Abstract
Purpose
We have diagnosed 35 cases of the supposedly rare condition metopic-sagittal synostosis in the past 20 years. Here, we introduce their clinical symptoms, neuroradiological findings, and surgical treatment methods, as well as discuss the relevant literature.
Methods
Subjects included 35 patients (33 boys and 2 girls; mean age 4.2 years; range 1–8 years). Magnetic resonance imaging (MRI) confirmed that there were no abnormal findings in the brain. Thirty patients presented with symptoms including speech delay, hyperactivity, autistic tendency, motor impairment, self-mutilation, and panic/temper tantrum behaviors. No other congenital malformation was observed, and all cases were considered to be the non-syndromic type. The final diagnosis was made using three-dimensional computed tomography (3D-CT) scans. The surgery was done the fronto-orbital advancement in addition to remove the large parts of sphenoid bones including sphenoid ridges at the skull base and trimmed the calvarium as necessary to reduce pressure.
Results
Surgical intervention improved clinical symptoms in nearly all 35 patients; cosmetic problems in patients with scaphocephaly were also corrected.
Conclusions
In the cases of child patients with metopic-sagittal synostosis who had clinical symptoms, surgical intervention improved such symptoms, suggesting its potential utility for metopic-sagittal synostosis with clinical symptoms. A surgical procedure focusing on the skull base was important for our successes. Based on the fact that metopic-sagittal synostosis was diagnosed in 35 patients at one institution over a relatively short period of time, this pathological condition may not be as rare as is currently believed.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
We have dealt with cases of mild trigonocephaly exhibiting various clinical symptoms since 1994 [20,21,22,23]; within these cases, we diagnosed cases where the metopic suture is fused with the sagittal suture, a phenomenon known as metopic-sagittal synostosis. This pathological condition is currently considered to be rare [3].
Currently, we have diagnosed 35 patients with metopic-sagittal synostosis. Patients presented with clinical symptoms that were improved to varying degrees following surgery in almost all cases.
No study of a large number of patients with this pathological condition has been reported, but the frequency of such patients being seen by our hospital suggests there may be many more cases than conventionally believed. Therefore, to improve the diagnosis and treatment of metopic-sagittal synostosis, we report here the analysis of clinical symptoms, neuroradiological diagnostic procedures, and surgical procedures and outcomes in our patients.
Methods
Subjects
The subjects were a total of 35 patients who were diagnosed with metopic-sagittal synostosis: 16 patients at Okinawa Prefectural Naha Hospital between November 1997 and March 2006 and 19 patients at Okinawa Prefectural Nanbu Medical Center/Nanbu Child Medical Center (formerly Okinawa Prefectural Naha Hospital) between April 2006 and January 2016. The subjects included 33 boys and 2 girls, and their ages ranged from 1 to 8 years (mean age = 4.2 years) (Fig. 1).
All the patients underwent chromosome testing, but no abnormal results were found. No other congenital malformation was observed, and all cases were considered to be the non-syndromic type.
In the family histories, two male siblings related to each of three patients and a female cousin of one patient had undergone surgery for mild trigonocephaly.
Clinical symptoms
All patients received a full clinical work-up that included neurological and psychiatric tests to assess behavior, cognition, and motor function and to detect the presence of specific or global developmental delays. In our assessments, we found no developmental delays in five patients. In 30 patients, we noted various symptoms; speech delay was the primary developmental delay. Speech delay and subsequent improvement were assessed according to the degrees of verbal capability established by the National Rehabilitation Center Sign-Significance Test (NRC S-S test) [12].
Diagnosis
A physical examination and visual inspection was used to initially diagnose the patients; subjects were inspected for ridges at the metopic and sagittal sutures either visually or by palpation. The diagnosis was confirmed by using a three-dimensional computed tomography (3D-CT) scan to confirm fusion of the metopic ridge and sagittal suture, as well as to visually confirm a sagittal ridge first identified by palpation. We used MRI to confirm that no congenital malformations or abnormal structural problems were present. Finally, we obtained plain skull X-rays before surgery in all patients to verify that the metopic and sagittal sutures were closed.
Monitoring of intracranial pressure
Intracranial pressure (ICP) was measured during surgery in 28 patients. After the scalp was flipped, a burr hole was prepared in the forehead and a sensor (Camino’s Monitoring System, Integra Lifesciences Corporation, NJ, USA) inserted into the epidural space. PCO2 was held at approximately 30 mmHg for the first measurement and 38–42 mmHg for the second, during which measurements were taken for several minutes.
Surgical procedure
Patients underwent one or both of the procedures described below, depending on their diagnoses. The anterior procedure was only performed in the patients with the non-scaphocephalic type; additionally, some fracture lines were introduced into the parietal bone (Fig. 2). The patients with the scaphocephalic type underwent both the anterior and posterior procedures.
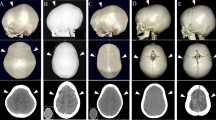
Surgical views and 3D-CT imaging findings immediately following surgery. a After removing the frontal calvarium and orbital bar. b The very wide sphenoid ridge (arrows) was removed up to the meningo-orbital band. c, d After the posterior craniectomies, pieces of bone were replaced in floating fashion, and the orbital bar was sutured to the lateral orbit. e The posterior craniectomies were done on the bilateral parietal bones. The most posterior points were several centimeters posterior to lambda. f 3D-CT imaging immediately following surgery for the scaphocephalic type. g 3D-CT imaging immediately following surgery for the non-scaphocephalic type
Anterior
We performed a bifrontal craniotomy in which the open-end was approximately 3 cm posterior to the coronal sutures. The frontal calvarium was extracted as one piece. Next, the lesser and greater wings of the sphenoid bone on both sides were partially removed, after which the sphenoid ridge was excised up to the meningo-orbital band. In most patients, the sphenoid ridge was very thin and wide and entered between the frontal and temporal lobes. Finally, the supraorbital bar with orbital roofs was removed as one piece. The extracted frontal calvarium was cut into four parts and returned to the forehead after trimming. The orbital bar was returned and fixed outside the orbit with 4–0 nylon suture.
Posterior
A linear craniectomy was performed by connecting four burr holes (bilateral parasagittal burr holes and bilateral burr holes made in the outermost part of the parietal bone) that were several centimeters posterior to lambda. The craniotomy was performed on both sides of the parietal bone, leaving the median bone of the sagittal suture. The extracted parietal bone was cut anteroposteriorly into three parts, and one part was returned. Three fracture lines were introduced in each side of the temporal bone. The returned parietal bone was allowed to float (Fig. 2).
The width of craniectomy approximately was 1–3 cm at the anterior part and 3–4 cm at the posterior part in patients under 4 years old. In 5 years of age or older, its width was narrower.
Results
Clinical assessment
Table 1 provides the clinical features of the patients and their outcomes (if known). Speech delay of various degrees was observed in 27 patients and described as follows: no meaningful word (n = 7), 20 words or fewer (n = 7), 100 words or more (n = 1), speaking in two-word sentences (n = 8), and speaking in three-word sentences (n = 4). Hyperactivity was observed in 15 patients and included excessive locomotion, inability to sit still, restlessness, and poor concentration. In 10 patients, autistic behavioral tendencies were observed, including reluctance to make eye contact, communication problems with other children, and spinning or other stereotypical movements. Seven patients had impaired motor function, primarily identified as being unable to walk by oneself or poor balance. Self-mutilation (mainly head banging) was observed in seven patients. Temper tantrums or panic were observed in eight patients. For example, one patient panicked and cried for over 30 min. Two patients presented with repetitive headache and vomiting. Cognitive regression was found in three patients, for whom previously spoken words or cognitive behaviors were lost over time.
Diagnostic assessments
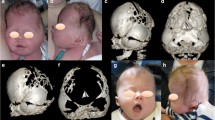
Twenty-seven patients were diagnosed with scaphocephaly by visual inspection (type 1); in eight of those patients, a sagittal ridge was observed by palpation. Eight patients were normocephalic (type 2) and lacked a sagittal ridge. A metopic ridge was observed by visual inspection or palpation in all 35 patients, as well as mild frontal bossing. Hollow temples were also noted in all patients (Fig. 3).
Two types of metopic-sagittal synostosis. a Type 1, scaphocephalic type. Upper column: with the sagittal ridge (eight cases). Lower column: without the ridge (19 cases). b Type 2, non-scaphocephalic (eight cases total; no sagittal ridges evident). A metopic ridge was confirmed in all patients. c The smaller anterior fossa, the very thin and wider sphenoid ridge (arrows), and marked digital markings
Definitive diagnoses were performed by 3D-CT scans, in which the fusion of the metopic ridge and sagittal suture was confirmed in all patients. The sagittal ridges observed by palpation were also shown by a 3D-CT scan. Intracranial 3D-CT imaging revealed narrowing of the anterior cranial fossa in almost all patients. The imaging of the internal table of the calvaria revealed digital impressions in the whole skull in 26 patients and in the parietal area or reaching up to the forehead in six patients (Table 1). With MRI, we found one patient whose cerebellar tonsil extended to the spinal canal; no other brain malformations or abnormalities were found.
We have recently attempted imaging the sphenoid ridge. The ridge is obviously thin, and the ability to observe it varies widely between patients (Fig. 3). In one case, we found that although the sagittal suture was evidently present at the age of 8 months, closure mostly occurred at the age of 1 year and 3 months, with complete closure at 1 year and 8 months (Fig. 4).
Measurement of ICP
The mean values of PCO2 and ICP for the first measurement were 31.2 and 8.3 mmHg, respectively, and those for the second were 39.2 and 19 mmHg, respectively. The second ICP measurements ranged from ≤10 mmHg (n = 1), 11–15 mmHg (n = 4), to ≥16 mmHg (n = 30) and are given in Table 1). The ICP values were high in the patients having digital impressions over 75% or more of the skull, except for one patient (4 mmHg). However, ICP values were also high in two patients whose digital impressions were limited only to the occipital and temporal lobes.
Results of the surgical procedure
No complications were observed in the 35 patients. Cosmetically, the forehead was widened and the frontal bossing eliminated in all patients. The degree of the hollow in the temples was also improved. All of the patients with type 1 scaphocephaly were normocephalic after surgery (Fig. 5). Since bone regeneration was so fast, in 2 years post-op. bone defect almost closed in all patients.
Clinical outcomes
As seen in Table 1, speech delays improved in 25 of 27 patients. Of seven patients who used no meaningful words before surgery, two patients have not shown any change as of the most recent follow-up, but one and two patients improved so as to speak in two- and three-word sentences, respectively, and two other patients were able to have daily conversations without difficulty. In the seven patients who spoke ≤20 words, improvements included a slight increase in the number of words (n = 1), speaking in two-word sentences (n = 2), speaking in three-word sentences (n = 1), and no difficulty having ordinary conversations (n = 3). One patient who initially spoke ≥100 words was subsequently able to have ordinary conversation without difficulty. Of the eight patients who had spoken in two-word sentences before surgery, two patients improved to speaking in three-word sentences and six patients became able to have ordinary conversations. All four patients who had spoken in three-word sentences or more became able to have ordinary conversations (Fig. 6).
Improvements were observed in all 15 patients with hyperactivity, 8 of 10 patients with autistic tendencies, all 7 patients with motor impairments, all 7 patients with self-mutilation behaviors, and all 8 patients with panic/temper tantrum symptoms. In the two patients having repetitive headache and vomiting, these symptoms improved immediately after surgery. Moreover, surgery improved a sleep disorder in one patient, enabling the reduction of the patient’s medicine for the disorder. Five patients whose development had been considered normal before surgery but who may have had minor speech delays and hyperactivity showed an increase in word expression and decrease in hyperactivity.
Improvements in clinical symptoms became evident within 3 months after surgery in most patients. There was no change in one patient who had undergone surgery at the age of 2 years and 4 months. This patient had speech delay (no meaningful words), high levels of hyperactivity, and autism-like symptoms before surgery. We were unable to contact two patients. Those patients whose symptoms improved soon after surgery have continued to thrive (Table 1).
In all patients, 3D-CT scans revealed the enlargement of the forehead and anterior cranial fossa. The type 1 patients (those with scaphocephaly) became normocephalic after surgery (Fig. 7). Scans demonstrated a decrease in digital impressions in all patients at 6 months after surgery, though this was not quantified. Bone regeneration allowed patients to experience daily life without difficulty by 1 to 1.5 years after surgery. MRI also showed the enlargement of the frontal lobe and the change was from the scaphocephalic to normocephalic skull (Fig. 8). Interestingly, the tonsillar herniation was observed before surgery in one patient disappeared after surgery (Fig. 8).
Changes induced by surgical intervention as identified by 3D computed tomography. In both a and b, the top row of images demonstrates the pre-operative condition, and the bottom row demonstrates the post-operative condition 6 months after surgery. a Scaphocephalic type (type 1). Note the enlargement of the frontal area and the anterior fossa, decrease of digital markings, and more normocephalic shape in the post-operative images. b Normocephalic type (type 2). Note the enlargement of the frontal area and the anterior fossa and decrease of digital markings in the post-operative images
Changes after surgery identified by MRI. a Note the scaphocephalic shape and smaller frontal lobes in prior to surgery. The normocephalic shape allowed for bigger frontal lobes, as seen in the post-operative MRI. b Note the finding of a tonsillar herniation (arrow) in this case prior to surgery (upper column), and the herniation completely disappeared after surgery (lower column)
Discussion
After cranioplasty in a child patient with mild trigonocephaly, language delay, and hyperactivity led to symptomatic improvement in 1994, we have reported that a number of clinical symptoms in patients with mild trigonocephaly improve with surgery [20,21,22,23]. Notably, modern imaging has increased our identification of closure of the metopic and sagittal sutures in suspected mild trigonocephaly, as well as the fused metopic suture in patients with suspected scaphocephaly. This pathology is described as metopic-sagittal synostosis, which is recognized as a rare disease [3, 7]. Chumas et al. [3] only reported seven cases as previously unclassified cases of craniosynostosis. In our cases, all patients were diagnosed at the age of 1 year and older rather than in infancy. Although the patients who were diagnosed with scaphocephaly had a particular head shape that could have been diagnosed in infancy, the diagnosis was delayed long enough for most patients to exhibit symptoms. Because of these conditions, the degree of recognition concerning general craniosynostosis may suffer.
We made definitive diagnoses after observing a ridge of the metopic suture, as well as the fused sagittal suture, with 3D-CT scans. The sagittal suture usually does not form a ridge in most patients who have a normocephalic or scaphocephalic shape; this fact presumably makes diagnosis without neuroimaging difficult. However, the condition can be easily diagnosed via 3D-CT scan, which is additionally considered the best diagnostic tool to capture basilar changes as well [5].
It is thought that despite the closure of the sagittal suture (see Fig. 3), which would potentially impart a scaphocephalic head shape, the gradual fusion of the sagittal suture after birth allows for development of a normocephalic head shape in some of these patients [16].
We feel that the findings of the forehead are consistent with mild trigonocephaly in all 35 patients; however, the narrowing of the forehead and anterior cranial fossa and the wide sphenoid ridge were considered to be abnormal findings.
In 32 patients, 3D-CT scans showed digital impressions in the area of ≥3/4 of the skull. In the cases with more digital impressions, the measured mean ICP value was high (≥10 mmHg). These results suggest the two phenomena may be positively correlated. In fact, there was a decrease in digital impressions after surgery, and in many of the patients for whom the impressions were decreased, ICP was also reduced. The tonsillar herniation in one patient, which was eliminated by the surgery, was further evidence of high ICP in at least some members of the group; it too was eliminated by the surgery. Thompson et al. [26] reported that overnight subdural intracranial pressure monitoring showed an increase in ICP relatively frequently in sagittal and metopic suture synostosis. Since most of their patients were ≤2 years old, and most of our patients were ≥2 years old, ICP may tend to increase as patients develop.
Concerning the surgical procedure for the anterior part, we used a procedure that had been performed for mild trigonocephaly, in which the upper orbit was extracted as a bar and shifted slightly anteriorward, the sphenoid bone was removed, and the abnormally broad sphenoid ridge was then also sufficiently removed. Some earlier studies pointed out the importance of an approach to the anterior cranial fossa in brachycephaly and plagiocephaly [1, 6, 13, 14, 17, 19], and we have followed this procedure. We performed osteotomy in the coronal section, allowing for a sufficient reduction in pressure. Fast bone regeneration is an interesting component of this pathological condition [13]. Indeed, we have had phenomena where bone regeneration occurred very early, even if a large osteotomy was performed in infancy. This phenomenon may be a cause of the recurrence of the pathology.
We selected this procedure in order to achieve a sufficient decrease in intracranial pressure and prevent the post-operative shape from regressing to the original shape. In fact, the patients with scaphocephaly became normocephalic after surgery, had no cosmetic problems, and had markedly reduced digital impressions suggestive of a decrease in intracranial pressure. Bone regeneration by 1 to 1.5 years after surgery had progressed well enough for patients to live their daily lives without difficulty.
Clinical symptoms had developed in 30 patients, including language delay, hyperactivity, autistic tendencies, motor impairment, self-mutilation, and panic/temper tantrum; all symptoms improved at high rates after surgery. In the patients showing improvement, some symptoms started to improve immediately after surgery and steadily improved thereafter. The enlargement of the forehead and anterior cranial fossa due to surgery caused the enlargement of the volume of the frontal lobe, which is considered to contribute to the reduction in symptoms. Moreover, an abnormally broad sphenoid ridge, which had been observed in our previous cases of mild trigonocephaly, was also seen in the patients of the present study. McCarthy et al. [14, 15] described this abnormal sphenoid ridge as a heavily overgrown sphenoid ridge and recommended excising it. Because decreased blood flow in the operculum has been associated with autism spectrum disorders in children [4], we believe that the reduction in autistic tendencies observed in our patients was caused by the decompression created by the removal of the sphenoid ridge contacting the operculum [23].
Since headache and vomiting, symptoms of high ICP, disappeared immediately after surgery, the elevated intracranial pressure was apparently improved after the decompressive cranioplasty. We believe the sleep disorder noted in one patient was also rescued by the surgical relief of the patient’s high ICP, given the published evidence of abnormally high ICP during sleep in cases of craniosynostosis [18, 26]. Faster development after surgery in the five seemingly normally developing patients (all scaphocephalic) was also presumably caused by the improvement in the poor intracranial environment.
Neurodevelopmental problems in various types of craniosynostosis have been mainly described by psychologists [2, 9, 11, 24, 25, 27]. Although the patients in the present study had at most minor changes in brain morphology, the narrowed forehead and elevated intracranial pressure are considered to have negatively affected the brain, thus producing the symptoms. Since Kapp-Simon et al. [10] indicated that even minor brain deformity can cause clinical symptoms after a long period of time; such symptoms in our cases would be expected.
Considering the above descriptions, definitive surgical indications for metopic and sagittal synostosis diagnosed based on 3D-CT scans should include any symptoms produced by synostosis-induced, long-standing morphological changes and the increased intracranial pressure suggested by the marked digital impressions.
Aside from our reports [20,21,22,23] and a study by Inagaki [8], no studies have shown that symptoms were improved by adding a surgical procedure to the therapeutic course. Thus, the evidence of reducing symptoms surgically is scant [25]. However, we believe that metopic-sagittal synostosis may be more common than once thought, so further evidence in support of decompression surgery may be forthcoming. Neurosurgeons dealing with various craniosynostosis must know that patients with these pathologies might develop not only cognitive impairment but also behavioral problems such as hyperactivity and autistic tendency. In this report, psychological tests were not applied. In the future, to evaluate these symptoms, developmental tests must be applied, a language assay, the child behavior checklist, and the childhood autism rating scale. Finally, we designate this pathological condition for type 1 as trigono-scaphocephaly.
References
Anderson FM (1981) Treatment of coronal and metopic synostosis: 107 cases. Neurosurgery 8:143–149
Bottero L, Lajeunie E, Arnard E, Marchac D, Renier D (1988) Functional outcome after surgery for trigonocephaly. Plast Reconstr Surg 102:952–958
Chumas PD, Cinalli G, Arnaud E, Marchac D, Renier D (1997) Classification of previously unclassified cases of craniosynostosis. J Neurosurg 86:177–181
Dapretto M, Davies MS, Pfeifer JH, Scott AA, Sigman M, Bookheimer SY, Iacoboni M (2006) Understanding emotions in others: mirror neuron dysfunction in children with autism spectrum disorders. Nat Neurosci 9:28–30
Genitori L, Lena GL, Dollo C, Choux M (1991–92) Skull base in trigonocephaly. Pediatr Neurosurg 17:175–181
Hoffman HJ, Hendrick EB (1979) Early neurosurgical repair in craniofacial dysmorphism. J Neurosurg 51:796–803
Inagaki T, Kyutoku S, Kawamoto K (2009) Study of showed a rare cranial form craniosynostosis cases (in Japanese). Nerv Syst Chidren 34:56–60
Inagaki T, Kyutoku S, Kawamoto K, Seno T, Kawaguchi T, Yamahara T, Oshige H, Yamanouchi Y, Kawamoto K (2007) The intracranial pressure of the patients with mild form of craniosynostosis. Childs Nerv Syst 23:1455–1459
Kapp-Simon KA (1998) Mental development and learning disorders in children with single suture craniosynostosis. Cleft Palate Craniofac J 35:197–203
Kapp-Simon KA, Speltz ML, Cunningham ML, Patel PK, Tomita T (2007) Neurodevelopment of children with single suture craniosynostosis; a review. Childs Nerv Syst 23:269–281
Kelleher MO, Murray DJ, McGillivray A, Kamel MH, Allcutt D, Earley MJ (2006) Behavioral, developmental, and educational problems in children with nonsyndromic trigonocephaly. J Neurosurg 105:382–384
Kodera T, Kurai N, Satake T (2010) National Rehabilitation Center Sign-Significance Test (S-S test) test manual, 4th edn. Escor. Co. Ltd., Chiba (in Japanese)
Marchac D (1978) Radical forehead remodeling for craniosynostosis. Plast Reconstr Surg 61:823–835
McCarthy JG, Coccaro PJ, Epstein F, Converse JM (1978) Early skeletal release in the infant with craniofacial dysostosis. Plast Reconstr Surg 62:335–346
McCarthy JG (1979) New concepts in the surgical treatment of the craniofacial synostosis in the infant. Clin Plast Surg 6:201–226
Morritt DG, Yeh FJ, Wall SA, Richards PG, Jayamohan J, Johnson D (2010) Management of isolated sagittal synostosis in the absence of scaphocephaly: a series of eight cases. Plast Reconstr Surg 126:572–580
Raimondi AJ, Gutierrez FA (1977) A new surgical approach to the treatment of coronal synostosis. J Neurosurg 46:210–214
Renier D, Sainte-Rose C, Marchac D, Hirsch JF (1982) Intracranial pressure in craniosynostosis. J Neurosurg 57:370–377
Seeger JF, Gabrielsen TO (1971) Premature closure of the frontosphenoidal suture in synostosis of the coronal suture. Radiology 101:631–635
Shimoji T, Shimabukuro S, Sugama S, Ochiai Y (2002) Mild trigonocephaly with clinical symptoms: analysis of surgical results in 65 patients. Childs Nerv Syst 18:215–224
Shimoji T, Tomiyama N (2004) Mild trigonocephaly and intracranial pressure: report of 56 patients. Childs Nerv Syst 20:749–756
Shimoji T, Shimoji K, Yamashiro K, Nagamine T, Kawakubo J (2009) Mild trigonocephaly—report of 300 operative cases. Nerv Sys Chidren 34:63–73
Shimoji T, Tominaga D, Shimoji K, Miyajima M, Tasato K (2015) Analysis of pre- and post-operative symptoms of patients with mild trigonocephaly using several developmental and psychological tests. Childs Nerv Syst 31:433–440
Sidoti EJ Jr, Marsh JF, Marty-Grames L, Noetzel MJ (1996) Long-term studies of metopic synostosis: frequency of cognitive impairment and behavioral disturbances. Plast Reconstr Surg 97:276–281
Speltz ML, Kapp-Simon KA, Cunningham M, Marsch J, Dawson G (2004) Single-suture craniosynostosis: a review of neurobehavioral research and theory. J Pediatr Psychol 29:651–668
Thompson DNP, Malcolm GP, Jones BM, Harkness WJ, Hayward RD (1995) Intracranial pressure in single-suture craniosynostosis. Pediatr Neurosurg 22:235–240
van der Meulen J (2012) Metopic synostosis. Childs Nerv Syst 28:1359–1367
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflict.
Rights and permissions
About this article
Cite this article
Shimoji, T., Kimura, T., Shimoji, K. et al. The metopic-sagittal craniosynostosis—report of 35 operative cases. Childs Nerv Syst 33, 1335–1348 (2017). https://doi.org/10.1007/s00381-017-3430-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-017-3430-1