Abstract
The relationship between activation of the sympathetic nervous system (SNS) and improvement of left ventricular (LV) function and how this correlates with clinical outcomes are not fully explored in Takotsubo syndrome (TS). The purpose of this study is to evaluate the relationship between activation of the SNS and LV function improvement and how this correlates with clinical outcomes in TS. Patients with TS were retrospectively identified. Patients were divided into two groups according to the timing of LV function improvement: < 1 month (S group) and ≥ 1 month (L group). Activation of the SNS was assessed by plasma catecholamine measurement and Iodine-123 meta-iodobenzylguanidine (I123-MIBG) scintigraphy. In-hospital complications included heart failure, cardiogenic shock, the use of invasive or noninvasive ventilation, life-threatening arrhythmia, cerebrovascular event and all-cause death. A total of 90 patients with TS were enrolled. Of these, 39 patients were in the S group and 51 in the L group. There were no significant differences between the two groups in clinical demographics. The L group was characterized by enhanced SNS activation, including higher levels of catecholamines and lower late heart–mediastinum ratio followed by higher washout rate in I123-MIBG scintigraphy, compared with the S group. In-hospital complications were increased in the L group (56% vs. 33.3%, p = 0.03), including higher rates of heart failure (45% vs. 23%, p = 0.03) and in-hospital death (8.0% vs. 0%, p = 0.03). In patients with TS, high activity of the SNS was observed in patients with delayed LV function recovery, which was associated with in-hospital adverse outcomes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Takotsubo syndrome (TS) is characterized by chest pain, electrocardiographic changes and transient left ventricular (LV) dysfunction with apical and mid-ventricular segments akinesis or dyskinesis in the absence of obstructive coronary artery disease which mimics acute coronary syndromes [1,2,3,4,5]. Patients with TS show significant improvement of systolic function within a week and achieve complete recovery by a couple of months after onset [6]. Although the underlying pathophysiological mechanism of TS is not well defined, several studies suggest that TS may be caused by an excess of catecholamines [7]. In the acute phase of TS, iodine-123 meta-iodobenzylguanidine (I123-MIBG) scintigraphy shows impaired apical myocardial uptake of I123-MIBG on planar images, which correlate with the impaired LV segments [8]. Interestingly, some parameters reflecting augmented sympathetic nerves system (SNS) activation, such as increased plasma catecholamine levels and decreased uptake of I123-MIBG in affected LV segments, have been observed in the acute phase of TS, whereas improvement findings (reduced plasma catecholamine levels and heightened uptake of I123-MIBG) have been found 3 months after the onset of TS [9].
TS generally has favorable outcomes, but some patients are prone to have severe in-hospital complications, including heart failure, cardiogenic shock, or life-threatening arrhythmias [10]. Recently, several studies have investigated the relationship between clinical factors and clinical outcomes [11, 12]. Some studies have reported that male gender and physical stress were predictors of in-hospital complications and all-cause mortality in patients with TS [13]. However, the relationship between SNS and recovery of LV function and how this correlates with clinical outcomes have not been fully explored. Therefore, the aim of this study was to investigate the impact of SNS activity on LV function improvement and its association with clinical outcomes in patients with TS.
Methods
Study population and study design
Patients with TS diagnosed using both echocardiography and coronary angiography at Kindai University Hospital were retrospectively identified from April 2001 to March 2018. Of these, patients who had serial echocardiographic follow-up were included in this study. The diagnosis of TS was based on the following Mayo Clinic criteria [14]:
-
1.
transient hypokinesis, akinesis or dyskinesis of the LV midsegment with or without apical involvement; the regional wall motion abnormalities extending beyond a single epicardial vascular distribution; a stressful trigger often but not always present;
-
2.
the absence of obstructive coronary disease or angiographic evidence of acute plaque rupture;
-
3.
new electrocardiographic abnormalities (either ST-segment elevation and/or T-wave inversion) or modest elevation in cardiac troponin; and
-
4.
the absence of pheochromocytoma or myocarditis.
All patients underwent echocardiography, electrocardiogram (ECG) and invasive/non-invasive coronary angiography, which were reviewed by two experienced independent cardiologists to evaluate the diagnosis of TS. The majority of patients underwent I123-MIBG scintigraphy during the acute phase to assess adrenergic activity.
During the clinical course, LV function was assessed by echocardiography using the American Heart Association 17-segment model. In each segment, wall motion was classified into normokinesia, hypokinesis, akinesia and dyskinesia. Echocardiogram was performed at approximately 1-week interval until 4 weeks, and after that, it was performed at approximately 1-month interval until LV wall motion was completely normalized for each segment [15]. LV function improvement was defined when LV wall motion was completely normalized or back to the baseline for each segment. Patients were divided into two groups according to the timing of LV function improvement: < 1 month (S group) and ≥ 1 month (L group).
This study was conducted in compliance with the Declaration of Helsinki with regard to investigations in human subjects, and the study protocol was approved by the institutional review board of the Kindai University Faculty of Medicine.
Blood sample collection
Blood samples were collected on admission for assessment of clinical hematology and biochemistry including brain natriuretic peptide (BNP), cardiac enzymes and plasma catecholamine levels. Cardiac enzymes including creatine kinase (CK), CK-MB, troponin I were measured at 3-h intervals until peak levels were reached. Plasma concentration of dopamine, epinephrine, and norepinephrine was measured under resting condition. The normal reference values for plasma epinephrine, norepinephrine, and dopamine were < 100 pg/ml, < 450 pg/ml and < 20 pg/ml.
I123-MIBG scintigraphy
MIBG scintigraphy was carried out as soon as possible after admission. For each patient, heart–mediastinum ratio (H/M) and washout rate (WR) were calculated. Patients were given 50-mg potassium iodine after breakfast of the examination day to block thyroid iodine uptake. Planar anterior images of the chest were obtained 15 and 240 min after injection of 111 MBq of MIBG. The following systems were used; a Philips Bright View gamma camera; a Cardiac High Resolution collimator; a 256 × 256 matrix; and an acquisition time of 180 s. Image interpretation was done using the smartMIBG (FUJIFILM RI Pharma Co, Ltd.) [16].
Clinical outcomes
In-hospital events including acute heart failure, use of invasive/non-invasive ventilation, cardiogenic shock, life-threatening arrhythmia, cerebrovascular event, and in-hospital death were assessed. An acute heart failure was defined by the presence of pulmonary edema in a patient requiring oxygen therapy, while cardiogenic shock was diagnosed in a patient with sustained systolic pressure < 90 mmHg. Life-threatening arrhythmia was defined as any ventricular arrhythmia that resulted in syncope or hypotension and required medical intervention. Long-term mortality was assessed based on chart review and/or telephone interview.
Statistical analysis
Continuous variables were analyzed for a normal distribution with the Kolmogorov–Smirnov test and presented as mean ± standard deviation (SD) or as median and interquartile range (IQR) if a normal distribution was present or not, respectively. Student’s t test or Mann–Whitney’s U test were used for comparisons of continuous variables where appropriate. Categorical variables are expressed as frequencies and percentages. Categorical variables were tested by means of the Chi-square test or Fisher’s exact test. Control for potential confounders and analysis of independent correlates of LV function improvement ≥ 1 month and incidence of in-hospital complications were performed with a logistic regression model. Odds ratio (OR) and 95% confidence interval (CI) were calculated. All univariate variables p < 0.1 and those deemed of clinical interest were included in the statistical model.
The log-rank test was used to compare the survival curves between the two patient groups classified the duration of LV function improvement: < 1 month (S group) and ≥ 1 month (L group). Factors with p < 0.1 on univariate analysis were entered into the Cox multivariate regression to define independent risk factors for the outcome. A two-tailed p value of less than 0.05 was considered to indicate a statistically significant difference for all the analyses performed. Statistical analysis was performed using JMPv13.0 software (SAS Institute Inc., Cary, NC, USA).
Results
Study population
From April 2001 to March 2018, a total of 95 TS patients were admitted to our hospital. Of these, five patients were excluded due to lack of echocardiographic follow-up. Therefore, a total of 90 patients were included in this study. The baseline clinical characteristics are shown in Table 1. The average age was 76.5 ± 8.9 years and most patients were females. The most common symptom and trigger were chest pain and physical stress. I123-MIBG scintigraphy was performed in 64 patients during the acute phase (7.3 ± 4.1 days after onset). The mean timing of LV function improvement was 4.2 ± 3.8 weeks.
Clinical characteristics and laboratory/scintigraphy findings related to the duration of LV function improvement
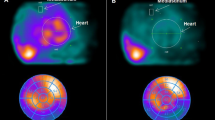
According to the timing of LV function improvement, 39 and 51 patients were in the S and L group, respectively. There were no significant differences in clinical demographics including age, gender, the presence of coronary risk factors, ECG feature, type of trigger, type of symptoms and ejection fraction (EF) on admission (Table 1). Figure 1 illustrates representative images of I123-MIBG scintigrams of two patients according to timing of LV function improvement.
Representative images of I123-MIBG scintigrams according to the timing of left ventricular function improvement. Examples of a patient with Takotsubo syndrome (TS) according to the timing of LV function improvement: < 1 month (S group) and ≥ 1 month (L group) with images of the iodine-123 meta-iodobenzylguanidine (I123-MIBG) scintigrams, as well as data for left ventricular ejection fraction (LVEF), late heart–mediastinum ratio (H/M late), washout rate, and plasma epinephrine, norepinephrine and dopamine levels
Regarding the laboratory findings, there were significantly higher levels of troponin I (5.4 ± 7.9 vs. 2.5 ± 3.9 ng/ml, p = 0.04) and higher levels of catecholamines in the L group (epinephrine: 112.5 ± 112.2 vs. 80.3 ± 95.7 pg/ml, p = 0.25, norepinephrine: 847.9 ± 590.2 vs. 576.4 ± 300.9 pg/ml, p = 0.03 and dopamine: 35.7 ± 35.6 vs. 20.1 ± 12.7 pg/ml, p = 0.03) (Fig. 2). In addition, I123-MIBG scintigraphy findings revealed lower late H/M (2.09 ± 0.45 vs. 2.45 ± 0.44, p = 0.01) and higher washout rate (33.9 ± 13.8 vs. 26.4 ± 10.2, p = 0.02) in the L group compared with the S group (Fig. 2).
Assessment of sympathetic nervous system activity. a–c The comparison of plasma catecholamine levels according to the timing of left ventricular function improvement. d, e The comparison of I123-MIBG scintigraphy findings according to the timing of left ventricular function improvement. Lines within boxes represent median values, the upper and lower boxes lines represent the 25th and 75th percentiles, respectively, and whiskers indicate minimal and maximal values. L group LV function improvement: ≥ 1 month, S group LV function improvement: < 1 month
A multivariable logistic regression analysis including age, gender, EF on admission, norepinephrine, and troponin I as covariates showed that the levels of norepinephrine (OR 1.003, 95% CI 1.000–1.005, p = 0.01) and troponin I (OR 1.540, 95% CI 1.079–2.197, p = 0.01) were independent predictors of delayed LV function improvement.
Clinical outcomes
Table 2 shows the prevalence of in-hospital complications. The median duration of hospitalization was 18.5 days in the study population. It was comparable between the S and L groups (median days and IQR: 18 [12,13,14,15,16,17,18,19,20,21,22,23,24,25,26] vs. 19 (13–37), p = 0.25). The incidence of in-hospital complications was significantly higher in the L group (56% vs. 33.3%, p = 0.03), including heart failure (45% vs. 23%, p = 0.03) and in-hospital death (8.0% vs. 0%, p = 0.03). In a univariate analysis, the occurrence of in-hospital complications was associated with lower body mass index (BMI) (19.35 ± 3.65 vs 20.84 ± 3.07, p = 0.04), lower EF (39.86 ± 13.07% vs 50.12 ± 12.43%, p = 0.01), higher levels of catecholamines (epinephrine: 167.9 ± 161.6 vs. 70.9 ± 53.7 pg/ml, p = 0.01, norepinephrine: 1091.2 ± 694.4 vs. 587.8 ± 310.3 pg/ml, p = 0.01, dopamine: 44.8 ± 46.6 vs. 23.1 ± 15.6 pg/ml, p = 0.01) and LV function recovery ≥ 1 month (54.9% vs 33.3%, p = 0.04). A multivariable logistic regression analysis including age, gender, EF, epinephrine, and the timing of LV function improvement as covariates showed that the lower BMI, lower EF, higher levels of epinephrine and delayed LV functional recovery were independent predictors of incidence of in-hospital complications (Table 2).
In the analysis of long-term mortality, 12 patients were excluded due to lost to follow-up. A total of 78 patients were included in the Kaplan–Meier analysis. Median follow-up time was 790.5 days (IQR 9–6046 days). A total of 22 patients died during follow-up period. No statistically significant differences were observed between the two groups (L group: 59.6% vs. S group: 40.4%, p = 0.14) (Fig. 3). However, significantly more male patients died compared to female patients (52.0% vs 16.9%, p = 0.01).
Using the multivariable cox regression analysis including age, gender, EF, the presence of cancer, the independent predictors of all-cause mortality were age (hazard ratio 1.05, 95% CI 1.01–1.08; p = 0.01) and EF (hazard ratio 0.97, 95% CI 0.95–0.99; p < 0.01) and the presence of cancer (hazard ratio 2.04, 95% CI 1.02–3.83; p = 0.03).
Discussion
This is the first study, to the best of our knowledge, to evaluate clinical features and outcomes of TS focusing on the timing of LV function recovery. The key findings of our investigation were as follows: (a) patients with delayed improvement of LV function in TS had significantly higher levels of catecholamine and higher washout rate in I123-MIBG; (b) delayed improvement of LV function in patients with TS was associated with in-hospital complications, which did not affect long-term mortality. These findings indicate delayed LV functional recovery was associated with activation of the SNS in patients with TS, which may lead to poor clinical outcomes in the acute phase.
The causes of TS are not fully understood, although several mechanisms have been proposed. Stress has represented the main factor in TS, with more than 85% of cases precipitated by either a physically or emotionally stressful event that prefaces the start of symptoms [17]. Indeed, in our study population, approximately 80% of patients had physical or emotional stress. It has been hypothesized that catecholamine surges accompanied by cellular calcium overload result in regional microvascular dysfunction in susceptible patients [18]. The myocardial stunning supposedly caused by direct catecholamine-induced myocardial toxicity in TS patients has been attributed to the ensuing diffuse catecholamine-induced microvascular spasm or dysfunction [19]. Lyon et al. hypothesized that high levels of epinephrine trigger a switch in the intracellular signal trafficking from Gs (stimulated) to Gi (inhibited) protein signaling in β2 adrenoreceptor, which affects the apical myocardium, explaining the apical ballooning in TS [20]. While hyperactivation of SNS is thought to be pathogenic in TS, how this affects degree of LV dysfunction and the duration of LV function improvement is not fully explored [21, 22].
MIBG scintigraphy has been established as an important technique to evaluate cardiac SNS function [23]. The decrease of cardiac I123-MIBG uptake is thought to reflect impaired adrenergic function or denervation in various cardiac diseases. The increase of the WR might imply some kind of myocardial damage that results in an increase of cardiac sympathetic activity to compensate for the dysfunction. Thomas et al. evaluated cardiac SNS activity with MIBG scintigraphy and plasma catecholamine samples in 32 patients with TS and 20 control subjects [9]. In particular, this study showed lower late H/M and higher WR in MIBG scintigraphy and concomitantly increased plasma levels of epinephrine in the subacute phase of TS compared with the control group in the subacute state. At follow-up, no differences in MIBG parameters were observed between the TS and control groups. The authors concluded that norepinephrine reuptake is blocked because of the high circulating levels of norepinephrine, which may account for the low H/M in late phase and high WR on MIBG imaging in the acute state of TS [9]. In comparison with previous investigations, our study evaluated not only the relationship between MIBG scintigraphy and plasma catecholamine, but also the effect on timing of LV function improvement and clinical outcomes. In our study population, the L group showed significantly lower late H/M and higher WR compared to the S group, which was also associated with poor clinical outcomes. Based on our findings, some of these mechanisms might be explained by the fact that patients with hyperactivation of local cardiac SNS and higher levels of norepinephrine had longer exposure to these surges, leading to delayed improvement of LV function.
TS generally has a favorable outcome. However, a variety of complications occur in the acute phase [24,25,26,27]. Indeed, the results of our study showed high rates of in-hospital complications (45.6% of all patients), while they were observed in 52% of the patients in a similar sample size TS registry [28]. Previous studies have shown that several factors may be associated with clinical outcomes in patients with TS. Ansari et al. demonstrated that TS patients receiving catecholamine therapy have poor in-hospital outcomes in comparison to patients not receiving any form of catecholamine support [29]. Giannakopoulos et al. reported that the in-hospital morbidity rate was significantly higher in TS patients with physical stressors [30]. Interestingly, TS patients suffering from physical stress had significant higher level of catecholamine and higher incidence of in-hospital complications as compared to patients with emotional stress in our study population (data not shown). Increased SNS activity could affect in-hospital clinical outcomes in two ways: (1) a direct effect mediated by the augmented concentrations of catecholamines (e.g., provoking acute LV outflow obstruction and increased ventricular afterload, as well as inducing ventricular arrhythmias and subsequent sudden cardiac death); and (2) an indirect effect through a delayed LV function recovery, which might be associated with complications due to heart failure or ventricular arrhythmias. However, further studies are warranted to determine the underlying mechanisms behind the association of augmented activation of SNS and impaired in-hospital outcomes in TS.
Long-term mortality has been reported in several investigations. Weidner et al. reported that males suffering from TS revealed a higher risk for all-cause mortality than females over a 5-year follow-up period [31]. A similar trend was observed in our study. 16 of 28 males (57.1%) had in-hospital complications and 13 (46.4%) had died during long-term follow-up, while 25 patients of 62 females (40.3%) had in-hospital complications and 9 (14.5%) had died. Most of the previous studies reported that higher age, male, and lower EF were independent predictors of long-term mortality in TS patients [32, 33]. In the multivariate Cox regression analysis of our study population, similar findings were observed. There was a trend towards increased long-term mortality in the group with delayed LV function recovery, without reaching statistical significance, which could be due to the small sample size of the present investigation. These hypotheses must be validated in larger cohort studies.
Study limitations
Several limitations of our study should be mentioned. First, this is a retrospective study from a single center. Seconds, some data were unavailable such as plasma catecholamine levels and MIBG scintigraphy. Third, we did not compare plasma catecholamine levels between TS and the other acute medical illness as a control group. In addition, 12 patients were lost to follow-up. Finally, since long-term clinical outcome was assessed based on telephone interview in some cases, the cause of death was uncertain.
Conclusions
In patients with TS, high activity of the SNS was observed in patients with delayed LV function recovery, which was associated with in-hospital adverse outcomes.
References
Hurst RT, Prasad A, Askew JW 3rd, Senqupta PP, Tajik AJ (2010) Takotsubo cardiomyopathy: a unique cardiomyopathy with variable ventricular morphology. JACC Cardiovasc Imaging 3:641–649
Bielecka-Dabrowa A, Mikhailidis DP, Hannam S, Rysz J, Michalska M, Akashi YJ, Banach M (2010) Takotsubo cardiomyopathy: the current state of knowledge. Int J Cardiol 142:120–125
Pelliccia F, Parodi G, Greco C, Antoniucci D, Brenner R, Bossone E, Cacciotti L, Capucci A, Citro R, Delmas C, Guerra F, Ionescu CN, Lairez O, Larrauri-Reyes M, Lee PH, Mansencal N, Marazzi G, Mihos CG, Morel O, Nef HM, Nunez Gil IJ, Passaseo I, Pineda AM, Rosano G, Santana O, Schneck F, Song BG, Song JK, Teh AW, Ungprasert P, Valbusa A, Wahl A, Yoshida T, Gaudio C, Kaski JC (2015) Comorbidities frequency in Takotsubo syndrome: an international collaborative systematic review including 1109 patients. Am J Med 128:654.e11–654.e19
Templin C, Ghadri JR, Diekmann J, Napp LC, Bataiosu DR, Jaguszewski M, Cammann VL, Sarcon A, Geyer V, Neumann CA, Seifert B, Hellermann J, Schwyzer M, Eisenhardt K, Jenewein J, Franke J, Katus HA, Burgdorf C, Schunkert H, Moeller C, Thiele H, Bauersachs J, Tschöpe C, Schultheiss HP, Laney CA, Rajan L, Michels G, Pfister R, Ukena C, Böhm M, Erbel R, Cuneo A, Kuck KH, Jacobshagen C, Hasenfuss G, Karakas M, Koenig W, Rottbauer W, Said SM, Braun-Dullaeus RC, Cuculi F, Banning A, Fischer TA, Vasankari T, Airaksinen KE, Fijalkowski M, Rynkiewicz A, Pawlak M, Opolski G, Dworakowski R, MacCarthy P, Kaiser C, Osswald S, Galiuto L, Crea F, Dichtl W, Franz WM, Empen K, Felix SB, Delmas C, Lairez O, Erne P, Bax JJ, Ford I, Ruschitzka F, Prasad A, Lüscher TF (2015) Clinical features and outcomes of Takotsubo (Stress) cardiomyopathy. N Engl J Med 373:929–938
Tsuchihashi K, Ueshima K, Uchida K, Oh-mura N, Kimura K, Owa M, Yoshiyama M, Miyazaki S, Haze K, Ogawa H, Honda T, Hase M, Kai R, Morii I (2001) Transient left ventricular apical ballooning without coronary artery stenosis: novel heart syndrome mimicking acute myocardial infarction. Angina Pectoris-Myocardial Infarction Investigation in Japan. J Am Coll Cardiol 38:11–18
Elesber A, Lerman A, Bybee KA, Murphy JG, Barsness G, Singh M, Rihal CS, Prasad A (2006) Myocardial perfusion in apical ballooning syndrome correlate of myocardial injury. Am Heart J 152(3):469.e9–469.e13
Sharkey SW, Windenburg DC, Lesser JR, Maron MS, Hauser RG, Lesser JN, Haas TS, Hodges JS, Maron BJ (2010) Natural history and expansive clinical profile of stress (tako-tsubo) cardiomyopathy. J Am Coll Cardiol 55:333–341
Cimarelli S, Sauer F, Morel O, Ohlmann P, Constantinesco A, Imperiale A (2010) Transient left ventricular dysfunction syndrome: patho-physiological bases through nuclear medicine imaging. Int J Cardiol 144:212–218
Christensen TE, Bang LE, Holmvang L, Skovgaard DC, Oturai DB, Søholm H, Thomsen JH, Andersson HB, Ghotbi AA, Ihlemann N, Kjaer A, Hasbak P (2016) 123I-MIBG scintigraphy in the subacute state of Takotsubo cardiomyopathy. JACC Cardiovasc Imaging 9:982–990
Akashi YJ, Goldstein DS, Barbaro G, Ueyama T (2008) Takotsubo cardiomyopathy: a new form of acute, reversible heart failure. Circulation 118:2754–2762
Mahmoud AN, Al-Ani M, Saad M, Lgendy AY, Elgendy IY (2016) Development and validation of simple integer risk score for prediction of in-hospital mortality following Takotsubo syndrome. Heart Lung 45:510–514
Krishnamoorthy P, Garg J, Sharma A, Palaniswamy C, Shah N, Lanier G, Patel NC, Lavie CJ, Ahmad H (2015) Gender differences and predictors of mortality in Takotsubo cardiomyopathy: analysis from the national inpatient sample 2009–2010 datebase. Cardiology 132:131–136
Sobue Y, Watanabe E, Ichikawa T, Koshikawa M, Yamamoto M, Harada M, Ozaki Y (2017) Physically triggered Takotsubo cardiomyopathy has a higher in-hospital mortality rate. Int J Cardiol 235:87–93
Prasad A, Lerman A, Rihal CS (2008) Apical ballooning syndrome (Tako-tsubo of stress cardiomyopathy): a mimic of acute myocardial infarction. Am Heart J 155:408–417
Lang RM, Badano LP, Mor-Avi V, Afilalo J, Armstrong A, Ernande L, Flachskampf FA, Foster E, Goldstein SA, Kuznetsova T, Lancellotti P, Muraru D, Picard MH, Rietzschel ER, Rudski L, Spencer KT, Tsang W, Voigt JU (2015) Recommendation for cardiac chamber quantification by echocardiography in adults: an Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 28(1):1–39.e14
Nakajima K, Nakata T (2015) Cardiac 123I-MIBG imaging for clinical decision making: 22-year experience in Japan. J Nucl Med 56(Suppl 4):11S–19S
Sharkey SW, Lesser JR, Maron BJ (2011) Cardiology patient page. Takotsubo (stress) cardiomyopathy. Circulation 124(18):e460–e462
Nef HM, Möllmann H, Troidl C, Kostin S, Voss S, Hilpert P, Behrens CB, Rolf A, Rixe J, Weber M, Hamm CW, Elsasser A (2009) Abnormalities in intracellular Ca2+ regulation contribute to the pathomechanism of Tako-Tsubo cardiomyopathy. Eur Heart J 30:2155–2164
Basso C, Theine G (2006) The pathophysiology of myocardial reperfusion: a pathologist`s perspective. Heart 92:1559–1562
Lyon AR, Ress PS, Prasad S, Poole-Wilson PA, Harding SE (2008) Stress (takotsubo) cardiomyopathy a novel pathophysiological hypothesis to explain cathecolamine-induced acute myocardial stunning. Nat Clin Pract Cardiovasc Med 5:22–29
Shams Y, De Palma R (2017) Contemporary review on the pathogenesis of takotsubo syndrome: the heart shedding tears: Norepinephrine churn and foam at the cardiac sympathetic nerve terminal. Int J Cardiol 228:528–536
Shams Y (2018) Plasma epinephrine levels and its causal link to takotsubo syndrome revisited: Critical review with a diverse conclusion. Cardiovasc Revasc Med. https://doi.org/10.1016/j.carrev.2018.10.026
Verberne HJ, van der Heijiden D, van Eck-Smit BLF, Somsen GA (2009) Persisting myocardial sympathetic dysfunction in takotsubo cardiomyopathy. J Nucl Cardiol 16:321–324
Murakami T, Yoshikawa T, Maekawa Y, Ueda T, Isogai T, Konishi Y, Sakata K, Nagao K, Yamamoto T, Takayama M (2014) Characterization of predictors of in-hospital cardiac complications of Takotsubo cardiomyopathy: multi-center registry from Tokyo CCU Network. J Cardiol 63:269–273
Kato K, Sakai Y, Ishibashi I, Himi T, Kobayashi Y (2018) Predictors of in-hospital cardiac complications in patients with Takotsubo syndrome. Heart Vessels. https://doi.org/10.1007/s00380-018-1172-y
Singh K, Carson K, Shah R, Sawhney G, Singh B, Parsaik A, Gilutz H, Usmani Z, Horowitz J (2014) Meta-analysis of clinical correlates of acute mortality in Takotsubo cardiomyopathy. Am J Cardiol 113:1420–1428
Lyon AR, Bossone E, Schneider B, Sechtem U, Citro R, Underwood SR, Sheppard MN, Figtree GA, Parodi G, Akashi YJ, Ruschitzka F, Filippatos G, Mebazaa A, Omerovic E (2016) Current state of knowledge on Takotsubo syndrome: a Position Statement from the Taskforce on Takotsubo Syndrome of the Heart Failure Association of the European Society of Cardiology. Eur J Heart Fail 18:8–27
Schneider B, Athanasiadis A, Schwab J, Pistner W, Gottwald U, Schoeller R, Toepel W, Winter KD, Stellbrink C, Müller-Honold T, Wegner C, Sechtem U (2014) Complications in the clinical course of tako-tsubo cardiomyopathy. Int J Cardiol 176:199–205
Ansari U, El-Battrawy I, Fastner C, Behnes M, Sattler K, Huseynov A, Baumann S, Tülümen E, Borggrefe M, Akin I (2018) Clinical outcomes associated with catecholamine use in patients diagnosed with Takotsubo cardiomyopathy. BMC Cardiovasc Disord 18:54
Konstantinos G, El-Battrawy I, Katja S, Uzair A, Ursula H, Martin B, Ibrahim A (2017) Comparison and outcome analysis with takotsubo cardiomyopathy triggered by emotional stress or physical stress. Front Psychol 8:527
Weidner KJ, El-Battrawy I, Behnes M, Schramm K, Fastner C, Kuschyk J, Hoffmann U, Ansari U, Borggrefe M, Akin I (2017) Sex differences of in-hospital outcomes and long-term mortality in patients with Takotsubo cardiomyopathy. Ther Clin Risk Manag 13:863–869
Huseynov A, El-Battrawy I, Ansari U, Schramm K, Zhou X, Lang S, Borggrefe M, Akin I (2017) Age related differences and outcomes of patients with Takotsubo syndrome. J Geriatr Cardiol 14:632–638
Stiermaier T, Moeller C, Oehler K, Desch S, Graf T, Eitel C, Vonthein R, Schuler G, Thiele H, Eitel I (2016) Long-term excess mortality in takotsubo cardiomyopathy: predictors, causes and clinical consequences. Eur J Heart Fail 18:650–656
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Takero Matsuura: has no conflict of interest to report. Masafumi Ueno: has no conflict of interest to report. Yoshitaka Iwanaga: has no conflict of interest to report. Shunichi Miyazaki: has no conflict of interest to report.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Matsuura, T., Ueno, M., Iwanaga, Y. et al. Importance of sympathetic nervous system activity during left ventricular functional recovery and its association with in-hospital complications in Takotsubo syndrome. Heart Vessels 34, 1317–1324 (2019). https://doi.org/10.1007/s00380-019-01359-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00380-019-01359-4