Abstract
There are increasing demands to reduce greenhouse gas emissions from agricultural soils worldwide. A significant portion of these emissions occur in cold regions during soil’s freezing and thawing. Focusing on over-winter cropland nitrous oxide (N2O) emissions, a review of 21 relevant peer-reviewed studies with a total of 88 comparisons was conducted to quantify the efficacy of field management practices (no-till, cover crops (CCs), nitrification, and urease inhibitors (NI + UI)) in reducing emissions. We also assessed these mitigation practices’ efficacy across soil types and between cold humid and cold dry areas. The ratio of non-growing season emissions to full-year N2O emissions reported in the studies used in this review ranged between 5 and 91%. No-till significantly reduced N2O emissions by 28%, and this effect was more pronounced in drier climates. NI + UI also significantly reduced over-winter emissions by 23% compared to conventional fertilizers, and this effect was more evident in medium-textured soils than coarse soils. CCs showed an overall reduction potential of 18%; however, this effect was not significant. This review showed that under the CC practice, N2O emissions were reduced overall in humid climates but increased in drier climates, while no-till and NI + UI practices effectively reduced over-winter emissions in both dry and humid winter regions and all soil types.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Agricultural soils emit trace gases that increase atmospheric concentrations of nitrous oxide (N2O), a greenhouse gas that contributes to global climate change (Davidson and Kanter 2014). Studies investigating N2O emissions from agricultural soils have vastly focused on growing season. Nonetheless, the contribution of over-winter N2O emissions to annual emissions that occur in mid-high to high latitude regions, such as Canada and the Mid-West US where soil freezing and thawing occur over a 5-month period, could reach up to 90% ( Risk et al. 2014). The bulk of over-winter emissions mostly occurs in the early spring partly due to the anaerobic conditions created during the thawing of soils enhancing N2O production (Chantigny et al. 2016; Ejack and Whalen 2021; Risk et al. 2014; Wagner-Riddle et al. 2007). Understanding the factors including substrate availability, snow cover, freeze–thaw dynamics, soil temperature, soil type, and moisture conditions that impact the magnitude of over-winter emissions is becoming the focus of recent studies (Chen et al. 2016; Smith et al. 2010; Zhe et al. 2018).
Appropriate agronomic management practices could help achieve a reduction in over-winter emissions. These practices include those tailored towards minimizing the substrates available for emission during the spring thaw such as the use of nitrification inhibitors during fall manure application and incorporation of cover crops. The use of nitrification inhibitors to prevent oxidation of \({\mathrm{NH}}_{4}^{+}\) to \({\mathrm{NO}}_{3}^{-}\) has been suggested as an effective method for reducing N2O losses, especially from fall-applied manure, which carries N mostly in the form of ammonium (NH3) or organic N (Dong et al. 2018; Vallejo et al. 2001; VanderZaag et al. 2011; Zhang et al. 2018). Apart from lowering substrate availability, practices that can alter soil temperature or moisture, such as the presence of cover crop and no-tillage, have been shown to lessen over-winter emissions (Congreves et al. 2017; Preza-Fontes et al. 2020; Wagner-Riddle et al. 2007). The use of non-legume cover crops (CC) as a mitigation practice for N2O emissions is centered on the hypothesis that cover-crops will absorb more moisture and \({\mathrm{NO}}_{3}^{-}\) from the soil, lowering their levels and resulting in reduced N2O emissions (Shackelford et al. 2019). The major difference between legume CC and non-legume CC is that while legume CC can fix nitrogen from the atmosphere to the soil, non-legume CC cannot, but can absorb large quantities of N from the soil. Having a vegetation cover atop the soil can also slow down the thawing of frozen soils and lower the freeze–thaw cycles, thereby reducing emission rates (Chen et al. 2020). Tillage could affect emissions through its influence on soil aeration, structure, temperature, moisture content, microbial activity, and gas diffusion through the subsoil to the surface (Gregorich et al. 2015; Signor and Cerri 2013). No-till field management could reduce N2O emissions by lowering freezing intensity and soil moisture (Wagner-Riddle et al. 2007). However, the efficacy of no-till and the use of non-legume cover crops remains under debate as some studies have also reported no consistent effects of these practices (Behnke and Villamil 2019; Elmi et al. 2003; Thomas et al. 2017). It is essential to clarify the controversies since farmers’ and policymakers’ willingness to adopt and implement recommended strategies will depend on data-driven evidence of reduced emissions, without hampering farm productivity.
Meta-analyses have been used to investigate the efficacy of agronomic practices including tillage, cover crops, and nitrification and urease inhibitors in mitigating N2O emissions. Earlier meta-analyses studies focusing majorly on growing season N2O losses showed no significant differences in N2O emissions between CT and no-till or reduced tillage (NT/RT) (Decock 2014; Feng et al. 2018; Six et al. 2004; van Kessel et al. 2013). Other studies reported significantly greater emissions by 12% (Shakoor et al. 2021) and 10% (Huang et al. 2018) in NT/RT treatments compared to CT. Concerning nitrification and urease inhibitors, previous meta-analysis studies have shown that the use of the inhibitors could reduce N2O losses by up to 44% (Feng et al. 2016; Qiao et al. 2015; Thapa et al. 2016; Yang et al. 2016). Investigating the overall effect of cover crops on N2O emissions, Basche et al. (2014) showed that 60% of the 106 observations utilized in their meta-analysis study positively affected N2O emissions, while 40% indicated negative effects.
Although these meta-analysis studies quantified the above management practices’ effectiveness on N2O emissions, they mainly included individual studies that reported growing season emissions and have not adequately accounted for the sizeable N2O losses that occur over winter. For instance, only 14% of the observations used by Decock (2014) were all year round measurement. We still do not have a clear overview of these strategies’ effectiveness in mitigating the considerable N2O losses that occur over-winter. Accordingly, this work is aimed to review studies to quantify the effectiveness of nitrification and urease inhibitors, tillage, and cover crops in reducing non-growing season N2O emissions in cold temperate regions. We also assessed the efficacy of these mitigation practices across soil types and aridity zones (cold-dry vs. cold-humid areas).
Mechanism of over-winter N2O emissions
The potential mechanism of over-winter N2O emissions in this study is discussed for two distinct periods: the freezing period and the spring-thaw period. The frozen soil reduces the microbial activities it harbors, although it does not completely inhibit them. After the initial lysis of some microbial cells that may occur at subzero temperatures (Maljanen et al. 2007), soil microorganisms gradually acclimatize to low temperatures (Smith et al. 2010). The relatively warm soil under snow cover further supports a level of their activities. For instance, decomposition of soil organic matter occurs during the freezing period, evidenced by the low rates of CO2 accumulation (Maljanen et al. 2007). However, the CO2 fluxes were lower compared to the fluxes observed during pre-freezing period. Likewise, mineralization of organic N also takes place, as shown by high levels of ammonium \({(\mathrm{NH}}_{4}^{+}\)) in these same soils (Maljanen et al. 2007). However, a significant concomitant increase in \({\mathrm{NO}}_{3}^{-}\) is not always observed following the soil thaw (Chen et al. 2016; Maljanen et al. 2007). This condition has been attributed to low levels of nitrifying bacteria found in the soil, as a result of their greater susceptibility to cold temperatures (Smith et al. 2010) or the possible rapid denitrification of \({\mathrm{NO}}_{3}^{-}\) — generated from a portion of the accumulated \({\mathrm{NH}}_{4}^{+}\) — to N2O and N2 (Maljanen et al. 2007; Zhe et al. 2018). Contrary to the effect of cold temperature on nitrifier populations that made their detection in mid-winter difficult, cold temperatures have a lesser effect on the denitrifier community and did not hamper their detection (Smith et al. 2010). Gases produced in the freezing period are trapped in the soil, given that ice blocks the pores of soil surface, resulting in increased concentrations of N2O and CO2 in the soil over time. However, partial release of such trapped gases can sometimes occur during the freezing period through cracks caused by increases in temperature and slight thawing of the soil (Teepe et al. 2001).
In early spring during the soil thaw, the ice barriers that trapped gases begin to give away, causing the physical release of these gases (Burton and Beauchamp, 1994; Teepe et al. 2001). As the soil thaws further, the frozen subsoil melts, enhancing soil drainage, thereby creating the aerobic topsoil required for nitrification of available \({\mathrm{NH}}_{4}^{+}\). Although more N2O is produced in the deeper soil layers over the freezing period, as evidenced by its greater concentrations at these depths, as well as the depletion of \({\mathrm{NH}}_{4}^{+}\) and organic N, a substantial proportion of these N2O molecules are converted to N2 by the time they reach the surface (Wagner-Riddle et al. 2008). One study by Van Groenigen et al. (2005) showed that while the subsoil is characterized by depleted δ15N indicating N2O production, the topsoil consumed N2O, as shown by a greater δ15N. Consumption of N2O is generally typical for soils which are anoxic and rich in OM, but have depleted \({\mathrm{NO}}_{3}^{-}\). This depletion of \({\mathrm{NO}}_{3}^{-}\) in the subsoil usually occurs at the onset of thawing, resulting in greater N2 than N2O fluxes (Ludwig et al. 2006). In addition, more recent studies suggest the release of trapped gases and newly produced N2O occurs at the onset of thawing. Such newly produced N2O fluxes could be up to five times the amount observed during the release of trapped gases (Risk et al. 2014). De novo production results from favorable conditions for nitrification or denitrification processes during thawing. Identified as the principal source of N2O emitted during the thawing process, the denitrification process requires anaerobic conditions, nitrates, and simple C compounds (Priemé and Christensen 2001; Sehy et al. 2004; van Groenigen et al. 2005). Anaerobic conditions are enhanced by a frozen subsoil which hinders drainage while creating saturated soil conditions (Nyborg et al. 1997). Additionally, the thaw period, characterized with high soil moisture from the snow melt, also enhances anaerobic conditions. The C and N required for denitrification could arise from the death of microbes due to freeze–thaw stresses, as well as residual C and N available in the soil (Chen et al. 2020; Pelster et al. 2013; Teepe et al. 2001).
A few studies have evaluated the role of nitrifiers and denitrifiers on over-winter N2O emissions. Using PCR-DGGE analysis, Smith et al. (2010) observed significant changes in the diversity of the nitrifier and denitrifier populations observed between the pre- and post-spring thaw period. While this method only observes changes in dominant microbial populations compared to amplicon-sequencing or metagenomics, the authors alluded that these variations in microbial community composition suggest that microbes must be active immediately after spring thaw. Significant N2O flushes that occurred during the thaw in their study were primarily attributed to denitrification. Similarly, Ludwig et al. (2004) used 15 N as a tracer to show that denitrification contributed 83% of N2O production in organic soils immediately after the soil began to thaw. A microcosm study (Sharma et al. 2006) observed an apparent increase in denitrifying bacteria and a larger amount of nitrate reductase (napA) and nitrite reductase (nirS) gene copies soon after the soil thaw began, followed by a decrease in gene copies. Their study concluded that the release of spring N2O fluxes was influenced by increased microbial activities and the expression of denitrifying genes. Using next-generation sequencing, Bent et al. (2016) showed over-winter N2O fluxes to relate to community composition of the denitrification genes (nirS/nirK and nosZ) in a residue-retained field, and both nitrifiers (bacterial amoA) and denitrifiers (nirK and nosZ) in a residue-removed field. However, a recent study has suggested that although changes in the nitrifier and denitrifier populations were observed during the winter period, these variations in the abundance of functional genes were not correlated with the changes in over-winter N2O fluxes (Yin et al. 2019). The lack of consensus on the role of denitrifiers in spring thaw N2O fluxes may stem from the detection limits of these sequencing methods. Additionally, these studies rely on gene copy numbers as a parameter for denitrifying activity, which does not directly translate to expression since these genes may be present in dormant species’ DNA (Nannipieri et al. 2020). Thus, future investigations could utilize an improved PCR primer set (Zhang et al. 2021) developed for better detection of denitrifying genes as well as metatranscriptomics to determine denitrifying activity. Furthermore, studies could use 15 N analysis of site preference to understand the role of denitrifying and nitrifying genes on over-winter N2O emissions.
Magnitude of over-winter N2O emissions
Table 1 provides the summary of the studies used for the review in the present study. It shows the magnitude of the non-growing season N2O emissions from the various studies, also expressed as a percentage of total annual emissions. Additionally, it includes the major treatments of the studies and the measurement period of the over-winter fluxes. More details of the data can be found in the supplementary data (S1). In this review (21 studies with a total of 88 comparisons), the ratio of the non-growing season to full-year N2O emissions ranged between 5 and 91%, highlighting high variation and the significance of these emissions. The over-winter flux measurement varied among studies. For example, in the North Atlantic region of the USA and Canada, and the Mid-West US, snow cover appears in early December, and peaks between February and early March, with the spring melt occurring within 2–3 weeks between late March and early April (Chantigny et al. 2016). Some of the studies started earlier or ended later to capture the entire winter and early spring period. As snow accumulation varies from year to year, the timing and intensity of winter-time soil fluxes may vary, as would the temporal occurrence and magnitude of peak fluxes.
Mitigation potential of no-till/reduced tillage
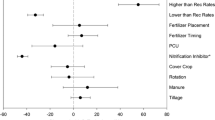
Using the meta-analysis approach, we quantified the effectiveness of mitigation practices in reducing non-growing season N2O emissions in cold temperate regions (more details of the method can be found in the supplementary information). In the winter and early spring, the effect of no-till/reduced tillage (NT/RT) compared with conventional tillage (CT) was − 28% (95% confidence interval = − 39 to − 16%), indicating a significant reduction of N2O emissions (Fig. 1; Table 2). Under the tillage practice, dry areas emitted significantly lower emissions than the humid areas (35% less), while soil texture did not significantly affect N2O emissions (Fig. 2; Table S2). Although among the studies there were years when no-till did not reduce N2O emissions over winter, in some other cases, studies reported that mean N2O fluxes under NT were one to six times lower than in CT plots (Congreves et al. 2017; Lemke et al. 1999; Wagner-Riddle et al. 2007). The micrometeorological method used in gas emission measurements in some studies (Congreves et al. 2017; Wagner-Riddle et al. 2007), allowing for more frequent sampling, would have accorded the researchers a greater chance of capturing distinct peak fluxes.
Forest plot (random effects model) indicating the overall effect size estimates of management practices: no-till/reduced tillage (RT) vs. tillage treatments; cover crops (CCs) vs. no cover crops (no-CCs); nitrification and urease inhibitors (NI + UI) vs. no inhibitors, on over-winter N2O emissions. Numbers in the bracket indicate the number of observations utilized in the analysis. The center circle represents the summary estimate. Confidence intervals are also reported. CI crossing the null (zero) line indicates no significant change in effect or that both treatments show equivalent effects
Forest plot (random effects model) indicating subgroup (climate and soil type) effects of no-tillage/reduced tillage (no-till/RT) vs. tillage treatments (CT). Confidence intervals are also reported. CI crossing the null (zero) line indicates no significant change in effect or that both treatments show equivalent effects
The lower N2O emissions under NT/RT can be attributed to the combined insulating effects of crop residues and snow cover that lowers the cumulative degree hours below 0 °C, an indicator of freezing intensity and duration (Wagner-Riddle et al. 2007). Wagner-Riddle et al. (2007) also suggested that crop residues that are retained on NT/RT plots can effectively trap snow. Due to their low heat of diffusivity, snow and residues insulate the soil and decrease the transfer of heat from the soil to the atmosphere, resulting in higher soil temperature in RT/NT than in CT (Wagner-Riddle et al. 2007). Under CT, soil temperatures varied over a wider range, increasing the degree of freezing than under NT/RT plots. The magnitude of over-winter N2O emissions has been linked to the number of degree hours below 0 °C. An exponential to plateau relationship model defined this link and showed that N2O emissions increased exponentially with cumulative freezing degree days until a plateau was reached (Wagner-Riddle et al. 2017). The wider temperature variation reported for CT (vs. NT/RT) soils (Wagner-Riddle et al. 2007; Smith et al. 2010) could lead to greater microbes’ die-off. When freezing stresses kill off some soil microbes, microbial polymers return to the soil C pool, while amino acids and other organic monomers are returned to the dissolved organic nitrogen (DON) pool (Schimel and Bennett 2004). Living microbes in the frozen soil’s liquid film then utilize these substances to stimulate N2O emissions (Chen et al. 2020; Pelster et al. 2013). Additionally, Ferrari Machado et al. (2021) suggested that higher N2O emission under CT may have been induced by mechanisms potentially resulting from lower temperatures such as the breakdown of soil aggregates and subsequent release of previously protected soil organic C. Lower temperatures could therefore cause an exponential rise in N2O fluxes for a few days resulting from the death of microbes and subsequent release of \({\mathrm{NO}}_{3}^{-}\) (Larsen et al. 2002; Maljanen et al. 2007; Sharma et al. 2006). This observation was also confirmed by Smith et al. (2010) who suggested that the soil’s lower nutrient availability in NT/RT plots due to lower freezing intensity could lower N2O emissions than CT plots. Smith et al. (2010) also implied that the significant differences in the composition of nitrifier and denitrifier communities observed soon after spring thaw could explain the differences in emissions between the CT and NT/RT treatments.
The difference in the establishment period of NT/RT from CT is an important factor to consider when investigating the effect size of NT/RT practice on N2O emissions. Six et al. (2004) showed that the potential to reduce N2O emissions with NT is only achieved with long-term NT practice. Elevated N2O emissions from newly adopted NT/RT plots slowly diminish due to the development of macropores, improved soil structure and aeration status, and decreased formation of anaerobic microsites. Studies have shown that the elevated N2O emissions observed under NT/RT only occurred within the first 10 years following the shift from CT to NT/RT, after which a significant reduction in N2O emissions was observed under NT/RT (Six et al. 2004; Feng et al. 2018; van Kessel et al. 2013). In our meta-analysis, 62% of the tillage studies had \(\ge\) 10 years of NT/RT and this could be one of the reasons for the significant reduction in N2O emissions observed under NT/RT.
This review revealed that relatively drier areas such as the low annual precipitation areas in Western Canada or China’s moist continental climate with dry winter periods emitted significantly lower emissions than the humid winter areas (35% less). Elevated soil moisture during the winter and especially in the spring thaw period favors the denitrification process (Maljanen et al. 2007), the main process contributing to freeze–thaw N2O emissions. Antecedent fall moisture conditions have also been shown to affect spring thaw N2O emissions, with a greater fall water-filled pore space (WFPS) leading to greater spring fluxes (Chen et al. 2016; Li et al. 2012; Priemé and Christensen 2001). Following a distinctly wet field growing season, Chen et al. (2016) observed relatively high subsequent spring thaw N2O fluxes. These elevated spring emissions, accounting for up to 49% of the non-growing season fluxes, were attributed to the soil’s high moisture condition, which led to a rise in soil WFPS to 54%, compared to the 28% measured in a previous drier year when no significant thaw emissions were observed. A confirmatory lab experiment within the same study showed that freeze–thaw cycles did not induce distinct fluxes at a 20% WFPS, but that emissions increased by an order of magnitude at a WFPS of 80% (Chen et al. 2016). Subsequent to a dry antecedent fall and low winter air temperatures, Li et al. (2012) measured limited spring thaw emissions — 6.6% of annual emissions, in contrast to about 70% from the previous wetter and warmer preceding fall and winter. Dry fall soil conditions and the presence of less unfrozen water during the winter would have limited denitrification. Therefore, dry areas which are likely to have lower antecedent (fall) soil moisture prior to the soil freezing up in the winter could emit significantly lower spring thaw emissions than humid areas.
Although our meta-analysis showed that soil texture did not significantly impact the emissions (Fig. 2; Table S2), coarse soils tended to lower emissions more than medium soils under tillage practices. This observation reveals that the lower N2O levels observed may be partially associated with soil texture influence on soil moisture since coarse soils have lower WFPS than medium soils. Water-filled pore space and soil moisture affect growing season emissions. For instance, Linn and Doran (1984) showed NT/RT fields to have a greater average water-filled pore space (WFPS = 62%) than CT fields (WFPS = 44%), leading them to relate the greater emissions to anaerobic metabolism occurring in the NT/RT plots. However, the influence of soil texture on WFPS under tillage practices was not significant over the winter period as soils would tend to be saturated, no matter the soil type.
Mitigation potential of cover crops
In our meta-analysis, the effect of cover crop treatment across the trials was − 18% (95% CI = − 35 to 3%) (Fig. 1; Table 2). This result indicates that although cover crops showed a tendency to reduce emissions overall (− 18%), the change in their effect on over-winter N2O emissions compared to the absence of a cover crop was not significantly different. In humid regions, the use of CCs significantly resulted in lower emissions (89% less) than in the dry areas, while soil texture did not significantly affect N2O emissions (Fig. 3; Table S2). This finding concurs with Basche et al. (2014) findings that cover crops did not significantly affect direct N2O emissions during the winter period. Their study also showed a significantly positive impact for data points measured during the decomposition of the cover crops compared to the impact during the cover crop growth period, which showed no significant difference. Although CCs can assimilate \({\mathrm{NO}}_{3}^{-}\) over winter, they have also been observed to produce elevated N2O concentrations in the rhizosphere by drawing on a supply of labile C and N generated by decomposing tissues and living roots (Thomas et al. 2017; Wertz et al. 2016). Therefore, while CCs have a better potential to mitigate N2O emissions by depleting \({\mathrm{NO}}_{3}^{-}\), this potential could be countered by the production of denitrification substrates in the cover crops’ root zone.
Forest plot (random effects model) indicating subgroup (climate and soil type) effects of cover crop (CC) vs. no cover crop treatments (no-CC). Confidence intervals are also reported. CI crossing the null (zero) line indicates no significant change in effect or that both treatments show equivalent effects
The capacity of cover crops to reduce over-winter N2O emissions through the assimilation of \({\mathrm{NO}}_{3}^{-}\) largely depends on whether the cover crop is adequately established before the winter. This could be why a clear distinction was observed between the humid and dry climates in our meta-analysis. In cold and dry areas, cover crops sometimes develop poorly, suffer winter-kill, and cannot produce spring biomass. This situation was the case in a study by Behnke and Villamil (2019) where N2O emissions (1.64 kg N ha−1) in the first 2 years of their study, when the cover crop was poorly established, were significantly larger than emissions (0.32 kg N ha−1) observed in the last 2 years, which had warmer temperatures and early November precipitation. Similarly, Dietzel et al. (2011) found that after a harsh winter, a winter rye cover crop (vs. the absence of a cover crop) had no significant effect on over-winter N2O emissions; however, after the following year’s milder winter, the cover crop resulted in lower N2O fluxes. When simulated in growth chambers compared to field conditions where CC did not reduce over-winter N2O emissions, Jarecki et al. (2009) observed that cover crops resulted in N2O reduction due to better growing conditions in the growth chambers, which allowed for the development of roots, faster growths, and N uptake. Our analysis showed that although not significantly different, emissions under the CC practice were lower in coarse soils than in medium soils. We adduce that the ease of plant growth in coarse soils, which facilitates good plant establishment, may explain why lower N2O emissions were observed in coarse soils. These observations point to the potential influence of CC developmental status on their overall impact on over-winter emissions.
Although cover crops tend to reduce spring thaw emissions in humid climates, and when their roots are well established, they have been observed to cause a significant increase in the growing season N2O emissions after the following crop has been planted and fertilizer applied (Mitchell et al. 2013; Preza-Fontes et al. 2020). This increase in N2O emissions is likely due to the denitrification substrates produced in the cover crops’ rhizosphere, as well as the decomposition and subsequent release of N2O when CCs are incorporated into the soil. Consequently, to improve N efficiency in CC plots, fertilizer inputs should be reduced after plowing (Guardia et al. 2016). The selected studies show no consensus about the cover crops’ effects on either spring thaw or growing season N2O emissions (Iqbal et al. 2015; Parkin et al. 2016). However, cover crops have other benefits, including reducing NH3, \({\mathrm{NO}}_{3}^{-}\), DOC, and suspended solids in leachate (Gillette et al. 2018; Smukler et al. 2012). Cover crops also increase soil organic C sequestration (Basche et al. 2014) and enhance microbial abundance (Kim et al. 2020).
Mitigation potential of nitrification and urease inhibitors
Our analysis on nitrification and urease inhibitors’ (NI + UIs’) effect on over-winter N2O emissions shows a summary result of − 23% (95% CI = − 28 to − 16%) (Fig. 1; Table 2), indicating that on average, NI + UI significantly reduced N2O emissions. Under NI + UI, no significant difference was observed in N2O emissions between humid and dry climates, although emissions were lower in humid areas. However, soil texture significantly impacted emissions under NI + UI (Fig. 4; Table S2). This effect was more evident in medium-textured soils than in coarse soils. This meta-analysis result concurs with a modelling study (Grant et al. 2020) which showed that nitrification inhibitors (NI) could reduce N2O emissions from fall-applied slurry by up to 33%. Our review included various types of NI and UI — 3,4-dimethyl pyrazole phosphate (DMPP), nitrapyrin, dicyandiamide (DCD), N-(n-butyl) thiophosphoric triamide (NBPT), hydroquinone (HQ). It has been shown that some inhibitor types could be more effective in reducing N2O emissions than others. For instance, Lin et al. (2017) revealed that the reduction coefficients for DMPP and nitrapyrin were 81% and 57%, respectively. Ding et al. (2015) also showed that DCD achieved more N2O reduction (78.6%) than NBPT (50%).
Forest plot (random effects model) indicating subgroup (climate and soil type) effects of nitrification and urease inhibitors (NI + UI) vs. no inhibitors. Confidence intervals are also reported. CI crossing the null (zero) line indicates no significant change in effect or that both treatments show equivalent effects. Only data for groups with more than one study is presented
Our findings together with earlier meta-analysis that focused majorly on the growing season (Feng et al. 2016; Qiao et al. 2015; Thapa et al. 2016; Yang et al. 2016) all agree that the use of NI + UI to prevent oxidation of \({\mathrm{NH}}_{4}^{+}\) to N2O is an effective method for reducing N2O losses. The significantly lower over-winter N2O emissions observed with NI + UI application could result from low soil temperatures during winter since the efficacy of NI + UI for reducing N2O emissions has been shown to depend on temperature. For instance, in their study of UK soils, McGeough et al. (2016) measured 89-, 37-, and 18-day DCD inhibitors half-lives (t½) in soils at 5 °C, 15 °C, and 25 °C, respectively. Similarly, Kelliher et al. (2008) found DCD inhibitors’ t½ values in the soil to be 110 days at 5 °C, compared to 20 days at 25 °C. NI + UI’s more prolonged efficacy at colder temperatures ensures the NI stays effective, particularly during the spring thaw period when N loss potential is typically high.
We further separated fall fertilizer + inhibitor application studies (Ding et al. 2015; Lin et al. 2017; Parkin and Hatfield 2010) from the rest of the inhibitor studies to investigate the effect size of fall application vs. prior spring application. We found that fall and prior spring application of inhibitors significantly reduced over-winter emissions by 12 and 14%, respectively. Fall-applied inhibitors likely remained active by the following spring given their shorter duration and the low temperature during winter, explaining their effectiveness in significantly lowering over-winter N2O emissions. However, the reason for the effectiveness of inhibitors applied in prior spring remains unclear given that the inhibitors should have degraded by the winter season. Additionally, it is hypothesized that the use of inhibitors, causing more N uptake by plants, could lower the C:N ratio in residues after harvest (Grant et al. 2020; Pfab et al. 2012). Ferrari Machado et al. (2020) suggested that the carryover effect of inhibitor treatments increasing soil N levels could be negligible after observing low N2O fluxes during both the growing and non-growing seasons compared to the conventional N sources. Other researchers have attributed lower over-winter emissions in NI + UI plots to the reduction of denitrifying bacteria rather than \({\mathrm{NO}}_{3}^{-}\) availability, since \({\mathrm{NO}}_{3}^{-}\) levels were the same as those found in conventional plots (Pfab et al. 2012). However, a contradictory finding shows a NI such as DMPP to have significantly reduced \({\mathrm{NH}}_{4}^{+}\) oxidation on grasslands, though it showed no adverse effect on the denitrifier community’s growth and activity (Duan et al. 2017). More studies are needed to understand the carryover effect of spring-applied inhibitors on over-winter N2O emissions. Further research could focus on the effect of inhibitors on the microbial community.
The result of our meta-analysis showed that soils with medium texture significantly influenced the efficacy of NI + UI in reducing N2O emissions more than coarse soils (Fig. 4). Medium-textured soils have larger surface area which allows the soil to hold more water. NI + UI’s effectiveness has been tied to WFPS, Lin et al. (2017) showing a wetter site to exhibit a noticeable beneficial effect of NI compared to a less wet site. This scenario was due to increased N2O production in the control plots resulting from high soil WFPS compared with the lower N2O production in NI plots where fall-added NIs prevented the oxidation of manures. This impact of WFPS on NI + UI’s efficacy may also be the reason for the lower N2O emissions observed in humid areas compared to dry areas. Similarly, Lin and Hernandez-Ramirez (2020) observed a greater NI efficacy in soils with increasing moisture content when the potential for N2O production was high. However, after a few days, when WFPS surpassed field capacity, NI’s potential to reduce N2O emissions from the soil diminished. This reduced efficiency was possibly due to the complete denitrification of \({\mathrm{NO}}_{3}^{-}\) to N2 in control plots leading to lower N2O emissions or the lapsing of NI effectiveness in NI treatment plots, giving rise to the possible oxidation of \({\mathrm{NH}}_{4}^{+}\) to N2O. Increasing NI rates has been recommended to ensure NI’s continued efficacy in soils with high moisture content. For instance, the most efficient NI rates were achieved by increasing the nitrapyrin rate by 25% as soil moisture increased from 60 to 80% WFPS (Lin and Hernandez-Ramirez 2020).
However, caution must be applied interpreting these results as NI + UI efficacy to inhibit \({\mathrm{NO}}_{3}^{-}\) production may also be affected by other soil properties, including organic matter, clay content, soil Cu, and total N (McGeough et al. 2016). For instance, NI’s lower inhibitory efficiency has been reported to result from high rates of adsorption by clay and SOC (Zhu et al. 2019). Therefore, it may be the case that there are variations in environmental and soil factors such as moisture, temperature, substrate availability, and management decisions such as timing, quantity, and placement of the NI + UI, that could affect the efficacy of NI + UI. Further studies investigating how the combination of these factors interacts with various NI rates over winter are needed. Given the potential trade-offs of NI application, including an increase in NH3 emissions and possible NI contamination, it is essential that studies also account for volatilization and N-leaching in addition to N2O measurements. Overall, NI + UI practice significantly reduces both over-winter and growing season N2O emissions. NI + UIs have also been shown to increase NUE use by 13% and boost yield by an average of 7.5%, and by adopting NI + UI practice, farmers could potentially gain up to US$6 per ha (Kanter and Searchinger 2018).
Conclusions
Occurring predominantly at the onset of the winter freezing period and in early spring when the soil begins to thaw, over-winter N2O emissions make up a significant portion (up to 91%) of annual cropland emissions. We discussed the overall efficacy of nitrification and urease inhibitors (NI + UI), no-till, and cover crops (CCs) to mitigate emissions. We show that while no-till and the use of NI + UI significantly reduced over-winter N2O emissions by 28% and 23%, respectively, the use of CCs overall did not cause any significant change in the emissions. Although CCs have the potential to mitigate N2O emissions by assimilating \({\mathrm{NO}}_{3}^{-}\) over winter, the production of denitrification substrates in the cover crops’ root zone could hinder this potential. This review revealed that cold-dry soil conditions, such as in Western Canada, could prevent CCs from being fully established in some years, hampering this management practice’s effectiveness for reducing N2O emissions in these regions. To effectively mitigate the significant freeze–thaw emissions, we recommend the enactment of policies that will encourage farmers’ adoption of NI + UI in addition to other management practices such as no-till and the use of cover crops. We highlight the need for more extensive research focusing on the effect of the interaction of soil and climate conditions with field practices on the magnitude of emissions. Further studies could focus on the roles of denitrifying and nitrifying genes on over-winter N2O emissions.
Data availability
The datasets generated and analyzed during this study are included in the manuscript. Any queries for the datasets can be directed to the corresponding author.
Code availability
The R software package metafor 2.4–0 was used for all analyses performed in this study. The code can be made available by the corresponding author upon reasonable request.
References
Asgedom H, Tenuta M, Flaten DN, Gao X, Kebreab E (2014) Nitrous oxide emissions from a clay soil receiving granular urea formulations and dairy manure. Agron J 106:732–744. https://doi.org/10.2134/agronj2013.0096
Basche AD, Miguez FE, Kaspar TC, Castellano MJ (2014) Do cover crops increase or decrease nitrous oxide emissions? A meta-analysis. J Soil Wat Conserv 69:471–482. https://doi.org/10.2489/jswc.69.6.471
Behnke GD, Villamil MB (2019) Cover crop rotations affect greenhouse gas emissions and crop production in Illinois, USA. Field Crops Res 241:107580. https://doi.org/10.1016/j.fcr.2019.107580
Bent E, Németh D, Wagner-Riddle C, Dunfield K (2016) Residue management leading to higher field-scale N2O flux is associated with different soil bacterial nitrifier and denitrifier gene community structures. Appl Soil Ecol 108:288–299. https://doi.org/10.1016/j.apsoil.2016.09.008
Burton DL, Beauchamp EG (1994) Profile nitrous oxide and carbon dioxide concentrations in a soil subject to freezing. Soil Sci Soc Am J 58:115–122. https://doi.org/10.2136/sssaj1994.03615995005800010016x
Chantigny MH, Rochette P, Angers DA, Goyer C, Brin LD, Bertrand N (2016) Nongrowing season N2O and CO2 emissions — temporal dynamics and influence of soil texture and fall-applied manure. Can J Soil Sci 97:452–464. https://doi.org/10.1139/cjss-2016-0110
Chen L, Chen Z, Jia G, Zhou J, Zhao J, Zhang Z (2020) Influences of forest cover on soil freeze-thaw dynamics and greenhouse gas emissions through the regulation of snow regimes: a comparison study of the farmland and forest plantation. Sci Tot Environ 726:138403. https://doi.org/10.1016/j.scitotenv.2020.138403
Chen Z, Ding W, Xu Y, Müller C, Yu H, Fan J (2016) Increased N2O emissions during soil drying after waterlogging and spring thaw in a record wet year. Soil Biol Biochem 101:152–164. https://doi.org/10.1016/j.soilbio.2016.07.016
Congreves KA, Brown SE, Németh DD, Dunfield KE, Wagner-Riddle C (2017) Differences in field-scale N2O flux linked to crop residue removal under two tillage systems in cold climates. GCB Bioenerg 9:666–680. https://doi.org/10.1111/gcbb.12354
Davidson EA, Kanter D (2014) Inventories and scenarios of nitrous oxide emissions. Environ Res Lett 9:105012. https://doi.org/10.1088/1748-9326/9/10/105012
Decock C (2014) Mitigating nitrous oxide emissions from corn cropping systems in the Midwestern U.S.: potential and data gaps. Environ Sci Technol 48:4247–4256. https://doi.org/10.1021/es4055324
Dietzel R, Wolfe D, Thies JE (2011) The influence of winter soil cover on spring nitrous oxide emissions from an agricultural soil. Soil Biol Biochem 43:1989–1991. https://doi.org/10.1016/j.soilbio.2011.05.017
Ding WX, Chen ZM, Yu HY, Luo JF, Yoo GY, Xiang J, Zhang HJ, Yuan JJ (2015) Nitrous oxide emission and nitrogen use efficiency in response to nitrophosphate, N-(n-butyl) thiophosphoric triamide and dicyandiamide of a wheat cultivated soil under sub-humid monsoon conditions. Biogeosciences 12:803–815. https://doi.org/10.5194/bg-12-803-2015
Dong D, Kou Y, Yang W, Chen G, Xu H (2018) Effects of urease and nitrification inhibitors on nitrous oxide emissions and nitrifying/denitrifying microbial communities in a rainfed maize soil: a 6-year field observation. Soil till Res 180:82–90. https://doi.org/10.1016/j.still.2018.02.010
Duan YF, Kong XW, Schramm A, Labouriau R, Eriksen J, Petersen SO (2017) Microbial N transformations and N2O emission after simulated grassland cultivation: effects of the nitrification. Appl Environ Microbiol 83:1–17. https://doi.org/10.1016/j.tetasy.2008.05.028
Ejack L, Whalen JK (2021) Freeze-thaw cycles release nitrous oxide produced in frozen agricultural soils. Biol Fertil Soils 57:389–398. https://doi.org/10.1007/s00374-020-01537-x
Elmi AA, Madramootoo C, Hamel C, Liu A (2003) Denitrification and nitrous oxide to nitrous oxide plus dinitrogen ratios in the soil profile under three tillage systems. Biol Fertil Soils 38:340–348. https://doi.org/10.1007/s00374-003-0663-9
Feng J, Li F, Deng A, Feng X, Fang F, Zhang W (2016) Integrated assessment of the impact of enhanced-efficiency nitrogen fertilizer on N2O emission and crop yield. Agric Ecosyst Environ 231:218–228. https://doi.org/10.1016/j.agee.2016.06.038
Ferrari Machado PV, Farrell RE, Bell G, Taveira CJ, Congreves KA, Voroney RP, Deen W, Wagner-Riddle C (2021) Crop residues contribute minimally to spring-thaw nitrous oxide emissions under contrasting tillage and crop rotations. Soil Biol Biochem 152:108057. https://doi.org/10.1016/j.soilbio.2020.108057
Ferrari Machado PV, Neufeld K, Brown SE, Voroney PR, Bruulsema TW, Wagner-Riddle C (2020) High temporal resolution nitrous oxide fluxes from corn (Zea mays L.) in response to the combined use of nitrification and urease inhibitors. Agric Ecosyst Environ 300:106996. https://doi.org/10.1016/j.agee.2020.106996
Feng J, Li F, Zhou X, Xu C, Ji L, Chen Z, Fang F (2018) Impact of agronomy practices on the effects of reduced tillage systems on CH4 and N2O emissions from agricultural fields: A global meta-analysis. PLoS ONE 13(5):e0196703. https://doi.org/10.1371/journal.pone.0196703
Gillette K, Malone RW, Kaspar TC, Ma L, Parkin TB, Jaynes DB, Fang QX, Hatfield JL, Feyereisen GW, Kersebaum KC (2018) N loss to drain flow and N2O emissions from a corn-soybean rotation with winter rye. Sci Tot Environ 618:982–997. https://doi.org/10.1016/j.scitotenv.2017.09.054
Grant RF, Lin S, Hernandez-Ramirez G (2020) Modelling nitrification inhibitor effects on N2O emissions after fall-and spring-applied slurry by reducing nitrifier NH4+ oxidation rate. Biogeosciences 17:2021–2039. https://doi.org/10.5194/bg-17-2021-2020
Gregorich E, Janzenx HH, Helgason B, Ellert B (2015) Nitrogenous gas emissions from soils and greenhouse gas effects. Adv in Agron 132:39–74. https://doi.org/10.1016/bs.agron.2015.02.004
Guardia G, Abalos D, García-Marco S, Quemada M, Alonso-Ayuso M, Cárdenas LM, Dixon ER, Vallejo A (2016) Effect of cover crops on greenhouse gas emissions in an irrigated field under integrated soil fertility management. Biogeosciences 13:5245–5257. https://doi.org/10.5194/bg-13-5245-2016
Halvorson AD, Del Grosso SJ (2012) Nitrogen source and placement effects on soil nitrous oxide emissions from no-till corn. J Environ Qual 41:1349–1360. https://doi.org/10.2134/jeq2012.0129
Huang Y, Ren W, Wang L, Hui D, Grove JH, Yang X, Tao B, Goff B (2018) Greenhouse gas emissions and crop yield in no-tillage systems: a meta-analysis. Agric Ecosyst Environ 268:144–153. https://doi.org/10.1016/j.agee.2018.09.002
Hung CY, Ejack L, Whalen JK (2021) Fall-applied manure with cover crop did not increase nitrous oxide emissions during spring freeze-thaw periods. Appl Soil Ecol 158:103786. https://doi.org/10.1016/j.apsoil.2020.103786
Iqbal J, Mitchell DC, Barker DW, Miguez F, Sawyer JE, Pantoja J, Castellano MJ (2015) Does nitrogen fertilizer application rate to corn affect nitrous oxide emissions from the rotated soybean crop? J Environ Qual 44:711–719. https://doi.org/10.2134/jeq2014.09.0378
Jarecki MK, Parkin TB, Chan AS, Kaspar TC, Moorman TB, Singer JW, Kerr BJ, Hatfield JL, Jones R (2009) Cover crop effects on nitrous oxide emission from a manure-treated Mollisol. Agric, Ecosyst Environ 134:29–35. https://doi.org/10.1016/j.agee.2009.05.008
Kanter DR, Searchinger TD (2018) A technology-forcing approach to reduce nitrogen pollution. Nat Sustain 1:544–552. https://doi.org/10.1038/s41893-018-0143-8
Kelliher FM, Clough TJ, Clark H, Rys G, Sedcole JR (2008) The temperature dependence of dicyandiamide (DCD) degradation in soils: a data synthesis. Soil Biol Biochem 40:1878–1882. https://doi.org/10.1016/j.soilbio.2008.03.013
Kim N, Zabaloy MC, Guan K, Villamil MB (2020) Do cover crops benefit soil microbiome? A meta-analysis of current research. Soil Biol Biochem 142:107701. https://doi.org/10.1016/j.soilbio.2019.107701
Larsen KS, Jonasson S, Michelsen A (2002) Repeated freeze-thaw cycles and their effects on biological processes in two arctic ecosystem types. Appl Soil Ecol 21:187–195. https://doi.org/10.1016/S0929-1393(02)00093-8
Lemke RL, Izaurralde RC, Nyborg M, Solberg ED (1999) Tillage and N source influence soil-emitted nitrous oxide in the Alberta Parkland region. Can J Soil Sci 79:15–24. https://doi.org/10.4141/S98-013
Li K, Gong Y, Song W, Lv J, Chang Y, Hu Y, Tian C, Christie P, Liu X (2012) No significant nitrous oxide emissions during spring thaw under grazing and nitrogen addition in an alpine grassland. Glob Chang Biol 18:2546–2554. https://doi.org/10.1111/j.1365-2486.2012.02704.x
Lin S, Hernandez-Ramirez G (2020) Nitrous oxide emissions from manured soils as a function of various nitrification inhibitor rates and soil moisture contents. Sci Tot Environ 738:139669. https://doi.org/10.1016/j.scitotenv.2020.139669
Lin S, Hernandez-Ramirez G, Kryzanowski L, Wallace T, Grant R, Degenhardt R, Berger N, Lohstraeter G, Powers L (2017) Timing of manure injection and nitrification inhibitors impacts on nitrous oxide emissions and nitrogen transformations in a barley crop. Soil Sci Soc Am J 81:1595–1605. https://doi.org/10.2136/sssaj2017.03.0093
Linn DM, Doran JW (1984) Effect of water-filled pore space on carbon dioxide and nitrous oxide production in tilled and nontilled soils. Soil Sci Soc Am J 48:1267–1272. https://doi.org/10.2136/sssaj1984.03615995004800060013x
Ludwig B, Teepe R, Lopes de Gerenyu V, Flessa H (2006) CO2 and N2O emissions from gleyic soils in the Russian tundra and a German forest during freeze-thaw periods-a microcosm study. Soil Biol Biochem 38:3516–3519. https://doi.org/10.1016/j.soilbio.2006.06.006
Ludwig B, Wolf I, Teepe R (2004). Contribution of nitrification and denitrification to the emission of N2O in a freeze-thaw event in an agricultural soil. J Plant Nutr Soil Sci 167: 678–684. https://doi.org/10.1002/jpln.200421462
Maljanen M, Kohonen AR, Virkajärvi P, Martikainen PJ (2007) Fluxes and production of N2O, CO2 and CH4 in boreal agricultural soil during winter as affected by snow cover. Tellus Ser B Chem Phys Meteorol 59:853–859. https://doi.org/10.1111/j.1600-0889.2007.00304.x
McGeough KL, Watson CJ, Müller C, Laughlin RJ, Chadwick DR (2016) Evidence that the efficacy of the nitrification inhibitor dicyandiamide (DCD) is affected by soil properties in UK soils. Soil Biol Biochem 94:222–232. https://doi.org/10.1016/j.soilbio.2015.11.017
Mitchell DC, Castellano MJ, Sawyer JE, Pantoja J (2013) Cover crop effects on nitrous oxide emissions: role of mineralizable carbon. Soil Sci Soc Am J 77:1765–1773. https://doi.org/10.2136/sssaj2013.02.0074
Mutegi JK, Munkholm LJ, Petersen BM, Hansen EM, Petersen SO (2010) Nitrous oxide emissions and controls as influenced by tillage and crop residue management strategy. Soil Biol Biochem 42:1701–1711. https://doi.org/10.1016/j.soilbio.2010.06.004
Nannipieri P, Ascher-Jenull J, Ceccherini MT, Pietramellara G, Renella G, Schloter M (2020) Beyond microbial diversity for predicting soil functions: a mini review. Pedosphere 30:5–17. https://doi.org/10.1016/S1002-0160(19)60824-6
Nyborg M, Laidlaw JW, Solberg ED, Malhi SS (1997) Denitrification and nitrous oxide emissions from a black chernozemic soil during spring thaw in Alberta. Can J Soil Sci 77:153–160. https://doi.org/10.4141/S96-105
Parkin TB, Hatfield JL (2010) Influence of nitrapyrin on N2O losses from soil receiving fall-applied anhydrous ammonia. Agric, Ecosyst Environ 136:81–86. https://doi.org/10.1016/j.agee.2009.11.014
Parkin TB, Kaspar TC, Jaynes DB, Moorman TB (2016) Rye cover crop effects on direct and indirect nitrous oxide emissions. Soil Sci Soc Am J 80:1551–1559. https://doi.org/10.2136/sssaj2016.04.0120
Pelster DE, Chantigny MH, Rochette P, Angers DA, Laganière J, Zebarth B, Goyer C (2013) Crop residue incorporation alters soil nitrous oxide emissions during freeze-thaw cycles. Can J Soil Sci 93:415–425. https://doi.org/10.4141/CJSS2012-043
Petersen SO, Mutegi JK, Hansen EM, Munkholm LJ (2011) Tillage effects on N2O emissions as influenced by a winter cover crop. Soil Biol Biochem 43:1509–1517. https://doi.org/10.1016/j.soilbio.2011.03.028
Pfab H, Palmer I, Buegger F, Fiedler S, Müller T, Ruser R (2012) Influence of a nitrification inhibitor and of placed N-fertilization on N2O fluxes from a vegetable cropped loamy soil. Agric Ecosyst Environ 150:91–101. https://doi.org/10.1016/j.agee.2012.01.001
Preza-Fontes G, Tomlinson PJ, Roozeboom KL, Warren J, Ruiz Diaz DA (2020) Nitrogen fertilization offsets the N2O mitigating effects of cover-crops and double-crop soybean in a wheat–sorghum system. Agron J 112:772–785. https://doi.org/10.1002/agj2.20095
Priemé A, Christensen S (2001) Natural perturbations, drying-wetting and freezing-thawing cycles, and the emission of nitrous oxide, carbon dioxide and methane from farmed organic soils. Soil Biol Biochem 33:2083–2091. https://doi.org/10.1016/S0038-0717(01)00140-7
Qiao C, Liu L, Hu S, Compton JE, Greaver TL, Li Q (2015) How inhibiting nitrification affects nitrogen cycle and reduces environmental impacts of anthropogenic nitrogen input. Glob Chang Biol 21:1249–1257. https://doi.org/10.1111/gcb.12802
Risk N, Wagner-Riddle C, Furon A, Warland J, Blodau C (2014) Comparison of simultaneous soil profile N2O concentration and surface N2O flux measurements overwinter and at spring thaw in an agricultural soil. Soil Sci Soc Am J 78:180–193. https://doi.org/10.2136/sssaj2013.06.0221
Schimel JP, Bennett J (2004) Nitrogen mineralization: challenges of a changing paradigm. Ecol 85:591–602
Sehy U, Dyckmans J, Ruser R, Munch JC (2004) Adding dissolved organic carbon to simulate freeze-thaw related N 2O emissions from soil. J Plant Nutr Soil Sci 167:471–478. https://doi.org/10.1002/jpln.200421393
Shackelford GE, Kelsey R, Dicks LV (2019) Effects of cover crops on multiple ecosystem services: ten meta-analyses of data from arable farmland in California and the Mediterranean. Land Use Policy 88:104204. https://doi.org/10.1016/j.landusepol.2019.104204
Shakoor A, Shahbaz M, Farooq TH, Sahar NE, Shahzad SM, Altaf MM, Ashraf M (2021) A global meta-analysis of greenhouse gases emission and crop yield under no-tillage as compared to conventional tillage. Sci Tot Environ 750:142299. https://doi.org/10.1016/j.scitotenv.2020.142299
Sharma S, Szele Z, Schilling R, Munch JC, Schloter M (2006) Influence of freeze-thaw stress on the structure and function of microbial communities and denitrifying populations in soil. Appl Environ Microbiol 72:2148–2154. https://doi.org/10.1128/AEM.72.3.2148
Signor D, Cerri CEP (2013) Nitrous oxide emissions in agricultural soils: a review. Pesqui Agropecu Trop 43:322–338. https://doi.org/10.1590/S1983-40632013000300014
Six J, Ogle SM, Breidt FJ, Conant RT, Mosiers AR, Paustian K (2004) The potential to mitigate global warming with no-tillage management is only realized when practised in the long term. Glob Chang Biol 10:155–160. https://doi.org/10.1111/j.1529-8817.2003.00730.x
Smith J, Wagner-Riddle C, Dunfield K (2010) Season and management related changes in the diversity of nitrifying and denitrifying bacteria over winter and spring. Appl Soil Ecol 44:138–146. https://doi.org/10.1016/j.apsoil.2009.11.004
Smukler SM, O’Geen AT, Jackson LE (2012) Assessment of best management practices for nutrient cycling: a case study on an organic farm in a Mediterranean-type climate. J Soil Wat Conserv 67:16–31. https://doi.org/10.2489/jswc.67.1.16
Teepe R, Brumme R, Beese F (2001) Nitrous oxide emissions from soil during freezing and thawing periods. Soil Biol Biochem 33:1269–75. https://doi.org/10.1016/S0038-0717(01)00084-0
Thapa R, Chatterjee A, Awale R, McGranahan DA, Daigh A (2016) Effect of enhanced efficiency fertilizers on nitrous oxide emissions and crop yields: a meta-analysis. Soil Sci Soc Am J 80:1121–1134. https://doi.org/10.2136/sssaj2016.06.0179
Thomas BW, Hao X, Larney FJ, Goyer C, Chantigny MH, Charles A (2017) Non-legume cover crops can increase non-growing season nitrous oxide emissions. Soil Sci Soc Ame J 81:189–199. https://doi.org/10.2136/sssaj2016.08.0269
Ussiri DAN, Lal R, Jarecki MK (2009) Nitrous oxide and methane emissions from long-term tillage under a continuous corn cropping system in Ohio. Soil till Res 104:247–255. https://doi.org/10.1016/j.still.2009.03.001
Vallejo A, Diez JA, López-Valdivia LM, Gascó A, Jiménez C (2001) Nitrous oxide emission and denitrification nitrogen losses from soils treated with isobutylenediurea and urea plus dicyandiamide. Biol Fertil Soils 34:248–257. https://doi.org/10.1007/s003740100409
van Groenigen JW, Zwart KB, Harris D, van Kessel C (2005) Vertical gradients of Δ15N and Δ18O in soil atmospheric N2O - temporal dynamics in a sandy soil. Rapid Commun Mass Spectrom 19:1289–1295. https://doi.org/10.1002/rcm.1929
van Kessel C, Venterea R, Six J, Adviento-Borbe MA, Linquist B, van Groenigen KJ (2013) Climate, duration, and N placement determine N2O emissions in reduced tillage systems: a meta-analysis. Glob Chang Biol 19:33–44. https://doi.org/10.1111/j.1365-2486.2012.02779.x
VanderZaag AC, Jayasundara S, Wagner-Riddle C (2011) Strategies to mitigate nitrous oxide emissions from land applied manure. Anim Feed Sci Technol 166–167:464–479. https://doi.org/10.1016/j.anifeedsci.2011.04.034
Wagner-Riddle C, Congreves KA, Abalos D, Berg AA, Brown SE, Ambadan JT, Gao X, Tenuta M (2017) Globally important nitrous oxide emissions from croplands induced by freeze-thaw cycles. Nat Geosci 10:279–283. https://doi.org/10.1038/ngeo2907
Wagner-Riddle C, Furon A, Mclaughlin NL, Lee I, Barbeau J, Jayasundara S, Parkin G, Bertoldi P, Warland J (2007) Intensive measurement of nitrous oxide emissions from a corn-soybean-wheat rotation under two contrasting management systems over 5 years. Glob Chang Biol 13:1722–1736. https://doi.org/10.1111/j.1365-2486.2007.01388.x
Wagner-Riddle C, Hu QC, van Bochove E, Jayasundara S (2008) Linking nitrous oxide flux during spring thaw to nitrate denitrification in the soil profile. Soil Sci Soc Am J 72:908–916. https://doi.org/10.2136/sssaj2007.0353
Wertz S, Goyer C, Zebarth BJ, Tatti E, Burton DL, Chantigny MH, Filion M (2016) The amplitude of soil freeze-thaw cycles influences temporal dynamics of N2O emissions and denitrifier transcriptional activity and community composition. Biol Fertil Soils 52:1149–1162. https://doi.org/10.1007/s00374-016-1146-0
Yang M, Fang Y, Sun D, Shi Y (2016) Efficiency of two nitrification inhibitors (dicyandiamide and 3, 4-dimethypyrazole phosphate) on soil nitrogen transformations and plant productivity: a meta-analysis. Sci Rep 6:1–10. https://doi.org/10.1038/srep22075
Yin M, Gao X, Tenuta M, Gui D, Zeng F (2019) Presence of spring-thaw N2O emissions are not linked to functional gene abundance in a drip-fertigated cropped soil in arid Northwestern China. Sci Tot Environ 695:133670. https://doi.org/10.1016/j.scitotenv.2019.133670
Zhang B, Penton CR, Yu Z, Xue C, Chen Q, Chen Z, Yan C, Zhang Q, Zhao M, Quensen JF, Tiedje JM (2021) A new primer set for clade I nosZ that recovers genes from a broader range of taxa. Biol Fertil Soils 57:523–531. https://doi.org/10.1007/s00374-021-01544-6
Zhang M, Wang W, Tang L, Heenan M, Xu Z (2018) Effects of nitrification inhibitor and herbicides on nitrification, nitrite and nitrate consumptions and nitrous oxide emission in an Australian sugarcane soil. Biol Fertil Soils 54:697–706
Zhe C, Shi-qi Y, Ai-ping Z, Xin J, Wei-min S, Zhao-rong MI, Qing Z (2018) Nitrous oxide emissions following seasonal freeze-thaw events from arable soils in Northeast China. J Integr Agric 17:231–246. https://doi.org/10.1016/S2095-3119(17)61738-6
Zhu G, Ju X, Zhang J, Müller C, Rees RM, Thorman RE, Sylvester-Bradley R (2019) Effects of the nitrification inhibitor DMPP (3,4-dimethylpyrazole phosphate) on gross N transformation rates and N2O emissions. Biol Fertil Soils 55:603–615. https://doi.org/10.1007/s00374-019-01375-6
Acknowledgements
We express our gratitude to the authors of the studies from which data used in this review were extracted. We thank Aidan De Sena and Georges T. Dodds for reviewing the manuscript. We also thank Agriculture and Agri-Food Canada (AAFC) for funding the project.
Funding
The research project to produce these results was the Agricultural Greenhouse Gases Program (AGGP). This project’s fund was disseminated by Agriculture and Agri-Food Canada (AAFC) – Project Number: AGGP-020.
Author information
Authors and Affiliations
Contributions
Kosoluchukwu C. Ekwunife curated data, performed the analysis, and wrote the paper. Chandra A. Madramootoo outlined scope of the study and reviewed and edited all drafts. Naeem A. Abbasi curated data and provided technical comments. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ekwunife, K.C., Madramootoo, C.A. & Abbasi, N.A. Assessing the impacts of tillage, cover crops, nitrification, and urease inhibitors on nitrous oxide emissions over winter and early spring. Biol Fertil Soils 58, 195–206 (2022). https://doi.org/10.1007/s00374-021-01605-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-021-01605-w