https://doi.org/10.1258/shorts.2012.011174
Peng B, Li D (2012) Fetus in fetu in the back. BMJ Case Rep. https://doi.org/10.1136/bcr.03.2012.6150
Mustafa G, Mirza B, Iqbal S, Sheikh A (2012) A case of fetus in fetu. APSP J Case Rep 3:9
Reddy RK, Kannaiyan L, Srirampur S et al (2012) Thoracic fetus in fetu. J Indian Assoc Pediatr Surg 17:178–179. https://doi.org/10.4103/0971-9261.102344
Sharma A, Goyal A, Sharma S (2012) Fetus in fetu: a rare case report. J Res Med Sci 17:491–494
Cingel V, Durdik S, Babala J, Polak S, Varga I (2012) Fetus in fetu from newborn’s mediastinum: case report and a review of literature. Surg Radiol Anat 34:197–202. https://doi.org/10.1007/s00276-011-0868-9
Sinha S, Sarin YK, Khurana N (2012) Prenatally diagnosed retroperitoneal fetus-in-fetu with ipsilateral testicular atrophy: a case report. J Neonatal Surg 1:41
Prakash A, Parelkar SV, Oak SN, Gupta RK, Sanghvi BV (2012) Giant epignathus with midline mandibular cleft: insights in embryology and management. Ann Maxillofac Surg 2:56–59. https://doi.org/10.4103/2231-0746.95322
Huddle LN, Fuller C, Powell T et al (2012) Intraventricular twin fetuses in fetu. J Neurosurg Pediatr 9:17–23. https://doi.org/10.3171/2011.10.PEDS11196
Has R, Kalelioglu IH, Esmer AC, Demirbas R, Yuksel A, Yavuz E (2013) Prenatal sonographic diagnosis of fetus in fetu. J Ultrasound Med 32:2212–2214. https://doi.org/10.7863/ultra.32.12.2212
Dutta HK, Thomas JK, Sahewala NK, Patgiri DK (2013) Fetus in fetu in a neonate: report of a case. Surg Today 43:547–549. https://doi.org/10.1007/s00595-012-0242-5
Kehal H, Billing S, Sharma BK, Mittal P (2013) Fetus-in-fetu: mimicking as teratoma on antenatal ultrasound. Indian J Surg 75:412–414. https://doi.org/10.1007/s12262-012-0745-8
Huang Y, Zhang Q, Feng JF, Liu J (2013) Fetus in fetu: a rare presentation. J Res Med Sci 18:924–925
Ji Y, Chen S, Zhong L et al (2014) Fetus in fetu: two case reports and literature review. BMC Pediatr 14:88. https://doi.org/10.1186/1471-2431-14-88
Maryńczak L, Adamek D, Drabik G, Kwiatkowski S, Herman-Sucharska I, Lankosz-Lauterbach J (2014) Fetus in fetu: a medical curiosity–considerations based upon an intracranially located case. Childs Nerv Syst 30:357–360. https://doi.org/10.1007/s00381-013-2191-8
Saikia P, Choudhury D, Kalita K (2014) Oro-facial dysmorphism with visible glossoepiglottic fold in a heteropagus: first description. Indian J Anaesth 58:223–224. https://doi.org/10.4103/0019-5049.130845
Ji Y, Song B, Chen S et al (2015) Fetus in fetu in the scrotal sac: case report and literature review. Medicine (Baltimore) 94:e1322. https://doi.org/10.1097/MD.0000000000001322
Pang KK, Chao NS, Tsang TK et al (2015) From observation to aetiology: a case report of a twin fetus-in-fetu and a revisit of the known rarity. Hong Kong Med J 21:80–83. https://doi.org/10.12809/hkmj133925
Padwal AD, Devi BI, Ramachandran S, Bhat DI, Shukla D, Ramu R (2016) Occipitocervical fetus in fetu with extracalvarial extension: a case report. Pediatr Neurosurg 51:87–92. https://doi.org/10.1159/000441035
Ullal S, Sanyal P, Kamath N, Rao S (2016) A Rare Case of Fetus within a Fetus. J Clin Diagn Res 10:TD03–TD04. https://doi.org/10.7860/JCDR/2016/19513.7804
Tiwari C, Shah H, Kumbhar V, Sandlas G, Jayaswal S (2016) Fetus in fetu: two cases and literature review. Dev Period Med 20:174–177
Song QY, Jiang XP, Jiang Y, Ning G, Yang TZ (2016) Fetus in fetu: from prenatal sonographic diagnosis to postnatal confirmation. Fetal Diagn Ther 39:158–160. https://doi.org/10.1159/000363056
Yaacob R, Zainal Mokhtar A, Abang Jamari D, Jaafar N (2017) The entrapped twin: a case of fetus-in-fetu. BMJ Case Rep. https://doi.org/10.1136/bcr-2017-220801
Enaud R, Lavrand F, Le Manh C, Rullier A, Lamireau T (2017) Intraperitoneal fetus in fetu: a rare cause of abdominal mass. J Pediatr Gastroenterol Nutr 65:e69. https://doi.org/10.1097/MPG.0000000000001027
Sewell EK, Massa-Buck B, Rubio EI et al (2017) Impact of prenatal diagnosis of fetus-in-fetu. J Neonatal Perinatal Med 10:333–338. https://doi.org/10.3233/NPM-16101
Denney JM, Stanley C, Armstrong LA, Marshall J, Settle B, Haldeman-Englert C (2017) Fetus in fetu in lieu of a sacrococcygeal teratoma: a case illuminating the utility of serial prenatal sonographic examinations in diagnosis. J Ultrasound Med 36:453–455. https://doi.org/10.7863/ultra.16.02040
Sitharama SA, Jindal B, Vuriti MK, Naredi BK, Krishnamurthy S, Subramania DB (2017) Fetus in fetu: case report and brief review of literature on embryologic origin, clinical presentation, imaging and differential diagnosis. Pol J Radiol 82:46–49. https://doi.org/10.12659/PJR.899956
Yu YR, Espinoza J, Mehta DK, Keswani SG, Lee TC (2018) Perinatal diagnosis and management of oropharyngeal fetus in fetu: a case report. J Clin Ultrasound 46:286–291. https://doi.org/10.1002/jcu.22528
Traisrisilp K, Srisupundit K, Suwansirikul S, Norasetthada T, Kosarat S, Tongsong T (2018) Intracranial fetus-in-fetu with numerous fully developed organs. J Clin Ultrasound 46:487–493. https://doi.org/10.1002/jcu.22566
Taher H, Abdellatif M, Wishahy A et al (2020) Fetus in fetu: lessons learned from a large multicenter cohort study. Eur J Pediatr Surg 30:343–349. https://doi.org/10.1055/s-0039-1698765
Harigovind D, Babu Sp H, Nair SV, Sangram N (2019) Fetus in fetu—a rare developmental anomaly. Radiol Case Rep 14:333–336. https://doi.org/10.1016/j.radcr.2018.11.020
Issa M (2019) Fetus in fetu: a rare case of intra-abdominal mass. Radiol Case Rep 14:1171–1174. https://doi.org/10.1016/j.radcr.2019.07.003
Kumar A, Paswan SS, Kumar B, Kumar P (2019) Fetus in fetu in an adult woman. BMJ Case Rep. https://doi.org/10.1136/bcr-2019-230835
Matsubara N, Akasaka Y, Kanagaki M, Okamoto S (2020) A case report of fetus in fetu with an aorta-like structure visualized by contrast-enhanced CT. Radiol Case Rep 15:2645–2648. https://doi.org/10.1016/j.radcr.2020.10.006
Wu Y, Jin X, Wu C, Wei G (2021) Cardiac arrest in infant due to giant fetus-in-fetu. Thorax 76(1):100–101. https://doi.org/10.1136/thoraxjnl-2020-215307
Sherbaf FG, Tavallaei N, Ghanbarinasab Z et al (2020) First case report of adnexal fetus in fetu. J Pediatr Adolesc Gynecol 33:745–747. https://doi.org/10.1016/j.jpag.2020.07.019
Surana A, Aggarwal A, Krishnan V, Malik A, Misra RN (2020) Intracranial fetus in fetu-a pediatric rarity. World Neurosurg 139:286–288. https://doi.org/10.1016/j.wneu.2020.03.156
Zhu K (2020) Prenatal and postnatal MRI imaging findings of intracranial parasitic fetus: a case report. Childs Nerv Syst. https://doi.org/10.1007/s00381-020-04891-1
Yan H, Liu J, Luo Y, Wu Y, Du L (2020) Preoperative diagnosis of a “humanoid” fetus in fetu using multimode ultrasound: a case report. BMC Pediatr 20:483. https://doi.org/10.1186/s12887-020-02389-y
Gupta DK, Lall A, Bajpai M (2001) Epigastric heteropagus twins—a report of four cases. Pediatr Surg Int 17:481–482. https://doi.org/10.1007/s003830000473
Spencer R (2001) Parasitic conjoined twins: external, internal (fetuses in fetu and teratomas), and detached (acardiacs). Clin Anat 14:428–444. https://doi.org/10.1002/ca.1079
Wang YZ, Cao LR, Cai CQ (2017) Accessory penis and scrotum in a male infant[J]. J Child Sci 1:E24–E26. https://doi.org/10.1055/s-0037-1604158
Kaufman MH (2004) The embryology of conjoined twins. Childs Nerv Syst 20:508–525. https://doi.org/10.1007/s00381-004-0985-4
Spencer R (2000) Theoretical and analytical embryology of conjoined twins: part I: embryogenesis. Clin Anat 13:36–53. https://doi.org/10.1002/(SICI)1098-2353(2000)13:1%3c36::AID-CA5%3e3.0.CO;2-3
Ratan SK, Rattan KN, Magu S, Gupta S, Narang R, Arora B (2008) Thoracopagus parasites in two sets of twins: evidence for the fusion theory. Pediatr Surg Int 24:1255–1259. https://doi.org/10.1007/s00383-008-2248-z
Logroño R, Garcia-Lithgow C, Harris C, Kent M, Meisner L (1997) Heteropagus conjoined twins due to fusion of two embryos: report and review. Am J Med Genet 73:239–243
Satter E, Tomita S (2008) A case report of an omphalopagus heteropagus (parasitic) twin. J Pediatr Surg 43:E37-39. https://doi.org/10.1016/j.jpedsurg.2008.01.071
Mahajan JK, Devendra K, Mainak D, Rao KL (2002) Asymmetric conjoined twins: atypical ischiopagus parasite. J Pediatr Surg 37:E33. https://doi.org/10.1053/jpsu.2002.35446
Kapur VK, Kulkarni MS, Shenoy MU (1997) Asymmetric conjoined twins. Pediatr Surg Int 12(4):308–309. https://doi.org/10.1007/BF01372158
Gul A, Aslan H, Ceylan Y (2004) Prenatal diagnosis of pygopagus tetrapus parasitic twin: case report. BMC Pregnancy Childbirth 4:13. https://doi.org/10.1186/1471-2393-4-13
Sharma G, Mobin SS, Lypka M, Urata M (2010) Heteropagus (parasitic) twins: a review. J Pediatr Surg 45:2454–2463. https://doi.org/10.1016/j.jpedsurg.2010.07.002
Tan A, Lee SL (2002) Prenatal diagnosis of parasitic twins using three-dimensional ultrasound: a case report. Ultrasound Obstet Gynecol 20:192–193. https://doi.org/10.1046/j.1469-0705.2002.00740.x
Johnson R, Weir P (2016) Separation of craniopagus twins. Camb Q Healthc Ethics 25:38–49. https://doi.org/10.1017/S0963180115000274
Acknowledgements
Not applicable.
Funding
This work was supported by the National Natural Science Foundation of China (grant number 81771589) and the Program of Tianjin Science and Technology Plan (grant number 18ZXDBSY00170).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. BH, XZ were responsible to design and conduct the study. Material preparation, data collection and analysis were performed by CG, LP, JC and YF. The first draft of the manuscript was written by XZ. CC was in charge of critical revisions and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial or proprietary interests in any material discussed in this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhi, X., Hu, B., Zhao, X. et al. A cohort of five cases with asymmetric conjoined twining and literature review. Pediatr Surg Int 38, 169–181 (2022). https://doi.org/10.1007/s00383-021-05006-w
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-021-05006-w