Abstract
Pit building antlions Euroleon nostras have been submitted to artificial cues in order to delineate their faculty to localize a prey. Series of propagating pulses in sand have been created from an extended source made of 10 piezoelectric transducers equally spaced on a line and located at a large distance from the pit. The envelope of each pulse encompasses six oscillations at a carrier frequency of 1250 Hz and up to eight oscillations at 1666 Hz. In one set of experiments, the first wave front is followed by similar wave fronts and the antlions respond to the cue by throwing sand in the opposite direction of the wave front propagation direction. In another set of experiments, the first wave front is randomly spatially structured while the propagation of the wave fronts inside the envelope of the pulse are not. In that case, the antlions respond less to the cue by throwing sand, and when they do, their sand throwing is more randomly distributed in direction. The finding shows that the localization of vibration signal by antlions are based on the equivalent for hearing animals of interaural time difference in which the onset has more significance than the interaural phase difference.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
All vertebrates possess auditory senses and have been extensively studied in their capacity to hear, select and differentiate auditory sources (Popper and Fay 2014). The human hearing is particularly studied for obvious anthropocentric reasons that encompass not only health and medicine but also military purposes and more than all the video and audio business (Blauert and Braasch 2020). Some vertebrates enrich also their audio sensory life with an extension to vibration senses (e.g., some marsupials, golden moles, some rodents and elephants (Narins et al. 2016). Arthropods on the other hand do not all possess sensory organs for airborne vibrations but some are equipped for the detection of vibrations propagating at the surface of solid or liquid media (Strauß et al. 2021). The functional reasons for such sensing remain quite similar to the vertebrate life: for intraspecific communication, to distinct between friend or foe and to identify a source of vibration i.e. to identify and localize a prey. To identify a prey suggests that a prey possesses its own auditory or vibratory signature, to localize it requires a spatially extended sensory device that enables the recognition of a spatial environment. In that matter arthropods sensitive to airborne vibration possess different physiological responses compared with human ears. With insects one has to consider two categories of organs: those that respond to particle velocity components, the others responding to pressure. One has also to keep in mind the huge scale difference between the wavelength of the signal being way larger than the their head or body spatial extent (Romer 2020). While directionality of airborne mechanical signals in arthropods has been well studied, localization of sources of mechanical signals transmitted by fluid or solid media was given less attention (Cocroft et al. 2014). Several studies demonstrate that arthropod species are able to localize vibrational signal source in solid media (Virant-Doberlet et al. 2006; Hager et al. 2019). Two main strategies are employed by arthropods in this regard, klinotactic directional orientation (comparing measurements over time) or tropotactic directional orientation (simultaneous comparison of signals arriving at two or more receptors) (Hager et al. 2019).
We address in the present paper an analysis of the localization capacity of substrate borne vibrations of the antlion larvae Euroleon nostras when the source of vibration is at large distance from the prey. The larvae are sit and wait predators that live on catching prey falling down into their trap. Alerted by the avalanche caused by this fall, the arrival of solid material (sand particles) on any part of the body of the antlion larvae makes a good enough signal. Often the success of catching prey relies on the determination of throwing sand from the bottom of the trap, apparently in any direction, which works sufficiently well for the first shovelling since the sand funnel is quite unstable but does not work that well in a repeated gesture of sand throwing, unless the antlions aims at the prey. It is worth noting that cues external to the pit also induce an attack behavior (Devetak et al. 2007; Martinez et al. 2020). Aiming the prey means that the antlion recognizes where its signal comes from. To address this question, experiments have been performed with natural prey placed outside of the sand funnel with antlions that have been obliterated of their visual sense (Mencinger-Vračko and Devetak 2008). Also, experiments with artificial cues placed inside the sand trap have been performed by Fertin and Casas (2007). In both experiments, the antlion throws sand in the direction of the prey or the cue. Those preys or cues are fairly modelized as point sources. These experiments conclude to a faculty of stereo sensing the vibrations emitted by the prey, i.e. tropotaxis. Tufts of hair on lateral parts of the mesothorax and methathorax of antlions are very likely to detect vibrational signals (Devetak 2014).
The present experiment brings a refined conception of spatially discriminating a vibration signal by the use of an artificial cue produced by a spatially extended source. The antlions were submitted to an artificial cue coming from a line of emittors. Each emittor is a piezo-electric transducer (PZT) with each PZT equaly distant to another. The ensemble is similar of a phased array used in radar systems or ultrasonics sonar systems. In a first set of experiment all of the PZT were driven in phase and produced a plane wave. In a second set of experiment the same device was used with a phase delay between the PZT in order to structure the first wave front of the signal. For each set of experiment the attack behaviour, i.e. sand tossing in response of the cue was observed and analysed in its direction and occurrence. These two sets of experiments differed essentially by one feature: or all the wave fronts inside the envelope of an emitted pulse followed each other identically, that is the first set of experiment, or the wave fronts inside a pulse followed identically, except for the wave front ahead of the pulse. This difference was at the basis of this study: to discriminate in the time difference of arrival of the signal considered to be the support of the spatial localization. If it is the time interval between two successive wavefronts, i.e. a phase time delay, the equivalent of the interaural phase difference (IPD) in airborne acoustic, or if it is the time interval of arrival of the first outbreak, i.e. the onset time difference (OTD), that is operating in the spatial localization. One can find similarities in the method developed in the present paper with those used in a recent study of the faculty of directional hearing in the fly Ormia ochracea (Mason 2021). While in this last study the responses to stimuli were investigated in behavior, mechanical and neural physiology, our study was restricted to the behavioral responses only. One emphasizes that, counter to airborne acoustic, the physical specificity of soilborne signal propagation had to be taken into account. In particular, the phase velocity differs from the energy propagation velocity of the wave. In this extend, physical considerations of interest in the field of biotremology can be found in Michelsen (2014), Mortimer (2017), Prešern et al. (2018).
Material and methods
Construction of the artificial cue
The device is presented in Fig. 1: the extended source of vibration is a line of ten piezo electric transducers (PZT) (Murata PKMCS0909E4000-R1) soldered on an electronic stripboard (Fig. 1A). The centers of the PZT are \(10.3 \, \textrm{mm}\) mutually distant. The line of PZTs, now referred as the antenna, was placed at an extremity of a container (size: \(52 \, \textrm{cm} \times 24 \, \textrm{cm}\) filled with \(7 \, \textrm{cm}\) deep sand with the PZTs facing down the sand surface (Fig. 1B). The experiments were conducted in parallell in Villetaneuse (France) and Maribor (Slovenia). In Villetaneuse, two similar copies of this device have been made and installed in a \(40 \, \textrm{dB}\) sound isolated chamber with an anechoic wall separating each device. Two similar copies of the device were used in Maribor as well.
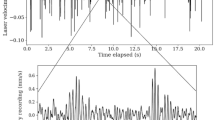
The PZTs were submitted to a square signal with a 600 or a 800 microsecond period. The ensemble was connected to two microprocessors (Arduino Uno), one driving a digital potentiometer defining a time window, the other producing a square signal for each PZT (see Supplementary Material-Stimuli). Considering that the frequency range of surface wave propagation in sand is upper bounded around \(4 \, \textrm{kHz}\) (Podlesnik et al. 2019), all harmonics of the square signal were damped, ensuring that only the fundamental frequency propagates in the medium. The fundamental frequencies of both square signals are within the range of the carrier frequency of the vibration signal produced by an ant walking on the sand surface (Fertin and Casas 2007; Devetak et al. 2007; Martinez et al. 2020). The amplitude of vibration of each PZT has been calibrated with a PDV 100 laser vibrometer (Polytec, GmbH, Waldbronn, Germany). The ensemble was assuring a vibration amplitude of a few nanometers at a distance of \(15 \, \textrm{cm}\) away from the antenna. Considering a phase velocity of \(120 \, \mathrm {m \, s^{-1}}\) at the frequency of \(1666 \, \textrm{Hz} \; (600 \, \upmu \textrm{s})\) period or \(1250 \, \textrm{Hz} \; (800 \, \upmu \textrm{s})\) period the wavelength of the propagating signal is between 7 to \(10 \, \textrm{cm}\). The amplitude of each PZT has been measured by laser velocimetry and leveled to a same amplitude. The overall amplitude of the resulting surface wave was kept in the 1–5 nanometer level, a value for which one is ensured of the response of the antlion within minutes at the start of the cue (Martinez et al. 2020).
Like in optics, albeit each PZT emittor is a source radiating equally in any direction, the sum of all these waves results in a propagation of a plane wave at distance of a few wavelengths beyond the PZT array. The cue was built with temporal windows opened in a duration of 5 milliseconds. Inside this temporal window a number of wave fronts propagate. The number of wave fronts are 6 in the case of a signal of \(1250 \, \textrm{Hz}\) carrier frequency and is 8 in the case of a signal of \(1666 \, \textrm{Hz}\) carrier frequency. These figures are consistent with the number of wave fronts occurring in the oscillation of the sand layer caused by the gait of an ant (Fertin and Casas 2007). This wave form is thereafter designated as a pulse (see Fig. 2 and Fig. SM2). The pulses were separated by time intervals randomly distributed between 20 and \(30 \, \textrm{ms}\). These time intervals has also been inspired by Fertin and Casas (2007). The details of the procedure are given in Supplementary Material-Stimuli.
Figure 3 is a simulation of the wave front propagation at the instant where the incoming wave front hits the antlion. In Fig. 3a, one sees the first wave front coming at the z coordinate where the antlion sits. Figure 3b is at a time successive to the first wave front arrival. Both situations inform about the same directionality. It is on that account impossible to discriminate between the OTD or IPD triggering the reaction of the antlion.
A plane wave is constructed by the synchronous vibration with a period of \(600 \,\upmu \textrm{s}\) of 10 PZTs aligned with the horizontal axis and regularly placed \(1.03 \,\textrm{cm}\) apart one for each other. The propagation of the plane wave is mainly in the direction perpendicular to the line of the PZTs. a the first wave front arrives at the level of the antlion body; b another wave front arrives at the level antlion body
On the other hand, when the extended source of vibration is used as a phased array (Visser 2005), by the fact that the pulse time interval does not produce an equiphase surface at the entrance (as in the exit) of the pulse, the signals for OTD and IPD differ qualitatively. Such phased array works on the principle of an addition of a given phase to the signal driving each emitter with respect to the adjacent emitter on one side. This phase shift is transmitted along the line: here each PZT vibrates with a delay of \(100 \,\upmu \textrm{s}\) with respect to the one on its side. This causes, by the addition of the individual wave fronts, a propagation of a corrugated wave front directed with an angle to the antenna axis. Figure 4 and Supplementary Material—Animation show the aspect of the first surface wave hitting the body of the antlion. Distinctively from the wave front propagation with all the PZTs vibrating in phase, this wave front is structured along the body of the antlion. The subsequent wave fronts are showing more regularity. In Fig. 4 the position of the antlion is chosen to exhibit the maximum of difference in order to illustrate the following consequence of this wave front construction. By the fact that the propagation is coming by pulses with randomized time interval between pulses, the initial phases of the pulses are equally distributed. As a consequence a pulse like the one illustrated in Fig. 4a can be followed by another pulse with an opposite phase and hence an opposite contrast of amplitude when it hits the antlion, as displayed in Fig. 4b. De facto, the succession of the global phase of the propagating wave is random in the succession of pulses. Hence, the amplitudes ridges of the onset waves are randomly distributed around the body of the antlion. If the information of localization was based on time difference of arrival of successive wave fronts on the respective sensory receptors, the jamming of the first arrival of the wave front would be of little consequence since it is followed by five to seven plane wave fronts. On the other hand if this time difference of arrival was based on the onset time difference, the signal received by the antlion being different from one pulse to another, the retrieved information would be equivocal. This is the basic principle of the tests on which the antlions have been submitted.
A wave is constructed by the synchronous vibration of 10 PZTs aligned as explained in the caption of Fig. 2. Here a time delay of 100 microseconds is imposed between to adjacent PZTs. As a result, the wave propagation is deformed with respect to the normal of the PZT line. a Propagation with some initial phase; b propagation with a \(180 ^\circ\) phase with respect to the phase in a
Experimental protocol
Experiments have been performed in Maribor with 25 antlions Euroleon nostras in sand of granular size 230–\(540 \,\upmu \textrm{m}\) and in Villetaneuse with 46 antlions Euroleon nostras in sand of granular size 160–\(200 \, \upmu \textrm{m}\), all in their third larvae stage. The antlion larvae develops through three stages. We kept on the third stage exclusively in order to align the results between the Villetaneuse and the Maribor experiments, though we realized afterwards that there was a difference of environment: the sand grains in Maribor was of larger size compared to the one used in Villetaneuse. This difference of size could have had a consequence in terms of rate of responses, as it has been studied by Klokočovnik et al. (2012). Both experiments used the same source of vibration placed 10 to 15 cm away of a single antlion larva.
An assay consisted to install one antlion inside a \(10 \, \textrm{cm}\) diameter plastic ring, placed \(15 \,\textrm{cm}\) away from the antenna and wait until it had created a pit. A minimum of five minutes later the plastic ring was removed of the sand surface. The video recording of was then started and the signal was triggered 40 min after. A signal consisted in five minutes repetition of a low amplitude signal pattern (around 1 nanometer amplitude) followed by five minutes repetition of a higher amplitude pattern (around 10 nanometer amplitude). The signal pattern is described in the section construction of the artificial cue.
In Villetaneuse, 36 individuals have experienced a control test for both situations (antenna with PZT oscillating in phase, antenna with PZT oscillating with a time delay). This control test consisted in setting the voltage to zero for the command of the pulse time window: hence, all pulses were with zero amplitude: this was to ensure that, despite eventual noises generated by the microprocessor and microcomputer powered up, no stimuli were present. All the experiments in Villetaneuse have been realized inside an acoustic chamber with 40 dB isolation for frequencies above 1000 Hz. In Maribor, the experiment was carried out in an anechoic chamber and the containers with sand were placed on vibration-free table. The measurement consisted in two observations: (i) the direction of sand tossing, considering the sand tossing correlated to the cue if this action was taking place in the time interval of the cue, (ii) the orientation, abdomen to head, of the antlion at the moment of sand tossing. The angular reference was the normal to the line of the antenna with the zero angle corresponding to sand tossed towards the antenna. Angles are positive in the clockwise direction.
The Villetaneuse experiment has been performed during five months, from July to November 2019. With the antenna in phase, 28 antlions have been submitted to the cue with the antenna emitting in phase generating a total of 443 sand tossing events while 28 antlions have been submitted to the dephased cue generating a total of 360 sand tossing events. Ten antlions have experienced both cues. Both assays were identical in numbers. The Maribor experiment has been performed during three months, from July to September 2020. 25 antlions have answered to the cue with the antenna emitting in phase with a total of 322 sand tossing events.
Data analysis
The GLMM analysis (R Core Team 2022) was used to confirm that the frequency of sand tossing by the antlion larvae was in correlation with artificial vibratory cue presented to the animals in comparison to the control test, in which the signal was played with zero amplitude (\(N=36\)). A random sample consensus (RANSAC) algorithm (Fischler and Bolles 1981) has been applied to the entire data respectively for the assays realized with the antenna emitting in phase and for dephased signal (time delay between the PZTs). It demonstrated a strong correlation between the sand tossing directions and the abdomen-to-head orientations: one is mainly in the opposite direction to the other, irrespective of the direction of propagation of the cue.
Circular statistics have been performed with the JASP circular statistics module (JASP 2023) and the astropy.stats. circstats module (Astropy Collaboration 2022) for the abdomen-to-head orientations of the antlion larvae and the sand throwing directions. We analysed data for the assays realized with the antenna emitting in phase (with carrier frequencies of 1250 and \(1666 \, \textrm{Hz}\)) and with a time delay between the PZTs, separately for a carrier frequency of \(1250\, \textrm{Hz}\) and a carrier frequency of \(1666 \, \textrm{Hz}\). The variables that we retained are angle mean value, mean resultant length, variance, Rayleigh test of uniformity, estimated \(\kappa\) parameter of the von Mises distribution and correlation between the abdomen-to-head direction and the sand tossing direction.
Results
For 36 antlions submitted to the control test and the actual test, the GLMM analysis based on this binomial variable has a \(\chi _1^2 = 14.02\) with \(P< 0.001\). The control test evidences that the sand tossing actions measured in the time window of the cue are correlated with the cue. Figure 5 represents the sand tossing direction versus the antlion abdomen to head orientation when the cue produced by the antenna corresponds to a surface wave propagation with all the emitters oscillating in phase, as explained in Fig. 2. At the carrier frequency of \(1666 \, \textrm{Hz}\), 285 events are considered (all recorded in Villetaneuse) and at the carrier frequency of \(1250 \,\textrm{Hz}\), 480 events are considered (158 in Villetaneuse and 322 in Maribor). One expects to observe a correlation between the sand tossing direction and the body orientation of the antlion. The question is to see if this correlation is regardless of the propagation direction of the cue. It has been observed in a precedent experiment that the projections of sands are mainly head to abdomen with an aperture angle of \(120 ^\circ\) angular (Martinez et al. 2020). If there is some plasticity in this behavior, one would notice some discrepancy to this behavior. The RANSAC algorithm evidences a reflexive head to abdomen sand tossing movement in both situations, irrespective of the body orientation of the antlion and of the type of cue. The results are shown in Fig. 5: (b) is similar to (a) with a cue tilted in orientation as explained in Fig. 4.
Sand tossing direction versus abdomen to head body orientation of the antlion. \(0 ^\circ\) is the direction of the normal to the antenna pointing towards the antenna. The dark blue line is the fitting of all data by random sample consensus The dark blue line is the fitting of all data by random sample consensus (RANSAC). a For cues emitted by the antenna with all PZT oscillating either at the frequency of 1666 Hz or \(1250 \, \textrm{Hz}\). b with the antenna oscillating with a time delay of 100 microseconds; the carrier frequency was either 1666 or \(1250 \, \textrm{Hz}\)
In the case of the antenna emitting in phase, polar distribution in the form of stacked points of abdomen to head body orientation for fixed sand tossing directions have been considered. Figure 6 are the polar distributions of the abdomen to head orientation for the four sand tossing directions \(0 ^\circ\), \(90 ^\circ\), \(180 ^\circ\) and \(270 ^\circ\) (see Table 1 for the descriptive statistics). The \(0 \, ^\circ\) projection direction bears the largest amount of sand tossing and has the more unimodal distribution for the abdomen to head position: for 207 tossings, 135 are aiming at one direction only, the other 72 are a part of a multimodal action (a repetition of sand tossing alternating between two or more directions). On the opposite direction, i.e., when the sand is tossed in the \(180 ^\circ\) direction, the distribution of body orientations looks bimodal. In this case, over 26 sand tossings, only one aims at one direction only, all the others are one of two directions of sand tossing that actually alternate in directions. For the antenna in phase experiment, among the total of 765 events, 390 are made of more than two repetitions in one direction, 200 are bimodal and 174 are multimodal (a repetition of sand tossing alternating between 3 directions or more). Complementarily, for a given body orientation, the alternate movement of sand tossing is highly dependent on the orientation of the antlion body with respect to the direction of the wave propagation. Only when the antlion orients itself with the head opposite to the phased array does it throws sand repetitively in a single direction. All other body orientations yield an alternate sand tossing fairly symmetric with respect to the body orientation and with a lesser amount of activity. The results for some angle of abdomen to head directions are summarized in 7. For the clarity of the data display, all body orientations within a \(10 ^\circ\) angular spread around the designed body direction have been taken into account and have been converged to a single direction with the accompanying angular shifts for the sand tossing directions.
Polar distribution by stacked points of body orientations in correspondence with sand tossing directions for the respective directions: \(0 ^\circ\), \(90 ^\circ\), \(180 ^\circ\), \(270 \, ^\circ\). The sand tossing directions are defined within \(\pm 18 \, ^\circ\). (a) \(0 ^\circ\) (number of sand tossing events = 207), (b) \(90 ^\circ\) (number of sand tossing events = 49), (c) \(180 ^\circ\) (number of sand tossing events = 26), (d) \(270 ^\circ\) (number of sand tossing events = 78)
Figure 8 displays the polar histograms for the abdomen to head antlion positions (left part) and the sand throwing directions (right part) respectively (a) for the antenna emitting in phase, (b) emitting with a time delay of 100 microseconds between adjacent PZTs with a carrier frequency of \(1250 \, \textrm{Hz}\) and (c) with a carrier frequency of \(1666 \, \textrm{Hz}\). The same results are displayed in polar diagrams of stacked points in the Supplementary Material, figure SM4. If sensitive to the direction of the wave front propagation, the expect mean direction vector for sand tossing for the (b) and (c) situations are most likely within the \(306 ^\circ \!{-}\!342^\circ\) sectors.
From the polar histogram and the circular statistics (see Table 2), one retains that the p value of the Rayleigh test of uniformity is zero for the assays in phase while the p values for the assays with the corrugated wave front, though remaining low, point to a uniform angular distribution. The \(\kappa\) parameters of the von Mises distribution have a similar progression: from high value for the assays in phase to low values for the assays with the corrugated wave front.
Polar histograms with surface of the sectors proportional to the events. The binning is 20: a sector emcompasses \(18 ^\circ\). Left part: abdomen to head antlion positions visualized at the moments of sand tossing action. Right part: sand tossing direction. a The PZTs of the antenna emit in phase, b the PZTs of the antenna emit with a time delay of 100 \(\upmu\)s and the carrier frequency is \(1250 \, \textrm{Hz}\), c the PZTs of the antenna emit with a time delay of 100 microseconds and the carrier frequency is \(1666 \, \textrm{Hz}\)
Discussion
Cues for directional sensing
Antlions have a comprehensive sense of the direction of propagation of the plane surface wave when it is produced by the phased array with the PZTs oscillating in phase. Compared with the results obtained in Mencinger-Vračko and Devetak (2008) and Fertin and Casas (2007), one might question the lack of focus to the proper direction in this experiment albeit the construction of the propagation direction is the one given by a plane wave. What makes the difference—or differences—between this experiment and Mencinger-Vračko and Devetak (2008) and Fertin and Casas (2007)? In the Fertin and Casas (2007) experiment, a needle is oscillating inside the pit. It is here most likely that the localization is based on the difference of intensity on each side of the antlion body since, for a planar radiation emitted from a point source, the intensity of vibration decreases with distance from the source in the inverse law, which means a drop down of 3 dB for every each doubling distance. This effect is prominent when the distance between the receptors is in the scale of the distance from the emitter. On the other hand, in the Mencinger-Vračko and Devetak (2008) experiment, the natural preys were placed 10 to 20 cm away from the pit: in this situation the difference of intensity received on either side of the body receptor is negligible and cannot be the origin of a spatial discrimination. In the case studied in the present paper, the plane wave construction corresponds to a prey present far away from the pit, a situation similar to the Mencinger-Vračko and Devetak experiment. In these conditions, one has to consider two types of physical information: (i) the intensity imbalance produced by the screening and diffraction of the wave by the body, (ii) the time difference of arrival of wave front crossing the body over. (i) is disregarded since the wavelength of the signal is 7–\(10 \, \textrm{cm}\), that is more than ten times bigger than the distance between the tufts of bristles on the lateral sides of the thorax, that are supposed to be the mechanoreceptors, (ii) involves two categories: the onset time difference (OTD) and the interaural phase difference (IPD) or its equivalent for vibroreception. What the results show for the response to the in phase cue is a definite answer to it by aiming at the prey despite the fact that it is less systematic in the present experiment, compared with the Mencinger-Vračko and Devetak experiment. Still the circular statistics (Fig. 8; Table 2) confirm the relative propensity to toss sand in the reversed direction of the plane wave propagation.
Behavioral responses
The correlation between the sand tossing direction and the abdomen to head (last column of Table 2) and RANSAC adjustments of the data for both cues (Fig. 5) exhibit an explicit preference for a backward movement of the mandibles. In the case of answering back to the signal in phase, the angular distribution of sand tossing points in the expected direction while the correlation between body orientation sand tossing direction is the same as for the other cues. From this observation it appears that the antlion orients itself before entering into action. On the opposite direction of sand throwing, the number of events is much less and the body orientation is not opposite to the direction of sand throwing. This observation advocates for the existence of a mechanism that allows to discriminate a vibration propagating back to front from a vibration propagating front to back. Throwing sand repetitively to a single direction - what we call a unimodal sand tossing action - is associated with a behavior of attack. This behavior is absent when the antlion throws sand in the same direction as the cue signal propagation. The bimodal or multimodal sand tossings are generally not associated to an attack behavior but more on the reconstruction or maintenance of the sand pit. It seems that receiving a signal coming from a considerable distance from the pit may also induce this behavior: to get set before the attack. That would explain the large number of events that answer to the cue in multimodal way. We understand that the behavioral response to the vibrational cues belongs to the tropotactic category since the alternate sand tossing in direction does not bring any information about the directionality of the cue.
Responses to the onset of a signal
The differences in descriptives and uniformity tests of the circular statistics are significant between the results for the in phase cue and the dephased cues. On one hand, for the answers to the in phase cues, the Rayleigh p value are way below the 0.0001 critical value and the estimated \(\kappa\) parameter of the von Mises distribution reaches 1 while the statistics for answering the dephased cue are opposite (though the statistics of the 1250 Hz dephased cues are more mitigated: here the mean average length of sand tossing vector is 0.31 but pointing to a direction not associated with any physical significance). By the fact that the onset of the wave has been disarrayed in the dephased cues, the activity of sand tossing has also strongly diminished, which one relates to the absence of capacity to identify the origin of the vibration. From these observations one sustains the idea that onset time delay is more important than phase time delay. If phase time delay was involved in the process of spatial recognition, the repetition of the wave fronts passing through the body of the antlion (five to eight) would have bring a sufficient amount of information. The response to the onset time delay rather than the phase time delay is also grounded with what an antlion can perceive, and the use it makes of it. Any prey is the source of sequences of pulses by its pace on the surface of the media propagating a vibration. The carrier frequency of these pulses that are mainly due to nature of the substrate may be of less importance.
Encoding time delays
A back of an envelope calculation assess that a 10 degrees accuracy in discriminating direction near the azimuth suggests that the neural system must be able to code time delays in the tens of microsecond scale. Some insects are documented to possess such a capacity (Robert 2005). This so-called hyperacute directional hearing measured in the fly Ormia ochracea is explained by a neuronal mechanism involving single neurons (Mason et al. 2001) while, at least in mammals, the detection of temporal features is identified as a coincidence detector mechanism involving several neurons (Joris et al. 1998). The question of the number of sensors makes also the question of directional information processing through vibration perception differ from sound perception. In contrast to animals with two ears-system the antlion has four receptors-system, i.e. paired tufts of hairs on lateral parts of the mesothorax and methatorax. The external morphology of these hairs has long been known (e.g. Lipovsek et al. 2009; Ramos et al. 2020). In arthropods possessing more than two vibration receptors, e.g. four receptors: backswimmer, Notonecta (Wiese 1974); six receptors: water strider, Gerris (Murphey 1971); eight receptors: sand scorpion, Paruroctonus (Brownell and Farley 1979; Brownell and van Hemmen 2001), the direction of the turn movement is determined directly from the central excitation-inhibition gradient originating in the central nervous system as it was shown for integration of vibratory information from different legs of Locusta migratoria (Cokl et al. 1985). In the seminal experiment of prey localization capacity by the mechanoreceptive system investigated by the method of phase shifting the four legs of Notonecta were directly stimulated with time delays (Wiese 1974). This experiment demonstrated that four mechanoreceptors are better than two to lessen the phase ambiguities at some angles. On other experiments in which vibrations were applied to the legs with independently controled platforms, desert scorpions (Brownell and Farley 1979), termites (Hager and Kirchner 2014) and leafcutter ants (Hager et al. 2017) have shown similar minimal distinguishable time differences, in the order of \(0.2 \, \textrm{ms}\). In a more remote signal transmission through a leaf stalks, it was evidenced that the sting bug Nezara viridula was receiving orientation information by a time delay of arrival of the vibration signal to the legs (Prešern et al. 2018). Here also the minimal time delay between cue arrivals that determines a behavior of orientation recognition is in the order of \(0.3 \, \textrm{ms}\). All these experiments suggest that the microsecond time delay is not quite the time intervals involved in discerning orientation by more than two sensors. A debate is then opened about the minimal time difference that a multisensory insect is able to integrate.
We emphasize that our method is not restricted to the study of the physiology of sensing vibrations and can easily be enlarged to the measure of interaural time difference reception for many species, when there is to explicit the role of onset time difference or phase time difference in the localization of sound or vibration.
References
Astropy Collaboration (2022) The astropy project: sustaining and growing a community-oriented open-source project and the latest major release (v.50) of the core package. ApJ 935(2):167. https://doi.org/10.3847/1538-4357/ac7c74
Blauert J, Braasch J (2020) The technology of binaural understanding. Springer Cham, Cham. https://doi.org/10.1007/978-3-030-00386-9
Brownell P, Farley RD (1979) Orientation to vibrations in sand by the nocturnal scorpion Paruroctonus mesaensis: mechanism of target localization. J Comp Physiol A 131(1):31–38. https://doi.org/10.1007/BF00613081
Brownell PH, van Hemmen JL (2001) Vibration sensitivity and a computational theory for prey-localizing behavior in sand scorpions. Am Zool 41(5):1229–1240. https://doi.org/10.1093/icb/41.5.1229
Cocroft RB, Gogala M, Hill PS, Wessel A (2014) Fostering research progress in a rapidly growing field. Studying vibrational communication, animal signals and communication, vol 3. Springer, Berlin, pp 3–12. https://doi.org/10.1007/978-3-662-43607-3
Cokl A, Otto C, Kalmring K (1985) The processing of directional vibratory signals in the ventral nerve cord of Locusta migratoria. J Comp Physiol A 156:45–52. https://doi.org/10.1007/BF00610665
Devetak D (2014) Sand-borne vibrations in prey detection and orientation of antlions. In: Cocroft RB, Gogala M, Hill PS, Wessel A (eds) Studying vibrational communication, animal signals and communication, vol 3. Springer, Berlin, pp 319–330. https://doi.org/10.1007/978-3-662-43607-3
Devetak D, Mencinger-Vračko B, Devetak M, Marhl M, Špernjak A (2007) Sand as a medium for transmission of vibratory signals of prey in antlions Euroleon nostras (Neuroptera: Myrmeleontidae). Physiol Entomol 32(3):268–274. https://doi.org/10.1111/j.1365-3032.2007.00580.x
Fertin A, Casas J (2007) Orientation towards prey in antlions: efficient use of wave propagation in sand. J Exp Biol 210:3337–3343. https://doi.org/10.1242/jeb.004473
Fischler MA, Bolles RC (1981) Random sample consensus: a paradigm for model fitting with applications to image analysis and automated cartography. Commun ACM 24(6):381–395. https://doi.org/10.1145/358669.358692
Hager FA, Kirchner WH (2014) Directional vibration sensing in the termite Macrotermes natalensis. J Exp Biol 217(14):2526–2530. https://doi.org/10.1242/jeb.103184
Hager FA, Kirchner L, Kirchner WH (2017) Directional vibration sensing in the leafcutter ant Atta sexdens. Biol Open 6(12):1949–1952. https://doi.org/10.1242/bio.029587
Hager FA, Krausa K, Kirchner WH (2019) Directionality in insect vibration sensing: behavioral studies of vibrational orientation. In: Hill P, Lakes-Harlan R, Mazzoni V, Narins P, Virant-Doberlet M, Wessel A (eds) Biotremology: studying vibrational behavior, vol 6. Springer, Cham, pp 235–255. https://doi.org/10.1007/978-3-030-22293-2_12
JASP Team (2023) JASP (Version 0.17) [Computer software]. https://jasp-stats.org/
Joris PX, Smith PH, Yin TC (1998) Coincidence detection in the auditory system: 50 years after Jeffress. Neuron 21(6):1235–1238. https://doi.org/10.1016/s0896-6273(00)80643-1
Klokočovnik V, Devetak D, Orlačnik M (2012) Behavioral plasticity and variation in pit construction of antlion larvae in substrates with different particle sizes. Ethology 118:1102–1110. https://doi.org/10.1111/eth.12012
Lipovšek DS, Pabst MA, Devetak D (2009) Morphology of the eyes and sensilla in the antlion larvae (Neuroptera: Myrmeleontidae). In: Pabst MA, Zellnig G (eds) Microscopy Conference 2009 [proceedings], Life Sciences. Verlag der Technischen Universität, vol 2, pp 403–404
Martinez V, Nowbahari E, Sillam-Dussès D, Lorent V (2020) Antlions are sensitive to subnanometer amplitude vibrations carried by sand substrates. J Comp Physiol A 206(5):783–791. https://doi.org/10.1007/s00359-020-01437-3
Mason AC (2021) Cues for directional hearing in the fly Ormia ochracea. Front Ecol Evol 9:1–10. https://doi.org/10.3389/fevo.2021.679064
Mason AC, Oshinsky ML, Hoy RR (2001) Hyperacurate directional hearing. Nature 410:686–690. https://doi.org/10.1038/35070564
Mencinger-Vračko B, Devetak D (2008) Orientation of the pit-building antlion larva Euroleon (Neuroptera, Myrmeleontidae) to the direction of substrate vibrations caused by prey. Zoology 111:2–8. https://doi.org/10.1016/j.zool.2007.05.002
Michelsen A (2014) Physical aspects of vibrational communication. In: Cocroft RB, Gogala M, Hill PSM, Wessel A (eds) Studying vibrational communication, animal signals and communication, vol 3. Springer, Berlin, pp 199–213
Mortimer B (2017) Biotremology: do physical constraints limit the propagation of vibrational information? Anim Behav 130:165–174. https://doi.org/10.1016/j.anbehav.2017.06.015
Murphey R (1971) Sensory aspects of the control of orientation to prey by waterstrider, Gerris remigis. Z Vergl Physiol 72:168–185. https://doi.org/10.1007/BF00297820
Narins PM, Stoeger AS, O’Connell-Rodwell C (2016) Infrasonic and seismic communication in the vertebrates with special emphasis on the afrotheria: an update and future directions. In: Suthers R, Fitch W, Fay R, Popper A (eds) Vertebrate sound production and acoustic communication, vol 53. Springer, Cham, pp 191–227
Podlesnik J, Klokočovnik V, Lorent V, Devetak D (2019) Prey detection in antlions: propagation of vibrational signals deep into the sand. Physiol Entomol 44:215–221. https://doi.org/10.1111/phen.12295
Popper AN, Fay RR (2014) Perspectives on auditory research (Springer handbook of auditory research vol 50). Springer New York, New York
Prešern J, Polajnar J, De Groot M, Zorović M, Virant-Doberlet M (2018) On the spot: utilization of directional cues in vibrational communication of a stink bug. Sci Rep 8:5418. https://doi.org/10.1038/s41598-018-23710-x
R Core Team (2022) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Ramos FA, Monserrat VJ, Contreras-Ramos A, Pérez-González S (2020) Comparative study of sensilla and other tegumentary structures of Myrmeleontidae larvae (Insecta, Neuroptera). J Morph 281:1191–1209. https://doi.org/10.1002/jmor.21240
Robert D (2005) Directional hearing in insects. In: Popper AN, Fay RR (eds) Sound source localization. Springer New York, New York, pp 6–35
Römer H (2020) Directional hearing in insects: biophysical, physiological and ecological challenges. J Exp Biol 223:jeb203224. https://doi.org/10.1242/jeb.203224
Strauß J, Stritih-Peljhan N, Nieri R, Virant-Doberlet M, Mazzoni V (2021) Communication by substrate-borne mechanical waves in insects: from basic to applied biotremology. Ad Insect Phys 61:189–307. https://doi.org/10.1016/bs.aiip.2021.08.002
Virant-Doberlet M, Čokl A, Zorović M (2006) Use of substrate vibrations for orientation: from behaviour to physiology. Insect sounds and communication: physiology, behaviour, ecology and evolution. Taylor & Francis Group, Boca Raton, pp 81–97
Visser HJ (2005) Array and phased array antenna basics. Wiley, Hoboken
Wiese K (1974) The mechanoreceptive system of prey localization in Notonecta—ii. The principle of prey localization. J Comp Physiol 92(3):317–325
Acknowledgements
We thank Nataša Stritih Peljhan from National Institute of Biology (Slovenia) and Elise Nowbahari from the Laboratoire d’Ethologie Expérimentale et Comparée for the careful and critical reading of the manuscript, Fabrice Wiotte from the Laboratoire de physique des lasers for the realization of printed circuits.
Funding
This work was supported by the BQR 2014/2015 from the Université Sorbonne Paris Nord. VM acknowledges the support of the École doctorale Galilée - Université Sorbonne Paris Nord. This work was partially supported by the Slovenian Research Agency with the Research Programme Computationally Intensive Complex Systems (Grant no. P1-0403) and partially by the Ministry of Education, Science and Sport of the Republic of Slovenia and by the European Regional Development Fund of the EU within the Project “Development of Research Infrastructure for the International Competitiveness of the Slovenian RRI Space - RI-SI-LifeWatch”.
Author information
Authors and Affiliations
Contributions
Contributions. Project definition DD, JP, VL, Conception and design VL, data collection VM, JP, data analysis VM, VL, JP, discussion and interpretation of data VL, VM, JP, DD, DSD, writing of first draft, VL, VM and revisions, VL, DD, JP, VM, DSD.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Laboratory animal care
Experiments complied with the principles of Laboratory Animal Care.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Handling Editor: Andrea Megela Simmons.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Martinez, V., Sillam-Dussès, D., Devetak, D. et al. Antlion larvae localize long distant preys by a mechanism based on time difference. J Comp Physiol A 210, 35–45 (2024). https://doi.org/10.1007/s00359-023-01641-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-023-01641-x