Abstract
Background
Bladder perforation by ventriculoperitoneal shunt is a rare complication that has been describe in 19 cases in prior literature.
Case description
This work describes the case of a 4-month-old baby who presented with extrusion of the distal catheter through the urethra. The patient underwent a laparotomy; the catheter was cut close to the bladder wall and repositioned into the peritoneal cavity. The bladder wall was sutured, and the remaining distal portion of the catheter was removed through the urethra.
Discussion and conclusion
Based on this single experience and a literature review, the authors classified the clinical signs and symptoms of bladder perforation by the ventriculoperitoneal shunt catheter. Finally, the authors propose a more conservative approach for this rare complication.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Perforation of an abdominal viscera and/or extrusion of the distal catheter of ventriculoperitoneal (VP) shunts are rare complications that have been described in prior literature. Colonic perforation is less infrequent than bladder or stomach perforations. Catheter extrusions may occur through the anus, vagina, urethra, mouth, scrotal skin, or umbilicus [1, 2].
Based on a literature review, 19 reported cases of bladder perforation have been found so far. This rare complication has an unknown pathophysiological mechanism and diverse clinical manifestations. Because of its rare frequency, bladder lesion treatment and shunt management are not consensual. However, the majority of authors recommend the replacement of the shunt system and treatment with antibiotics [1, 3–19].
The present study reports on a patient whose peritoneal catheter was exteriorized through the urethra. The approach proved to be more conservative and successful. Based on this single experience and a literature review, a different type of management for this rare complication is proposed.
Background
Bladder perforation by the shunt catheter is a very rare complication. The authors of this study, therefore, did a literature review in PubMed, using the following keywords: bladder perforation and ventriculoperitoneal shunt. The references of the selected articles completed the research. Twenty articles were identified, but three were discarded (one case of iatrogenic bladder perforation, one case of ventriculovesical shunt, and another in which the bladder perforations were not related to the VP shunt). In total, 19 reported cases have been found in the literature so far. These 19 cases, plus the present study, are summarized in Tables 1 and 2 and compose the literature data used in the sections below [1, 3–19].
Clinical manifestation
This complication can be seen in both sexes and at all ages, and tends to be more common in children. However, this higher incidence is mostly related to the fact that the VP shunt is more prevalent at this age. The present case is the youngest patient in the literature.
The etiology of hydrocephalus has no relation to this complication. Hydrocephalus associated to spina bifida appears more frequently, but many other etiologies are also described. Some authors [2, 6, 7, 11] have reported that the bladder perforation may be associated with a higher volume within the bladder, but this feature was present in only five (25 %) of the patients. The present case had a normal bladder. The time interval between the last shunt surgery and the occurrence of the complication is quite different in the literature, varying from 2 months (the present case) to 8 years. Bladder perforation occurred after the first shunt insertion in four out of the nine patients whose data were available. In three cases, many revisions have been reported. Thus, it can be speculated that previous surgeries do not increase the chance of this complication.
According to the collected data, it can be concluded that bladder perforation is a rare, unpredictable, and unavoidable event. However, once it occurs, what would be its natural evolution? How might it appear, so that doctors can suspect and diagnose it?
Inside the bladder, the catheter may stay for a given period and/or exteriorize through the urethra. Catheter extrusion was present in 12 cases (60 %). Based on the literature review, it is not possible to conclude the exact time interval between the moment the catheter perforated the bladder wall and when the diagnosis is established. Four articles reported the presence of calculus inside the bladder, at least two of which were large. Considering the fact that the formation of calculus is a long-term process, one can speculate that the catheter may stay assymptomatic inside the bladder for a long period before being diagnosed. Ten (50 %) patients reported one or more of the following signs and symptoms, not necessarily related to urinary infection: dysuria, urgency, frequency, urinary retention, hematuria, and abdominal pain. How long these signs and symptoms lasted was poorly reported. Some articles mentioned a few days, but one [15] reported that they lasted for 3 months. It can therefore be speculated that these signs and symptoms may be related to the presence of the catheter inside the bladder. If the perforation process is painful or if it can appear in some specific way is difficult to pinpoint. However, these reactions seem to be silent, given that six patients (half of those with exteriorized catheters) presented a urethral extrusion of the catheter with no other symptoms. It is not possible to say how long the catheter stayed inside the bladder before its exteriorization.
Meningitis was observed in two of the 20 (10 %) reported cases. Upon admission to the hospital, both patients showed signs and symptoms of central nervous system infection. They presented no urinary infection, but the catheter had extruded through the urethra in both cases. The articles do not describe how long the catheter had been in contact with the exterior before the diagnosis. This could be important information, because it can be speculated that the longer the exposure time, the higher the chance of an ascending infection. On the other hand, considering that the tubing exteriorization is quickly noticed and most likely causes some degree of distress, one believe that all of the patients sought medical assistance on the same day of externalization, as occurred in the present case. An abdominal abscess was detected in one patient, who also had a urinary infection. Urinary infection was present in another two cases. In summary, urinary infection was present in three cases, none of which had catheter exteriorization or meningitis. Based on this information, one may suppose that the pathophysiological mechanism for shunt infection is ascending germs after external catheter contact. When the tubing remains inside the bladder, the chance of shunt infection is unlikely, particularly because this cavity is sterile.
Diagnosis
Based on data from the literature and the present case, one can divide the clinical manifestation of bladder perforation by VP shunt into two main categories: (1) exteriorization of the peritoneal catheter; and (2) urinary symptoms: dysuria, frequency and urgency, urinary retention, and lower abdominal pain. For those patients in the first category, the diagnosis is straightforward. For patients harboring VP shunts that present with urinary symptoms, bladder perforation should be considered as a differential diagnosis, and an ultrasonography should be performed. It is important to stress that no patient presented a shunt dysfunction.
Management and outcome
The management of this complication consists of (1) treatment of the bladder lesion and (2) management of the VP shunt. The surgical approach was performed by laparotomy in six cases and cystoscopy in five cases. In three cases, the peritoneal catheter was cut at its subcutaneous passage on the thorax, and the distal portion was removed through the urethra. In six reports, these data were missing. The bladder was repaired in eight cases, but not repaired in another eight patients. In four cases, this information was not available. The four cases presenting intravesical calculus were treated by cystoscopy in order to fragment the stone and remove the catheter. However, in one of these cases, the authors failed and had to switch to a laparotomy, perform resection and repair the bladder wall. The cystoscopic approach seems to be less invasive and, consequently, more attractive. However, this does not allow for bladder wall repair. No author reported urinary fistula or the presence of urine inside the peritoneal cavity. During the surgery of the present case, it was noted that the bladder orifice was exactly the same as that of the catheter. Considering that the bladder wall is mainly composed of muscular fibers and if one keeps the bladder empty during the postoperative period by inserting a Foley catheter, one could imagine that the perforation can close spontaneously. On the other hand, one can also suppose that the trajectory through the bladder can be epithelized and not amenable for a proper sealing. Moreover, tractioning the tubing and cutting it close to the inner surface of the bladder would not allow the surgeon to be sure of the exact position of the remaining catheter inside the abdomen. Finally, literature on bladder trauma recommends the suturing of the bladder wall in cases of intraperitoneal lesions [20]. Analysis from the National Trauma Data Bank concluded that the mortality with or without surgical repair was 2.4 and 7.7 %, respectively. Compared with no repair, surgical suturing was associated with improved survival in both closed and open injuries [21]. Considering that presented above, the authors propose two kinds of approaches according to the clinical presentation. (1) In case of catheter exteriorization through the urethra (majority of patients), we recommend a median suprapubic laparotomy, an inventory of the abdomen cavity, slight catheter traction, tube cutting close to the bladder, its repositioning inside the peritoneal cavity, and bladder repair. (2) In case of catheter retention inside the bladder, the authors recommend a cystoscopic approach in order to remove the catheter, cutting the tube close to the inner wall of the bladder, and the fragmentation of any apparent calculi. Whatever the approach, it should be followed by the insertion of a Foley catheter. The time period for vesical catheterization ranged from 5 to 15 days in the literature, but the majority of authors maintained it for 5 days. The authors believe this short period is enough to allow the bladder wall to heal and would not increase the chance of urinary tract infection or prolong the patient’s hospital stay. Contrary to all other authors who reported the use and length of antibiotic administration, we opted to administer antibiotics for 24 h as a prophylatic, as is common in any other ordinary surgery. The authors suggest a longer use antibiotics only in cases of infection and, considering its site (urinary, meningeal, or abdomen) and germ, to determine the specific drug to be used and its length of use.
Regarding the management of the shunt, the present case followed the simplest approach when compared to the other authors. The hardware was not removed, and the proximal components were not touched. The literature review showed the removal of nine shunts, four externalizations or implantations of external ventricular drainage, and two cases of shunt revision. Analyzing the 20 cases from the literature (including the present one), only two cases of shunt infection were found, and in both, the patients presented signs and symptoms of central nervous system infection. Based on these data, the authors propose, in the case of bladder perforation, that if the patient shows no sign or symptom of shunt infection, then the surgeon should cut the peritoneal catheter close to the bladder perforation and redirect the catheter tip in the abdominal cavity, leaving the entire system in place. At the same time, the surgeon should collect CSF samples for biochemical analysis and culture. If the CSF shows any infection, use the appropriate course of antibiotics and change all the hardware afterwards. If the patient presents a shunt infection, exteriorize the distal catheter after cutting it close the bladder, with future replacement of the entire hardware when the infectious process has ended.
Exemplary case
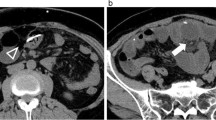
A 4-month-old girl was brought by her mother to the Emergency Ward of Hospital das Clínicas at the Federal University of Minas Gerais. That morning, the mother had noted the extrusion of the VS shunt catheter through the baby’s urethra. According to the mother, the child was irritable and had not been sleeping well for the last few days. She was a pre-term baby that had been shunted at the age of 2 months due to congenital hydrocephalus. The postoperative course had proven uneventful. Upon physical examination, the baby was alert, reactive, with no focal deficit. No meningeal signs or fever was observed. The head’s circumference and the anterior fontanel were normal. The abdomen was soft and insensitive upon palpation, with normal peristaltic sounds. A 10-cm-length catheter was protruding through the urethra. A computerized tomography scan of the head revealed an optimally positioned ventricular catheter inside a normal sized ventricular system. Radiographies of the skull, thorax, and abdomen showed a normal positioning of the shunt system. The distal catheter crossed the pelvis, reaching the proximal third of the thigh.
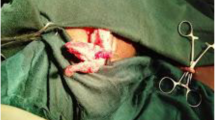
After having obtained the parents’ consent and under general anesthesia, the child underwent an infraumbilical, median laparotomy. The cavity inventory showed no liquid or signs of peritonitis; however, the surgeons could see the site where the peritoneal catheter had penetrated the bladder wall (Fig. 1). No urinary fistula could be observed. A few centimeters of the catheter was removed and cut close to the bladder wall. A CSF sample was collected, and the catheter tip was repositioned inside the peritoneal cavity. The remaining distal portion of the catheter was removed through the urethra, and the bladder wall was closed in two layers by absorbable suture. The abdomen wall was closed, and a Foley catheter was inserted. Cefazolin was administered for 24 h. The outcome of the CSF analysis was normal. The mother reported that the child slept better after the procedure. The Foley catheter was removed on the seventh postoperative day, and the child was discharged from the hospital the following day. The two-month follow-up period has shown no abnormalities.
Conclusion
Bladder perforation by VP shunt catheter is a rare, unpredictable, and unavoidable event. It may appear in two forms. The first and most common is catheter extrusion through the urethra, making the diagnosis straightforward. The second is by urinary signs and symptoms, inducing the physician to consider this complication as a differential diagnosis in this clinical scenario. The confirmatory diagnosis can be established by an abdominal ultrasound. Associated shunt infection is uncommon. Its treatment should be developed according to the infection’s clinical presentation. If catheter extrusion is present, the authors propose the performance of a laparotomy, cutting the catheter close to the bladder perforation site, followed by the redirectioning of the catheter tip and suture of the bladder wall. The remaining portion of the catheter should be removed at the urethra exit. If the catheter is retained inside the bladder, the approach should be cystoscopic. Exteriorization of the shunt should be reserved for uncommon cases of infection. The time course of antibiotics is determined by the presence and type of infection; otherwise, it should be administered as a prophylactic for 24 h only.
References
Grosfeld JL, Cooney DR, Smith J, Campbell RL (1974) Intra-abdominal complications following ventriculoperitoneal shunt procedures. Pediatrics 54:791–796
Yilmaz MB, Egemen E, Tonge M, Kaymaz M (2013) Transoral protrusion of a peritoneal catheter due to gastric perforation 10 years after a ventriculoperitoneal shunting: case report and review of the literature. Turkish Neuorsurgery 23:285–288
Binning MJ, Ragel BT, Walker ML, Kestle JRW (2006) Retained peritoneal shunt tubbing causing hematuria. Case ilustration. J Neurosurg Pediatr 104:434
Butler L, Keys C, Lam JPH (2013) Bladder calculus formation on the tip of a migrated disused ventriculoperitoneal shunt. J Pediatr Surg 48:E1–E3
Chen TH, Lin MS, Kung WM, Hung KS, Chiang YH, Chen CH (2011) Combined ventriculoperitoneal shunt blockage, viscus perforation and migration into urethra, presenting with repeated urinary tract infection. Ann R Coll Surg Engl 93:e151–e153
De Aguiar GB, Mizrahi C, Aquino JHW, Tavares CM, Telles C, Nigri F, et al. (2011) Urethral extrusion of a peritoenal catheter in a patient with neobladder: a rare complication of shunt insertion. Neuropediatrics 42:124–127
De Jong L, Van Der AAF, De Ridder D, Van Calenbergh F (2011) Extrusion of ventriculoperitoneal shunt catheter through an appendicovesicostomy. Br J Neurosurg 25:115–116
Eichel L, Allende R, Mevorach RA, Hulbert WC, Rabinowitz R (2002) Bladder calculus formation and urinary retention secondary to perforation of a normal bladder by a ventriculoperitoneal shunt. Urology 60:344iv–3344v
Ibrahim AK (2014) Urinary bladder stone complicating ventriculovesical shunt. Sultan Qaboos Univ Med 14:e142–e144
Kataria R, Sinha VD, Chopra S, Gupta A, Vyas N (2013) ) Urinary bladder perforation, intra-corporeal knotting and per-urethral extrusion of ventriculoperitoneal shunt in a single patient: case report and review of literature. Childs Nerv Syst 29:693–697
Mevorach RA, Hulbert WC, Merguerian PA, Rabinowitz R (1992) Perforation and intravesical erosion of a ventriculoperitoneal shunt in a child with an augmentation cystoplasty. J Urol 147:433–434
Mihajlovic M, Tasic G, Raicevic M, Mrdak M, Petrovic B, Radlovic V (2012) Asymptomatic perforation of large bowel and urinary bladder as a complication of ventriculoperitoenal shunt: report of two cases. Srp Arh Celok Led 140:211–215
Pohlman GD, Wilcox DT, Hankinson TC (2011) Erosive bladder perforation as a complication of ventriculoperitoneal shunt with extrusion from the urethral meatus: case report and literature review. Pediatr Neurosurg 47:223–226
Prasad VSSV, Krishna AM, Gupta PK (1995) Extrusion of peritoneal catheter of ventriculoperitoneal shunt through the urethra. Br J Neurosurg 95:209–210
Ramana Murthy KV, Jayaram Reddy S, Prasad DVSRK (2009) Perforation of the distal end of the ventriculoperitoenal shunt into the bladder with calculus formation. Pediatr Neurosurg 45:53–55
Surchev J, Georgiev K, Enchev Y, Avramov R (2002) Extremely rare complications in cerebrospinal fluid shunt operations. J Neurosurg Sci 46:100–102
Ueda Y, Kakino S, Hashmoto O, Imoto K (1998) Perforation of the bladder by a peritoenal catheter: an unusual late complication of ventriculo-peritoneal shunt. No Shinkei Geka 26:413–416
Yazar U, Kanat A, Akca N, Gazioglu G, Arda I, Kazdal H (2012) Urethral protrusion of the abdominal catheter of ventriculoperitoenal shunt: case report of extremely rare complication. J Pediatr Neurosci 7:111–113
Yerkes EB, Rink RC, Cain MP, Luerssen TG, Casale AJ (2001) Shunt infection and malfuntion after augmentation cystoplasty. J Urol 165:2262–2264
Gomez RG, Ceballos L, Coburn M, Corriere Jr JN, Dixon CM, Lobel B, McAninch J (2004) Consensus on genitourinary trauma. BJU Int 94:27–32
Deibert CM, Glassberg KI, Spencer BA (2012) Repair of pediatric bladder rupture improves survival: results from the National Trauma Data Bank. J Pediatr Surg 47:1677–1681
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Rights and permissions
About this article
Cite this article
Miranda, M.E., de Sousa, M.B., Tatsuo, E.S. et al. Bladder perforation by ventriculoperitoneal shunt. Childs Nerv Syst 32, 2321–2326 (2016). https://doi.org/10.1007/s00381-016-3202-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00381-016-3202-3