Abstract
The tympanal ear is an evolutionary acquisition which helps moths survive predation from bats. The greater diversity of bats and echolocation strategies in the Neotropics compared with temperate zones would be expected to impose different sensory requirements on the neotropical moths. However, even given some variability among moth assemblages, the frequencies of best hearing of moths from different climate zones studied to date have been roughly the same: between 20 and 60 kHz. We have analyzed the auditory characteristics of tympanate moths from Cuba, a neotropical island with high levels of bat diversity and a high incidence of echolocation frequencies above those commonly at the upper limit of moths’ hearing sensitivity. Moths of the superfamilies Noctuoidea, Geometroidea and Pyraloidea were examined. Audiograms were determined by non-invasively measuring distortion-product otoacoustic emissions. We also quantified the frequency spectrum of the echolocation sounds to which this moth community is exposed. The hearing ranges of moths in our study showed best frequencies between 36 and 94 kHz. High sensitivity to frequencies above 50 kHz suggests that the auditory sensitivity of moths is suited to the sounds used by sympatric echolocating bat fauna. Biodiversity characterizes predators and prey in the Neotropics, but the bat–moth acoustic interaction keeps spectrally matched.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The tympanal ears of moths sense incoming sounds and enable the evaluation of acoustical signals from predators or mates. The evolution of moths ears took place under the selective pressure imposed by predation from sympatric bats (Roeder 1967; Fullard and Yack 1993). Moth hearing has the primary role of detecting these predators; therefore, moths commonly sense a broad range of ultrasonic frequencies with maximal sensitivity in the spectral range most used in bats’ echolocation (20–60 kHz) (Roeder and Treat 1957; Fullard 1988; Nakano et al. 2015). Additionally, some species of moths have incorporated sound-emitting capabilities as a secondary acquisition (Conner 1999; Nakano et al. 2009); they produce ultrasound in mate-calling (Spangler 1988; Takanashi et al. 2010) or as predator defense (Barber and Conner 2007; Corcoran et al. 2009). Ultrasound has become a survival tool for moth species capable of detecting it or producing it.
The selective pressure imposed by bats on moth hearing is shown by studies that compare moth species that differ in their sympatric bat fauna. ter Hofstede et al. (2013) suggested that moths from the UK are more sensitive to high frequencies than moths from Canada and Denmark, possibly as a consequence of the horseshoe bats’ predation on British moths. Horseshoe bats echolocate at frequencies above 80 kHz. Moths in neotropical areas are challenged by an acoustic environment made complex not only by a rich diversity of bat species but also by a large diversity of echolocation strategies, including low and high frequencies with several temporal designs (Fullard 1982; Kalko et al. 1996). Moths surrounded by a high diversity of bats have higher acoustic sensitivity and broader threshold curves compared with moths from sites of low bat diversity (Fenton and Fullard 1979; Fullard 1982). However, the frequencies of highest sensitivity of tropical moths still appear to be mostly under 60 kHz (Fullard and Thomas 1981; Fullard 1988; Fullard et al. 1997). The echolocation spectrum that acts as predation pressure on those moth communities includes call frequencies that are clearly above 60 kHz (Fullard and Belwood 1988). However, those frequencies do not constitute the primary selection pressure since they are produced by bats that are not primarily insectivorous or are less common species. Habitats with an assemblage of insectivorous bats employing a wide range of echolocation frequencies having similar energy over most of the spectrum may reveal a correspondingly high diversity of hearing capabilities in moths, with best frequencies covering a wide ultrasound range.
The richest bat fauna in the West Indies is found in Cuba, a subtropical island with 26 species, 70% of which are insectivorous (Silva 1979). Cuban bats illustrate the diversity of echolocation systems, and seven species echolocate at frequencies above those commonly limiting the hearing sensitivity of moths (Macías and Mora 2003; Macías et al. 2006; Mora and Macías 2011). It is unknown if these high echolocation frequencies constitute a significant predation pressure for this moth community. The echolocation assemblage most strongly affecting moths’ auditory sensitivity depends not only on the echolocation frequencies, but also on certain ecological factors, such as feeding habits and population levels (Fullard and Belwood 1988). Previous bioacoustic (Mora et al. 2005; Macías et al. 2006; Mora and Torres 2008) and ecological (Silva 1979; Mancina et al. 2007) studies of Cuban bats could help to estimate the bat predation potential on the island. The distribution and population levels of insectivorous bats within this habitat, and the spectral analysis of their echolocation signals make it possible to characterize the Cuban echolocation assemblage.
The study of moth ears is essential for understanding the bat–moth interaction. Traditional studies analyzing hearing capabilities of moths have used neurophysiological recordings to evaluate tympanal nerve responses (Roeder and Treat 1957; Surlykke and Treat 1995; Fullard et al. 2003). Other studies have included behavioral criteria (Rydell et al. 1997; Dawson et al. 2004; Ratcliffe et al. 2009). An alternative method for evaluating the best hearing range in moths could be the noninvasive recording of distortion-product otoacoustic emissions (DPOAE) (Coro and Kössl 1998; Mora et al. 2013). DPOAEs are faint sounds that appear when the ear is stimulated simultaneously with two tones; they reach their largest amplitudes at frequencies in the best hearing range of each species and reflect mechanical characteristics of sensory transduction (Kössl et al. 2008). Moth ears emit conspicuous DPOAEs, which are similar to those generated by vertebrate ears (Coro and Kössl 1998; Kössl et al. 2007). In the present work, we describe the hearing capabilities of a neotropical moth community by measuring DPOAE audiograms. We analyze the acoustic relationships between the frequencies that constitute relevant predation pressure and the auditory characteristics of sympatric moths.
Materials and methods
Animal preparation
Adult individuals of the superfamilies Noctuoidea, Geometroidea and Pyraloidea (species names in Fig. 2) were collected from fluorescent lights or from host plants in diverse localities of Havana. We also reared Galleria mellonella from the larval stage. These pyralid specimens were fed with a mixture of honey and wheat bran. All adult moths were fed ad libitum with a 30% sucrose solution and kept at ambient light and temperature in the Bioacoustics Lab at the Faculty of Biology, University of Havana. Each specimen was photographed and identified by comparison with specimens and vouchers previously deposited in the Institute for Ecology and Systematics and the Museum of Natural History “Felipe Poey”. All animals were tested within two days of their capture or emergence.
For auditory evaluation, specimens were tethered with modeling clay to a cork platform covered by wax. Neither dissection nor anesthetics were used. Moths with thoracic ears were positioned dorsal side up and slightly tilted to the left. Specimens with abdominal ears were placed ventral side up with the thorax slightly pulled back. To allow free coupler approach to the tympanum, wings were held to the sides and the external auditory structures were carefully displaced but not dissected. Only the right ear was examined per animal. Experiments were carried out at room temperature of 24–28 °C and relative humidity of 45–65%, inside a Faraday cage that also was acoustically isolated. The physiological state of the moths during recordings was assessed by monitoring spontaneous movements of the antennae.
Distortion-product otoacoustic emissions (DPOAEs) recording and analysis
To measure DPOAEs, a custom-built acoustic coupler was placed about 1 mm from the tympanum in an open sound system configuration. The acoustic coupler included two ½ inch microphone capsules used as loudspeakers (Microtech Gefell MK 202) for delivering tones, and a ¼ inch microphone (Microtech Gefell MK301) connected to a preamplifier (Microtech Gefell MV302) for monitoring sound pressure at the tympanum. The coupler had three channels and an overall tip diameter of 1.5 mm; it was connected to the microphone capsules through conical speculums. The acoustic signal from the microphone was amplified using a conditioning amplifier (Brüel & Kjær Nexus 2690) and digitized with a Fireface 400 sound card. A sampling rate of 192 kHz was used for stimulus generation and microphone recording. The sound system was calibrated in situ for frequencies between 10 and 96 kHz using white noise. Nonlinear distortions of the recording setup were evaluated by performing control measurements in five individuals with the coupler in front of a cuticular body surface 5 mm distant from the tympanum. Sound pressure levels used in the present study are expressed in dB SPL (dB re. 20 µPa).
The stimuli used were two simultaneous pure tones of 42.7 ms duration (1 ms rise/fall time, 0° sine phase difference between the onset of both) with a pause of 1 s. In the present study we deliberately chose short stimulus durations to avoid frequency up-tuning of the tympanum (Windmill et al. 2006; Mora et al. 2015). The frequencies and levels of these primary tones are denoted as f 1, f 2, L 1 and L 2, respectively. A fixed f 2/f 1 ratio of 1.09 and a level combination of L 1 = L 2+10 dB were used since these stimulus parameters evoke large DPOAE levels in moths (Coro and Kössl 1998; Kössl and Coro 2006). For each moth, a matrix of 861 frequency-level combinations (41 frequencies × 21 levels) was randomly presented with f 2 values from 14 to 94 kHz (2 kHz steps), and L 2 values from 30 to 70 dB SPL (2 dB steps). The microphone signal was analyzed by performing an FFT (rectangular window, 8192 data points) with a frequency resolution of 23.4 Hz. From the spectrum were measured the amplitude of 2f 1 − f 2 DPOAE and the noise floor, calculated as the mean amplitude of 10 FFT bins at both sides and 1 kHz from the DPOAE frequency. The DPOAE audiogram was constructed by representing the levels of 2f 1 − f 2 for each stimulus frequency-level combination in a color map. The auditory threshold was defined as the L 2 level necessary to elicit 2f 1 − f 2 levels equivalent to 15 dB SPL. Since the spectral analysis was performed directly on the short signal recorded by the microphone without averaging, the noise floor was higher than in previous measurements where we averaged over several seconds (e.g., Kössl and Coro 2006). Consequently, we determined the mechanical tuning curve of the moth as the 15 dB SPL iso-contour line calculated by linear interpolation in the frequency-level response matrix using the Matlab contour function. From the median species audiogram, we evaluated: (1) best frequency (BF, kHz), i.e. the f 2 frequency of maximum sensitivity, (2) minimum threshold (MTr, dB SPL), i.e., threshold at best frequency, (3) frequency range of best sensitivity delimited by the highest (Fmax, kHz) and lowest frequencies (Fmin, kHz) at +10 dB re MTr, and (4) tuning sharpness (Q10 dB = BF/(Fmax − Fmin)). Stimulation control, data acquisition, and data analysis were performed using software written in Matlab R2009 (The MathWorks, Inc., Natick, MA, USA).
Bat call analysis
Recordings of Cuban bats were obtained from the Bat Call Archive of the Research Group in Bioacoustics and Neuroethology, University of Havana. Recordings were made using an ultrasound detector (Ultrasound Advice U30; frequency response ±3 dB, 20–120 kHz). The signals were digitized at a sampling rate of 312 kHz using a 16-bit A/D converter (Measurement Computing PC-CARD-DAS 16/330) controlled with the software BatSound 2.1 (Pettersson Elektronik). Echolocation calls were resampled at 250 kHz and spectrally analyzed using Avisoft-SASLab Pro (FFT 512 points, 93.75% overlap, Hamming window).
To define the echolocation assemblage that acts as selective pressure on tympanate Cuban moths, we combined the normalized frequency spectra of the calls emitted by 15 species of insectivorous bats. Search calls from ten different individuals of each species were analyzed. To determine the assemblage spectrum, the averaged power spectrum calculated per species was weighted by a “predation factor” (Pf) that was used as a measure of their potential as predators. To calculate Pf, we took into account the distribution (D) and population abundance (PA) indexes proposed by Mancina et al. (2007) for Cuban bats. For each index these authors defined three classes to which numeric values were assigned. For species abundance they proposed: 0 common, 1 scarce and 2 rare. To categorize the geographical distribution they defined: 0 widespread, 1 moderate and 2 restricted. In the present study we propose Pf = 5 − (PA + D). The cumulative maximum that the two indices can generate is 4, which indicates a rare and restricted species that, as we assume, exerts only a low predation pressure on the moths. Pf constitutes a semiquantitative assessment using the two indices that includes subtraction from 5 to avoid zero factors for the weighting of the spectral amplitudes.
Results
Moth auditory analyses
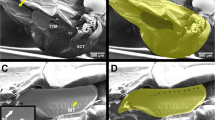
To describe the hearing of this neotropical moth community, distortion products were recorded from the tympanum of 99 individuals of 31 species. The recording of 2f 1 − f 2 level in a matrix of frequency-level combinations allowed us to determine DPOAE audiograms for each specimen. Examples of six individual DPOAE audiograms, showing a high diversity of spectral selectivity, are illustrated in Fig. 1. Moth ears were sensitive to ultrasonic frequencies from 14 to 94 kHz. Some species showed asymmetric threshold curves (Fig. 1c, d) with steeper slopes for frequencies below the BF. However, it is noteworthy that several audiograms were limited by the highest frequency measurable by the equipment, i.e., 94 kHz (Fig. 1e, f) and, in fact, were so sensitive to highest the ultrasonic frequencies we could measure, that it was not possible to define the entire threshold curve. A similar picture emerged when assessing the frequency range of best hearing sensitivity for all species (Fig. 2). The maximum frequency (Fmax +10 dB re MTr) was coincident with the 94 kHz limit of our recording range in 18 species (58%), suggesting a probable extension of the spectral hearing range of the moths to much higher frequencies.
Hearing diversity in neotropical moths. Examples of six individual DPOAE audiograms are represented as color maps of 2f 1 − f 2 levels. As stimuli, two simultaneous pure tones f 1 and f 2 of 42.7 ms duration were used, at sound levels of L 1=L 2+10 dB and at a fixed f 2/f 1 ratio of 1.09. Auditory threshold curves (white lines) were defined by the L 2 levels necessary to elicit 2f 1 − f 2 levels equivalent to 15 dB SPL. Species names are specified above each audiogram
Spectral ranges of best hearing sensitivity. Individual or median (for species with sample size >1) frequency ranges are represented as a function of f 2 or 2f 1 − f 2 frequencies. Horizontal lines depict the bandwidth, i.e. difference between the highest (Fmax) and lowest (Fmin) frequencies at +10 dB re minimum threshold. Best frequencies are represented by black circles. The spectral range of highest sensitivity previously defined for moths (Fullard 1998; Nakano et al. 2015) is delimited with a gray rectangle. Species names—N are specified to the right. The upper frequency limit imposed by the equipment (94 kHz) is highlighted in red
The frequencies of maximum sensitivity were between 36 and 94 kHz, with a majority in the typical hearing range of most moths (ca., 20–60 kHz), although a surprising number showed best tuning to about 60 kHz (Fig. 2). Moreover, it is also noteworthy that in 14 species (45%) the BF of the median audiogram was above 60 kHz. The asymmetries of the threshold curves were also evident in this bandwidth analysis; BFs were closer to Fmin in several species (see Fig. 2). The ears were broadly tuned, Q10 dB values were generally <2 (Table 1); in only eight species, were Q10 dB values >2 recorded. In those eight species the BFs were at higher frequencies and the Fmax was cut off at 94 kHz. Minimum thresholds were found to be between 31 and 56 dB SPL (Table 1), with a median value of 48 dB SPL.
The echolocation assemblage
Representative echolocation calls of Cuban insectivorous bats are presented in Fig. 3a. The diversity of call designs as well as their spectral characteristics is evident. These species echolocate at frequencies from 15 to 100 kHz, but most energy is focused between 30–90 kHz, as illustrates the average spectrum (Fig. 3b, dark grey area). The echolocation assemblage that resulted from weighting by the “predation factor” (Fig. 3b, light grey area) gave a more equal distribution among frequencies with a small notch around 75 kHz. Seven bat species echolocate with frequencies above 50 kHz, four of which are mormoopids, a predominant group in Cuba (Silva 1979). Frequencies corresponding to Pteronotus parnellii calls, a mormoopid bat echolocating with a long (~15 ms) constant-frequency component of about 60 kHz, is highlighted as a small peak in the echolocation assemblage. With Pf equivalent to five, mormoopids species contribute to the predominance of high frequencies in the weighted average spectrum.
Spectral correlation between moths’ hearing range and the Cuban echolocation assemblage. a Spectrograms of representative search calls of each insectivorous Cuban bat: a, Eumops ferox; b, Nyctinomops macrotis; c, Tadarida brasiliensis; d, Molossus molossus; e, Mormopterus minutus; f, Eptesicus fuscus; g, Nycticeius cubanus; h, Lasiurus pfeifferi; i, Pteronotus macleayii; j, P. parnellii; k, P. quadridens; l, Mormoops blainvillei; m, Nyctiellus lepidus; n, Natalus primus; o, Macrotus waterhousii. b Non-weighted average power spectrum (dark gray) and spectrum weighted by the “predation factor” (see text, light gray). c Median threshold curve estimated from 31 species of moths is correlated with the weighted spectrum of bat echolocation
Bat–moth acoustic interactions in the neotropics
The median threshold curve from all evaluated moths was estimated from the median curves for each species (Fig. 3c). This integrative analysis from ears tuned to a great diversity of frequency ranges yielded a broad curve which expands across the spectrum. Nevertheless, tuning to frequencies above 50 kHz prevails in this neotropical moth community The spectral relation between the threshold curve of moths and the echolocation spectrum of bats (Fig. 3c) shows that Cuban insectivorous bats, as a group, echolocate with maximum energy at frequencies to which moths’ ears are most sensitive.
Discussion
The simple ears of moths, with their small number of auditory neurons shares an essential feature with the vertebrate ears: nonlinear mechanical sound processing generates distinct distortion-product otoacoustic emissions (Kössl et al. 2008). DPOAEs have been previously reported in three species of Noctuoidea: Ascalapha odorata, Empyreuma pugione, Ptilodon cucullina (Coro and Kössl 1998; Kössl et al. 2007) and one species of Geometroidea, Urania boisduvalii (Barro 2001). The present study extends DPOAE recordings to 31 moth species from a neotropical location. Threshold curves based on a 2f 1 − f 2 iso-level criterion allowed us to define the hearing range for each species. Our study shows a high diversity of frequency tuning with maximum sensitivity at frequencies between 36 and 94 kHz (or higher, since the recording system did not go above 94 kHz.).
Diversity begins with the design of the ears. There are many structural differences among the ears of the three sampled superfamilies (Minet and Surlykke 2003). Interspecific variations in the mechanical properties of the tympanic membrane, the internal cavities and the external tympanic recess may be correlated with differences in auditory tuning characteristics (Adams 1972). Differences in tuning might also be related to the structural variations between entire scolopidia, or their components (Yack 2004). From our results, it is not possible to determine whether the position of the tympanic organ or the number of sensory cells are related to the overall frequency response. Moths from the superfamilies Geometroidea and Pyraloidea, both with abdominal ears and four scolopidia (Minet and Surlykke 2003), showed a tendency to be selective to the high frequencies (see Fig. 2). Previous vibrometric, electrophysiological and behavioral studies support this observation for pyralid moths (Pérez and Zhantiev 1976; Spangler 1987; Heller and Krahe 1994; Rodríguez et al. 2005) However, best sensitivities at frequencies below 50 kHz have also been described in both superfamilies (Surlykke and Filskov 1997; Takanashi et al. 2010). In order to evaluate these parameters, it will be necessary to increase the sample size for each taxonomic group. It is worth noting, moreover, that there is a great deal of diversity in hearing range among the noctuids that have been studied, all of them with metathoraxic ears and only two scolopidia.
All audiograms were broadly tuned as is expected for neotropical moths having to cope with a diverse bat assemblage (Fullard 1982). Broad threshold curves increase the chance of detecting potential predators emitting signals with different spectral content. Tuning curves estimated from DPOAE recordings are comparable in overall sensitivity to neuronal thresholds, but intraspecific comparisons reveal slight differences. As an example, for the species A. odorata the minimum threshold was 42 dB SPL, which is higher than the threshold previously defined (around 30 dB SPL) by electrophysiological recordings (Fullard 1984; Pérez and Coro 1985). In insects, 2f 1 − f 2 DPOAE usually can be elicited with stimuli levels near the species-specific auditory threshold (Kössl et al. 2008). However, DPOAEs are faint sounds whose detectability depends on the background noise. In our experiments, a threshold equivalent to 15 dB SPL was used based on the mean background noise level plus two standard deviations. In order to avoid the frequency up-tuning that appears at the acoustically stimulated moths’ tympanum (Windmill et al. 2006; Mora et al. 2015), we did not use multiple stimulus presentations and response averaging. Instead, a 42 ms single stimulus was presented. The high background noise level precludes us from evaluating 2f 1 − f 2 response below 15 dB SPL, resulting in lower sensitivities (higher minimum thresholds).
Previous studies in moths from diverse locations have consistently found highest sensitivity at frequencies between 20 and 60 kHz (Fullard 1982; Fullard et al. 1997; ter Hofstede et al. 2013). The Cuban moth community exhibited a slightly different response spectrum when assessed for DPOAE responses. A wide diversity of DPOAE audiograms demonstrates that most Cuban moths have best sensitivities to frequencies above 50 kHz. Methodological issues could be related to these differences. However, our results are in accordance with Fullard’s observations regarding tropical locations; the increased use of higher frequencies by the echolocating bat communities appears to be matched by the greater sensitivities of sympatric moths in the high frequency range (Fullard 1982). A great diversity of bats using a variety of echolocation strategies imposes different sensory requirements to sympatric moths. Mormoopid bats are predominant in Cuba and constitute the principal source of the high frequencies that put predation pressure on tympanate moths (Silva 1979; Mancina et al. 2007).
We found a high incidence of moths with threshold minima at about 60 kHz, close to the dominant second harmonic in the echolocation calls of P. parnellii. During flight, the mustached bat compensates for upward Doppler shifts of echo frequency by lowering the frequency of the emitted calls, so that returning echoes are stabilized at a frequency that is slightly higher than 61 kHz (Keating et al. 1994). P. parnellii is the only New World species that has evolved high-duty cycle echolocation (Fenton et al. 2012). This species is abundant and widespread throughout Cuba (Silva 1979). It seems that the specific properties and resonances of the P. parnellii ear that make it most sensitive and sharply tuned to ~61 kHz are mirrored by a corresponding, although less sharply tuned, development in some moth ears. In short, our observations are consistent with the conclusion that the ears of many neotropical moths have evolved their frequency sensitivity to maximize detection and avoidance of mustached bats.
Hearing research in moths has been mainly based on a neurophysiological approach. The neuronal threshold curve parallels the behavioral or vibrational audiograms for some species (Skals and Surlykke 2000; ter Hofstede et al. 2011), and this is also expected from the mechanical evaluation of the tympanum from OAEs responses. Several studies in vertebrates have demonstrated a correlation between the minimal thresholds in the neuronal audiogram and lowest thresholds for distortion-product generation (Kössl 1992; Manley et al. 1993; Meenderink et al. 2005). In insects, too, 2f 1 − f 2 distortion products reach maximal amplitudes at frequencies of highest auditory sensitivity (Coro and Kössl 1998; Kössl and Boyan 1998). However, the hearing range defined by electrophysiological recordings for species from the genera Achaea, Heliothis, Leucania, Melipotis, Mocis, Ophisma, Spodoptera (Agee 1967; Fullard 1994; Fullard et al. 1997) are not consistent with the spectral range that we found by evaluating DPOAE generation. Best frequencies for these genera previously reported from neurophysiological studies, are below 40 kHz, which contrasts to the tuning of DPOAE audiograms to higher frequencies (>50 kHz) found in the present study. Certain issues related to methodology may explain the discrepancies between the results. Exposing the auditory nerve requires dissection of the thorax and partial removal of the flight musculature (Roeder and Treat 1957). Furthermore, the previously cited authors recorded in decapitated moths with wings removed. In contrast, DPOAE recording permits noninvasive determination of the frequency dependence of ear sensitivity. This approach helps to preserve the mechanical properties of the acoustical system and keeps intact potential effectors associated with the tympanic organ. Simultaneous recording of mechanical (DPOAE) and electrophysiological responses of the peripheral auditory system in specimens with intact reflex pathways could help to elucidate causes of this frequency mismatch.
Conclusions
Despite their morphological simplicity, the ears of moths show highly diverse tuning capabilities. The behavioral context in which they have evolved seems to be an essential factor to enrich the frequency-sound level space that is shaping the moths’ tuning curves. The high density of Cuban bat fauna compared with temperate faunas places greater predation pressure at the higher frequencies of the spectrum. The ears of Cuban moths are tuned to the frequency range used by the collection of bats on the island. The overlap between the bats’ echolocation frequencies and the hearing range of sympatric moths described for this neotropical community is comparable to the overlap previously found in temperate areas, but with a shifted spectral range. Future studies using noninvasive approaches are needed for reconciling DPOAE and neuronal recordings.
References
Adams WB (1972) Mechanical tuning of the acoustic receptor of Prodenia eridania (Cramer)(Noctuidae). J Exp Biol 57(2):297–304
Agee H (1967) Response of acoustic sense cell of the bollworm and tobacco budworm to ultrasound. J Econ Entomol 60(2):366–369
Barber JR, Conner WE (2007) Acoustic mimicry in a predator–prey interaction. Proc Natl Acad Sci USA 104(22):9331–9334
Barro A (2001) Historia natural y bioacústica de Urania boisduvalii (Guenée) (Lepidoptera: Uraniidae). Master Thesis, Universidad de La Habana, La Habana, Cuba
Conner WE (1999) ‘Un chant d’appel amoureux’: acoustic communication in moths. J Exp Biol 202(13):1711–1723
Corcoran AJ, Barber JR, Conner WE (2009) Tiger moth jams bat sonar. Science 325(5938):325–327
Coro F, Kössl M (1998) Distortion-product otoacoustic emissions from the tympanic organ in two noctuoid moths. J Comp Physiol A 183(4):525–531. doi:10.1007/s003590050278
Dawson JW, Kutsch W, Robertson RM (2004) Auditory-evoked evasive manoeuvres in free-flying locusts and moths. J Comp Physiol A 190(1):69–84. doi:10.1007/s00359-003-0474-3
Fenton MB, Fullard JH (1979) Influence of moth hearing on bat echolocation strategies. J Comp Physiol A 132(1):77–86
Fenton MB, Faure PA, Ratcliffe JM (2012) Evolution of high duty cycle echolocation in bats. J Exp Biol 215(17):2935–2944
Fullard JH (1982) Echolocation assemblages and their effects on moth auditory systems. Can J Zool 60(11):2572–2576
Fullard JH (1984) Acoustic relationships between tympanate moths and the Hawaiian hoary bat (Lasiurus cinereus semotus). J Comp Physiol A 155(6):795–801
Fullard JH (1988) The tuning of moth ears. Experientia 44(5):423–428
Fullard JH (1994) Auditory changes in noctuid moths endemic to a bat-free habitat. J Evol Biol 7(4):435–445
Fullard JH (1998) The sensory coevolution of moths and bats. In: Hoy R, Popper A, Fay R (eds) Comparative hearing: insects. Springer, New York, pp 279–326
Fullard JH, Belwood JJ (1988) The echolocation assemblage: acoustic ensembles in a neotropical habitat. In: Nachtigall P, Moore PB (eds) Animal Sonar. NATO ASI Science, vol 156. Springer, New York, pp 639–643. doi:10.1007/978-1-4684-7493-0_68
Fullard JH, Thomas DW (1981) Detection of certain African, insectivorous bats by sympatric, tympanate moths. J Comp Physiol 143(3):363–368
Fullard JH, Yack JE (1993) The evolutionary biology of insect hearing. Trends Ecol Evol 8(7):248–252. doi:10.1016/0169-5347(93)90200-9
Fullard JH, Dawson JW, Otero L, Surlykke A (1997) Bat-deafness in day-flying moths (Lepidoptera, Notodontidae, Dioptinae). J Comp Physiol A 181(5):477–483
Fullard JH, Dawson JW, Jacobs DS (2003) Auditory encoding during the last moment of a moth’s life. J Exp Biol 206(Pt 2):281–294
Heller KG, Krahe R (1994) Sound production and hearing in the pyralid moth Symmoracma minoralis. J Exp Biol 187(1):101–111
Kalko EK, Handley CO, Handley D (1996) Organization, diversity, and long-term dynamics of a Neotropical bat community. In: Long-term studies of vertebrate communities, pp 503–553
Keating A, Henson O, Henson M, Lancaster W, Xie D (1994) Doppler-shift compensation by the mustached bat: quantitative data. J Exp Biol 188(1):115–129
Kössl M (1992) High frequency distortion products from the ears of two bat species, Megaderma lyra and Carollia perspicillata. Hear Res 60(2):156–164. doi:10.1016/0378-5955(92)90018-I
Kössl M, Boyan GS (1998) Acoustic distortion products from the ear of a grasshopper. J Acoust Soc Am 104(1):326–335
Kössl M, Coro F (2006) L1, L2 maps of distortion-product otoacoustic emissions from a moth ear with only two auditory receptor neurons. J Acoust Soc Am 120(6):3822–3831
Kössl M, Coro F, Seyfarth EA, Nassig WA (2007) Otoacoustic emissions from insect ears having just one auditory neuron. J Comp Physiol A 193(8):909–915. doi:10.1007/s00359-007-0244-8
Kössl M, Möckel D, Weber M, Seyfarth EA (2008) Otoacoustic emissions from insect ears: evidence of active hearing? J Comp Physiol A 194(7):597–609. doi:10.1007/s00359-008-0344-0
Macías S, Mora EC (2003) Variation of echolocation calls of Pteronotus quadridens (Chiroptera: Mormoopidae) in Cuba. J Mammal 84(4):1428–1436
Macías S, Mora EC, García A (2006) Acoustic identification of mormoopid bats: a survey during the evening exodus. J Mammal 87(2):324–330
Mancina CA, Echenique-Díaz LM, Tejedor A, García L, Daniel-Álvarez Á, Ortega-Huerta MA (2007) Endemics under threat: an assessment of the conservation status of Cuban bats. Hystrix It J Mamm 18(1):3–15
Manley GA, Köppl C, Johnstone BM (1993) Distortion-product otoacoustic emissions in the Bobtail Lizard. 1. General-Characteristics. J Acoust Soc Am 93(5):2820–2833. doi:10.1121/1.405803
Meenderink SWF, van Dijk P, Narins PM (2005) Comparison between distortion product otoacoustic emissions and nerve fiber responses from the basilar papilla of the frog. J Acoust Soc Am 117(5):3165–3173. doi:10.1121/1.1871752
Minet J, Surlykke A (2003) Auditory and sound producing organs. In: Kristensen NP (ed) Handbook of Zoology. Lepidoptera, Moths and Butterflies. Morphology, Physiology and Development, vol 2. Walter de Gruyter, Berlin, pp 289–323
Mora EC, Macías S (2011) Short Cf-Fm and Fm-short CF calls in the echolocation behavior of Pteronotus macleayii (Chiroptera: Mormoopidae). Acta Chiropterol 13(2):457–463
Mora EC, Torres L (2008) Echolocation in the large molossid bats Eumops glaucinus and Nyctinomops macrotis. Zool Sci 25(1):6–13
Mora EC, Rodriguez A, Macías S, Quiñonez I, Mellado MM (2005) The echolocation behaviour of Nycticeius cubanus (Chiroptera: Vespertilionidae): Inter-and intra-individual plasticity in vocal signatures. Bioacoustics 15(2):175–193
Mora EC, Cobo-Cuan A, Macias-Escriva FD, Pérez M, Nowotny M, Kössl M (2013) Mechanical tuning of the moth ear: distortion-product otoacoustic emissions and tympanal vibrations. J Exp Biol 216(Pt 20):3863–3872. doi:10.1242/jeb.085902
Mora EC, Cobo-Cuan A, Macías-Escrivá F, Kössl M (2015) Unexpected dynamic up-tuning of auditory organs in day-flying moths. J Comp Physiol A 201:1–10
Nakano R, Takanashi T, Fujii T, Skals N, Surlykke A, Ishikawa Y (2009) Moths are not silent, but whisper ultrasonic courtship songs. J Exp Biol 212(pt 24):4072–4078. doi:10.1242/jeb.032466
Nakano R, Takanashi T, Surlykke A (2015) Moth hearing and sound communication. J Comp Physiol A 201(1):111–121
Pérez M, Coro F (1985) Physiological characteristics of the tympanic organ in noctuoid moths II. Responses to 45 ms and 5 s acoustic stimuli. J Comp Physiol A 156(5):689–696
Pérez M, Zhantiev RD (1976) Functional organization of the tympanal organ of the flour moth, Ephestia kuehniella. J Insect Physiol 22(9):1267–1273. doi:10.1016/0022-1910(76)90105-0
Ratcliffe JM, Fullard JH, Arthur BJ, Hoy RR (2009) Tiger moths and the threat of bats: decision-making based on the activity of a single sensory neuron. Biol Lett 5(3):368–371. doi:10.1098/rsbl.2009.0079
Rodríguez RL, Schul J, Cocroft RB, Greenfield MD (2005) The contribution of tympanic transmission to fine temporal signal evaluation in an ultrasonic moth. J Exp Biol 208(21):4159–4165. doi:10.1242/Jeb.01893
Roeder KD (1967) Nerve cells and insect behavior. Harvard University Press, Cambridge
Roeder KD, Treat AE (1957) Ultrasonic reception by the tympanic organ of noctuid moths. J Exp Zool 134(1):127–157
Rydell J, Skals N, Surlykke A, Svensson MG (1997) Hearing and bat defence in geometrid winter moths. Proc R Soc Lond B 264(1378):83–88. doi:10.1098/rspb.1997.0012
Silva G (1979) Los murciélagos de Cuba. Editorial Academia, La Habana
Skals N, Surlykke A (2000) Hearing and evasive behaviour in the greater wax moth, Galleria mellonella (Pyralidae). Physiol Entomol 25(4):354–362. doi:10.1046/j.1365-3032.2000.00204.x
Spangler HG (1987) Ultrasonic communication in Corcyra cephalonica (Stainton) (Lepidoptera: Pyralidae). J Stored Prod Res 23(4):203–211
Spangler HG (1988) Sound and the moths that infest beehives. Fla Entomol:467-477
Surlykke A, Filskov M (1997) Hearing in geometrid moths. Naturwissenschaften 84(8):356–359
Surlykke A, Treat AE (1995) Hearing in Wintermoths. Naturwissenschaften 82(8):382–384. doi:10.1007/s001140050204
Takanashi T, Nakano R, Surlykke A, Tatsuta H, Tabata J, Ishikawa Y, Skals N (2010) Variation in courtship ultrasounds of three Ostrinia moths with different sex pheromones. PLoS One 5(10):e13144. doi:10.1371/journal.pone.0013144
ter Hofstede HM, Goerlitz HR, Montealegre ZF, Robert D, Holderied MW (2011) Tympanal mechanics and neural responses in the ears of a noctuid moth. Naturwissenschaften 98(12):1057–1061. doi:10.1007/s00114-011-0851-7
ter Hofstede HM, Goerlitz HR, Ratcliffe JM, Holderied MW, Surlykke A (2013) The simple ears of noctuoid moths are tuned to the calls of their sympatric bat community. J Exp Biol 216(21):3954–3962. doi:10.1242/Jeb.093294
Windmill JFC, Jackson JC, Tuck EJ, Robert D (2006) Keeping up with bats: dynamic auditory tuning in a moth. Curr Biol 16(24):2418–2423. doi:10.1016/j.cub.2006.09.066
Yack JE (2004) The structure and function of auditory chordotonal organs in insects. Microsc Res Techniq 63(6):315–337. doi:10.1002/Jemt.20051
Acknowledgements
We thank the students at the Faculty of Biology for their assistance collecting moths. Identification of the moths would not have been possible without the expert help of Rayner Nuñez and Alejandro Barro. Frank Macias-Escriva provided software for data processing and a valuable logistical support. Special thanks to A. Grinnell for his help with the revision of the manuscript. We thank the two anonymous reviewers for their valuable comments. This work was supported by the Alexander von Humboldt Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Cobo-Cuan, A., Kössl, M. & Mora, E.C. Hearing diversity in moths confronting a neotropical bat assemblage. J Comp Physiol A 203, 707–715 (2017). https://doi.org/10.1007/s00359-017-1170-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00359-017-1170-z