Abstract
Despite the importance of diverse plant growth-promoting endophytes in agricultural production, their biotechnological and agricultural applications are not well-documented. The diversity of microbial communities interacting with the endosphere contributes to plant functions and immunity, leading to higher productivity. Plant-microbe interactions range from beneficial in terms of influencing plant growth to harmful, as they also cause plant diseases. Microbial survival in the internal tissues of plants depends on their colonization tendencies and their ability to compete with the indigenous plant microflora. The infiltration of microbes through the external soil-root environment into the plant endosphere significantly enhances growth-promoting attributes of plants such as antibiosis, siderophore production, induced systemic resistance, bioremediation and growth hormones synthesis. However, the growth and diversity of endophytic microbes are influenced by the availability of soil nutrients, presence of pathogens, plant growth stages, plant genome, and other abiotic factors. Knowledge and understanding of the possible use and biotechnological relevance of endosphere communities in sustainable agriculture cannot be overemphasized. Hence, this review discusses the importance of endophytic microbes in agriculture for enhancing crop productivity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Agrochemical use in various agricultural practices such as fertilizer and pesticide applications for enhancing crop yield has been the conventional means of crop production for a very long time (Qaswar et al. 2020). However, the long-term use of agrochemicals deteriorates soil parameters, alters soil ecological stability and reduces plant performance (Gouda et al. 2018). Research into plant endosphere communities is gaining attention, and the use of agriculturally important microbes to mitigate plant stressors has yielded promising results. Hence, there is a need to optimize soil conditions to favor the resident microorganisms in the biotransformation of organic matter; this will make soil nutrients available to plants and therefore improve the soil’s health (Uzoh and Babalola 2018). Maximum agricultural food production for the world’s growing population can be achieved through agricultural intensification with modern technologies.
Different elemental nutrients such as nitrogen, iron, carbon, phosphorus and potassium are recycled by microorganisms inhabiting the root endosphere (Adegbeye et al. 2020). They are involved in the overall performance of plant growth by contributing to the acquisition of nutrients and carbon as energy sources in a given niche. The agricultural importance of soil microorganisms in crop productivity cannot be overemphasized and this has led to research in plant endosphere colonizers (Alster et al. 2020). Generally, soil microbes form a close association with the host plant either by inhabiting the rhizosphere or colonizing the internal tissues, called the endosphere (Truyens et al. 2015; Alawiye and Babalola 2019). The endosphere is the region within the tissues of plants that harbor diverse microbial communities comprising bacteria, fungi and archaea with complex multifunctional properties (Adeleke and Babalola 2020). Different microbial colonizers inhabit the external surface of plants compared to intracellular space within the tissues of the host plant (Potshangbam et al. 2017). The microbes that colonize the internal tissues of the host plants, referred to as microbial endophytes (Gupta et al. 2020b), have the potential to establish mutual, harmful, or neutral relationships with diverse plant species (Dobereiner 1992; Hardoim et al. 2015). Beneficial microbial endophytes symbiotically colonize the intracellular space of the plant tissues without causing harm or eliciting disease conditions in the host plants (Kandel et al. 2017).
Endophytes are regarded as a subset of rhizosphere microbes that infiltrate plant tissues by a vertical method via seed inoculation or by horizontal networking from the rhizosphere (Adeleke and Babalola 2021). In either of these methods, plant-microbe interactions have either mutual benefits or pathological effects on the host plant (Huang et al. 2016). Endophyte infiltration is facilitated by exudate secretion containing signal molecules in exchange for nutrients in-and-out of the soil-root environment (Glick and Bashan 1997; Alawiye and Babalola 2019). The complex root structure determines the number of exudates secreted. Exudates contain biomolecules such as fatty acids, oligosaccharides, sugars, phenolic, carbohydrates, enzymes, flavonoids, amino acids, volatile compounds, mucilage, nucleotides, sterol, vitamins, and structural carbohydrates, which provide carbon-energy sources for the microorganisms (Jambon et al. 2018). The endophytic community uses these multifunctional compounds as nutrient sources (Adeleke and Babalola 2020). The number of endophytes found in plants can influence plant physiology, affecting plant growth and fitness (Verma and White 2018).
The root endophyte community facilitates the exchange of molecules in response to interactions between microbes and chemical substances secreted from the plant roots. This exchange enhances plant health and growth (Santoyo et al. 2016). Under certain environmental conditions, endophytic microbes are responsible for reducing the effects of drought, high temperature, the stress of nutritional deficiency, and the infestation of phytopathogens on crops (Jha and Subramanian 2018; Adeleke and Babalola 2021). Thus, adequate utilization of plant nutrients by plant-associated microorganisms enables them to exert diverse beneficial effects on the host plant.
Soil and endosphere microbial populations may vary in terms of diversity and relative abundance due to geographical locations (Lin et al. 2020), age of plants, weather conditions (winter or summer) (Hashem et al. 2019), and agricultural practices such as tilling and fertilizer application (Wang et al. 2018). The survival of microorganisms within the host plant depends on their colonization potential in establishing a cell biomass community. Factors including geographical location, plant type, soil quality, genotype, and tissue, presence of pathogens such as bacteria, viruses, nematodes, plant growth stages, and mechanisms of colonization pathway influence the endophytic population in plants (Zhang et al. 2020a).
The use of endophytes as bioinoculants as an alternative to conventional crop improvement and plant disease prevention techniques in agriculture has gained prominence in protecting crops against phytopathogens, thereby enhancing healthy plant growth (Alawiye and Babalola 2019). The use of bioformulants (biofertilizers/biopesticides/bioherbicides) derived from microbial endophytes are useful in crop management to control weeds (Souza et al. 2017). Since endophytic microbes are involved in the cycling of mineral elements and can boost plant immunity, their introduction through biotechnological approaches into the endosphere community may eventually reduce reliance on the use of chemical fertilizers and pesticides by replacing them with helpful agricultural microbes.
It is imperative to understand the diverse potentials of the microbial communities inhabiting the plant endosphere to ensure sustainable improvement in crop productivity through agricultural intensification (Adeleke and Babalola 2020). Based on the varied plant growth-promoting attributes of endophytes, it is necessary to explore the potential application of endophytes in agriculture and understand how they influence plant physiology. This review will discuss microbial endophyte ecology and highlight their role in agriculture.
Endosphere Microbial Communities Associated with Plants
The root, stem, leaf, and seed endosphere harbor diverse microbial communities that form close interactions with the host plant, i.e., promoting plant growth and health, as plants shelter the endophytes (Mukherjee et al. 2020). Mechanisms of endophyte colonization with the host plant are not yet fully understood. Endophytic microbes are predominantly associated with most economical plants such as maize, wheat, sorghum, wheat, and soybean. The culturable microbial endophytes occupy the discreet regions in the endosphere (Adeleke and Babalola 2020). The major bacteria group dominating the plant endosphere include Gram-negative and Gram-positive bacteria like rhizobacteria while endophytic mycorrhiza fungi found in the soil also form a close association with the roots of the plants (Saharan and Nehra 2011). Fungal and bacterial endophytes in the endosphere of different plant organs have been characterized (Tyc et al. 2020). For example, microbial endophytic genera such as Bacillus, Lysinibacillus, Brevibacillus, Paenibacillus, Nocardioides, Curtobacterium, Microbacterium, Brachybacterium, Kocuria, Micrococcus, Fusarium, Xylaria, Cladosporium, Acremonium, Epicoccum, Curvularia have been identified in seeds, leaves, and stems of common bean (Phaseolus vulgaris) and Vitis vinifera (Parsa et al. 2016). The common examples of endophytic bacterial and fungal genera associated with plants are shown in Table 1.
Harnessing bioincentives from microbial endophytes isolated from legumes and non-legume plants such as cotton wool (Populous deltoids), sweet potato (Ipomoea batata), soybean (Glycine max), and tomato (Solanum lycopersicum), and their application have contributed to crop yields (Mashiane et al. 2017). Similarly, the co-existence between bacteria genera Methylobacterium, Pseudomonas, Caulobacter, Erwinia, Acidovorax, Chryseobacterium, Paenibacillus, Sphingomonas, Corynebacterium, Acinetobacter, Devosia, and Brevundimonas; and fungi genera Verticillium, Aspergillus, Eurotium, Fusarium, Wallemia, Septoria, Pestalotiopsis, Penicillium, Trichosporon, and Cryptococcus in sprout, stems, and roots of rice (Oryza sativa) have supported plant growth and improvement in rice production (Wang et al. 2016a). The importance of endophytic microbes in plant growth promotion depends on their ability to fix nitrogen, synthesize indole-3-acetic acid, and produce siderophores, phytohormones, and antimicrobial compounds (Pham et al. 2017). Examples of such endophytes include Azoarcus spp BH72 and Pseudomonas stutzeri A1501 from rice (Wang et al. 2016b; Pham et al. 2017), Gluconacetobacter diazotrophicus PaI5 from sugarcane (James et al. 2001), Azospirillum lipoferum 4B from maize (Garcia et al. 2017), Burkholderia phytofirmans PsJN from grapevine (Compant et al. 2008), and Enterobacter cloacae ENHKU01 from pepper (Santoyo et al. 2016).
The endophytic community found in the root zone easily derived all-inclusive multifunctional compounds as nutrient sources (Dubey et al. 2021). Most microbes found in the endosphere might display competent novel genes in promoting plant growth. However, only 1% of the total microbial population in the endosphere has been isolated and identified by conventional isolation techniques. Thus, further research into their isolation can make them useful tools in future agricultural production (Hiruma et al. 2018).
Microbial endophytes can directly influence plant growth by nitrogen fixation, production of growth-promoting stimulators like phytohormones (cytokinins, gibberellins, and auxin), phosphate solubilization, synthesis of biological control agents in combating soil and plant pathogens, production of secondary metabolites such as antibiotics, siderophores, ammonia and hydrogen cyanide (Verma et al. 2015). Additionally, microbial endophytes play an important role in plant response to withstand harsh environmental conditions by inducing systemic resistance (Zhang et al. 2020a).
The survival of bacterial endophytes in the root region or other plant organs can support plant adaptation to different environments by synthesizing metabolic chemical compounds and lytic enzymes (Bashan et al. 1995). The metabolic activities contribute to the development, growth, and immunity of plants. For instance, auxin production from endophytes can mobilize or activate plant growth by cell division, elongation, and differentiation depending on the plant response to environmental stimuli (Shi et al. 2009). 1-Aminocyclopropane-1-carboxylate deaminase (ACCD) synthesis by plant endophytes can enhance plant growth under stress conditions by lowering the rate of ethylene production and reducing pathogen effects on the host plants (Glick 2014). Ethylene production in the endosphere modulates plant physiology and is an emerging strategy for microbial signaling in a host plant (Santoyo et al. 2016). The strong affinity, i.e., the chelating potential of endophytic microbes to Fe(III) by synthesizing siderophores also enables them to limit plant pathogen infestation on crops (Jiao et al. 2020). Plant endophytes function in phosphate solubilization, further aided by organic acid secretion to facilitate the conversion of insoluble phosphates into soluble phosphate derivative (monobasic or dibasic) ions, thus making it accessible and utilizable by plants (Shahid et al. 2015).
The production of specific metabolites by fungal endophytes acts as biocontrol (Mane and Vedamurthy 2018). These fungi positively influence plant growth through the secretion of biological control agents, which act against potential plant pathogens (Bashan and De-Bashan 2010). Like bacterial endophytes, endophytic fungi have been isolated from plants. The study of fungal endophytes has attracted mycologists, soil ecologists, and environmental microbiologists because of their inherent importance in agriculture (Adeleke and Babalola 2021; Afzal et al. 2019).
Below-ground and Above-ground Microbial Endophyte Inhabitants
The plant types and nutrients available in a microhabitat facilitated the recruitment and establishment of different microbial communities in the endosphere (van Overbeek and Saikkonen 2016). Diverse microbial communities inhabit different ecological niches, including below-ground and above-ground habitats that provide unique environments in an ecosystem (Oliveira et al. 2002).
These below-ground and above-ground habitats contain agriculturally important microorganisms, and their exploration is promising in agricultural productivity (Cole et al. 2018). The microorganisms from these habitats may resemble each other based on their morphological features but have different genetic compositions that determine their functions (Adeleke and Babalola 2020). The genetic composition and mechanisms exhibited by endophytic microbes are important in eliciting their functions on the host plant as different functions are linked with the specific endophyte domain (Table 2).
Harnessing plant-microbe resources is appropriate in ensuring sustainable agriculture. By implication, the exploitation of microbial endophytes is a key factor in addressing environmental challenges derived from soil stressors (Sandargo et al. 2019). Current findings on plant endophytic communities, diversities and their functions have addressed several ecological challenges that make them important in sustainable agriculture.
Various studies have identified endophytic bacterial phyla, Actinobacteria, Bacteroidetes, Firmicutes, and Proteobacteria from Arabidopsis thaliana, Brassica chinensis, G. max, V. vinifera, and Zea mays (Reeve et al. 2015; Truong et al. 2017). The advancement of molecular tools and bioinformatics as applied in microbial genomic studies has revealed information on the genomic content of plant-associated microbial endophytes (Levy et al. 2018). Research using modern molecular tools is still underway to obtain further knowledge of the functions of bacterial endophytes that would aid their commercialization as bioinoculants.
Plant Roots Structural Complexity and Endophytes Colonization
Changes in environmental conditions can influence microbial populations in the host plants. The plant roots which are responsible for skeletal support and absorbing and transporting soil nutrients needed for plant growth, also harbor highly diverse numbers of microorganisms due to excess release of exudates. This has resulted in microbial diffusion from the rhizosphere into the endosphere, by vertical or horizontal means (Adeleke and Babalola 2020). The colonization tendencies of bacterial endophytes into the plant roots depend on many factors which include the age of the plant, root architecture and environment; plant species, availability of soil nutrients, and plant developmental stages (Afzal et al. 2019). Bacterial endophyte attachment is aided by certain lytic enzymes that disrupt the plant cell wall, thus creating permissible pathways for easy penetration and colonization within the plant tissues by specialized organelles such as flagella or pili (Zheng et al. 2015). However, the presence of harmful microorganisms such as nematodes, viruses, and certain bacteria are factors that negatively affect the colonization tendencies of bacterial endophytes.
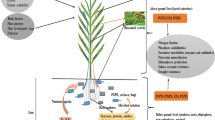
The plant-microbe interactions and infiltration into the root zone are complex (Fig. 1). Endophytic bacteria positively contribute to plant nutrition, facilitate root nutrients absorption and the release of metabolites, chelates, or enzymes to mobilize insoluble nutrients (Alawiye and Babalola 2019). The mutual association between PGPE and mycorrhizal fungi benefits plants, even under stress conditions, thus making them potential candidates in the formulation of biofertilizers. PGPE with known promotional potential for plant growth includes Bradyrhizobium, Pseudomonas, Bacillus, Paenibacillus, Burkholderia, Agrobacterium, Klebsiella, Streptomyces, Rhizobium, Arthrobacter, Brevibacillus, Alcaligenes, Azotobacter, Enterobacter, Serratia, and Azospirillum (Bashan 1999; Kaur et al. 2016; Nehra et al. 2016).
A diagram of endophytes interactions and infiltration into plant roots. a Endophytes colonizing the intracellular spaces of plant root; b root hair; c exudates secretion to the surrounding root environment; d the rhizosphere region; e microbial attachment and colonization onto the root; f signal networking of plant endophytic microbes interaction; g leaf-associated endophytes
The biofertilizer produced from PGPE conserves and influences other soil microorganisms in stimulating plant growth and other physiological functions when applied to the soil (Hong et al. 2018). PGPE and arbuscular mycorrhizal (AM) fungi are further categorized as subgroups of plant growth-promoting microorganisms (PGPM). Plant growth-promoting rhizobacteria (PGPR) such as Chryseobacterium humid, Pseudomonas reactants, and AM such as Rhizophagus irregularis improve crop yields (Moreira et al. 2016). PGPE and AM can be applied in combination with organic manure as a good alternative to chemical fertilizers and pesticides to maximize crop productivity and reduce environmental pollution. Biofertilizers containing bacteria or fungi can be applied singularly or combined to agricultural soil for higher crop productivity (Gouda et al. 2018).
The lateral root structure of a plant provides a pathway of entry for bacteria into the host tissue, functioning analogously to the plant root system (Dong et al. 2018). Plant hormones such as auxin initiate biological processes involving lateral and adventitious root formation and elongation (López-Bucio 2003). The roots of plants penetrate deeply below ground level, providing mechanical supports needed by plants to absorb nutrients from the soil by capillary networking in channeling nutrients up to the stem and other parts of the plants through plant supporting tissues (xylem and phloem) (Dobbelaere et al. 2003).
Plant roots naturally surround with nutrients, releasing exudates containing monosaccharide sugars, necessitating the build-up of microbial activities entering the plants at the early developmental stage. These nutrients also support the growth of mycorrhizal fungi that enable them to produce spores, promote mycelial growth, accelerate bioremediation of pollutants, and enhance root protection against infection. Also, secretion of plant hormones that cause root development and supply of nutrients for plant growth contribute to their ability to survive in a nutrient-limiting environment. The secretion of these metabolites, such as sugars, amino acids, and water supports, can greatly contribute to the viability of microbes in the endo-rhizosphere (Alawiye and Babalola 2019). Hence, plant root structures influence plant growth and microbial populations to survive external environmental stress (Enebe and Babalola 2018).
Plant Growth Stimulating Factors and Ecological Factor Consequences
Diverse groups of microorganisms exist in nature. They are found in different environments such as plants, soil, water and air. Plants stand as a major ecological niche for innumerable microorganisms inhabiting their surrounding soil and plant organs (Taghavi et al. 2009). Their strong affinity in establishing close association makes them inhabit the plant surface (rhizoplane) and colonize intracellular spaces within the plant tissues without causing harm (Adeleke and Babalola 2020). These microorganisms stimulate plant growth factors that enable them to exert growth effects on plants, as well as the source of nutrients for other beneficial microorganisms that might cohabit within the internal tissues of the host plant (Santoyo et al. 2016). Culturing endophytes in a culture medium under favorable conditions has made possible the exploration of developing unaltered microbial resources; and their production in large quantities to formulate bio-inoculants such as bio-fertilizer that can be adjuvants in enriching soil nutrients for higher crop productivity (Bashan 1998; Verma and White 2018). However, to formulate biofertilizers maximally, it is fundamental to have in place suitable biological carriers that can facilitate the proliferation of microbial cells and sustain microbial viability under storage conditions after production.
The survival of endophytic microorganisms in plants is largely affected by the limited supply of nutrient and environmental conditions such as rainfall, temperature, drought, and soil parameters (Edwards et al. 2015). Under certain conditions, plant beneficial endophytes can be opportunistic with the tendency of causing disease symptoms in plants. Therefore, an antagonistic mechanisms approach in the effective control of plant pathogens is crucial. Studies have shown that the biocontrol efficacy of native plant growth-promoting rhizobacteria against rhizome rot disease that cause light yellowing of leaf in turmeric plants (Chenniappan et al. 2019). Research findings on the disease conditions caused by Pseudomonas syringae in olive, green bean (leaf spot), tobacco (leaf spot), and tomato (soft rot) have been linked to their ability to produce toxins and resist antimicrobial substances (Xin et al. 2018). The cause of fire blight diseases in many ornamentals and fruit trees by Erwinia amylovora has resulted in yield loss and reduction in crop productivity (Doolotkeldieva and Bobusheva 2016). Ralstonia solanacearum, Xanthomonas spp, and Xylella fastidiosa have been classified as the potential etiological agent of wilt in potato and moko disease in banana (Wubshet 2018).
Endophytes isolated from the same plant cultivar might differ in genotype but share similar effects on plant physiology and biocontrol activities against plant pathogens. The endophytic mechanisms in a host plant can displace pathogens in the same ecosystem by instigating plant defense mechanisms and producing active biological compounds. The cause of disease symptoms in plants depends on host plant susceptibility, suitable growth conditions, plant microflora, and other pathogens (Eljounaidi et al. 2016).
Roles of PGPE in Enhancing Agricultural Crop Productivity
The biological and metabolic process involve the recycling of soil nutrients, biotransformation of environmental waste into soluble form for easy absorption by plants as well as the synthesis of certain biological molecules in the control of plants pathogens attack (Compant et al. 2010; Ojuederie et al. 2019). For example, the application of PGPE Azospirillum spp associated with the root of the Cactus plant and Cyanobacteria from drought-prone areas has contributed to soil quality, aeration, water-retention capacity, and soil elemental carbon to nitrogen ratio (C:N) (Romeh and Hendawi 2014).
Endophytes can develop vigor to the plant growth promotion effects, resistance to drought, and tolerance to poor soil conditions (Teixeira et al. 2019). The improvement of plant growth can be influenced by compounds like phytohormones produced by microbial endophytes. The indole-3-acetic acid (IAA) secreted by endophytic fungi Colletotrichum alatae, Fusarium spp, Cylindrocarpon spp, Cladosporium tenuissium, Trichoderma harzianum, and Hypoxylon spp isolated from Dendrobium moniliforme stimulate plant growth and protect them against pathogens (Glick et al. 2001; Shah et al. 2019). Apart from auxins, cytokinin, gibberellin, and organic acids produced as principal mechanisms by endophytes, other indirect mechanisms that aid plant growth are known. The cytokinins produced by endophytes isolated from Scots pine exerted beneficial effects by promoting plant growth. Other volatile compounds like acetoin and 2, 3-butanediol secreted by bacterial endophytes stimulated plant growth (Rath et al. 2018). The modulation effects of polyamines produced by mycorrhiza and Azospirillum brasilense on plant growth and development facilitate their cooperation with the host plant (Bashan and Levanony 1990; Kaushal and Wani 2016).
Plant growth regulators like auxins vitalized cell division, cell elongation, and cell differentiation. Endophytic bacteria and fungi potentially secrete physiologically active auxins that benefit plants. However, microorganisms colonizing various plant organs influenced the physiological functions and growth of plants by secreting growth parameters (Odelade and Babalola 2019). Additionally, PGPE exhibits a strong affinity in reducing metal toxicity under stress conditions, thus promoting plant growth (Zhang et al. 2020b). Highlights below are mechanisms employed by PGPE in enhancing agricultural crop productivity.
Nitrogen (N2) Fixation
Nitrogen accounts for 78% of atmospheric gases. It forms the main component of chemical fertilizers, and plant biomolecules for the synthesis of nucleic acids and proteins (Ferreira et al. 2016). When combined with other mineral elements such as oxygen or hydrogen, nitrogen produced complex nitrogenous compounds needed for soil amendment in ammonium (NH4+) and nitrate (NO3−) fertilizers. Nutrient-limiting soils affect the performance of plants in terms of flowering seed formation, growth, etc. PGPE in the genera Rhizobium, Pseudomonas, Sinorhizobium, Bradyrhizobium, Azorhizobium, Enterobacter, Klebsiella, Mesorhizobium, Allorhizobium, and Azospirillum symbiotically colonize the tissues of legumes and non-legume plants and fix atmospheric nitrogen in the form of ammonia (NH3) to the soil without interference to the soil nutrients supply to the host plant (Bashan et al. 2009; Nohwar et al. 2019). The isolation of bacterial endophytes Azospirillum from the roots of non-leguminous crops and their re-inoculation have contributed to plant growth (Döbereiner 1992; Mhatre et al. 2019).
Availability of nutrients needed for plant survival can enhance mutual patterns of microbes colonizing the root-endosphere in promoting plant growth and fixing nitrogen needed for plant functions. The mechanisms underlying the nitrogen fixation in non-leguminous plants by the potential non-rhizobia fixing bacteria are known, especially in sugarcane upon co-inoculation with endophytic bacteria such as Azospirillum amazonense, G. diazotrophicus, Herbaspirillum seropedicae, Herbaspirillum rubrisubalbicans, and Burkholderia tropica (Merasenla et al. 2016; Pedraza et al. 2010).
The ability of endophytes to produce genes coding nitrogenase may underline their nitrogen-fixing potential for improved plant performance in terms of height, root/shoot ratio, and height (Knoth et al. 2014). Some groups of root endophytes previously identified as nitrogen fixers include Rhodotorula graminis, Acetobacter diazotrophicus, Rhizobium tropici bv populus, Pseudomonas putida, Herbaspirillum spp, Burkholderia vietnamiensis, Sphingomonas yanoikuyae, and Azoarcus spp (Glick et al. 2001; Knoth et al. 2014). The nitrogen-fixing capability of these bacterial endophytes might be compared to the Rhizobium inhabiting the root nodule of leguminous plants. High efficiency of nitrogen fixating capability of bacterial endophytes G. diazotrophicus on Pinus flexilis (Limber pine) and Picea engelmannii (Engelmann spruce) growing in a nutrient-limiting environment have demonstrated the roles of G. diazotrophicus strains in nitrogen fixation and other physiological functions in those plants (Carrell and Frank 2014). An improvement in the seedling growth of canola (Brassica napus L.) inoculated with nitrogen-fixing bacterial endophytes Paenibacillus polymyxa P2b-2R has been tested with success (Puri et al. 2016). Also, the nitrogen-fixing potential of Pseudomonas stutzeri A15 and their growth-promoting effects on inoculated rice seedlings compared to uninoculated rice seedlings improved the plant yield (Pham et al. 2017). Nitrogen fixation by microorganisms can cause metabolic changes in plant growth and fitness, particularly in nitrogen-deficient soil environments. However, the amount of nitrogen fixation to the soil for plant use might vary depending on the number of microorganisms involved and their ability to produce nitrogen fixation genes (nifH) (Smercina et al. 2019).
Endophytic microbe interactions with the host plant and their nitrogenase production have efficiently made atmospheric nitrogen available to many plants. The nitrogen-fixing ability of endophytic microbes can ensure plant survival in a limiting nitrogen soil environment, thus promoting plant growth and immunity (Ke et al. 2019). The nitrogen-fixing ability of some bacterial endophytes such as Rahnella, Burkholderia, Acinetobacter, and Sphingomonas isolated from the stem part of Populus trichocarpa and Salix sitchensis have made nitrogen accessible for plants by stimulating their growth in nitrogen-deficient soils (Ma et al. 2016a). The persistence of nitrogen-fixing bacterial endophytes in a nitrogen-limiting soil environment might enhance nitrogen fixation rate and nitrogen accumulation in the host plants (Arora et al. 2019).
Phosphate (P) Solubilization
Phosphorus is one of the essential micronutrients required for the proper functioning in living organisms, viz; energy transport, root development, plant growth promotion as well as other physiological processes (Lobo et al. 2019). The application of phosphorus to alter soil had resulted in a phosphorus complex formation thereby making it inaccessible for plant absorption. In most agricultural soil, phosphate level ranges between 400 mg/kg and 1200 mg/kg, though, only 1 mg/kg of the soluble form of phosphate (HPO4−1 and H2PO4−2) is made available to plants (Rasul et al. 2019). Some endophytic microbes undergo several processes to make phosphorus available to plants via solubilization, acidification, chelation, ion exchange, organic acid production, and secretion of extracellular acid phosphatase (Santoyo et al. 2016). Endophytes assimilate soluble phosphate by preventing adsorption and this makes endophytes a reservoir of phosphorus, which enhances their phosphate solubilization potential in a phosphate limiting environment (Vidya et al. 2016).
Phosphate-solubilizing microbes play a major role in unlocking phosphate compounds in the soil, thus making them readily available for plant use (Rasul et al. 2019). The phosphate-solubilizing capability of endophytes enhances microbial survival and promote plant growth by synthesizing organic acids such as citric and gluconic acids that chelate the insoluble phosphate compounds at low pH to release protons. Different phosphate-solubilizing bacteria bacterial genera: Flavobacterium, Achromobacter, Pseudomonas, Streptomyces, Bacillus, Rhizobium, Burkholderia, Erwinia, Aereobacter, Enterobacter, Agrobacterium, and Micrococcus have been isolated from the endosphere and phyllosphere regions of plants (Pandey et al. 2016).
The media composition, types of substrates and growing conditions can contribute to the rate of phosphate production by microbial endophytes. However, not all bacterial isolates from plant rhizoplane, rhizosphere, or endosphere can stimulate plant growth or solubilize phosphate in the soil for plant use. For instance, phytase potential synthesis of endophyte bacteria isolated from Z. mays L. contributes to plant functions in acquiring soil nutrients (Hafsan et al. 2018). An increase in maize seedlings inoculated with Bacillus amyloliquefaciens FZB45 producing phytase under phosphorus limiting soil has been observed via phosphorus accessibility and plant uptake (Ramírez and Kloepper 2010).
Iron (Fe) Solubilization
Iron is one of the essential elements that forms life and exists in the soil in insoluble ferric form. Like phosphorus, iron exists in various forms such as iron (iii) oxide (ferrihydrite, goethite, and hematite) in the soil, and is not readily available for direct plant absorption. Iron availability in the soil might be due to chelating agents produced by microbial endophytes, thus making iron abundant in iron-deficient soils (Salam and Varma 2019). The low molecular weight siderophores developed a strong affinity for iron (iii) ions, as well as bind to other iron (ii) ions bivalent metal for easy assimilation by plants. The calcareous and alkaline nature of some agricultural soils limits plant access to elemental iron from the soil. Soil-root endophytes producing siderophores chelate iron by transforming them into a soluble form for plant use. Plants acquire iron from siderophores producers via direct mechanisms as determined by the ability of microorganisms to produce siderophores. Uptake of iron (Fe) from the iron-siderophore network across plant root is necessitated by chelate breakdown and solubilization of unavailable forms of iron by phytosiderophore synthesis (Sah et al. 2017).
The siderophore produced by microorganisms (microbial siderophores) generally displays more affinity than plant synthesized siderophores (phytosiderophores). Substructure plant growth in metal-contaminated soil amasses high iron that will support siderophore producing microbes (Ferreira et al. 2019). The microbial siderophores with high iron affinity are regarded as a significant means of making iron available to plants under metal-stressed conditions. Metal accumulation in the soil is made accessible for plant absorption when siderophore microbes are involved in the iron solubilization process. The actual role of siderophores produced by endophytes is unknown except by the induction of systemic resistance (Hardoim et al. 2015).
Some endophytes have elicited siderophore genes that modulate iron metabolism and transport in most plants. The siderophore gene potential of Epichloë festucae isolated from rhygrass has modulated the effects of siderophores on E. festucae infected plants. The induction of resistance due to siderophore synthesis by endophytic Methylobacterium strains inhibit pathogenic bacteria X. fastidiosa causing chlorosis in Citrus trees (Bucci 2018). A high siderophore production rate of 47% and 25% of total endophytic bacteria colonizing the endosphere than rhizosphere of silver birch (Betula pendula L.) and black alder (Alnus glutinosa L.) has dictated the level of iron in the soil (Złoch et al. 2016). Reduction in Cd2+ toxicity in the host plants has also been linked to an increase in iron acquisition. Iron uptake in plants is accelerated by ligand exchange, breaking down organic chelate, and direct formation of Fe (iii) siderophore complexes (Ma et al. 2016b). Bioaccumulation of heavy metals in Spartina maritima improves plant growth. The inability of endophytic bacteria in the uptake of Cd concentrates from polluted marshes soils has reduced their resistance ability (Mesa et al. 2015).
The phytosiderophores and siderophores produced by microorganisms can enhance iron-chelating capability in many plants. The activities of root endophytes in response to higher iron uptake in non-sterile calcareous soils than sterile soils due to soil variables enable them to form plant-microbe interactions (Otieno et al. 2015). The iron-chelating ability of Pseudomonas spp isolated from growing mung beans (Vigna radiata) and their growth effects have yielded positive responses regarding plant performance (Otieno et al. 2015). Besides, iron uptake by plants has contributed to leaf color pigmentation thereby preventing chlorosis. The potential of bacterial endophytes producing siderophores by inoculation with the indigenous soil microbes to form bacteria-siderophore complexes make iron resources available for the plant. Hence, the siderophore producing PGPE—P. putida can enhance plant iron nutrition (Sah et al. 2017).
Phytoremediation/Bioremediation
Various waste generated from different sources can serve as excellent nutrient sources for microbial growth. The over-accumulation of recalcitrant xenobiotic compounds has made them resistant to microbial degradation and thus negatively influences plant growth, crop yields, soil nutrients, diversity, and distribution of beneficial microorganisms in response to phytoremediation activities (Uzoh and Babalola 2018). Heavy metals that contaminate in a soil environment include selenium, copper, lead, zinc, cadmium, cobalt, chromium, and mercury. Other contaminants include; (i) inorganic compounds—phosphate, ammonia, nitrate, sodium, arsenic), radioactive compounds (strontium, uranium or cesium); (ii) organic compounds—chlorinated solvents like trichloroethylene; explosives such as petroleum hydrocarbons including toluene, xylene, and benzene, polycyclic aromatic hydrocarbons; trinitrotoluene and 1,3,5-trinitro-1,3,5-hexahydrotriazine; and (iii) pesticides—atrazine and bentazon (Salam and Varma 2019).
Screening of essential microbial traits has proven efficient in the degradation/removal of environmental pollutants or heavy metal from the contaminated soil (Rostami and Azhdarpoor 2019). The removal of pollutants from the soil environment can be accomplished by the activities of endophytic microbes involving rhizodegradation, transformation, and stabilization using lytic enzymes in breaking down complex organic molecules. The inability of plant microbes to degrade environmental pollutants due to their non-remediation properties can result in less biomass production (Enebe and Babalola 2018).
Some endophytic bacteria grow well under stress conditions and remediate environmental pollutants. The presence of recalcitrant and xenobiotic substances in the soil affects the microbial population and their metabolic activities. Inoculation of heavy metal-polluted soils with PGPE can facilitate the bioavailability of heavy metals, thus making them available for plant growth (Wang et al. 2019). Accumulation of various toxic chemicals in the soil can transiently migrate through the plants into the food chain system, causing human health issues and other related disease conditions. Waste removal from agricultural soil using endophytic microorganisms has prompted developing an eco-friendly biological approach in environmental pollution control (Gupta et al. 2020a).
Considerable numbers of bacterial species associated with endosphere degrading environmental pollutants have been isolated and identified (Rani et al. 2019). The use of PGPE in reducing heavy metal induction phytotoxicity enhances plant biomass production in metal-polluted soils (Chen et al. 2010). The role of PGPE on plant improvement and crop yields; and their potential in heavy metals bioremediation on agricultural-polluted soil can help in bioremediation processes (Ma et al. 2016b). Some examples of endophytes genera involved in bioremediation processes include Pseudomonas, Bacillus, Burkholderia, Stenotrophomonas, Pusillimonas, Terribacillus, Acinetobacter, Achromobacter, Staphylococcus, Curtobacteriu, Microbacterium, Arthrobater, Leifsonia, Paenibacillus, Enterobacter, Acinetobacter, Rahnella, Herbaspirillum, Serratia, Flavobacterium, Chryseobacterium, Sphingomonas, Methylobacterium and Variovorax (Ma et al. 2016b).
Siderophores Production
Siderophores are biological molecules produced by microorganisms, with a low molecular weight of 200 Da–2 KDa. They are primarily involved in iron-chelation in nutrient-limiting environments (Odelade and Babalola 2019). The activities of PGPE in the synthesis of siderophores are expressed by indirect mechanisms on plants (Omomowo and Babalola 2019). Many PGPEs reduce plant stress either by synthesizing biological control agents or by systemic induction of plant response against pathogens (Ferreira et al. 2019). Endophytes producing siderophores help control plant pathogens that affect crop yields and act as key determinants for plant-induced systemic resistance. Some endophytic microbes can synthesize substances that exert lethal effects on various pathogens. The substances include metabolites (volatile organic compounds), siderophores antibiotics, and enzymes. The siderophores produced by endophytic microbes are composed of chelating compounds, binding to iron, and improve plants' resistance to environmental stresses (Dimkpa et al. 2009).
Siderophores producing capability of the endophytic bacteria domain in the control of plant pathogens have contributed to plant health and immunity (Omomowo and Babalola 2019). For example, siderophores production from Bacillus antiquum in the control of Rhizoctonia solani that cause black scurf and stem canker and charcoal rot disease caused by Macrophomina phaseolina in sorghum have suggested their use as a biocontrol agent (Gopalakrishnan et al. 2011; Aloo et al. 2019). The singularly or combined use of antibiotics in biocontrol systems has been employed in disease control and suppression. Endophytic microbes can potentially outcompete pathogens colonizing the same ecological niche, thus reducing their detrimental effects on crop yield (Card et al. 2015).
Enzymes Production
Endophytes produce enzymes primarily to facilitate tissue colonization by utilizing polysaccharide compounds containing xylan and pectin present in the host plants (Fouda et al. 2021). Examples of enzymes produced by endophytes that facilitate tissue colonization include lipase, amylase, tannase, cellulase, glucanase, protease, laccase, xylanase, and chitinase (Sharma 2019). Like other metabolites, enzymes produced by endophytic microbes perform specific functions against invading plant pathogens and degradation of organic materials such as lignin, complex polysaccharides, cellulose, and hemicellulose (Chu et al. 2021). Despite the actions of lytic enzymes like lipase or cellulase on organic compounds, the host plants permit endophyte association within the tissue but recognize them as an obstacle for tissue colonization (Chen et al. 2016). Other metabolites such as essential oils and alkaloids secreted from plants can facilitate host colonization patterns (Korenblum and Aharoni 2019). Under favorable conditions, fungal endophytes can complete their life cycle within the host tissues by synthesizing enzymes that can cause alteration in the host plant defense mechanisms and decomposition of organic materials (Tiwari and Rana 2015; Adeleke and Babalola 2021).
Biocontrol Activities of Plant Endophytes
The PGPE colonizing plant endosphere capable of synthesizing biomolecules can enhance plant performance in stimulating plant resistance against pathogens (Adeleke and Babalola 2020). Plant protection against pathogens may be achieved directly through antagonistic interactions and induction of host resistance, or indirectly by suppressing plant pathogens and enhancing plant growth through various mechanisms (Orozco-Mosqueda et al. 2018). Such mechanisms include the ability of endophytes to secrete antimicrobial metabolites, enzymes degrading microbial cell walls, and hydrogen cyanide that represses the growth of microbial pathogens (Adeleke and Babalola 2020).
Endophytic microbes associating with plants live and grow inside the cell of the host plant without any disease symptoms. These microorganisms that colonize plant tissues may have evolved from plants and become non-pathogenic (Xu et al. 2019). During long-term co-evolution of endophytes and plants, the establishment of equilibrium can make beneficial endophytes turn pathogenic in host plants under stress conditions. Over time, plant-microbe forms ecological balancing when microbial activity and plant interactions are achieved (Nefzi et al. 2019). The mechanisms exhibited by endophytes as biocontrol agents against plant pathogens can directly or indirectly be achieved with significant ecological effects. The use of microbial endophytes in plant protection against pathogens may be due to their ability to produce phytoalexins (Kollakkodan et al. 2020).
PGPE plays a major role in the management of crops against microbial pathogens. The ability of endophytes to elicit biocontrol traits, antibiotic production, iron chelator siderophores, extracellular enzymes determine their biocontrol activities (Ye et al. 2019). The biocontrol activities of Aspergillus terreus and Penicillium citrinum isolated from sunflower plants against Sclerotium rolfsii with overall performance and improvement in sunflower yield have positively influenced sunflower growth (Potshangbam et al. 2017). The use of PGPE as biocontrol agents against plant pathogens is listed in Table 3.
A major challenge in the use of microbes as biocontrol agents can be based on its application. The exploitation of derivative fungal or bacterial resources as biological agents has been employed in the control of plant pathogens (Pan et al. 2015). The recruitment of endophytes in modern agricultural practices has made them a potential candidate in the formulation of biopesticides on application to suppress activities of phytopathogens (Sindhu et al. 2016). Reports on the fast and intense defense mechanisms of bacterial endophytes in the host plant, suitably in the control of entomopathogenic fungi that cause collar rot in plants have revealed their importance in growing pathogen-free plants (Jaber and Ownley 2018).
Metabolites secreted by Fusarium spp E4 and E5 in the control of plant diseases and for promoting the growth of Euphorbia pekinensis depend on increased terpenoid content (Nefzi et al. 2019). Muscodor albus isolated from Cinnamomum zeylanicum producing 28 volatile compounds have elicited their bactericidal and fungicidal activities on microbial pathogens (Praptiwi et al. 2015). M. crispansis, a fungus of Dwarf pineapple (Ananas ananassoides) from Bolivian Amazon Basin has also displayed antimicrobial efficacy due to volatile organic compounds secretions against fungal pathogens: Botrytis cinerea, Fusarium culmorum, Pythium ultimum, Alternaria helianthi, Verticillium dahlia, Phytophthora cinnamomi, Sclerotinia sclerotiorum, and R. solani; and bacteria Xanthomonas axonopodis (Hashem et al. 2019; Yuan et al. 2017). The use of bacterial endophytes in the control of Verticillium wilt and Fusarium wilts (wilts that cause vascular coloration, chlorosis and necrosis in the leaves of herbaceous plants such as vegetables, maize, sunflower, soybean, and cowpea) have contributed to plant health. Studies have shown the application of Pseudomonas, Clavibacter, Erwinia, and Ralstonia in the control of vascular wilt diseases in infected plants (Eljounaidi et al. 2016).
Several challenges surround the control of pathogens associated with most plants since there are no clear-cut measures in proffering curable treatments to the infected plants except by removing the infected parts from the plant body. The persistence of vascular wilt soil pathogens depends on their resistive structures that ensure their viable living for a long time, invariably making its control difficult (Yadeta and Thomma 2013). The use of chemical pesticides to control vascular wilt diseases may be cost-effective but inefficient as this negatively poses environmental and health challenges.
Consequently, it is imperative to find an alternative ecological friendly approach to the control of plant diseases. One of such promising approaches is the exploration of biocontrol agents from bacterial or fungal endophytes. For effective use of endophytes as biocontrol agents, it must be; (i) a natural means of exploration to ameliorate over-dependence on the use of fertilizers and pesticides, (ii) self-sustaining with the ability of proliferation and forms cellular biofilm after the establishment in a niche, and (iii) reduce the use of pesticides that permit prevention of long-term diseases in an eco-friendly manner (Eljounaidi et al. 2016).
Phytohormones Production
The phytohormones suppress metal stress in plants, thus suggesting that metal stress reduction by endophytic microbes can cumulatively exert combining nutritional and physiological effects on the plants (Afzal et al. 2019). The phytohormones either inhibit or promote plant growth depending on the concentration and increase nutrient uptake and improve crop productivity (Spaepen et al. 2009). The ability to produce certain plant growth hormones, which includes auxin, indole-3-acetic acid, cytokinin, gibberellin and abscisic acid have been attributed to two predominant endophytic bacteria genera, namely Bacillus and Pseudomonas (Hashem et al. 2019).
Indole-3-acetic acid (IAA), a major auxin synthesized by microorganisms partakes in different physiological processes in the plant such as regulation of plant growth and it functions as cell–cell signaling molecules (Crozier et al. 1988). The IAA concentration differs in various parts of the plants (Ismaila et al. 2018). This auxin produced by endophytes modulates plant-endophyte interactions and plant survival in metal-polluted soil. PGPE such as Enterobacter, Staphylococcus, Pseudomonas, Azospirillum, and Azotobacter from rhizosphere soil of faba bean (Vicia faba) and root of sugarcane (Saccharum officinarum) have been identified as phytohormone producers (Alemu 2016; Kruasuwan and Thamchaipenet 2016; Okon and Kapulnik 1986).
Phytohormone synthesis enhances root development in many plants (Santoyo et al. 2016). The low synthesis of IAA can stimulate primary root development and elongation, while high IAA synthesis by endophytes can stimulate adventitious root growth with impairment to the primary root growth. However, modification of phytohormones equilibrium through ACC synthesis can accelerate plant growth (Dobbelaere et al. 2003).
Ethylene is another phytohormones that contributes significantly to the growth and survival of plants under stress conditions. Its functions include cell elongation, leaf development (senescence), root initiation, root nodulation, abscission, and fruit ripening as well as auxin transport (Ji et al. 2020). Ethylene synthesis in plants is achieved through the production of enzyme S-adenosyl-L-methionine (SAM) synthetase, which catalyzes the conversion of methionine and adenosine triphosphate (ATP) to SAM; then ACC synthase mediating the hydrolysis of SAM to ACC and methylthioadenosine (MTA), and finally, oxidization of ACC by enzyme ACC oxidase to its products form, ethylene, carbon dioxide, and hydrogen cyanide (He et al. 2019). Growing plants in a metal stress environment stimulate ethylene production, thus inhibiting root elongation, root hair formation, and lateral root growth (Glick et al. 1998; Wu et al. 2020). Similarly, some endophytic microbes reduce the stress-regulated influence in plants by enzyme reaction in the hydrolysis of ACC and subsequently reduce ethylene production stress by plants (Santoyo et al. 2016).
Induced Systemic Resistance and Metabolites Secretion
Induce systemic resistance (ISR) is a systemic induction process responsible for plant stimuli in developing resistance against plant pathogens. The modern application of chemical and PGPBE metabolites as biocontrol measures is promising in crop pathogen control and agricultural sustainability (Alvin et al. 2016). The plant-associated endophytes can develop multiple mechanisms to stimulate plant defense systems through the expression of oxidative and nitrosative genes that are required for the activation of mechanisms such as superoxide dismutase, peroxidases, guaiacol, catalase, b-1,3-glucanases, and chitinase activity (Okon and Labandera-Gonzalez 1994). Consequently, it can instigate reactive oxygen species production in protecting cell organelles con cell oxidative shock and physiological stress that may have preset these actions due to endophytic microbe activity (Asghari et al. 2020).
The life cycle of endophytes in host plants is said to be shorter than the host plant itself; hence, endophytes proliferate rapidly and form microbial biomass within the host plants for selective antagonism in contributing to the resistance against other endophyte-like pathogens (Sahu et al. 2019). The induction of systemic resistance in endophytes enhanced their tolerance to other camouflaged endophyte pathogens. The mutualistic association between endophytes and host plants prompted the systemic responses in plants. The ability of endophytes to bypass host responses makes them successfully colonize the plants without suppressing the activity of the host plants (Xu et al. 2019).
Endophytic bacteria genera Pseudomonas and Bacillus are regarded to have caused systemic induction resistance in plants based on key attributes and their ability to possess attachment cell organelles, organic acids (salicylic acid, jasmonic acid) production, synthesis of volatile compounds and other biomolecules siderophores, lipopolysaccharides and N-acyl-homoserine lactones (Hardoim et al. 2015). The induction resistance of Methylobacterium spp strain IMBG290 isolated from potato shoot against potato pathogen Pectobacterium atrosepticum, owing to the vicissitude in the microbial community structure and diversity, boosts plant immunity against phytopathogens (Ardanov et al. 2016). The corresponding changes in the endophytes community to pathogen resistance significantly repress disease activity.
Less information is available on fungal endophytes plant protection through ISR. Like bacterial endophytes, fungal endophytes also produce certain metabolites compounds that inhibit plant pathogens. Some compounds include phenols, polyketones, peptides, steroids, flavonoids, quinols, alkaloids, terpenoids, and chlorinated compounds (Hardoim et al. 2015). The antibiotic substances 2-phenylethanol and 4-hydroxybenzoate produced by Enterobacter spp strain 638, and munumbicins, kakadumycins, and coronamycin produced by actinomycetes stimulate resistance against pathogens (Braga and Faria 2020). The antibacterial activity of chemical compounds multicyclic indolosesquiterpenes produced by Streptomyces spp HKI0595 isolated from mangrove tree (Kandelia candel) and spoxazomicins A to C produced from Streptosporangium oxazolinicum strain K07-0450 T isolated from orchid plants, importantly stimulate plant defense against pathogens (Chakravorty et al. 2020). The array of chemical compounds produced by bacterial and fungal endophytes are found applicable in agriculture; nevertheless, elucidation on the specific functions in plant–microbe interactions is required.
Future Prospects/Conclusions
The diverse endophytic microorganisms occupying the external and intracellular compartments of plants play an important role in plant ecology and physiology. These endophytic microbial diversities inhabiting different plant organs might exhibit genetic relatedness. Their exploration and applications in modern agriculture sustainability can guarantee maximum crop production and food safety. Though crop production is mostly affected by rainfall, drought, salinity, temperature as well as pathogens, the conventional application of chemical fertilizers and pesticides to improve agricultural productivity has been associated with various environmental or ecological effects.
The complex assemblage of microbial communities inhabiting the endosphere was reviewed in the present study. The survival and diversity of plant endophytes are influenced by environmental conditions and plant phenotype or genotype. Sometimes, the impact of endophytes on plant growth and crop productivity is monitored through laboratory routines involving isolation, identification, and inoculation of desirable microorganisms and monitoring under controlled greenhouse pots experiments.
In an ecosystem, the understanding of the genetic composition of plant endophytes is vital in developing environmentally friendly agriculture by harnessing microbial resources in the formulation of bioformulants or bioinoculants such as biofertilizers. The roles of endophytes in agriculture are numerous and include plant growth promotion, improving plant health and immunity, enhancing crop and agricultural productivity, plant protection against pathogens, plant tolerance to environmental stresses, bioremediation of organic pollutants, etc. Microbial endophytes have been used in the formulation of biopesticides toxic to plant pathogens as well as secretion of metabolite compounds such as antibiotics and hydrogen cyanide.
In conclusion, agricultural productivity can be enhanced using modern agricultural techniques that involve the synthesis of organic or microbial-based products in promoting plant growth and control of pathogens as the best alternative to chemical fertilizers and pesticides in developing environmentally friendly agriculture, using microbial resources for improved agriculture in a sustainable manner.
References
Abonyi DO, Eze PM, Abba CC, Ujam NT, Proksch P, Okoye FB, Esimone CO (2018) Biologically active phenolic acids produced by Aspergillus sp., an endophyte of Moringa oleifera. Eur J Biol Res 8:157–167
Adegbeye MJ, Ravi Kanth Reddy P, Obaisi AI, Elghandour MMMY, Oyebamiji KJ, Salem AZM, Morakinyo-Fasipe OT, Cipriano-Salazar M, Camacho-Díaz LM (2020) Sustainable agriculture options for production, greenhouse gasses and pollution alleviation, and nutrient recycling in emerging and transitional nations—an overview. J Clean Prod 242:118319. https://doi.org/10.1016/j.jclepro.2019.118319
Adeleke BS, Babalola OO (2020) The endosphere microbial communities, a great promise in agriculture. Int Microbiol 24:1–17. https://doi.org/10.1007/s10123-020-00140-2
Adeleke BS, Babalola OO (2021) Biotechnological overview of agriculturally important endophytic fungi. Hortic Environ Biotechnol 63:1–14
Afzal I, Shinwari ZK, Sikandar S, Shahzad S (2019) Plant beneficial endophytic bacteria: mechanisms, diversity, host range and genetic determinants. Microbiol Res 221:36–49. https://doi.org/10.1016/j.micres.2019.02.001
Alawiye TT, Babalola OO (2019) Bacterial diversity and community structure in typical plant rhizosphere. Diversity 11:179
Alemu F (2016) Isolation of Pseudomonas fluorescens species from faba Bean rhizospheric soil and assessment of indole acetic acid production: In vitro study, Ethiopia. Amer J Biosci 4:9–15
Aloo BN, Makumba BA, Mbega ER (2019) The potential of Bacilli rhizobacteria for sustainable crop production and environmental sustainability. Microbiol Res 219:26–39. https://doi.org/10.1016/j.micres.2018.10.011
Alster CJ, von Fischer JC, Allison SD, Treseder KK (2020) Embracing a new paradigm for temperature sensitivity of soil microbes. Glob Change Biol 26:3221–3229
Alvin A, Kalaitzis J, Sasia B, Neilan B (2016) Combined genetic and bioactivity-based prioritization leads to the isolation of an endophyte-derived antimycobacterial compound. J Appl Microbiol 120:1229–1239
Anisha C, Jishma P, Bilzamol VS, Radhakrishnan E (2018) Effect of ginger endophyte Rhizopycnis vagum on rhizome bud formation and protection from phytopathogens. Biocatal Agric Biotechnol 14:116–119
Arafa M, El-Batanony NH (2018) Growth, yield and chemical composition response of some legume crops to inoculation with non-rhizobial endophytic bacteria from Melilotus indicus (L.) all. Nodules. J Plant Prod 9:353–358
Ardanov P, Lyastchenko S, Karppinen K, Häggman H, Kozyrovska N, Pirttilä AM (2016) Effects of Methylobacterium sp. on emergence, yield, and disease prevalence in three cultivars of potato (Solanum tuberosum L.) were associated with the shift in endophytic microbial community. Plant Soil 405:299–310
Arora P, Wani ZA, Ahmad T, Sultan P, Gupta S, Riyaz-Ul-Hassan S (2019) Community structure, spatial distribution, diversity and functional characterization of culturable endophytic fungi associated with Glycyrrhiza glabra L. Fungal Biol 123:373–383. https://doi.org/10.1016/j.funbio.2019.02.003
Asghari S, Harighi B, Ashengroph M, Clement C, Aziz A, Esmaeel Q, Ait Barka E (2020) Induction of systemic resistance to Agrobacterium tumefaciens by endophytic bacteria in grapevine. Plant Pathol 69:827–837
Baiyee B, Ito S-i, Sunpapao A (2019) Trichoderma asperellum T1 mediated antifungal activity and induced defense response against leaf spot fungi in lettuce (Lactuca sativa L.). Physiol Mol Plant Pathol 106:96–101
Banik A, Mukhopadhaya SK, Dangar TK (2016) Characterization of N2-fixing plant growth promoting endophytic and epiphytic bacterial community of Indian cultivated and wild rice (Oryza spp.) genotypes. Planta 243:799–812
Bashan Y (1998) Inoculants of plant growth-promoting bacteria for use in agriculture. Biotechnol Adv 16:729–770
Bashan Y (1999) Interactions of Azospirillum spp. in soils: a review. Biol Fertil Soils 29:246–256
Bashan Y, De-Bashan LE (2010) How the plant growth-promoting bacterium Azospirillum promotes plant growth—a critical assessment. Adv Agron 108:77–136
Bashan Y, Levanony H (1990) Current status of Azospirillum inoculation technology: Azospirillum as a challenge for agriculture. Can J Microbiol 36:591–608
Bashan Y, Puente ME, Rodriguez-Mendoza MN, Toledo G, Holguin G, Ferrera-Cerrato R, Pedrin S (1995) Survival of Azospirillum brasilense in the bulk soil and rhizosphere of 23 soil types. Appl Environ Microbiol 61:1938–1945
Bashan Y, Salazar B, Puente ME, Bacilio M, Linderman R (2009) Enhanced establishment and growth of giant cardon cactus in an eroded field in the Sonoran Desert using native legume trees as nurse plants aided by plant growth-promoting microorganisms and compost. Biol Fertil Soils 45:585–594
Bergna A, Cernava T, Rändler M, Grosch R, Zachow C, Berg G (2018) Tomato seeds preferably transmit plant beneficial endophytes. Phytobiomes J 2:183–193
Braga A, Faria N (2020) Bioprocess optimization for the production of aromatic compounds with metabolically engineered hosts: recent developments and future challenges. Front Bioeng Biotechnol 8:96
Bucci EM (2018) Xylella fastidiosa, a new plant pathogen that threatens global farming: ecology, molecular biology, search for remedies. Biochem Biophy Res Comm 502:173–182
Card SD, Hume DE, Roodi D, McGill CR, Millner JP, Johnson RD (2015) Beneficial endophytic microorganisms of Brassica—a review. Biol Control 90:102–112. https://doi.org/10.1016/j.biocontrol.2015.06.001
Carrell AA, Frank AC (2014) Pinus flexilis and Picea engelmannii share a simple and consistent needle endophyte microbiota with a potential role in nitrogen fixation. Front Microbiol 5:333
Chakravorty P, Srivastava N, Ibeyaima A, Sarethy IP (2020) Antimicrobial and antioxidant compounds in endophyte isolate L-003 obtained from the aquatic plant Nelumbo nucifera. The Nat Prod J 10:139–144
Chen L, Luo S, Xiao X, Guo H, Chen J, Wan Y, Li B, Xu T, Xi Q, Rao C (2010) Application of plant growth-promoting endophytes (PGPE) isolated from Solanum nigrum L. for phytoextraction of Cd-polluted soils. Appl Soil Ecol 46:383–389
Chen L, Brookes PC, Xu J, Zhang J, Zhang C, Zhou X, Luo Y (2016) Structural and functional differentiation of the root-associated bacterial microbiomes of perennial ryegrass. Soil Biol Biochem 98:1–10. https://doi.org/10.1016/j.soilbio.2016.04.004
Chenniappan C, Narayanasamy M, Daniel G, Ramaraj G, Ponnusamy P, Sekar J, Ramalingam PV (2019) Biocontrol efficiency of native plant growth promoting rhizobacteria against rhizome rot disease of turmeric. Biol Control 129:55–64
Chu X, Awasthi MK, Liu Y, Cheng Q, Qu J, Sun Y (2021) Studies on the degradation of corn straw by combined bacterial cultures. Biores Technol 320:124174
Cole MB, Augustin MA, Robertson MJ, Manners JM (2018) The science of food security. npj Sci Food 2:1–8
Compant S, Clément C, Sessitsch A (2010) Plant growth-promoting bacteria in the rhizo-and endosphere of plants: their role, colonization, mechanisms involved and prospects for utilization. Soil Biol Biochem 42:669–678
Compant S, Kaplan H, Sessitch A, Nowak J, Ait Barka E, Clément C (2008) Endophytic colonization of Vitis vinifera L. by Burkholderia phytofirmans strain PsJN: from the rhizosphere to inflorescence tissues. FEMS Microbiol Ecol 63:84–93
Crozier A, Arruda P, Jasmim JM, Monteiro AM, Sandberg G (1988) Analysis of indole-3-acetic acid and related indoles in culture medium from Azospirillum lipoferum and Azospirillum brasilense. Appl Environ Microbiol 54:2833–2837
Dimkpa CO, Merten D, Svatoš A, Büchel G, Kothe E (2009) Metal-induced oxidative stress impacting plant growth in contaminated soil is alleviated by microbial siderophores. Soil Biol Biochem 41:154–162
Dobbelaere S, Vanderleyden J, Okon Y (2003) Plant growth-promoting effects of diazotrophs in the rhizosphere. Critl Rev Plant Sci 22:107–149
Dobereiner J (1992) History and new perspectives of diazotrophs in association with non-leguminous plants. Symbiosis 13:1–13
Döbereiner J (1992) Recent changes in concepts of plant bacteria interactions: endophytic N2 fixing bacteria. Ciência e Cultura 44:310–313
Dong L, Cheng R, Xiao L, Wei F, Wei G, Xu J, Wang Y, Guo X, Chen Z, Chen S (2018) Diversity and composition of bacterial endophytes among plant parts of Panax notoginseng. Chin Med 13:41
Doolotkeldieva T, Bobusheva S (2016) Fire blight disease caused by Erwinia amylovora on Rosaceae plants in Kyrgyzstan and biological agents to control this disease. Adv Microbiol 6:831
Dubey A, Saiyam D, Kumar A, Hashem A, Abd Allah EF, Khan ML (2021) Bacterial root endophytes: characterization of their competence and plant growth promotion in soybean (Glycine max (L.) Merr.) under drought stress. Int J Environ Res Public Health 18:931
Edwards J, Johnson C, Santos-Medellín C, Lurie E, Podishetty NK, Bhatnagar S, Eisen JA, Sundaresan V (2015) Structure, variation, and assembly of the root-associated microbiomes of rice. Proc Nat Acad Sci 112:911–920
Eljounaidi K, Lee SK, Bae H (2016) Bacterial endophytes as potential biocontrol agents of vascular wilt diseases–review and future prospects. Biol Control 103:62–68
Enebe MC, Babalola OO (2018) The influence of plant growth-promoting rhizobacteria in plant tolerance to abiotic stress: a survival strategy. Appl Microbiol Biotechnol 102:7821–7835
Feng N-X, Yu J, Zhao H-M, Cheng Y-T, Mo C-H, Cai Q-Y, Li Y-W, Li H, Wong M-H (2017) Efficient phytoremediation of organic contaminants in soils using plant–endophyte partnerships. Sci Total Environ 583:352–368. https://doi.org/10.1016/j.scitotenv.2017.01.075
Ferreira AS, Balbinot Junior AA, Werner F, Zucareli C, Franchini JC, Debiasi H (2016) Plant density and mineral nitrogen fertilization influencing yield, yield components and concentration of oil and protein in soybean grains. Bragantia 75:362–370
Ferreira MJ, Silva H, Cunha A (2019) Siderophore-producing rhizobacteria as a promising tool for empowering plants to cope with iron limitation in saline soils: a review. Pedosphere 29:409–420. https://doi.org/10.1016/S1002-0160(19)60810-6
Fouda A, Eid AM, Elsaied A, El-Belely EF, Barghoth MG, Azab E, Gobouri AA, Hassan SE-D (2021) Plant growth-promoting endophytic bacterial community inhabiting the leaves of Pulicaria incisa (Lam.) DC inherent to arid regions. Plants 10:76
Garcia MM, Pereira LC, Braccini AL, Angelotti P, Suzukawa AK, Marteli D, Felber PH, Bianchessi PA, Dametto IB (2017) Effects of Azospirillum brasilense on growth and yield compounds of maize grown at nitrogen limiting conditions. Rev Ciênc Agrár 40:353–362
Glick BR (2014) Bacteria with ACC deaminase can promote plant growth and help to feed the world. Microbiol Res 169:30–39
Glick BR, Bashan Y (1997) Genetic manipulation of plant growth-promoting bacteria to enhance biocontrol of phytopathogens. Biotechnol Adv 15:353–378
Glick BR, Penrose DM, Li J (1998) A model for the lowering of plant ethylene concentrations by plant growth-promoting bacteria. J Theor Biol 190:63–68
Glick BR, Penrose DM, Ma W (2001) Bacterial promotion of plant growth. Biotechnol Adv 19:135–138
Gopalakrishnan S, Humayun P, Kiran BK, Kannan IGK, Vidya MS, Deepthi K, Rupela O (2011) Evaluation of bacteria isolated from rice rhizosphere for biological control of charcoal rot of sorghum caused by Macrophomina phaseolina (Tassi) Goid. World J Microbiol Biotechnol 27:1313–1321
Gouda S, Kerry RG, Das G, Paramithiotis S, Shin H-S, Patra JK (2018) Revitalization of plant growth promoting rhizobacteria for sustainable development in agriculture. Microbiol Res 206:131–140
Gupta P, Kumar V, Usmani Z, Rani R, Chandra A, Gupta VK (2020) Implications of plant growth promoting Klebsiella sp. CPSB4 and Enterobacter sp. CPSB49 in luxuriant growth of tomato plants under chromium stress. Chemosphere 240:124944. https://doi.org/10.1016/j.chemosphere.2019.124944
Gupta S, Chaturvedi P, Kulkarni MG, Van Staden J (2020) A critical review on exploiting the pharmaceutical potential of plant endophytic fungi. Biotechnol Adv 39:107462
Hafsan H, Nurhikmah N, Harviyanti Y, Sukmawati E, Rasdianah I, Muthiadin C, Agustina L, Natsir A, Ahmad A (2018) The potential of endophyte bacteria isolated from Zea mays L as phytase producers. J Pure Appl Microbiol 12:1–4
Hamayun M, Hussain A, Khan SA, Kim H-Y, Khan AL, Waqas M, Irshad M, Iqbal A, Rehman G, Jan S (2017) Gibberellins producing endophytic fungus Porostereum spadiceum AGH786 rescues growth of salt affected soybean. Front Microbiol 8:686
Hardoim PR, Van Overbeek LS, Berg G, Pirttilä AM, Compant S, Campisano A, Döring M, Sessitsch A (2015) The hidden world within plants: ecological and evolutionary considerations for defining functioning of microbial endophytes. Microbiol Mol Biol Rev 79:293–320
Hashem A, Tabassum B, Fathi Abd Allah E (2019) Bacillus subtilis: a plant-growth promoting rhizobacterium that also impacts biotic stress. Saudi J Biol Sci 26:1291–1297. https://doi.org/10.1016/j.sjbs.2019.05.004
He W-S, Cui D, Li L, Tong L-T, Rui J, Li H, Zhang H, Liu X (2019) Cholesterol-reducing effect of ergosterol is modulated via inhibition of cholesterol absorption and promotion of cholesterol excretion. J Func Foods 57:488–496
Hiruma K, Kobae Y, Toju H (2018) Beneficial associations between Brassicaceae plants and fungal endophytes under nutrient-limiting conditions: evolutionary origins and host–symbiont molecular mechanisms. Curr Opin Plant Biol 44:145–154. https://doi.org/10.1016/j.pbi.2018.04.009
Hong CE, Kim JU, Lee JW, Lee SW, Jo I-H (2018) Diversity of bacterial endophytes in Panax ginseng and their protective effects against pathogens. Biotechnol 8:397
Huang Y, Kuang Z, Wang W, Cao L (2016) Exploring potential bacterial and fungal biocontrol agents transmitted from seeds to sprouts of wheat. Biol Control 98:27–33
Ismaila AH, Qadira M, Husnaa MI, Ahmadb A, Hamayuna M (2018) Endophytic fungi isolated from Citrullus colocynthesl leaves and their potential for secretion of indole acetic acid and gibberellin. J Appl Environ Biol Sci 8:80–84
Jaber LR, Ownley BH (2018) Can we use entomopathogenic fungi as endophytes for dual biological control of insect pests and plant pathogens? Biol Control 116:36–45. https://doi.org/10.1016/j.biocontrol.2017.01.018
Jambon I, Thijs S, Weyens N, Vangronsveld J (2018) Harnessing plant-bacteria-fungi interactions to improve plant growth and degradation of organic pollutants. J Plant Interact 13:119–130
James EK, Olivares FL, de Oliveira AL, dos Reis Jr FB, da Silva LG, Reis VM (2001) Further observations on the interaction between sugar cane and Gluconacetobacter diazotrophicus under laboratory and greenhouse conditions. J Exp Bot 52:747–760
Jha Y, Subramanian R (2018) Effect of root-associated bacteria on soluble sugar metabolism in plant under environmental stress. Plant metabolites and regulation under Environ mental stress. Elsevier, Amsterdam, pp 231–240
Ji J, Yuan D, Jin C, Wang G, Li X, Guan C (2020) Enhancement of growth and salt tolerance of rice seedlings (Oryza sativa L.) by regulating ethylene production with a novel halotolerant PGPR strain Glutamicibacter sp. YD01 containing ACC deaminase activity. Acta Physiol Plantarum 42:1–17
Jiao R, Munir S, He P, Yang H, Wu Y, Wang J, He P, Cai Y, Wang G, He Y (2020) Biocontrol potential of the endophytic Bacillus amyloliquefaciens YN201732 against tobacco powdery mildew and its growth promotion. Biol Control 143:104160
Kandel S, Joubert P, Doty S (2017) Bacterial endophyte colonization and distribution within plants. Microorganisms 5:77
Kaur H, Kaur J, Gera R (2016) Plant growth promoting rhizobacteria: a boon to agriculture. Int J Cell Sci Biotechnol 5:17–22
Kaushal M, Wani SP (2016) Plant-growth-promoting rhizobacteria: drought stress alleviators to ameliorate crop production in drylands. Ann Microbiol 66:35–42
Ke X, Feng S, Wang J, Lu W, Zhang W, Chen M, Lin M (2019) Effect of inoculation with nitrogen-fixing bacterium Pseudomonas stutzeri A1501 on maize plant growth and the microbiome indigenous to the rhizosphere. Syst Appl Microbiol 42:248–260. https://doi.org/10.1016/j.syapm.2018.10.010
Knoth JL, Kim SH, Ettl GJ, Doty SL (2014) Biological nitrogen fixation and biomass accumulation within poplar clones as a result of inoculations with diazotrophic endophyte consortia. New Phytol 201:599–609
Kollakkodan N, Anith K, Nysanth N (2020) Endophytic bacteria from Piper colubrinum suppress Phytophthora capsici infection in black pepper (Piper nigrum L.) and improve plant growth in the nursery. Arch Phytopathol Plant Prot 10(1080/03235408):1818493
Korenblum E, Aharoni A (2019) Phytobiome metabolism: beneficial soil microbes steer crop plants’ secondary metabolism. Pest Manag Sci 75:2378–2384
Kruasuwan W, Thamchaipenet A (2016) Diversity of culturable plant growth-promoting bacterial endophytes associated with sugarcane roots and their effect of growth by co-inoculation of diazotrophs and actinomycetes. J Plant Growth Reg 35:1074–1087
Larran S, Simon MR, Moreno MV, Siurana MS, Perelló A (2016) Endophytes from wheat as biocontrol agents against tan spot disease. Biol Control 92:17–23
Levy A, Conway JM, Dangl JL, Woyke T (2018) Elucidating bacterial gene functions in the plant microbiome. Cell Host Microbe 24:475–485
Lin G-Y, Lin C-Y, Chang S-J, Lin W-Y (2020) The dynamics of endophytic bacterial community structure in rice roots under different field management systems. Agronomy 10:1623
Lobo CB, Juárez Tomás MS, Viruel E, Ferrero MA, Lucca ME (2019) Development of low-cost formulations of plant growth-promoting bacteria to be used as inoculants in beneficial agricultural technologies. Microbiol Res 219:12–25. https://doi.org/10.1016/j.micres.2018.10.012
López-BucioCruz-Ramı́rez JA, Herrera-Estrella L (2003) The role of nutrient availability in regulating root architecture. Curr Opin Plant Biol 6:280–287. https://doi.org/10.1016/S1369-5266(03)00035-9
Ma T, Zeng W, Li Q, Wu J, Huang J (2016a) Effects of water, salt and nitrogen stress on sunflower (Helianthus annuus L.) at different growth stages. J Soil Sci Plant Nutri 16:1024–1037
Ma Y, Rajkumar M, Zhang C, Freitas H (2016b) Beneficial role of bacterial endophytes in heavy metal phytoremediation. J Environ Manag 174:14–25. https://doi.org/10.1016/j.jenvman.2016.02.047
Mane R, Vedamurthy A (2018) The fungal endophytes: sources and future prospects. J Med Plants Studies 6:121–126
Mashiane RA, Ezeokoli OT, Adeleke RA, Bezuidenhout CC (2017) Metagenomic analyses of bacterial endophytes associated with the phyllosphere of a Bt maize cultivar and its isogenic parental line from South Africa. World J Microbiol Biotechnol 33:80
Merasenla A, Gayan A, Buragohain S, Nath DJ (2016) Occurrences and characterization of endophytic Gluconacetobacter isolated from sugarcane tissues of North Eastern region of India. Ind J Agric Res 50:287–294
Mesa J, Mateos-Naranjo E, Caviedes MA, Redondo-Gómez S, Pajuelo E, Rodríguez-Llorente ID (2015) Endophytic cultivable bacteria of the metal bioaccumulator Spartina maritima improve plant growth but not metal uptake in polluted marshes soils. Front Microbiol 6:1450
Mhatre PH, Karthik C, Kadirvelu K, Divya KL, Venkatasalam EP, Srinivasan S, Ramkumar G, Saranya C, Shanmuganathan R (2019) Plant growth promoting rhizobacteria (PGPR): a potential alternative tool for nematodes bio-control. Biocatal Agric Biotechnol 17:119–128. https://doi.org/10.1016/j.bcab.2018.11.009
Moreira H, Pereira SI, Marques AP, Rangel AO, Castro PM (2016) Mine land valorization through energy maize production enhanced by the application of plant growth-promoting rhizobacteria and arbuscular mycorrhizal fungi. Environ Sci Pollut Res 23:6940–6950
Mukherjee A, Singh B, Verma JP (2020) Harnessing chickpea (Cicer arietinum L.) seed endophytes for enhancing plant growth attributes and bio-controlling against Fusarium sp. Microbiol Res 237:126469
Nefzi A, Abdallah RAB, Jabnoun-Khiareddine H, Ammar N, Daami-Remadi M (2019) Ability of endophytic fungi associated with Withania somnifera L. to control Fusarium crown and root rot and to promote growth in tomato. Braz J Microbiol 50:481–494
Nehra V, Saharan BS, Choudhary M (2016) Evaluation of Brevibacillus brevis as a potential plant growth promoting rhizobacteria for cotton (Gossypium hirsutum) crop. Springerplus 5:948
Nohwar N, Khandare RV, Desai NS (2019) Isolation and characterization of salinity tolerant nitrogen fixing bacteria from Sesbania sesban (L) root nodules. Biocatal Agric Biotechnol 21:101325. https://doi.org/10.1016/j.bcab.2019.101325
Odelade KA, Babalola OO (2019) Bacteria, fungi and archaea domains in rhizospheric soil and their effects in enhancing agricultural productivity. Int J Environ Res Public Health 16:3873
Ojuederie OB, Olanrewaju OS, Babalola OO (2019) Plant growth promoting rhizobacterial mitigation of drought stress in crop plants: implications for sustainable agriculture. Agronomy 9:712
Okon Y, Kapulnik Y (1986) Development and function of Azospirillum-inoculated roots. Plant Soil 90:3–16
Okon Y, Labandera-Gonzalez CA (1994) Agronomic applications of Azospirillum: an evaluation of 20 years worldwide field inoculation. Soil Biol Biochem 26:1591–1601
Oliveira Ad, Urquiaga S, Döbereiner J, Baldani J (2002) The effect of inoculating endophytic N2-fixing bacteria on micropropagated sugarcane plants. Plant Soil 242:205–215
Omomowo OI, Babalola OO (2019) Bacterial and fungal endophytes: tiny giants with immense beneficial potential for plant growth and sustainable agricultural productivity. Microorganisms 7:481
Orozco-Mosqueda MdC, Rocha-Granados MdC, Glick BR, Santoyo G (2018) Microbiome engineering to improve biocontrol and plant growth-promoting mechanisms. Microbiol Res 208:25–31. https://doi.org/10.1016/j.micres.2018.01.005
Otieno N, Lally RD, Kiwanuka S, Lloyd A, Ryan D, Germaine KJ, Dowling DN (2015) Plant growth promotion induced by phosphate solubilizing endophytic Pseudomonas isolates. Front Microbiol 6:745
Pan D, Mionetto A, Tiscornia S, Bettucci L (2015) Endophytic bacteria from wheat grain as biocontrol agents of Fusarium graminearum and deoxynivalenol production in wheat. Mycot Res 31:137–143
Pandey PK, Singh S, Singh AK, Samanta R, Yadav RNS, Singh MC (2016) Inside the plant: bacterial endophytes and abiotic stress alleviation. J Appl Nat Sci 8:1899–1904
Parsa S, García-Lemos AM, Castillo K, Ortiz V, López-Lavalle LAB, Braun J, Vega FE (2016) Fungal endophytes in germinated seeds of the common bean, Phaseolus vulgaris. Fungal Biol 120:783–790
Pedraza RO, Motok J, Salazar SM, Ragout AL, Mentel MI, Tortora ML, Guerrero-Molina MF, Winik BC, Díaz-Ricci JC (2010) Growth-promotion of strawberry plants inoculated with Azospirillum brasilense. World J Microbiol Biotechnol 26:265–272
Pham VT, Rediers H, Ghequire MG, Nguyen HH, De Mot R, Vanderleyden J, Spaepen S (2017) The plant growth-promoting effect of the nitrogen-fixing endophyte Pseudomonas stutzeri A15. Arch Microbiol 199:513–517
Potshangbam M, Devi SI, Sahoo D, Strobel GA (2017) Functional characterization of endophytic fungal community associated with Oryza sativa L. and Zea mays L. Front Microbiol 8:325
Praptiwi MI, Fathoni A, Wulansari D, Agusta A (2015) Antibacterial screening of the culture of endophytic fungal extracts isolated from cinnamon stick (Cinnamomum burmanii [Nees & T. Nees] Blume). J Teknol Indones 38:33–41
Puri A, Padda KP, Chanway CP (2016) Evidence of nitrogen fixation and growth promotion in canola (Brassica napus L.) by an endophytic diazotroph Paenibacillus polymyxa P2b–2R. Biol Fertil Soils 52:119–125
Qaswar M, Jing H, Ahmed W, Dongchu L, Shujun L, Lu Z, Cai A, Lisheng L, Yongmei X, Jusheng G (2020) Yield sustainability, soil organic carbon sequestration and nutrients balance under long-term combined application of manure and inorganic fertilizers in acidic paddy soil. Soil Tillage Res 198:104569
Ramírez CA, Kloepper JW (2010) Plant growth promotion by Bacillus amyloliquefaciens FZB45 depends on inoculum rate and P-related soil properties. Biol Fertil Soils 46:835–844
Rani R, Kumar V, Usmani Z, Gupta P, Chandra A (2019) Influence of plant growth promoting rhizobacterial strains Paenibacillus sp. IITISM08, Bacillus sp. PRB77 and Bacillus sp. PRB101 using Helianthus annuus on degradation of endosulfan from contaminated soil. Chemosphere 225:479–489
Rasul M, Yasmin S, Zubair M, Mahreen N, Yousaf S, Arif M, Sajid ZI, Mirza MS (2019) Phosphate solubilizers as antagonists for bacterial leaf blight with improved rice growth in phosphorus deficit soil. Biol Control 136:103997. https://doi.org/10.1016/j.biocontrol.2019.05.016
Rath M, Mitchell T, Gold S (2018) Volatiles produced by Bacillus mojavensis RRC101 act as plant growth modulators and are strongly culture-dependent. Microbiol Res 208:76–84
Reeve W, Ardley J, Tian R, Eshragi L, Yoon JW, Ngamwisetkun P, Seshadri R, Ivanova NN, Kyrpides NC (2015) A genomic encyclopedia of the root nodule bacteria: assessing genetic diversity through a systematic biogeographic survey. Stand Genom Sci 10:14
Romeh A, Hendawi M (2014) Bioremediation of certain organophosphorus pesticides by two biofertilizers, Paenibacillus (Bacillus) polymyxa (Prazmowski) and Azospirillum lipoferum (Beijerinck). J Agric Sci Technol 16:265–276
Rostami S, Azhdarpoor A (2019) The application of plant growth regulators to improve phytoremediation of contaminated soils: a review. Chemosphere 220:818–827. https://doi.org/10.1016/j.chemosphere.2018.12.203
Sah S, Singh N, Singh R (2017) Iron acquisition in maize (Zea mays L.) using Pseudomonas siderophore. 3 Biotech 7:121
Saharan B, Nehra V (2011) Plant growth promoting rhizobacteria: a critical review. Life Sci Med Res 21:30
Sahu PK, Singh S, Gupta A, Singh UB, Brahmaprakash G, Saxena AK (2019) Antagonistic potential of bacterial endophytes and induction of systemic resistance against collar rot pathogen Sclerotium rolfsii in tomato. Biol Control 137:104014
Salam M, Varma A (2019) Bacterial community structure in soils contaminated with electronic waste pollutants from Delhi NCR, India. Electron J Biotechnol 41:72–80. https://doi.org/10.1016/j.ejbt.2019.07.003
Sandargo B, Chepkirui C, Cheng T, Chaverra-Muñoz L, Thongbai B, Stadler M, Hüttel S (2019) Biological and chemical diversity go hand in hand: basidiomycota as source of new pharmaceuticals and agrochemicals. Biotechnol Adv 37:107344. https://doi.org/10.1016/j.biotechadv.2019.01.011
Santoyo G, Moreno-Hagelsieb G, del Carmen O-M, Glick BR (2016) Plant growth-promoting bacterial endophytes. Microbiol Res 183:92–99. https://doi.org/10.1016/j.micres.2015.11.008
Segaran G, Sathiavelu M (2019) Fungal endophytes: a potent biocontrol agent and a bioactive metabolites reservoir. Biocatal Agric Biotechnol 21:101284. https://doi.org/10.1016/j.bcab.2019.101284
Shah S, Shrestha R, Maharjan S, Selosse M-A, Pant B (2019) Isolation and characterization of plant growth-promoting endophytic fungi from the roots of Dendrobium moniliforme. Plants 8:5
Shahid M, Hameed S, Tariq M, Zafar M, Ali A, Ahmad N (2015) Characterization of mineral phosphate-solubilizing bacteria for enhanced sunflower growth and yield-attributing traits. Ann Microbiol 65:1525–1536
Sharma KP (2019) Tannin degradation by phytopathogen’s tannase: a plant’s defense perspective. Biocatal Agric Biotechnol 21:101342. https://doi.org/10.1016/j.bcab.2019.101342
Shehata H, Lyons E, Jordan K, Raizada M (2016) Bacterial endophytes from wild and ancient maize are able to suppress the fungal pathogen Sclerotinia homoeocarpa. J Appl Microbiol 120:756–769
Shi Y, Lou K, Li C (2009) Promotion of plant growth by phytohormone-producing endophytic microbes of sugar beet. Biology Fertil Soils 45:645–653
Sindhu SS, Sehrawat A, Sharma R, Dahiya A (2016) Biopesticides: use of rhizosphere bacteria for biological control of plant pathogens. Strain 90:166
Singh DK, Sharma VK, Kumar J, Mishra A, Verma SK, Sieber TN, Kharwar RN (2017) Diversity of endophytic mycobiota of tropical tree Tectona grandis Linn. f.: spatiotemporal and tissue type effects. Sci Rep 7:1–14
Smercina DN, Evans SE, Friesen ML, Tiemann LK (2019) To fix or not to fix: controls on free-living nitrogen fixation in the rhizosphere. Appl Environ Microbiol 85:e02518-e2546
Souza ARCd, Baldoni DB, Lima J, Porto V, Marcuz C, Machado C, Ferraz RC, Kuhn RC, Jacques RJ, Guedes JV (2017) Selection, isolation, and identification of fungi for bioherbicide production. Braz J Microbiol 48:101–108
Spaepen S, Vanderleyden J, Okon Y (2009) Plant growth-promoting actions of rhizobacteria. Adv Bot Res 51:283–320
Taghavi S, Garafola C, Monchy S, Newman L, Hoffman A, Weyens N, Barac T, Vangronsveld J, van der Lelie D (2009) Genome survey and characterization of endophytic bacteria exhibiting a beneficial effect on growth and development of poplar trees. Appl Environ Microbiol 75:748–757
Teixeira PJPL, Colaianni NR, Fitzpatrick CR, Dangl JL (2019) Beyond pathogens: microbiota interactions with the plant immune system. Curr Opin Microbiol 49:7–17. https://doi.org/10.1016/j.mib.2019.08.003
Tiwari R, Rana C (2015) Plant secondary metabolites: a review. Int J Eng Res Gen Sci 3:661–670
Tiwari K, Thakur HK (2014) Diversity and molecular characterization of dominant Bacillus amyloliquefaciens (JNU-001) endophytic bacterial strains isolated from native neem varieties of Sanganer region of Rajasthan. J Biodiv Bioprosp Dev 1:115
Truong DT, Tett A, Pasolli E, Huttenhower C, Segata N (2017) Microbial strain-level population structure and genetic diversity from metagenomes. Genome Res 27:626–638
Truyens S, Weyens N, Cuypers A, Vangronsveld J (2015) Bacterial seed endophytes: genera, vertical transmission and interaction with plants. Environ Microbiol Rep 7:40–50
Tyc O, Putra R, Gols R, Harvey JA, Garbeva P (2020) The ecological role of bacterial seed endophytes associated with wild cabbage in the United Kingdom. Microbiol Open 9:e00954
Uzoh IM, Babalola OO (2018) Rhizosphere biodiversity as a premise for application in bio-economy. Agric Ecosyst Environ 265:524–534. https://doi.org/10.1016/j.agee.2018.07.003
van Overbeek LS, Saikkonen K (2016) Impact of bacterial–fungal interactions on the colonization of the endosphere. Trends Plant Sci 21:230–242
Vaz AB, Fonseca PL, Badotti F, Skaltsas D, Tomé LM, Silva AC, Cunha MC, Soares MA, Santos VL, Oliveira G (2018) A multiscale study of fungal endophyte communities of the foliar endosphere of native rubber trees in Eastern Amazon. Sci Rep 8:1–11
Verma S, White J (2018) Indigenous endophytic seed bacteria promote seedling development and defend against fungal disease in browntop millet (Urochloa ramosa L.). J Appl Microbiol 124:764–778
Verma P, Yadav AN, Khannam KS, Panjiar N, Kumar S, Saxena AK, Suman A (2015) Assessment of genetic diversity and plant growth promoting attributes of psychrotolerant bacteria allied with wheat (Triticum aestivum) from the Northern hills zone of India. Ann Microbiol 65:1885–1899
Vidya P, Shintu P, Jayaram K (2016) Impact of phopshate solubilizing bacteria (Bacillus polymixa) on drought tolerance of green gram (Vigna radiata (L.) Wilczek). Ann Plant Sci 5:1318–1323
Wang W, Zhai Y, Cao L, Tan H, Zhang R (2016a) Endophytic bacterial and fungal microbiota in sprouts, roots and stems of rice (Oryza sativa L.). Microbiol Res 188–189:1–8. https://doi.org/10.1016/j.micres.2016.04.009
Wang W, Zhai Y, Cao L, Tan H, Zhang R (2016b) Endophytic bacterial and fungal microbiota in sprouts, roots and stems of rice (Oryza sativa L.). Microbiol Res 188:1–8
Wang Q, Jiang X, Guan D, Wei D, Zhao B, Ma M, Chen S, Li L, Cao F, Li J (2018) Long-term fertilization changes bacterial diversity and bacterial communities in the maize rhizosphere of Chinese Mollisols. Appl Soil Ecol 125:88–96
Wang G, Ye C, Zhang J, Koziol L, Bever JD, Li X (2019) Asymmetric facilitation induced by inoculation with arbuscular mycorrhizal fungi leads to overyielding in maize/faba bean intercropping. J Plant Interact 14:10–20
Wu Y, Ma L, Liu Q, Vestergård M, Topalovic O, Wang Q, Zhou Q, Huang L, Yang X, Feng Y (2020) The plant-growth promoting bacteria promote cadmium uptake by inducing a hormonal crosstalk and lateral root formation in a hyperaccumulator plant Sedum alfredii. J Hazard Mater 395:122661
Wubshet Z (2018) Economic importance and management of ginger bacterial wilt caused by Ralstonia solanacearum. Int J Res Study Agric Sci 4:1–11
Xin X-F, Kvitko B, He SY (2018) Pseudomonas syringae: what it takes to be a pathogen. Nat Rev Microbiol 16:316
Xu T, Cao L, Zeng J, Franco CM, Yang Y, Hu X, Liu Y, Wang X, Gao Y, Bu Z (2019) The antifungal action mode of the rice endophyte Streptomyces hygroscopicus OsiSh-2 as a potential biocontrol agent against the rice blast pathogen. Pesticide Biochem Physiol 160:58–69
Yadeta K, Thomma B (2013) The xylem as battleground for plant hosts and vascular wilt pathogens. Front Plant Sci 4:97
Ye D, Li T, Yi Y, Zhang X, Zou L (2019) Characteristics of endophytic fungi from polygonum hydropiper suggest potential application for P-phytoextraction. Fungal Ecol 41:126–136. https://doi.org/10.1016/j.funeco.2019.05.001
Yuan Y, Feng H, Wang L, Li Z, Shi Y, Zhao L, Feng Z, Zhu H (2017) Potential of endophytic fungi isolated from cotton roots for biological control against Verticillium wilt disease. PLoS One 12:e0170557
Zhang L, Zhang W, Li Q, Cui R, Wang Z, Wang Y, Zhang Y-Z, Ding W, Shen X (2020a) Deciphering the root endosphere microbiome of the desert plant Alhagi sparsifolia for drought resistance-promoting bacteria. Appl Environ Microbiol 86:e02863-e2819
Zhang X, Su C, Liu X, Liu Z, Liang X, Zhang Y, Feng Y (2020b) Effect of plant-growth-promoting rhizobacteria on phytoremediation efficiency of Scirpus triqueter in pyrene-Ni co-contaminated soils. Chemosphere 241:125027. https://doi.org/10.1016/j.chemosphere.2019.125027
Zheng H, Mao Y, Teng J, Zhu Q, Ling J, Zhong Z (2015) Flagellar-dependent motility in Mesorhizobium tianshanense is involved in the early stage of plant host interaction: study of an flgE mutant. Curr Microbiol 70:219–227
Zheng Y-K, Miao C-P, Chen H-H, Huang F-F, Xia Y-M, Chen Y-W, Zhao L-X (2017) Endophytic fungi harbored in Panax notoginseng: diversity and potential as biological control agents against host plant pathogens of root-rot disease. J Ginseng Res 41:353–360
Złoch M, Thiem D, Gadzała-Kopciuch R, Hrynkiewicz K (2016) Synthesis of siderophores by plant-associated metallotolerant bacteria under exposure to Cd2+. Chemosphere 156:312–325
Acknowledgements
B.S.A. is grateful to the National Research Foundation (NRF), South Africa/The World Academy of Science African Renaissance Ph.D. scholarship (Ref: UID: 116100) for giving him a stipend. O.O.B. acknowledges NRF for the grants (UID: 123634; 132595) that support research in her laboratory.
Funding
This study was funded by the National Research Foundation of South Africa (UID: 123634; 132595).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that there is no conflict of interest regarding the publication of this review paper.
Additional information
Handling Editor: Rhonda Peavy.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Adeleke, B.S., Babalola, O.O. Roles of Plant Endosphere Microbes in Agriculture-A Review. J Plant Growth Regul 41, 1411–1428 (2022). https://doi.org/10.1007/s00344-021-10406-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00344-021-10406-2