Abstract
Purpose
For patients with prostate cancer, validated and reliable instruments are essential for measuring patient-reported outcomes. The aim of this study was to validate the German version of the widely established Expanded Prostate Cancer Index Composite with 26 items (EPIC-26).
Methods
A German translation of the original questionnaire was tested in 3094 patients with localized or locally advanced (any T, any N and M0) prostate cancer with treatment intent (including radical prostatectomy, brachytherapy, active surveillance, watchful waiting). They completed the EPIC-26 questionnaire before treatment. A total of 521 of them also completed a questionnaire 12 months afterward. Internal consistency, sensitivity to change, and construct validity were assessed.
Results
The internal consistency of all domains was sufficient (Cronbach’s alpha between 0.64 and 0.93). Item-to-scale correlation coefficients showed acceptable associations between items and their domain score (all > 0.30), with the lowest scores for “bloody stools” (r = 0.37) and “breast problems” (r = 0.32). Confirmatory and exploratory factor analysis confirmed the five-dimension structure of the EPIC-26 (comparative fit index 0.95).
Conclusions
Psychometric evaluation suggests that the German version of the EPIC-26 is a well-constructed instrument for measuring patient-reported health-related symptoms in patients with prostate cancer.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Prostate cancer (PCa) is the most common cancer among men in Germany, with 57,370 newly diagnosed cases in 2014 [1]. As the survival rates are increasing [2] and the 5-year as well as the 10-year overall survival rates are at a high level (91% and 90%, respectively in 2014) [1], patient-reported outcome (PRO) measurements of PCa-related symptoms and functions are an important part of clinical outcome measurement. PCa patients often describe specific cancer-related and therapy-related adverse effects (AEs). Disease-related symptom measurement in PCa patients should therefore differentiate between illness-specific domains such as urinary incontinence, bowel function, sexual function, and endocrine function [3,4,5]. Measurement of these PCa-related symptoms is of interest not only in relation to investigation of individual patients’ AEs during PCa treatment, for example, but also for comparing patient outcomes across providers.
A widely established instrument for health-related quality of life (HRQoL) assessment in men with PCa is the Expanded Prostate Cancer Index Composite with 26 items (EPIC-26). The EPIC-26 questionnaire is an abbreviated version of the EPIC-50 questionnaire [6], which is used in many completed and ongoing studies. The original EPIC included a set of 50 questions. As there was a need to facilitate HRQoL measurement to allow more comprehensive research and clinical approaches, Szymanski et al. developed the EPIC-26 [7]. Subsequently, Schmidt et al. identified the EPIC-26 as being the HRQoL instrument with the best characteristics for managing change in PCa care [8]. Psychometric test results are available for several translations of the EPIC-26 (e.g., into Chinese, Norwegian, and Canadian French) [9,10,11].
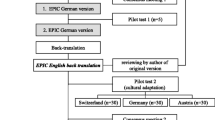
A German translation of the EPIC-26 was published by Beyer et al. [12]. Umbehr et al. [13] validated a German version of the EPIC questionnaire with 50 items (including the items in the shorter EPIC-26) using a translation different from that used by Beyer et al. as the authors were focusing more on a cultural-adaptational approach resulting in a questionnaire applicable not only in Germany, but also in the German-speaking parts of Austria and Switzerland. No results from psychometric testing of the validity and reliability of the German version of EPIC-26 as translated by Beyer et al. have yet been published. The aim of the present study was therefore to test the German version of EPIC-26 for reliability and validity using responses from patients with PCa who answered the questionnaire before and 1 year after definitive treatment. Data from the ongoing prospective, multicenter Prostate Cancer Outcomes (PCO) study (www.pco-study.com) [Kowalski et al. submitted] were used. The German PCO study is part of an ongoing international study (the TrueNTH Global Registry) that was initiated by the Movember Foundation. The aim of the study is to compare clinical and patient-reported outcomes internationally for patients with localized PCa [14]. Fourteen countries—Austria, Australia, Canada, the Czech Republic, Denmark, Finland, Germany, Ireland, Italy, Spain, the Netherlands, New Zealand, the UK, and the USA—are currently participating in the TrueNTH Global Registry.
Ethical approval
The PCO study was approved by the local ethics committee in Berlin (Eth-12/16). All of the patients included provided written informed consent.
Materials and methods
Patients
Patients with localized or advanced PCa (any T, any N, M0) are asked to complete the EPIC-26 questionnaire before and 1 year after definitive treatment. Patients treated with active surveillance (AS) or watchful waiting (WW) are asked to complete the follow-up questionnaire 12 months after being included in the study. PCa-specific clinical parameters (e.g., cancer stage according to the German Guideline for Prostate Cancer presented in Appendix 4, type of therapy, comorbidities) as well as patient-reported outcomes (PROs) measured using the EPIC-26 were collected in a setting of consecutive inclusion of patients between July 2016 and March 2018, in accordance with the reference guide of the International Consortium for Health Outcomes Measurement (ICHOM) standard set for localized prostate cancer [15, 16]. The study includes PCa patients receiving radical prostatectomy including cystoprostatectomy (RPE), radiation therapy (RT), watchful waiting (WW), active surveillance (AS), and other treatments (OT, such as androgen deprivation or focal therapy). All risk groups (low, medium, high, T3/T4, N1) are represented.
Questionnaire validation was conducted on the basis of the responses given by these patients in the participating German prostate cancer centers. Since this ongoing study started in July 2016, post-treatment data (12 months after study inclusion) were already available for patients who were recruited early during the study.
The German version of the EPIC-26
The EPIC-26 is a set of 26 items, which are categorized into one single item (overall urinary problems) and five subdomains consisting of four to six items: incontinence (four items), irritative and obstructive urinary problems (four items), bowel function (six items), sexuality (six items), and vitality/hormonal function (five items). Responses are measured on a Likert-type scale. The original and German versions of the EPIC-26 are included in the appendixes (Appendixes 2 and 3).
The German version of the EPIC-26 published by Beyer et al. [12] is used in this study. The process of translation and the evaluation of the translation are described in that paper.
Statistical analysis
Reliability was measured using the responses before definitive treatment by calculating interdomain correlations and Cronbach’s alpha coefficients [17] as an indicator of internal consistency. Item-to-scale correlations for each subdomain were carried out as described by Cohen [18]. Sensitivity to change was examined by calculating correlations between EPIC-26 scores before and 12 months after treatment for those patients with available data. Paired Student’s two-sample t test was performed.
Construct validity was analyzed on the basis of responses before definitive treatment by using a confirmatory factor analysis to examine the five-subdomain structure and the scales’ dimensionality in the EPIC-26. Beforehand, an exploratory factor analysis was performed to analyze whether a five-subdomain structure is statistically tenable. Maximum likelihood with robust correction was used.
The open-source software program RStudio 1.1.383 was used for all statistical analyses.
Results
Sample
A total of 3094 PCa patients from 44 German PCa centers were included. At the time of data transfer (April 2018), post-treatment data were already available for 521 of these patients (within the 1-year follow-up time frame, 839 patients were asked to answer the post-treatment questionnaire). The mean age at diagnosis in the total patient sample was 66 years. In all, 2608 patients were treated with RPE alone; 221 with RT; 172 patients with a combination of RPE and RT; 79 patients with AS or WW; and 14 patients received other treatment options, including radical cystectomy (cf. Kowalski et al. submitted) (Table 1).
Statistics
Reliability
Reliability was investigated before treatment (T0) on the basis of the total patient sample. Item-to-scale correlation ranged from 0.32 to 0.95, with the lowest coefficients for the items “bloody stools (#6d)” (r = 0.37) and “breast problems (#13b)” (r = 0.32) (Table 2). Internal consistency was assessed by calculating Cronbach’s alpha for the EPIC-26 subdomains, where a Cronbach’s alpha > 0.65 was considered sufficient [19]. This was possible for all but one subdomain; for the urinary irritative/obstructive subdomain score alone, Cronbach’s alpha was 0.64 (Table 3). Table 2 provides an overview of the characteristics of the subdomains (in comparison with selected findings from Szymanski et al. [7]).
Table 4 shows the interdomain correlation matrix. The low correlation scores (range between 0.11 and 0.32) indicate sufficient discrimination between the single scores and thus confirm the five-dimension structure of the EPIC-26 (all P < 0.001).
Sensitivity to change
Sensitivity to change was investigated in 521 patients for whom data were available for questionnaire responses 12 months after treatment. For all subdomains, significant changes over time were found (P < 0.001), with the greatest difference being seen in the sexual domain (Table 3).
Construct validity
Construct validity was investigated before treatment using the total patient sample. After factor extraction using Kaiser’s criterion (all factors with an eigenvalue greater than one are retained), solutions with up to six factors were possible. Exploratory factor analysis was therefore performed with four, five, and six latent variables. After extraction of five factors and factor rotation using the varimax method, the results showed a fit at χ2 = 1664.44 (n = 3094, degrees of freedom 185, P < 0.001), a standardized root mean square residual (SRMR) of 0.02, and a Tucker–Lewis index of 0.94. This five-factor solution was superior to the four-factor and six-factor solutions (cf. Appendix 1). For all exploratory factor analyses, item #5 was not taken into account, as it is not used in the original EPIC-26 or any validated translation of it. A confirmatory factor analysis with five latent variables (the five domains of the EPIC-26) was thus estimated using the robust maximum likelihood estimator. After 68 iterations, the results showed a fit at χ2 = 991.72 (n = 3094, degrees of freedom 265 after 60 iterations, P < 0.001), a comparative fit index (CFI) of 0.95, a root mean square error of approximation (RMSEA) of 0.04, and an SRMR of 0.04. Factor loadings ranged between 0.12 and 0.95, with the weakest association with their latent variable for the items “hematuria (#4c)” (B = 0.22), “bloody stools (#6d)” (B = 0.19) and “breast problems (#13b)” (B = 0.12) (Table 2).
Discussion
The aim of this study was to validate the German version of the EPIC-26 questionnaire. In general, the results of the psychometric tests described are similar to those for the original American questionnaire [7]. As a result, the findings suggest that the German version of the EPIC-26 can be used as a reliable and valid instrument to analyze symptoms and function in PCa patients.
The mean scores for the different time points were comparable to previously published results [7, 10, 11] (Table 3). However, differences between the study samples and timing of measurements in comparison with validations carried out for other languages need to be carefully discussed, as some of the published studies report results of questioning before treatment and some after it. Eighty-five percent of the patients included in the present sample, compared with 76% in the source population for these patients, were treated with RPE. Common AEs consequently include erectile dysfunction, for example, which is less prevalent in patients who undergo RT. The relatively low score for the sexual domain 12 months after treatment (mean = 24.28) in the sample therefore needs to be interpreted with these circumstances being taken into account. However, it may be assumed that the over-representation of patients with RPE in the study does not impede the quality of the questionnaire validation presented here.
The findings showed that the German version of the EPIC-26 questionnaire has sufficient reliability. The internal consistency of the scales (with Cronbach’s alpha ranging between 0.64 and 0.93) and item-to-scale correlation coefficients (all ≥ 0.30) were acceptable. A Cronbach’s alpha > 0.65 was considered satisfactory. Only the urinary irritative/obstructive subdomain had a Cronbach’s alpha below this threshold (α = 0.64). This has also been reported for other versions of the EPIC-26 [10, 11], suggesting weaker internal consistency for this subdomain in general that is mainly due to the item “hematuria.”
Psychometric testing commonly requires item-to-scale correlation coefficients to be over 0.40, or at least not less than 0.30, to confirm adequate reliability. As reported above, item-to-scale coefficients of between 0.30 and 0.40 were found for the items “bloody stools” and “breast problems.” This is consistent with previously published results [10, 11]. For the “breast problems” item, this low correlation with the vitality/hormonal domain was also reported for the original EPIC-26 questionnaire. As these findings have now been confirmed by several studies, it may be suggested that the construction of the vitality/hormonal domain should be revised, and providers may be advised to interpret the single items rather than the total score.
An acceptable level of sensitivity to change was found, analyzed by comparing the responses of patients before and 12 months after treatment with only the bowel and vitality/hormonal subdomain, showing low mean differences which are still statistically significant. This corresponds to clinically known AEs in PCa patients after treatment [4, 20]. Since only 33 of the 521 patients included in the analyses of sensitivity to change were treated with RT, the differences in EPIC-26 reported mostly involve RPE-related side effects.
Four-dimension, five-dimension, and six-dimension structures in EPIC-26 were compared using exploratory factor analysis. Kaiser’s criterion suggested six factors. However, Kaiser’s criterion often overestimates the number of factors [21]. After comparison of the three possible solutions, and without taking into consideration the item “overall urinary problem” (item #5), which is not used for domain construction in any of the existing translations, extracting five factors appeared to be the most convincing approach in relation to factor loadings, discriminatory power, and the authors’ intention to cover the five domains in the instrument. Exclusion of the item “overall urinary problem” was based on the questionnaire’s construction process. If the item is permitted for the factor analysis, a four-factor solution combing items #1 to #5 (all urinary items) is also statistically approved. The five-dimension structure of the EPIC-26 was also confirmed using confirmatory factor analysis. The weakest loading of an item and its postulated factor (e.g., subdomain) were found for the items “hematuria” and “breast problems.” As these findings are also reported in other studies, the importance of these two items for the domain may be questionable.
Limitations
Test–retest reliability and convergent validity were not examined in the present study. The large sample size (n = 3094) and the multicenter approach may be regarded as strengths of the study.
Conclusion
Measuring patient-reported outcomes is attracting increasing interest in oncological research and also in health-care provision. Valid and reliable psychometric instruments are therefore needed to evaluate disease-related symptoms in patients with prostate cancer. An international consensus group has strongly recommended using the EPIC-26 questionnaire for comparable and standardized evaluation of PROs and AEs in patients with localized prostate cancer [15]. The psychometric findings presented here mean that the German version of EPIC-26 is now available as a well-validated and reliable instrument for further PRO research and can be recommended for German-speaking patients with prostate cancer.
References
Zentrum für Krebsregisterdaten and Gesellschaft der epidemiologischen Krebsregister in Deutschland (2017) Krebs in Deutschland für 2013/2014. Robert Koch-Institut, Berlin
Trama A et al (2015) Survival of male genital cancers (prostate, testis and penis) in Europe 1999-2007: results from the EUROCARE-5 study. Eur J Cancer 51(15):2206–2216
Litwin MS et al (1995) Quality-of-life outcomes in men treated for localized prostate cancer. JAMA 273(2):129–135
Madalinska JB et al (2001) Health-related quality-of-life effects of radical prostatectomy and primary radiotherapy for screen-detected or clinically diagnosed localized prostate cancer. J Clin Oncol 19(6):1619–1628
Sanda MG et al (2008) Quality of life and satisfaction with outcome among prostate-cancer survivors. N Engl J Med 358(12):1250–1261
Wei JT et al (2000) Development and validation of the expanded prostate cancer index composite (EPIC) for comprehensive assessment of health-related quality of life in men with prostate cancer. Urology 56(6):899–905
Szymanski KM et al (2010) Development and validation of an abbreviated version of the expanded prostate cancer index composite instrument for measuring health-related quality of life among prostate cancer survivors. Urology 76(5):1245–1250
Schmidt S et al (2014) Assessing quality of life in patients with prostate cancer: a systematic and standardized comparison of available instruments. Qual Life Res 23(8):2169–2181
Lam WWT et al (2017) Psychometric assessment of the Chinese version of the abbreviated expanded prostate cancer index composite (EPIC-26) and the clinical practice version (EPIC-CP) in chinese men with prostate cancer. J Pain Symptom Manage 53(6):1085–1090
Fossa SD et al (2016) Psychometric testing of the Norwegian version of the expanded prostate cancer index composite 26-item version (EPIC-26). Scand J Urol 50(4):280–285
Vigneault E et al (2017) Validation of the French-Canadian version of the expanded prostate cancer index composite (EPIC) in a French-Canadian population. Can Urol Assoc J 11(12):404–410
Beyer B et al (2015) “Expanded prostate cancer index composite” (EPIC-26): Funktionelles Behandlungsergebnis bei Patienten mit lokalisiertem Prostatakarzinom. Urologe A 54(11):1591–1595
Umbehr MH et al (2018) The German version of the Expanded Prostate Cancer Index Composite (EPIC): translation, validation and minimal important difference estimation. Health Qual Life Outcomes 16(1):36
Evans SM et al (2017) Cohort profile: the TrueNTH Global Registry: an international registry to monitor and improve localised prostate cancer health outcomes. BMJ Open 7(11):e017006
Martin NE et al (2015) Defining a standard set of patient-centered outcomes for men with localized prostate cancer. Eur Urol 67(3):460–467
ICHOM ( 2017) Localized prostate cancer data collection reference guide. https://ichom.org/files/medical-conditions/localized-prostate-cancer/localized-prostate-cancer-reference-guide.pdf. Accessed 23 Sep 2019
Cronbach LJ (1951) Coefficient alpha and the internal structure of tests. Psychometrika 16:297–334
Cohen J (1988) Statistical power analysis for the behavioral sciences. N.J.L. Erlbaum Associates, Hillsdale
Nunnally CJ, Bernstein I (1994) Psychometric Theory. McGraw-Hill, New York
Reeve Bryce B et al (2018) Symptom and function profiles of men with localized prostate cancer. Cancer 124(13):2832–2840
Nunnally JC, Bernstein IH (1994) Psychometric theory, 3rd edn. McGraw-Hill, New York
German Guideline Program in Oncology (2018) ed. Interdisziplinäre Leitlinie der Qualität S3 zur Früherkennung, Diagnose und Therapie der verschiedenen Stadien des Prostatakarzinoms, Version 5.0, 1. Aktualisierung, AWMF-Registernummer: 043/022OL
Wei J, Dunn R, Litwin M, Sandler H, and Sanda M (2000) Development and validation of the expanded prostate cancer index composite (EPIC) for comprehensive assessment of health-related quality of life in men with prostate cancer. Urology 56:899–905
Funding
This study was funded by the Movember Foundation.
Author information
Authors and Affiliations
Contributions
NT Sibert: data analysis, data collection and management, manuscript writing/editing; S Dieng: protocol/project development, data collection and management, manuscript writing/editing; A Oesterle: data analysis, data collection and management; G Feick: protocol/project development, manuscript writing/editing; G Carl: protocol/project development, manuscript writing/editing; T Steiner: data collection and management, manuscript writing/editing; J Minner: data collection and management, manuscript writing/editing; F Roghmann: data collection and management, manuscript writing/editing; B Kaftan: data collection and management, manuscript writing/editing; F Zengerling: B Kaftan: data collection and management, manuscript writing/editing; A Hinkel: data collection and management, manuscript writing/editing; B Beyer: data collection and management, manuscript writing/editing; A Heidenreich: data collection and management, manuscript writing/editing; N Harke: data collection and management, manuscript writing/editing; B Brehmer: data collection and management, manuscript writing/editing; J Pfitzenmaier: data collection and management, manuscript writing/editing; J Fichtner: data collection and management, manuscript writing/editing; A Neisius: data collection and management, manuscript writing/editing; P Hammerer: data collection and management, manuscript writing/editing; S Wesselmann: protocol/project development, manuscript writing/editing; C Kowalski: protocol/project development, data analysis, manuscript writing/editing.
Corresponding author
Ethics declarations
Conflicts of interest
GC, GF, TS, JM, FR, FZ, AHi, BB, AHe, NH, BB, JP, JF, AN, and PH hereby declare that they have no potential conflicts of interest. NTS, CK, SD, AO, and SW are employees of the two institutions in charge of the certification system. BK receives consulting and lecturing honoraria from various companies that do not involve any conflicts with the research presented here. A comprehensive list is available on request.
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the ethics committee of the Medical Association of Berlin (Eth-12/16) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendix 1: Comparison of the four-, five- and six-factor solutions
Appendix 2: The EPIC-26—Expanded Prostate Cancer Index Composite Short Form [23]



Appendix 3: German translation of the EPIC-26 [12]



Appendix 4: Staging according to the German Guideline Prostate Cancer (version April 2018)
In this study we use the staging proposed by the German Guideline for Prostate Cancer [22]:
-
Localized prostate cancer: T1-2, N0 M0.
-
Localized prostate cancer with low risk: PSA ≤ 10 ng/ml and Gleason-Score 6 and cT1c or cT2a.
-
Localized prostate cancer with intermediate risk: PSA > 10 ng/ml–30 ng/ml or Gleason 7 or cT 2b.
-
Localized prostate cancer with high risk: PSA > 20 ng/ml or Gleason ≥ 8 or cT2c.
-
-
Locally advanced prostate cancer: T3-4 N0 M0.
-
Advanced prostate cancer: any T N1 and M0.
-
Metastasized prostate cancer: any T any N and M1.
Furthermore, localized prostate cancer with cT1a or cT1b is classified as localized prostate cancer with low risk.
Rights and permissions
About this article
Cite this article
Sibert, N.T., Dieng, S., Oesterle, A. et al. Psychometric validation of the German version of the EPIC-26 questionnaire for patients with localized and locally advanced prostate cancer. World J Urol 39, 11–25 (2021). https://doi.org/10.1007/s00345-019-02949-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00345-019-02949-7