Abstract
Objectives
The Scrotal and Penile Imaging Working Group (SPI-WG) appointed by the board of the European Society of Urogenital Radiology (ESUR) has produced recommendations for magnetic resonance imaging (MRI) of the scrotum.
Methods
The SPI-WG searched for original and review articles published before September 2016 using the Pubmed and Medline databases. Keywords used were ‘magnetic resonance imaging’, 'testis or testicle or testicular', 'scrotum', 'intratesticular', 'paratesticular', 'extratesticular' 'diffusion-weighted', 'dynamic MRI'. Consensus was obtained among the members of the subcommittee. The expert panel proposed recommendations using Oxford Centre for Evidence-Based Medicine 2011 Levels of Evidence.
Results
The recommended MRI protocol should include T1-, T2-weighted imaging, diffusion-weighted imaging and dynamic contrast-enhanced MRI. Scrotal MRI can be clinically applied for lesion characterisation (primary), including both intratesticular and paratesticular masses, differentiation between germ-cell and non-germ-cell neoplasms (evolving), characterisation of the histological type of testicular germ cell neoplasms (TGCNs, in selected cases), local staging of TGCNs (primary), acute scrotum (in selected cases), trauma (in selected cases) and undescended testes (primary).
Conclusions
The ESUR SPI-WG produced this consensus paper in which the existing literature on MRI of the scrotum is reviewed. The recommendations for the optimal imaging technique and clinical indications are presented.
Key points
• This report presents recommendations for magnetic resonance imaging (MRI) of the scrotum.
• Imaging acquisition protocols and clinical indications are provided.
• MRI is becoming established as a worthwhile second-line diagnostic tool for scrotal pathology.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Imaging the scrotal contents has advanced significantly in the last decade, initially with improvements in ultrasound (US) techniques and latterly with the application of magnetic resonance imaging (MRI). The drive behind adoption of the new technologies has been to reduce the number of unnecessary radical surgical procedures by better lesion characterisation.
Ultrasonography, including conventional grey-scale and colour Doppler ultrasonography (CDUS), remains the first choice for diagnostic imaging of the scrotum [1,2,3,4,5]. However, changes in echogenicity may be nonspecific and a confident characterisation of the nature of scrotal masses is not always possible [6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21].
MRI has proved to be a diagnostic tool of high performance for morphological assessment and tissue characterisation in the evaluation of scrotal masses, and may be used to reduce the incidence of unnecessary surgical explorations, especially in cases of inconclusive US findings or in patients in which the US findings are inconsistent with the clinical examination [6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52]. MRI is less operator-dependent than US and has a wide field of view and multiplanar capabilities, demonstrating in exquisite anatomical detail the entire scrotum [6,7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52]. Advances in MRI using a multiparametric approach, which combines anatomical and functional data, help in narrowing the differential diagnosis in cases of both intratesticular [8, 11,12,13,14,15,16,17,18,19,20,21, 42, 53,54,55,56,57,58,59,60,61,62,63,64,65,66,67] and paratesticular [8, 14,15,16,17,18,19,20, 43, 48, 49] masses. MRI findings correlate well with the histological characteristics of testicular germ cell neoplasms (TGCNs) [8, 15, 30, 68,69,70]. Accurate preoperative imaging evaluation of the local extent of the disease is provided in patients with TGCNs who may be candidates for testis-sparing surgery (TSS) [7, 13, 16, 42]. The visualization of the tunica albuginea is another distinct advantage of MRI, allowing the assessment in cases of trauma (surgical or conservative management depending on integrity) or testicular neoplasms (T staging, up until now restricted to histological analysis) [8, 13, 19, 24, 26].
However, data regarding optimal acquisition protocols and clinical indications for MRI of the scrotum are lacking. The aim of this work was to produce recommendations on (1) minimal acquisition standards for MRI of the scrotum, and (2) clinical indications from the Scrotal and Penile Imaging Working Group (SPI-WG) of the European Society of Urogenital Radiology (ESUR).
Method
For these recommendations, the ESUR SPI-WG searched for original and review articles published before September 2016, in the English-language literature, regarding studies on human subjects, using the Pubmed and Medline databases. Keywords used were ‘magnetic resonance imaging’, 'testis or testicle or testicular', 'scrotum', 'intratesticular', 'paratesticular', 'extratesticular', 'diffusion-weighted' and 'dynamic MRI'. Consensus was obtained among the members of the subcommittee through discussions at international congresses and by e-mail. When necessary, the body of scientific recommendations was supplemented by the combined expertise of the multinational group.
The expert panel proposed a final recommendation for each criterion using Oxford Centre for Evidence-Based Medicine (OCEBM) 2011 Levels of Evidence [LE] [71], defined as follows:
-
LE1: systematic review of cross-sectional studies, with consistently applied reference standard and blinding
-
LE2: individual cross-sectional studies, with consistently applied reference standard and blinding
-
LE3: non-consecutive studies, or studies without consistently applied reference standards
-
LE4: case-control studies, or poor or non-independent reference standard
-
LE5: mechanism-based reasoning (expert opinion).
The following grades were attributed: Grade A, consistent LE1 studies; Grade B, consistent LE2 or LE3 studies or extrapolations from LE1 studies; Grade C, LE4 studies or extrapolations from LE2 or LE3 studies; and Grade D, LE5 studies or troubling inconsistent or inconclusive studies of any level.
Results
Technical requirements
1.5 Tesla versus 3.0 Tesla systems
The majority of published articles use a 1.5 Tesla system. No data on a 3.0 Tesla magnet exist.
Coil selection
A circular surface coil is recommended for imaging the scrotum in most studies (LE2) [6, 8, 10, 12, 13, 15, 17, 25, 36, 38, 45, 55, 58, 60, 63]. Surface coils offer an increased signal-to-noise ratio, allowing thin-section imaging and small field of views (Fig. 1a). However, studies using phased-array multichannel coils also have reported satisfactory results in the evaluation of scrotal pathology (LE2) [19, 21, 24, 40, 41, 43, 59, 67], with the option of using parallel imaging methods. Phased-array coils also may be used to evaluate the abdomen and pelvis for staging testicular malignancies [12, 15].
Technical requirements for MRI of the scrotum: (a) Various circular surface coils 17, 11 and 8 cm in diameter. In the choice of a surface coil, patient's age, size of scrotum and imaging coverage should be taken into consideration. (b) Adhesive tape and gauge. (c) Surface coil is placed over the scrotum. (d) Patient positioning
Surface coils are recommended in the evaluation of the scrotum (Grade B). Phased-array coils also provide diagnostic information (Grade B).
Patient preparation and positioning
Adequate support and correct positioning of the scrotum is essential (LE2) [8, 12, 13, 15, 17, 19, 21, 45, 55, 58, 63]. The penis should be raised, covered with a light cloth such as gauze, and fixed to the lower abdominal wall. Scrotum should be elevated by placing a towel between the thighs. A second towel is placed over the scrotum and the coil is placed on top of the second towel. The towels ideally should be warm to reduce scrotal muscular contractions that would degrade the images. Bands may be used to appose the thighs as close as possible to maintain the scrotum on the thighs. A peripheral intravenous line with a 19-gauge needle is placed into the subcutaneous veins of the forearm or antecubital fossa. Finally, the patient is placed into the MRI unit in the supine position with the feet first (Figs. 1b-d).
Adequate support and correct positioning of the scrotum are essential key elements in performing MRI of the scrotum (Grade B).
Conventional (anatomical) imaging
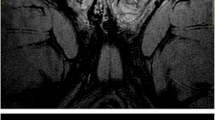
The testes should be examined in at least two orthogonal planes, along the testicular length and transverse axes. The coronal and axial plane is recommended. Both T1-weighted (T1WI) and T2-weighted (T2WI) sequences are essential (Fig. 2). T1WI provides information about scrotal anatomy and demonstrates increased signal in certain tissues, such as fat and methaemoglobin. T1WI in transverse orientation is recommended (LE2) [8, 12, 13, 15, 17, 19, 21, 45, 55, 58, 63]. MRI protocol in some studies included fat-saturated T1WI (LE4) [15, 17, 19] and dual-echo (in-phase and out-of-phase) sequences (LE4) [17].
Normal MRI findings in a 22-year-old man. Transverse (a) T1-weighted (T1WI) and (b) T2-weighted (T2WI). Normal testes are seen as sharply demarcated, homogeneous oval structures, with intermediate T1 and high T2 signal. The tunica albuginea appears as a thin hypointense halo, clearly defined on T2WI. The epididymis is slightly heterogeneous, isointense and hypointense (arrowhead) to the testis on T1WI and T2WI, respectively. (c) Coronal T2WI shows both epididymal heads (arrow), hypointense compared to normal testes. Bilateral small hydrocoele is also seen (normal finding). (d) Transverse diffusion-weighted image (DWI) (b = 900 s/mm2) and (e) the corresponding apparent diffusion coefficient (ADC) map. Normal testes are hyperintense and slightly hypointense on high b value DWI and ADC maps, respectively, due to the histological complexity of the normal testicular parenchyma. The ADC of normal testis has been reported to vary from 1.08 to 1.31 × 10-3 mm2/s, with an increase with advancing age [61]
T2WI in at least two planes, including the transverse and the coronal plane, should follow (LE2) [8, 12, 13, 15, 17, 19, 21, 45, 55, 58, 63]. These images are best for lesion detection, localisation and characterisation. Sagittal T2WI is optional, but is recommended in the following cases: evaluation of the epididymis; lesion localisation; depiction of small-sized lesions close to the anterior or posterior testicular surfaces (LE4) [6]; testicular rupture (L32) [8, 13, 15, 19, 24]; and local staging of testicular carcinomas (LE2) [7, 13, 15, 16, 42].
T2WI obtained with TE of 100–140 ms provides satisfactory T2 contrast between the testis, the epididymis, the spermatic cord and the surrounding fat tissue (LE2) [8, 12, 13, 15, 17, 19, 21, 45, 55, 58, 63]. Some studies have used chemical-selective fat-suppression T2WI (LE2) [19, 38, 45, 55, 59, 63], instead of conventional T2W1. Heavy T2WI also has been proposed for the evaluation of scrotal pathology (LE5) [45]. T2*-weighted gradient echo sequence has been advocated in patients suspected of testicular torsion due to their sensitivity in the detection of blood products (LE2) [45, 55, 63].
Optimal coverage is provided by thin sections (3–4 mm) without any gap and a 10- to 28-cm field of view. For very small testes, 2.5 mm without any gap can be performed. Superior and inferior saturation bands should be applied to eliminate ghosting artefacts from blood flow along the phase-encoding axis [12]. Respiratory compensation is not necessary [51]. Axial T1WI of the abdomen is recommended to search for metastatic lymphadenopathy [12], and as an overview in cases of testicular pain looking for causes of referred pain.
Axial T1WI, axial and coronal T2WI with thin sections are the minimum requirements for conventional MRI of the scrotum (Grade B). Fat-saturated T1WI and fat-saturated T2WI may be used as alternatives to standard sequences (Grade B). The addition of T2*-weighted gradient echo sequence is proposed in acute scrotum (Grade B).
Diffusion-weighted imaging
A few recent studies reported improvement in the characterisation of intratesticular lesions with the addition of diffusion-weighted imaging (DWI) (LE2) [58, 59]. The apparent diffusion coefficient (ADC) of testicular carcinomas has been reported to be lower than that of normal testis and various benign intratesticular lesions [58, 59]. DWI also has been reported useful in the diagnosis of testicular torsion (LE2) [63], the detection and localisation of nonpalpable undescended testes (LE2) [65], and the detection of fibrosis in testes with varicocele [67].
DWI should be acquired in the axial plane with an echo planar diffusion pulse sequence. At least three different b values should be acquired, including 0, 400–500 and 800–1,000 s/mm2. ADC maps can be generated on the MRI console, and should be analysed both qualitatively and quantitatively (Figs. 2d,e). For qualitative assessment, high b value (800–1,000) DWI and ADC maps should be used, evaluated in combination with T1WI and T2WI.
DWI is recommended in the evaluation of scrotal pathology (Grade B).
Dynamic contrast-enhanced MRI
Dynamic contrast-enhanced (DCE) MRI of the scrotum can be used when further tissue characterisation is needed or when patients present with acute scrotal symptoms (LE2) [21, 25, 45, 55, 56, 63]. The technique is useful in the diagnosis of testicular torsion (LE4) [23, 64] and the characterisation of scrotal lesions (LE2) [21, 55]. In a retrospective study of 44 men with various testicular lesions [56] three types of contrast enhancement were described according to the shape of time-signal-intensity (TSI) curves: type I, a gradual linear increase of enhancement throughout the examination, corresponding to the enhancement of normal testis; type II, a brisk upstroke enhancement, followed by either a plateau or a slight further increase of enhancement, representing benign lesions (Fig. 3); and type III, a brisk enhancement, followed by gradual washout of the contrast medium, representing malignancies (Fig. 4).
Left acute epididymo-orchitis in a 40-year-old man. T2-weighted image (T2WI) in (a) transverse and (b) coronal orientation show heterogeneity and hypointensity of the left testicular parechyma. (c) Coronal subtracted dynamic contrast-enhanced (DCE) image (early phase) depicts left testis avidly and heterogeneously enhancing. (d) TSI curves of acute orchitis (type II, blue) and contralateral normal testis (type I, red)
Left testicular seminoma in a 27-year-old man. T2-weighted image (T2WI) in sagittal (a) and axial (b) planes depict left intratesticular mass, mainly homogenous, of low signal intensity. The tumour invades the testicular tunicae and extends into the paratesticular space (long arrow), findings which subsequently were confirmed on pathology. (c) Transverse T1-weighted image (T1WI). The neoplasm is detected isointense compared to the contralateral testis. (d) Transverse apparent diffusion coefficient (ADC) (b = 900 s/mm2) map shows hypointense tumour, due to restricted diffusion. The ADC (x 10-3 mm2/s) of the neoplasm is 0.53. (e) TSI of the tumour (type III)
Although more technically demanding and time-consuming, subtraction DCE-MRI following the administration of gadolinium-based contrast medium is recommended for the evaluation of the scrotum (LE2) [21, 25, 45, 55, 56, 63]. The technique consists of a series of coronal three-dimensional (3D) fast field-echo sequences performed after a bolus injection (1–2 ml/s) of gadolinium-based contrast medium. The rapid injection of contrast agent should be performed within 5 s, followed by flush of 20 ml of physiological saline. Five to seven imaging sets should be consecutively acquired 15 s after the injection of contrast material. The actual examination time is 8 min and the duration of each set 50–60 s. Then, the slice-by-slice subtraction is performed to obtain subtraction DCE images. The data set obtained immediately before administration of contrast agent is used as a mask and subtracted section by section from each of the data sets obtained after contrast administration by using commercially available software.
Some studies have used post-contrast conventional TIWI with (LE4) [15, 19] and without fat saturation (LE4) [17, 60], in the axial and coronal plane, instead of DCE-MRI.
Subtracted DCE-MRI is recommended in the evaluation of scrotal diseases (Grade B). Conventional contrast-enhanced T1WI may be used as an alternative (Grade C).
Table 1 summarises the minimum requirements for MRI of the scrotum.
Clinical indications
Lesion localisation
The primary goal in the evaluation of a palpable scrotal mass is to determine its intra- or extratesticular origin. US remains the initial imaging modality in assessing the location of a scrotal mass. However, when the exact location is uncertain with US, MRI may be helpful (LE4) [12, 15,16,17, 19, 48, 49], especially in differentiating masses arising from the tunica albuginea rather than from the peripheral seminiferous tubules (Fig. 5). The delineation of the tunica albuginea, seen as a thin hypointense rim between the mass and the adjacent testis, helps to indicate the origin of the mass in these cases [19]. T2WI in three planes is recommended.
T2-weighted image (T2WI) in (a) coronal and (b) transverse orientation show small right paratesticular mass lesion (arrowhead), in close proximity to the testicular tunicae and the ipsilateral testis. The lesion appears slightly hypointense on T2WI. (c) Axial contrast-enhanced T1WI depicts lesion enhancing after gadolinium administration. Histology reported adenomatoid tumour attached at the visceral layer of the tunica vaginalis
MRI of the scrotum is recommended when differentiation between intratesticular and paratesticular masses is not possible, based on US findings (Grade C).
Lesion characterisation
Intratesticular masses
The recommended MRI protocol for characterisation of intratesticular masses should include T1WI, T2WI, DWI and subtracted DCE-MRI.
Benign versus malignant
Although most intratesticular masses are malignant, a possible diagnosis of various benign entities based on imaging features may improve patient care and decrease the number of unnecessary radical surgical procedures. In these cases, follow-up, biopsy, tumour enucleation and TSS may be justified [6,7,8,9,10,11,12,13,14,15,16,17,18,19,20, 39, 42]. MRI may provide important information in the preoperative characterisation of the histological nature of various benign intratesticular mass lesions in terms of morphological information and by showing the presence of fat, fluid, haemorrhage, fibrous tissue and solid contrast-enhancing tissue (LE2) [39, 42]. The technique can be used mainly as a problem-solving tool when US findings are equivocal (LE4) [6,7,8, 10,11,12,13,14,15,16,17,18,19,20, 42].
In patients with equivocal US, MRI is recommended as a second-line technique for characterisation of intratesticular masses (Grade C).
Germ cell neoplasms versus non-germ cell neoplasms
The conventional MRI criteria used to characterize TGCNs have been described [7, 8, 10, 12, 17, 20, 30, 39, 42] (Appendix 1).
The widespread use of US has resulted in an increase of incidentally discovered nonpalpable small solid testicular masses, which are up to 80% are benign, with Leydig cell tumours (LCTs) being the most frequent [72]. TSS is recommended in small LCTs [73]. Although the preoperative characterisation of these tumours is difficult, MRI features may be helpful (LE2) [21]. TSS is also recommended in small-sized Sertoli cell tumours [73]. No adequate data are available on MRI for the characterisation of these neoplasms [21, 42, 54].
MRI may be used to suggest the diagnosis of primary testicular lymphoma in the presence of a hypointense T2 mass, strongly and heterogeneously enhancing, detected over 60 years of age (LE4) [74].
MRI may help in the characterisation of LCTs (Grade B).
Seminomas versus nonseminomatous tumours
Radical orchidectomy is the therapy of choice in men with TGCNs and usually should be carried out without any delay. However, in cases of disseminated disease and/or life-threatening metastases, chemotherapy is the first treatment, followed by orchidectomy when the patient's clinical condition improves [73]. In these cases, the preoperative differentiation between seminomas and nonseminomatous tumours is helpful. MRI features have been found to closely correlate with the histopathological characteristics of TGCNs (LE4) (Fig. 6) (Appendix 1) [8, 15, 17, 68,69,70]. ADC may be used as an additional tool (LE4) for differential diagnosis [70].
Nonseminomatous germ cell neoplasm of the left testis (embryonal carcinoma, teratoma and yolk sac tumour) in a 20-year-old man. Coronal (a) T2-weighted image (T2WI) and (b) subtracted dynamic contrast-enhanced (DCE) images demonstrate large heterogeneous left testicular mass, strongly and inhomogeneously enhancing
MRI is recommended to differentiate seminomas from nonseminomatous testicular neoplasms in selected cases (Grade C).
Paratesticular masses
Primary solid tumours of the paratesticular space are uncommon. A confident characterisation of their nature may obviate unnecessary radical orchidectomy in cases of benign lesions. US findings often overlap, precluding a specific diagnosis [48, 49]. MRI features with respect to tumour location, morphological findings and tissue characteristics can help to narrow the differential diagnosis (LE4) [8, 14, 15, 17, 48, 49, 75]. In cases of malignancies, MRI can help in suggesting the diagnosis of an aggressive neoplasm, assessing the local extent and the presence or absence of distant metastases (LE4) [14, 15, 48, 49, 76].
MRI protocol should implement T1WI, T2WI in three planes, DWI and subtracted DCE-MRI.
MRI is recommended for characterisation of paratesticular masses (Grade C).
Local staging of testicular germ cell neoplasms (TGCNs)
Although radical orchidectomy is the treatment of choice for TGCNs, TSS can be attempted in special circumstances, including synchronous bilateral testicular tumours, metachronous contralateral tumours and a tumour in a solitary testis [73]. MRI performs well in the preoperative evaluation of the local stage T in TGCNs, in patients who are candidates for TSS. Accurate information regarding tumour dimensions, possible invasion of the rete testis, the paratesticular space and/or the spermatic cord and presence of a pseudocapsule helping in tumour enucleation are provided by MRI (LE4) [7, 16, 17]. MRI protocol should include T2WI in all three planes.
MRI is recommended for local staging of TGCNs (Grade C).
Acute scrotum
US with colour or power Doppler is the first imaging examination in the evaluation of acute scrotum, often allowing discrimination between causes that require emergency surgery, such as torsion from those that can be treated non-surgically [1,2,3,4,5, 77]. However, the detection of blood flow on US depends on the expertise of the investigator, and the sensitivity of the machine [11, 23, 26]. Patients with incomplete testicular torsion or those referred to a urologist several days after the onset of symptoms also represent problematic cases, both clinically and sonographically [8]. MRI may be used as a second-line examination in cases of inconclusive US findings or as a confirmatory study, helping plan appropriate treatment (LE2) [22, 25, 55, 63].
Segmental testicular infarction (STI) is a rare testicular disease, usually presenting with acute scrotal pain. CDUS findings are often diagnostic. MRI is excellent in suggesting the diagnosis of STI, when US findings are atypical (LE4) [15, 26, 41].
Despite satisfactory results, the disadvantages of MRI in an emergency setting should be acknowledged, including long examination time, high cost, frequent unavailability and need for sedation of young patients [25, 45].
The recommended MRI protocol should include T1WI, T2WI, DWI and subtracted DCE-MRI (LE2) [22, 25, 55, 63]. When the affected testis shows absent or diminished enhancement, T2*WI can be added to help in the diagnosis of haemorrhagic necrosis (LE4) [64].
MRI may be used as a complimentary examination in acute scrotal diseases (Grade B).
Trauma
Accurate characterisation of the type of injury in cases of blunt scrotal trauma is mandatory, so that appropriate treatment could be planned. US is the first-line diagnostic tool for the evaluation of blunt trauma, accurately assessing the integrity of the tunica albuginea in most cases [3]. In equivocal cases, MRI represents a useful alternative imaging modality [LE2] [24]. In cases of trauma, MRI protocol should implement T1WI, T2WI in all three planes and subtracted DCE-MRI [8, 13, 15, 19, 24, 26].
MRI is recommended as a second-line imaging examination in cases of scrotal trauma and non-diagnostic US findings (Grade B).
Undescended testes
US does not always reliably localize nonpalpable undescended testes and does not rule out an intra-abdominal testis [78]. Many studies have addressed on the diagnostic efficacy of MRI in locating undescended testes [LE2] [50,51,52, 79, 80]. Krishnaswami et al. [81] in a systematic review concluded that conventional MRI, including T1WI and T2WI, is poor at locating both atrophied and intra-abdominal testes but performs modestly well in locating those in the inguino-scrotal regions. However, the addition of fat-suppressed T2WI (LE2) [65] and DWI (LE2) [65, 66] improves the diagnostic performance of the technique in detecting nonpalpable testes.
The recommended protocol includes T1WI, axial and coronal fat-suppressed T2WI and DWI. In cases of intra-abdominal testis, coverage should be extended to the lower poles of the kidneys. MRI is recommended for locating nonpalpable undescended testes (Grade B).
Table 2 summarises clinical indications for MRI of the scrotum.
Conclusion
These recommendations show that MRI of the scrotum, including a multiparametric protocol, represents a valuable and efficient supplemental imaging technique in the evaluation of scrotal pathology. This is of great clinical importance because more precise treatment strategies can be determined and surgical explorations or orchidectomy avoided for more patients. The minimal requirements for the acquisition of MRI can be applied with the generally available 1.5 T MR systems.
Abbreviations
- 3D:
-
Three-dimensional
- ADC:
-
Apparent diffusion coefficient
- CDUS:
-
Colour Doppler ultrasonography
- DCE:
-
Dynamic contrast-enhanced
- DWI:
-
Diffusion-weighted imaging
- ESUR:
-
European Society of Urogenital Radiology
- LCTs:
-
Leydig cell tumours
- LE:
-
Levels of Evidence
- MRI:
-
Magnetic resonance imaging
- OCEBM:
-
Oxford Centre for Evidence-Based Medicine
- SPI-WG:
-
Scrotal and Penile Imaging Working Group
- T1WI:
-
T1-weighted
- T2WI:
-
T2-weighted
- TGCNs:
-
Testicular germ cell neoplasms
- TSI:
-
Testicular segmental infarction
- TSS:
-
Testis sparing surgery
- US:
-
Ultrasonography
References
Appelbaum L, Gaitini D, Dogra VS (2013) Scrotal ultrasound in adults. Semin Ultrasound CT MR 34:257–273
Bhatt S, Jafri SZ, Wasserman N, Dogra VS (2011) Imaging of non-neoplastic intratesticular masses. Diagn Interv Radiol 17:52–63
Bhatt S, Dogra VS (2008) Role of US in testicular and scrotal trauma. Radiographics 28:1617–1629
Dogra VS, Gottlieb RH, Oka M, Rubens DJ (2003) Sonography of the scrotum. Radiology 227:18–36
Hamm B (1997) Differential diagnosis of scrotal masses by ultrasound. Eur Radiol 7:668–679
Baker LL, Hajek PC, Burkhard TK (1987) MR imaging of the scrotum: pathologic conditions. Radiology 163:93–98
Thurnher S, Hricak H, Carroll PR, Pobiel RS, Filly RA (1988) Imaging the testis: comparison between MR imaging and US. Radiology 167:631–636
Cramer BM, Schlegel EA, Thueroff JW (1991) MR imaging in the differential diagnosis of scrotal and testicular disease. Radiographics 11:9–21
Sica GT, Teeger S (1996) MR imaging of scrotal, testicular, and penile diseases. Magn Reson Imaging Clin N Am 4:545–563
Serra AD, Hricak H, Coakley FV et al (1998) Inconclusive clinical and ultrasound evaluation of the scrotum: impact of magnetic resonance imaging on patient management and cost. Urology 51:1018–1021
Choyke PL (2000) Dynamic contrast-enhanced MR imaging of the scrotum: reality check. Radiology 217:14–15
Woodward PJ, Sohaey R, O'Donoghue MJ, Green DE (2002) From the archives of the AFIP: tumours and tumourlike lesions of the testis: radiologic-pathologic correlation. Radiographics 22:189–216
Andipa E, Liberopoulos K, Asvestis C (2004) Magnetic resonance imaging and ultrasound evaluation of penile and testicular masses. World J Urol 22:382–391
Gupta R, Alobaidi M, Jafri SZ, Bis K, Amendola M (2005) Correlation of US and MRI Findings of Intratesticular and Paratesticular Lesions: From Infants to Adults. Curr Prob Diagn Radiol 34:35–45
Kim W, Rosen MA, Langer JE, Banner MP, Siegelman ES, Ramchandani P (2007) US MR imaging correlation in pathologic conditions of the scrotum. Radiographics 27:1239–1253
Parenti GC, Feletti F, Brandini F et al (2009) Imaging of the scrotum: role of MRI. Radiol Med 114:414–424
Cassidy FH, Ishioka KM, McMahon CJ (2010) MR imaging of scrotal tumours and pseudotumours. Radiographics 30:665–683
Philips S, Nagar A, Dighe M, Vikram R, Sunnapwar A, Prasad S (2012) Benign non-cystic scrotal tumours and pseudotumours. Acta Radiol 53:102–111
Mohrs OK, Thoms H, Egner T et al (2012) MRI of patients with suspected scrotal or testicular lesions: diagnostic value in daily practice. AJR Am J Roentgenol 199:609–615
Tsili AC, Giannakis D, Sylakos A, Ntorkou A, Sofikitis N, Argyropoulou MI (2014) MR imaging of scrotum. Magn Reson Imaging Clin N Am 22:217–238
Manganaro L, Vinci V, Pozza C et al (2015) A prospective study on contrast-enhanced magnetic resonance imaging of testicular lesions: distinctive features of Leydig cell tumours. Eur Radiol 25:3586–3595
Trambert MA, Mattrey RF, Levine D, Berthoty DP (1990) Subacute scrotal pain: evaluation of torsion versus epididymitis with MR imaging. Radiology 175:53–56
Terai A, Yoshimura K, Ichioka K et al (2006) Dynamic contrast-enhanced subtraction magnetic resonance imaging in diagnostics of testicular torsion. Urology 67:1278–1282
Kim SH, Park S, Choi SH, Jeong WK, Choi JH (2009) The efficacy of magnetic resonance imaging for the diagnosis of testicular rupture: a prospective preliminary study. J Trauma 66:239–242
Mäkelä E, Lahdes-Vasama T, Ryymin R et al (2011) Magnetic resonance imaging of acute scrotum. Scand J Surg 100:196–201
Avery LL, Scheinfeld MH (2013) Imaging of penile and scrotal emergencies. Radiographics 33:721–740
Rholl KS, Lee JK, Ling D, Heiken JP, Glazer HS (1987) MR imaging of the scrotum with a high-resolution surface coil. Radiology 163:99–103
Seidenwurm D, Smathers RL, Lo RK, Carrol CL, Bassett J, Hoffman AR (1987) Testes and scrotum: MR imaging at 1.5 T. Radiology 164:393–398
Sohn M, Neuerburg J, Bohndorf K, Sikora R, Daus HJ (1989) The value of magnetic resonance imaging at 1.5 T in the evaluation of the scrotal content. Urol Int 44:284–291
Schultz-Lampel D, Bogaert G, Thüroff JW, Schlegel E, Cramer B (1991) MRI for evaluation of scrotal pathology. Urol Res 19:289–292
Mattrey RF (1991) Magnetic resonance imaging of the scrotum. Semin Ultrasound CT MR 12:95–108
Schnall M (1993) Magnetic resonance imaging of the scrotum. Semin Roentgenol 28:19–30
Oyen R, Verellen S, Drochmans A (1993) Value of MRI in the diagnosis and staging of testicular tumours. J Belge Radiol 76:84–89
Müller-Leisse C, Bohndorf K, Stargardt A (1994) Gadolinium-enhanced T1-weighted versus T2-weighted imaging of scrotal disorders: is there an indication for MR imaging? J Magn Reson Imaging 4:389–395
Kubik-Huch RA, Hailemariam S, Hamm B (1999) CT and MRI of the male genital tract: radiologic-pathologic correlation. Eur Radiol 9:16–28
Langer JE, Ramchandani P, Siegelman ES, Banner MP (1999) Epidermoid cysts of the testicle: sonographic and MR imaging features. AJR Am J Roentgenol 173:1295–1299
Watanabe Y (2002) Scrotal imaging. Curr Opin Urol 12:149–153
Avila NA, Premkumar A, Merke DP (1999) Testicular adrenal rest tissue in congenital adrenal hyperplasia: comparison of MR imaging and sonographic findings. AJR Am J Roentgenol 172:1003–1006
Muglia V, Tucci S Jr, Elias J Jr, Trad CS, Bilbey I, Cooperberg PL (2002) Magnetic resonance imaging of scrotal diseases: when it makes the difference. Urology 59:419–423
Stikkelbroeck NM, Suliman HM, Otten BJ, Hermus AR, Blickman JG, Jager GJ (2003) Testicular adrenal rest tumours in postpubertal males with congenital adrenal hyperplasia: sonographic and MR features. Eur Radiol 13:1597–1603
Fernandez-Perez GC, Tardaguila FM, Velasco M, Rivas C, Dos Santos J, Cambronero J et al (2005) Radiologic findings of segmental testicular infarction. AJR Am Roentgenol 184:1587–1593
Tsili AC, Argyropoulou MI, Giannakis D, Sofikitis N, Tsampoulas K (2010) MRI in the characterisation and local staging of testicular neoplasms. AJR Am J Roentgenol 194:682–689
Park SB, Lee WC, Kim JK et al (2011) Imaging features of benign solid testicular and paratesticular lesions. Eur Radiol 21:2226–2234
Aganovic L, Cassidy F (2012) Imaging of the scrotum. Radiol Clin North Am 50:1145–1165
Watanabe Y (2012) Instrumentation, technical requirements: MRI. In: Bertolotto M, Trombetta C (eds) Scrotal Pathology, Medical Radiology. Diagnostic Imaging. Springer-Verlag, Berlin, pp 17–26
Woldrich JM, Im RD, Hughes-Cassidy FM, Aganovic L, Sakamoto K (2013) Magnetic resonance imaging for intratesticular and extratesticular scrotal lesions. Can J Urol 20:6855–6859
Parker RA 3rd, Menias CO, Quazi R et al (2015) MR Imaging of the Penis and Scrotum. Radiographics 35:1033–1050
Woodward PJ, Schwab CM, Sesterhenn IA (2003) From the archives of the AFIP: extratesticular scrotal masses: radiologic-pathologic correlation. Radiographics 23:215–240
Akbar SA, Sayyed TA, Jafri SZ, Hasteh F, Neill JS (2003) Multimodality imaging of paratesticular neoplasms and their rare mimics. Radiographics 23:1461–1476
Kier R, McCarthy S, Rosenfield AT, Rosenfield NS, Rapoport S, Weiss RM (1988) Nonpalpable testes in young boys: evaluation with MR imaging. Radiology 169:429–433
Sarihan H, Sari A, Abeş M, Dinç H (1998) Nonpalpable undescended testis: value of magnetic resonance imaging. Minerva Urol Nefrol 50:233–236
Kanemoto K, Hayashi Y, Kojima Y, Maruyama T, Ito M, Kohri K (2005) Accuracy of ultrasonography and magnetic resonance imaging in the diagnosis of nonpalpable testis. Int J Urol 12:668–672
Tsitouridis I, Maskalidis C, Panagiotidou D, Kariki EP (2014) Eleven patients with testicular leydig cell tumours: clinical, imaging, and pathologic correlation. J Ultrasound Med 33:1855–1864
Reinges MHT, Kaiser WA, Miersch WD, Vogel J, Reiser M (1995) Dynamic MRI of benign and malignant testicular lesions: preliminary observations. Eur Radiol 5:615–622
Watanabe Y, Dohke M, Ohkubo K et al (2000) Scrotal disorders: evaluation of testicular enhancement patterns at dynamic contrast-enhanced subtraction MR imaging. Radiology 217:219–227
Tsili AC, Argyropoulou MI, Astrakas LG et al (2013) Dynamic contrast-enhanced subtraction MRI for characterizing intratesticular mass lesions. AJR Am J Roentgenol 200:578–585
Sanharawi JE, Correas JM, Glas L, Ferlicot S, Izard V, Ducot B et al (2016) Non-palpble incidentally found testicular tumours: Differentiation between benign, malignant and burned-out tumours using dynamic contrast-enhanced MRI. Eur J Radiol 85:2072–2082
Tsili AC, Argyropoulou MI, Giannakis D, Tsampalas S, Sofikitis N, Tsampoulas K (2012) Diffusion-weighted MR imaging of normal and abnormal scrotum: preliminary results. Asian J Androl 14:649–654
Algebally AM, Tantawy HI, Yousef RR, Szmigielski W, Darweesh A (2015) Advantage of Adding Diffusion Weighted Imaging to Routine MRI Examinations in the Diagnostics of Scrotal Lesions. Pol J Radiol 80:442–449
Sonmez G, Sivrioglu AK, Velioglu M et al (2012) Optimized imaging techniques for testicular masses: fast and with high accuracy. Wien Klin Wochenschr 124:704–708
Tsili AC, Giannakis D, Sylakos A et al (2014) Apparent diffusion coefficient values of normal testis and variations with age. Asian J Androl 16:493–497
Tsili AC, Ntorkou A, Baltogiannis D et al (2015) The role of apparent diffusion coefficient values in detecting testicular intraepithelial neoplasia: preliminary results. Eur J Radiol 84:828–833
Maki D, Watanabe Y, Nagayama M et al (2011) Diffusion-weighted magnetic resonance imaging in the detection of testicular torsion: feasibility study. J Magn Reson Imaging 34:1137–1142
Watanabe Y, Nagayama M, Okumura A et al (2007) MR imaging of testicular torsion: features of testicular hemorrhagic necrosis and clinical outcomes. J Magn Reson Imaging 26:100–108
Kato T, Kojima Y, Kamisawa H, Takeuchi M, Mizuno K, Sasaki S et al (2011) Findings of fat-suppressed T2-weighted and diffusion-weighted magnetic resonance imaging in the diagnosis of non-palpable testes. BJU Int 107:290–294
Kantarci M, Doganay S, Yalcin A, Aksoy Y, Yilmaz-Cankaya B, Salman B (2010) Diagnostic performance of diffusion-weighted MRI in the detection of nonpalpable undescended testes: comparison with conventional MRI and surgical findings. AJR Am J Roentgenol 195:W268–W273
Karakas E, Karakas O, Cullu N et al (2014) Diffusion-weighted MRI of the testes in patients with varicocele: a preliminary study. AJR Am J Roentgenol 202:324–328
Johnson JO, Mattrey RF, Phillipson J (1990) Differentiation of seminomatous from nonseminomaous testicular tumours with MR imaging. AJR A m J Roentgenol 154:539–543
Tsili AC, Tsampoulas C, Giannakopoulos X (2007) MRI in the histologic characterisation of testicular neoplasms. AJR Am J Roentgenol 189:W331–W337
Tsili AC, Sylakos A, Ntorkou A et al (2015) Apparent diffusion coefficient values and dynamic contrast enhancement patterns in differentiating seminomas from nonseminomatous testicular neoplasms. Eur J Radiol 84:1219–1226
OCEBM Levels of Evidence Working Group*. “The Oxford Levels of Evidence 2”. Oxford Centre for Evidence-Based Medicine. http://www.cebm.net/index.aspx?o=5653
Rocher L, Ramchandani P, Belfield J et al (2015) Incidentally detected non-palpable testicular tumours in adults at scrotal ultrasound: impact of radiological findings on management Radiologic review and recommendations of the ESUR scrotal imaging subcommittee. Eur Radiol. doi:10.1007/s00330-015-4059-7
Albers P, Albrecht W, Algaba F, Bokemeyer C, Cohn-Cedermark G, Fizazi K, Horwich A, Laguna MP, Nicolai N, Oldenburg J. European Association of Urology. Guidelines on testicular cancer. Available at: http://www.uroweb.org/wp-content/uploads/11 Testicular-Cancer_LR.pdf.
Tsili AC, Argyropoulou MI, Giannakis D, Sofikitis N, Tsampoulas K (2012) Primary diffuse large B-cell testicular lymphoma: magnetic resonance imaging findings. Andrologia 44:845–847
Bertolotto M, Boulay-Coletta I, Butini R, Dudea SM, Grenier N, Oltmanns G et al (2016) Imaging of mesothelioma of tunica vaginalis testis. Eur Radiol 26:631–638
Ap Dafydd D, Messiou C, Thway K, Strauss DC, Nicol DL, Moskovic E (2016) Paratesticular Sarcoma: Typical Presentation, Imaging Features and Clinical Challenges. Urology 14 [Epub ahead of print]
Bertolotto M, Cantisani V, Valentino M, Pavlica P, Derchi LE (2016) Pitfalls in Imaging for Acute Scrotal Pathology. Semin Roentgenol 51:60–69
Tasian GE, Copp HL (2011) Diagnostic performance of ultrasound in nonpalpable cryptorchidism: a systematic review and meta-analysis. Pediatrics 127:119–128
Miyano T, Kobayashi H, Shimomura H, Yamataka A, Tomita T (1991) Magnetic resonance imaging for localizing the nonpalpable undescended testis. J Pediatr Surg 26:607–609
Yeung CK, Tam YH, Chan YL, Lee KH, Metreweli C (1999) A new management algorithm for impalpable undescended testis with gadolinium enhanced magnetic resonance angiography. J Urol 162:998–1002
Krishnaswami S, Fonnesbeck C, Penson D, McPheeters ML (2013) Magnetic resonance imaging for locating nonpalpable undescended testicles: a meta-analysis. Pediatrics 131:e1908–e1916
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Guarantor
The scientific guarantor of this publication is Dr. Athina C. Tsili.
Conflict of interest
The authors of this manuscript declare no relationships with any companies whose products or services may be related to the subject matter of the article.
Funding
The authors state that this work has not received any funding.
Statistics and biometry
No complex statistical methods were necessary for this paper.
Informed consent
Written informed consent was not required for this study because no patients were included.
Ethical approval
Institutional Review Board approval was not required because this manuscript presents guidelines only and did not require the use of patient-sensitive data.
Methodology
Guidelines based on literature review and expert opinion.
Rights and permissions
About this article
Cite this article
Tsili, A.C., Bertolotto, M., Turgut, A.T. et al. MRI of the scrotum: Recommendations of the ESUR Scrotal and Penile Imaging Working Group. Eur Radiol 28, 31–43 (2018). https://doi.org/10.1007/s00330-017-4944-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00330-017-4944-3