Abstract
Purpose
The diagnosis of Hirschsprung’s disease (HD) was revolutionized by the introduction of rectal suction biopsy (RSB), allowing specimens to be taken without general anesthesia on the ward or as an out-patient procedure. However, insufficient tissue samples are not uncommon, and subsequently histopathologists often remain reluctant to confirm the presence or absence of enteric ganglion cells merely on the basis of submucosal RSBs. The aim of this study was to evaluate the current usage of RSB in the diagnostic work-up of HD based on an international survey.
Methods
A 15-item questionnaire was distributed among participants and faculty members at the 21st International Meeting of the Pediatric Colorectal Society.
Results
Eighty-seven pediatric surgeons from 30 countries completed the anonymous survey (response rate 70.2 %), grouped into 68 (78.2 %) staff surgeons and 19 (21.8 %) trainees, with a median work experience of 18 years (range 2–45 years). Of these, 74 (85.1 %) use RSB in the diagnostic work-up of patients with suspected HD, whereas 13 (14.9 %) prefer open full-thickness biopsy under general anesthesia. In total, 47 (63.5 %) respondents perform ≥20 RSBs (range 3–100 RSBs) per year. Five different RSB instruments were reported, the most common ones being rbi2 (65.0 %), Solo-RBT (15.0 %) and multipurpose suction biopsy kit (8.3 %). Only 22 (29.7 %) of the respondents use a defined negative suction pressure, with a median of 10 mL air (range 6–25 mL air). The most proximal reported biopsy site was located at a median of 2 cm (range 1–15 cm) above the pectinate line and a median of 2 (range 1–5) specimens are routinely taken, mainly from the posterior rectal wall. Insufficient tissue samples with need for repeat RSB were encountered in a median of 10 % (range 0–40 %). Most frequently used staining methods for rectal biopsies are hematoxylin/eosin (75.9 %), acetylcholinesterase (73.6 %), and calretinin (33.3 %). Overall, 36 (48.6 %) respondents had experienced RSB-related complications, including self-limiting rectal blood loss (n = 28), persistent rectal bleeding requiring blood transfusion (n = 9) and rectal perforation requiring surgical intervention (n = 7).
Conclusions
Although RSB is considered to be today’s gold standard for the diagnosis of HD, many aspects of its current usage are lacking consensus. Therefore, a prospective multi-center study or larger global audit appears warranted to identify if the present survey reflects common surgical practice and to establish universal standards for RSB.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hirschsprung’s disease (HD), also known as congenital megacolon, is a recognized developmental disorder of the enteric nervous system, characterized by the absence of intrinsic ganglion cells in the myenteric and submucosal plexuses of the distal colon, causing severe constipation and functional obstruction shortly after birth. Hence, the most important diagnostic features of HD require the histological evaluation of adequate rectal wall biopsies, showing a combination of aganglionosis and hypertrophic nerve trunks [1, 2]. Sampling of the necessary mucosal and submucosal tissue was revolutionized in 1965, when Dobbins and Bill performed the first rectal suction biopsy (RSB) [3], a relatively simple and less traumatic procedure to exclude the diagnosis of HD. This technique was further refined by Noblett in 1969 with the introduction of a specific RSB tube, particularly for the use in neonates and young infants [4]. Today, RSB is widely considered to be the gold standard in the diagnostic work-up of patients with suspected HD [5], allowing specimens to be taken safely without general anesthesia on the ward or as an out-patient procedure. However, some histopathologists remain reluctant to confirm the presence or absence of enteric ganglion cells merely on the basis of RSBs [6], as it necessitates a thick specimen with enough submucosal tissue to be considered as adequate [7]. Consequently, insufficient tissue samples are not uncommon and often result in a diagnostic delay with need for repeat RSB or full-thickness rectal biopsy, causing considerable parental anxiety, prolonged hospital stay and increased costs [8]. Although very rare, serious RSB-related complications, such as massive rectal bleeding [9, 10] and bowel perforation [11], have been reported, which in turn add a further risk to the patient. Nevertheless, a recent systematic review on RSB has shown that it is a safe and accurate method for the diagnosis of HD [12], but it remains unclear whether these results actually reflect the contemporary surgical practice. Therefore, the purpose of the present study was to evaluate the current usage of RSB as a diagnostic tool for HD, focusing on individual techniques and experiences based on the results from an international survey.
Methods
Participants and questionnaire-based survey
Following approval from the local organizing committee, a 15-item questionnaire was distributed among all participants and faculty members at the 21st International Meeting of the Pediatric Colorectal Society (Dublin, June 15–17, 2014) and was anonymously collected at the registration desk on the last day of the conference. In the first section of the questionnaire, respondents were asked regarding their position, years of work experience and country of practice. The following part comprised questions specifically related to the usage of RSB, including the clinical setting (e.g. under general anesthesia or as an out-patient procedure) and average number of biopsies performed per year. Furthermore, respondents were questioned about their individual preferences, such as RSB instrument, precise location of biopsy site, average number of specimens routinely taken and application of any defined negative suction pressure. In addition, respondents were asked to estimate the percentage of encountered insufficient tissue samples (i.e. containing only mucosa) with need for repeat RSB and what histological/immunohistochemical staining methods are currently in use at their institution. In the last section of the questionnaire, respondents were questioned if they had experienced any RSB-related complications and how they managed these.
Data extraction, statistical analysis and geomapping
All answers were extracted into an electronic datasheet in a standardized manner. Any ambiguities were resolved by consensus of both authors. The resulting data set was analyzed according to the options for each question with the PASW Statistics 18.0 software application (SPSS Inc., Chicago, USA) using descriptive statistics (i.e. frequency or percentage of predefined subgroups). amMap (https://www.amcharts.com/visited_countries), a JavaScript-based online mapping library was used for creation of a colored world cartogram.
Results
General information and characteristics of respondents
Eighty-seven pediatric surgeons from 30 countries (Fig. 1) completed the questionnaire (response rate 70.2 %): 68 (78.2 %) were staff surgeons and 19 (21.8 %) were trainees, with a median work experience of 18 years (range 2–45 years). Eighty-two (94.3 %) of the respondents stated that they are currently working in a university-affiliated teaching hospital and five (5.7 %) in a district general hospital.
Usage of RSB and individual techniques
Of the 87 surveyed pediatric surgeons, 74 (85.1 %) use RSB in the diagnostic work-up of patients with suspected HD, whereas 13 (14.9 %) prefer open full-thickness biopsy under general anesthesia. In total, 47 (63.5 %) respondents perform ≥20 RSBs (range 3–100 RSBs) per year. Five different types of RSB instruments were reported, the most common ones being rbi2 (Aus systems Pty Ltd, Allenby Gardens, Australia) (65.0 %), Solo-RBT (SAMO Biomedica, Bologna, Italy) (15.0 %), and multipurpose suction biopsy kit (Quinton Instrument Company, Seattle, USA) (8.3 %), followed by SBT-100 (Medical Measurements Inc., Hackensack, USA) (6.7 %) and various self-made devices (5.0 %). Fourteen (18.9 %) respondents did not state which RSB instrument they have in use at their institution. Only 22 (29.7 %) of the respondents use a defined negative suction pressure, with a median of 10 mL air (range 6–25 mL air). The most proximal reported biopsy site was located at a median of 2 cm (range 1–15 cm) above the pectinate line, and a median of 2 (range 1–5) specimens is routinely taken, mainly from the posterior rectal wall. Sixteen (21.6 %) respondents mentioned that they had to change the RSB device at least once in their career, because it was either too old (n = 6), no longer sterilizable (n = 5) or did not work properly (n = 5).
Percentage of insufficient RSB specimens
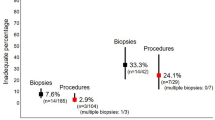
Insufficient tissue samples (i.e. containing only mucosa) with need for repeat RSB or open full-thickness biopsy was encountered in a median of 10 % (range 0–40 %).
Histological and immunohistochemical staining methods currently in use
Most frequently used staining methods for rectal biopsies are hematoxylin/eosin (75.9 %), acetylcholinesterase (73.6 %) and calretinin (33.3 %), whereas a few institutions have additional neuron-specific markers, such as NADPH-diaphorase (2.3 %) or lactate dehydrogenase (1.1 %) in use.
RSB-related complications
Overall, 36 (48.6 %) respondents had experienced one or more RSB-related complications, including self-limiting rectal blood loss (n = 28), persistent rectal bleeding requiring blood transfusion (n = 9) and rectal perforation requiring surgical intervention (n = 7). None of the respondents reported any sepsis or death after RSB.
Discussion
The definitive diagnosis of HD can be difficult, as it relies on histological demonstration of the complete absence of myenteric and submucosal ganglion cells in rectal biopsies from the distal colon. Prior to the introduction of RSB, open full-thickness biopsy was used to obtain specimens from newborn infants or young children that contain enough tissue (i.e. including muscle coats and submucosa) to make a relatively easy diagnosis of HD. Although the RSB technique made the procedure much less traumatic, allowing specimens be taken in a conscious patient without any form of speculum or proctoscope [13, 14], it made the diagnosis of HD much more difficult for histopathologists [15].
The present survey revealed that there are obvious discrepancies regarding the way rectal biopsies are currently obtained. The majority (85.1 %) of respondents stated that they prefer RSB in the diagnostic work-up of patients with suspected HD, whereas the remaining 14.9 % routinely perform an open full-thickness biopsy under general anesthesia. Interestingly, a recent systematic review [12] has reported that nearly 7.0 % of all RSBs are also carried out under general anesthesia (mainly due to the patient’s age), which in turn has implications for the clinical management. The location of the most proximal biopsy site varied widely, ranging between 1 and 15 cm above the pectinate line and mostly from the proximal rectal wall. A number of previous studies have demonstrated that the distal 1–2 cm of rectum is normally hypoganglionic, and thus sampling from this area may lead to a false impression of aganglionosis [16, 17]. Furthermore, there are significant differences regarding the number of specimens that are routinely taken, ranging from one to five samples per RSB. Several authors have recommended rectal biopsies from multiple levels (e.g. 1, 2, and 3 cm) to increase the likelihood that adequate tissue is obtained and to reduce the possibility that very short-segment disease may be missed [6, 18, 19]. Overall, the optimal biopsy site for RSB depends on the patient’s age at the time of procedure.
Although Campbell and Noblett [20] did not have any inadequate specimens in their series of 116 RSBs acquired from 45 patients of all ages, there is a recognized failure rate with many authors reporting high rates of insufficient biopsies [21–23], mainly because tissue is sampled blindly and some instruments were difficult to use. The respondents of the present survey stated that they have encountered inadequate tissue samples in up to 40 % of their cases with need for repeat RSB or open full-thickness biopsy. This high number of faulty procedures seems to be unacceptable in view of the relatively simple biopsy technique and the extensive amount of work load for the involved histopathologist. Pediatric surgeons should therefore only use RSB devices they are familiar with. The most important point for a successful RSB is an adequate and consistent suction level. Irrespective of whether a conventional syringe or wall/machine suction is used [24], it requires the appropriate technique of maintaining adequate suction and using a sharp, clean-cutting blade [10]. Surprisingly, less than 30 % of respondents use a defined negative suction pressure for obtaining a RSB, which can have a huge impact on the specimen size and patient safety. A manometric suction control guarantees not only the grasping of sufficient submucosal tissue, but also minimizes the risk of perforation. Suction efficacy can also be improved by gently approximating the capsule tip against the posterior rectal wall. It is not enough to insert the instrument into the rectum only. For accuracy and safety, some authors further recommend that RSB should be performed under direct vision after anal dilatation with the patient under general anesthesia [23, 25]. A further advantage of RSB being a relatively simple, but reliable procedure is that it can be easily taught also to trainees.
The International Working Group of the 2009 World Congress of Gastroenterology advocates that the diagnosis of HD requires a biopsy specimen of at least 3 mm diameter and a minimum of one-third of the sample should include submucosa to be considered adequate [7]. Interestingly, a recent study from the USA has demonstrated that tissue specimens obtained by RSB are significantly smaller than those obtained by full-thickness biopsy, especially in older children [26]. However, the authors also found that RSB and open rectal biopsy appear equivalent in their ability to provide sufficient submucosa [26]. When properly oriented and cut adequately 50–75 sections should be generally sufficient to exclude the presence of submucosal ganglion cells [6, 18]. This in turn requires an experienced histopathologist, who is familiar with the various staining methods used in the diagnostic work-up of HD, to avoid false-positive or false-negative results [27–29]. As a general rule, at least two formalin-fixed and paraffin-embedded samples are needed for routine histology with an additional specimen on saline wet gauze for enzyme immunohistochemistry of frozen sections. In 2006, Dutch researchers compared the accuracy of different tests for diagnosing HD and showed that RSB stained for acetylcholinesterase is the most accurate test for patients with suspected HD [30]. More recently, another systematic review has revealed that RSB provides in almost 90 % adequate tissue samples for the diagnosis of HD, with mean sensitivity and specificity rates of approximately 97 and 99 %, respectively [12]. On the other hand, it has been reported that RSB identifies HD in patients <39 days of age with a sensitivity of only 50 %, thus frequently leading to inconclusive results with need for additional biopsies [31]. In addition, RSB has been found to provide often inconsistent results in older infants and children, probably because their submucosal tissue is more fibrous [18], raising concern of a decreased diagnostic yield. However, with an adequacy of 90.5 % for patients >5 years in their series, Brady et al. [32] have proven that RSB is also effective in evaluating older children with suspected HD.
There have been a few reports on serious and potentially life-threatening adverse events associated with RSB [9–11, 21, 33, 34], but the overall number of complications in clinical practice appears to be very small. Immediately after the procedure, the patient may experience slight transient rectal bleeding, which usually settles spontaneously. Respondents of the present survey had experienced a total of nine incidents of persistent rectal bleeding requiring blood transfusion. Furthermore, rectal perforations requiring surgical intervention were reported by respondents in seven cases. Rectal perforations seem to be more probable in neonates and young infants, as demonstrated by the presence of portions of circular muscle layer in this age group compared with older patients. Rare, but devastating adverse events have been described in the literature, including injury to the common iliac artery [33], peripheral limb gangrene [34] and death [11]. None of the respondents in this survey had encountered any form of sepsis or fatality after RSB. Some authors have suggested performing RSB only in newborn infants with neonatal onset of symptoms [35, 36], whereas others also advocate this in patients that present later in life [37, 38]. A recent systematic review has indicated that the likelihood of RSB-related complications appears to be higher in newborns and young infants compared to older children [12]. In contrast, Keyzer-Dekker et al. [39] found that RSB can also be reliably and safely performed in preterm infants, which implies that there is no reason to postpone a RSB in these patients. Clearly, great care should always be taken, particularly when the procedure is performed in children younger than 1 year of age [40]. Moreover, it has been stated that the diagnosis of HD using RSB for acetylcholinesterase immunohistochemistry is only safe after 3 weeks of age [41]. Irrespective of the patient’s age, a thorough history and physical examination should be performed first to reliably identify patients unlikely to have HD, thus sparing them from an unnecessary RSB [35].
In conclusion, although RSB is considered to be today’s gold standard for the diagnosis of HD, many aspects of its current usage are lacking consensus. A prospective multi-center study or larger global audit, therefore appears to be warranted to identify if the present survey actually reflects common surgical practice and to establish universal standards for RSB.
References
Kapur RP (1999) Hirschsprung disease and other enteric dysganglionoses. Crit Rev Clin Lab Sci 36(3):225–273
Monforte-Muñoz H, Gonzalez-Gomez I, Rowland JM et al (1998) Increased submucosal nerve trunk caliber in aganglionosis: a “positive” and objective finding in suction biopsies and segmental resections in Hirschsprung’s disease. Arch Pathol Lab Med 122(8):721–725
Dobbins WO 3rd, Bill AH Jr (1965) Diagnosis of Hirschsprung’s disease excluded by rectal suction biopsy. N Engl J Med 272(13):990–993
Noblett HR (1969) A rectal suction biopsy tube for use in the diagnosis of Hirschsprung’s disease. J Pediatr Surg 4(4):406–409
Martucciello G, Pini Prato A, Puri P et al (2005) Controversies concerning diagnostic guidelines for anomalies of the enteric nervous system: a report from the fourth International Symposium on Hirschsprung’s disease and related neurocristopathies. J Pediatr Surg 40(10):1527–1531
Qualman SJ, Jaffe R, Bove KE et al (1999) Diagnosis of Hirschsprung disease using the rectal biopsy: multi-institutional survey. Pediatr Dev Pathol 2(6):588–596
Knowles CH, De Giorgio R, Kapur RP et al (2010) The London classification of gastrointestinal neuromuscular pathology: report on behalf of the Gastro 2009 International Working Group. Gut 59(7):882–887
Hall NJ, Kufeji D, Keshtgar A (2009) Out with the old and in with the new: a comparison of rectal suction biopsies with traditional and modern biopsy forceps. J Pediatr Surg 44(2):395–398
Dahshan A (2014) Serious rectal bleeding complicating suction rectal biopsy in a child. W V Med J 110(2):34–35
Pini-Prato A, Carlini C, Pesce F et al (2011) Massive bleeding after rectal suction biopsy: uncommon and unexpected delayed onset. World J Pediatr 7(1):83–85
Rees BI, Azmy A, Nigam M et al (1983) Complications of rectal suction biopsy. J Pediatr Surg 18(3):273–275
Friedmacher F, Puri P (2015) Rectal suction biopsy for the diagnosis of Hirschsprung’s disease: a systematic review of diagnostic accuracy and complications. Pediatr Surg Int 31(9):821–830
Kurer MH, Lawson JO, Pambakian H (1986) Suction biopsy in Hirschsprung’s disease. Arch Dis Child 61(1):83–84
Yunis EJ, Dibbins AW, Sherman FE (1976) Rectal suction biopsy in the diagnosis of Hirschsprung disease in infants. Arch Pathol Lab Med 100(6):329–333
Lake BD, Puri P, Nixon HH et al (1978) Hirschsprung’s disease: an appraisal of histochemically demonstrated acetylcholinesterase activity in suction rectal biopsy specimens as an aid to diagnosis. Arch Pathol Lab Med 102(5):244–247
Aldridge RT, Campbell PE (1968) Ganglion cell distribution in the normal rectum and anal canal. A basis for the diagnosis of Hirschsprung’s disease by anorectal biopsy. J Pediatr Surg 3(4):475–490
Venugopal S, Mancer K, Shandling B (1981) The validity of rectal biopsy in relation to morphology and distribution of ganglion cells. J Pediatr Surg 16(4):433–437
Kapur RR (2009) Practical pathology and genetics of Hirschsprung’s disease. Semin Pediatr Surg 18(4):212–223
Ohi RJ, Tseng SW, Kamiyama T et al (1990) Two-point rectal mucosal biopsy for selection of surgical treatment of Hirschsprung’s disease. J Pediatr Surg 25(5):527–530
Campbell PE, Noblett HR (1969) Experience with rectal suction biopsy in the diagnosis of Hirschsprung’s disease. J Pediatr Surg 4(4):410–415
Alizai NK, Batcup G, Dixon MF et al (1998) Rectal biopsy for Hirschsprung’s disease: what is the optimum method? Pediatr Surg Int 13(2–3):121–124
Athow AC, Filipe MI, Drake DP (1990) Problems and advantages of acetylcholinesterase histochemistry of rectal suction biopsies in the diagnosis of Hirschsprung’s disease. J Pediatr Surg 25(5):520–526
Kobayashi H, Li Z, Yamataka A et al (2002) Rectal biopsy: what is the optimal procedure? Pediatr Surg Int 18(8):753–756
Ali AE, Morecroft JA, Bowen JC et al (2006) Wall or machine suction rectal biopsy for Hirschsprung’s disease: a simple modified technique can improve the adequacy of biopsy. Pediatr Surg Int 22(8):681–682
Croffie JM, Davis MM, Faught PR et al (2007) At what age is a suction rectal biopsy less likely to provide adequate tissue for identification of ganglion cells? J Pediatr Gastroenterol Nutr 44(2):198–202
Muise ED, Hardee S, Morotti RA et al (2016) A comparison of suction and full-thickness rectal biopsy in children. J Surg Res 201(1):149–155
Martucciello G (2008) Hirschsprung’s disease, one of the most difficult diagnoses in pediatric surgery: a review of the problems from clinical practice to the bench. Eur J Pediatr Surg 18(3):140–149
Barshack I, Fridman E, Goldberg I et al (2004) The loss of calretinin expression indicates aganglionosis in Hirschsprung’s disease. J Clin Pathol 57(7):712–716
Meier-Ruge W, Lutterbeck PM, Herzog B et al (1972) Acetylcholinesterase activity in suction biopsies of the rectum in the diagnosis of Hirschsprung’s disease. J Pediatr Surg 7(1):11–17
de Lorijn F, Kremer LC, Reitsma JB et al (2006) Diagnostic tests in Hirschsprung disease: a systematic review. J Pediatr Gastroenterol Nutr 42(5):496–505
Meinds RJ, Kuiper GA, Parry K et al (2015) Infant’s age influences the accuracy of rectal suction biopsies for diagnosing of Hirschsprung’s disease. Clin Gastroenterol Hepatol 13(10):1801–1807
Brady AC, Saito JM, Lukas K et al (2016) Suction rectal biopsy yields adequate tissue in children. J Pediatr Surg 51(6):966–969
de Brito IA, Maksoud JG (1987) Evolution with age of the acetylcholinesterase activity in rectal suction biopsy in Hirschsprung’s disease. J Pediatr Surg 22(5):425–430
Jani BR, Brereton RJ, Dillon MJ (1989) Peripheral limb gangrene following rectal biopsy. Treatment with prostacyclin and exchange transfusion. Clin Pediatr (Phila) 28(12):585–588
Ghosh A, Griffiths DM (1998) Rectal biopsy in the investigation of constipation. Arch Dis Child 79(3):266–268
Lewis NA, Levitt MC, Zallen GS et al (2003) Diagnosing Hirschsprung’s disease: increasing the odds of a positive rectal biopsy result. J Pediatr Surg 38(3):412–416
Simpson BB, Ryan DP, Schnitzer JJ et al (1996) Surgical evaluation and management of refractory constipation in older children. J Pediatr Surg 31(8):1040–1042
Wheatley MJ, Wesley JR, Coran AG et al (1990) Hirschsprung’s disease in adolescents and adults. Dis Colon Rectum 33(7):622–629
Keyzer-Dekker CM, Sloots CE, Schokker-van Linschoten IK et al (2016) Effectiveness of rectal suction biopsy in diagnosing Hirschsprung disease. Eur J Pediatr Surg 26(1):100–105
Pini-Prato A, Martucciello G, Jasonni V (2006) Rectal suction biopsy in the diagnosis of intestinal dysganglionoses: 5-year experience with Solo-RBT in 389 patients. J Pediatr Surg 41(6):1043–1048
Goto S, Ikeda K, Toyohara T (1984) Histochemical confirmation of the acetylcholinesterase-activity in rectal suction biopsy from neonates with Hirschsprung’s disease. Z Kinderchir 39(4):246–249
Acknowledgments
The authors thank all the attendees and faculty members of the 21st International Meeting of the Pediatric Colorectal Society that participated in this survey.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that this research was conducted in the absence of any commercial or financial relationships that could be constructed as a potential conflict of interest.
Rights and permissions
About this article
Cite this article
Friedmacher, F., Puri, P. Current practice patterns of rectal suction biopsy in the diagnostic work-up of Hirschsprung’s disease: results from an international survey. Pediatr Surg Int 32, 717–722 (2016). https://doi.org/10.1007/s00383-016-3907-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00383-016-3907-0