Abstract
A photographic seabed survey conducted off the Antarctic Peninsula region provided the opportunity to study spatial patterns, abundance and behaviour of the notothenioid benthic fish fauna. Overall, a total of 12,715 images taken with the Ocean Floor Observation System (OFOS) along 26 transects in three ecoregions (Joinville Island, Bransfield Strait and Drake Passage) were analysed. The fish fauna consisted of at least 34 species belonging to four families of both low-Antarctic and high-Antarctic origin. Nototheniids showed the highest relative abundance and species richness, followed by channichthyids, bathydraconids and artedidraconids. Direct in-situ observations in OFOS seabed images allowed descriptions of fish behaviour, such as aggregation of individuals (Notothenia coriiceps), specific body postures (Cygnodraco mawsoni and Cryodraco antarcticus) and parental care (Chaenodraco wilsoni, Chionodraco rastrospinosus, Pagetopsis macropterus and Trematomus hansoni). Fish density and species richness was primarily correlated with the occurrence of bryozoans, ascidians, and large cup-shaped sponges, providing a three-dimensional habitat suitable for fish settling, foraging, breeding and refuge from predators. Fish diversity was higher (a) off Joinville Island and in Bransfield Strait than in Drake Passage, where almost exclusively low-Antarctic species were recorded, and (b) between 100 and 600 m than at greater depths. Overall, the benthic fish fauna off the northern Antarctic Peninsula is zoogeographically composite and widespread, with well-structured spatial partitioning.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The benthic fish fauna off the northern Antarctic Peninsula and neighbouring islands is overwhelmingly dominated by species from the suborder Notothenioidei, both in terms of biomass and numbers, at approximately 85% of species and over 95% of individuals (Kock and Jones 2005). Driven by the different environmental conditions in the area, and based on their own latitudinal distribution, the fish fauna consists primarily of low-Antarctic species, such as nototheniids (Notothenia, Gobionotothen and Lepidonotothen) and some channichthyids, and high-Antarctic species, such as nototheniids (Trematomus) and bathydraconids that are abundant in waters deeper than 300 m but of negligible biomass (Kock and Stransky 2000). Based on several trawl surveys carried out in this area in the last two decades, the species composition and their relative abundance are relatively well-known (Kock et al. 2000; Jones et al. 2003; Arana et al. 2020). Nevertheless, the use of benthic trawling as investigating methodology inherently do not allow to document-specific habitat preferences and fish behaviour.
In response to climate change, Southern Ocean ecosystems have rapidly changed for at least the last three decades (Turner et al. 2014), in particular off the Antarctic Peninsula, where the changes are faster and more pronounced than elsewhere in the Southern Ocean (Clarke et al. 2007). Ocean warming has produced multiple effects in this area, including increases in ice melting rate, iceberg scouring of the seabed and sediment inflow, all contributing to considerably reduce the extent of sea-ice habitats and alter seafloor geomorphology (Siegert et al. 2019; Brasier et al. 2021). During their evolutionary history, marine organisms living in the Southern Ocean acquired a wide array of behavioural, biological and physiological adaptations to cope with the unique environmental conditions (Peck 2018). Their resilience capacity largely depends on their adaptation and acclimation potential. In Antarctic biotas, these adaptations are complex, extremely variable, and have not been subjected to the modern climate regime (Gutt et al. 2020). Climate change effects can be also diverse, affecting behavioural and physiological adaptations, spatial distribution and, ultimately, marine biodiversity (Siegert et al. 2019; Rogers et al. 2020). Baseline surveys are necessary to properly document and describe Antarctic biodiversity, and can be used as starting point to plan and develop long-term monitoring programmes. The comparison of data acquired over a long time period will enable to assess the impacts of environmental changes and to foster conservation practices (Convey and Peck 2019).
The recent employment of video cameras mounted on remotely operated vehicles allows the collection of a large amount of scientific information on seafloor ecosystems, including in-situ views of seabed habitats and quantitative data on the species composition, abundance, and spatial distribution of epibenthic fauna. Compared to trawl surveys, the image-based methods have inherent advantages, because they cause less disturbance to seabed habitats and biotas and allow analysis at a finer-scale for spatial structure and species associations. This innovative and low impact approach based on underwater photographic images analyses has been used to study the demersal fish assemblages of some Antarctic areas, including the Weddell Sea (Ekau and Gutt 1991; La Mesa et al. 2019), the Lazarev Sea (Gutt and Ekau 1996), the western Antarctic Peninsula (Amsler et al. 2016) and the Amundsen Sea (Eastman et al. 2013).
In the framework of the research project Dynamics of Antarctic Marine Shelf Ecosystems (DynAMo), aimed at investigating the epibenthic megafauna and its seafloor habitats in areas differently affected by the ongoing recent climate change, a seabed imaging survey was carried out on the shelf off the northern Antarctic Peninsula using the ocean floor observation system (OFOS) developed at the Alfred Wegener Institute, Helmholtz Centre for Polar and Marine Research in Bremerhaven (AWI) (Piepenburg et al. 2017). During the survey, the faunistic composition of mega-epibenthic communities and biota-environment relationships were explored at multiple spatial scales (Gutt et al. 2019). Based on a huge amount of environmental data like bottom water temperature, seasonal summer primary production and averaged sea-ice cover, and macrobenthic community studies, three different ecoregions were identified: an area east of the tip of the Antarctic Peninsula in the Weddell Sea (WS), another in the Bransfield Strait (BS) and the last located in the Drake Passage (DP) (Dorschel et al. 2016).
Following a similar approach and complementing the published data on the invertebrate benthic fauna (Gutt et al. 2019), we performed a multivariate analysis of the seabed images to investigate spatial patterns, abundance and behavioural aspects of notothenioid fishes, which dominated the benthic fish fauna of this area. The main question we would like to address was if and how these three different ecoregions influenced the patterns of distribution of the local demersal fish fauna, as well as the role of environmental drivers at a smaller spatial scale (i.e., habitat and depth) within each of them. Present results would represent baseline data for future investigation aimed at assessing changes in fish biodiversity and spatial distribution driven by the ongoing climate change.
Materials and methods
Oceanographic characteristics of the study area
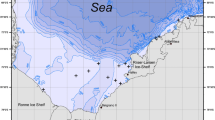
Off the northern tip of the Antarctic Peninsula, the Bransfield Strait and the neighbouring areas, including the western shelf of the South Shetland Islands and Joinville Island groups, have a complex hydrographic structure, consisting of different current pathways and water masses (Huneke et al. 2016) (Fig. 1). The southern and western sides of Joinville Island are directly intersected by cold water masses of Weddell Sea origin, which inflow into the Bransfield Strait forming the Transitional Zonal Water with Weddell Sea influence (TWW) through the shelf current (SC) around the tip of the Antarctic Peninsula. The western part of the Bransfield Strait is characterized by the Bransfield Current (BC), which flows north-westerly forming the relatively warm Transitional Zonal Water with Bellingshausen influence (TBW). Finally, the western shelf of the South Shetland Islands along the southernmost part of the Drake Passage is fringed by the southern boundary of the Antarctic Circumpolar Current (ACC) and is subjected to the TBW and modified Circumpolar Deep Water influence (Huneke et al. 2016). The bottom topography of this entire area is complex as well, characterized by wide and flat shelves with shallow banks incised by deep canyons or inner-shelf depressions, and slopes of variable steepness and ruggedness (Dorschel et al. 2016).
Schematic diagram of the main water masses and hydrography of the Bransfield Strait (a) and cross section of the upper few 100 m (b) (partially redrawn from Huneke et al. 2016). AP Antarctic Peninsula, SSI South Shetland Islands, JI Joinville Island, EI Elephant Island, BC Bransfield Current, SC shelf current, TBW Transitional Zonal Water with Bellingshausen influence, TWW Transitional Zonal Water with Weddell Sea influence, mCDW modified Circumpolar Deep Water
Field sampling
A seabed imaging survey aimed at investigating the epibenthic megafauna and seafloor habitats was conducted during Polarstern cruise ANT-XXIX/3 (PS81) off the northern Antarctic Peninsula between 26 January and 12 March 2013 (Gutt et al. 2013). Seabed images were taken from a total of 31 stations at water depths between 100 and 784 m along drift profiles (transects) by the AWI OFOS. For the benthic fish analyses, we selected 26 stations (Table 1), to avoid redundancy or mismatch between them. OFOS was a surface-powered gear equipped with a high-resolution, wide-angle still camera (Canon®EOS 5D Mark III). The system was vertically deployed from the star-board side of the ship until it hovered approximately 1.5 m above the seabed, then towed slowly over the ground at a speed of approximately 0.5 kn (0.25 ms−1) (Piepenburg et al. 2017). Three lasers placed beside the still camera projected on the bottom as red light points forming an equilateral triangle with a side length of 50 cm. For each image, the seabed area depicted was estimated using altimeter-derived height above seafloor and the laser’s scale. Seabed images each depicting an area of approximately 3.45 m2, were taken every 30 s along each transect, with length varying depending on the tow duration (Piepenburg et al. 2017). In this study, transects ranged in duration from 1:35 to 4:31 h and in distance over ground from 1.83 to 4.14 km, being strongly influenced by local weather conditions (Table 1).
The transects were located in the three ecoregions mentioned above (Fig. 2). Within each ecoregion, a series of depth-related habitats were selected along a bathymetric intermediate-scale shelf-slope gradient, including Bank, Upper slope, Slope, and Deep/canyon (Gutt et al. 2016). Based on mean depth within each transect, the following depth strata were selected: (1) 100–200 m, (2) 200–300 m, (3) 300–400 m, (4) 400–500 m, (5) 500–600 m and (6) 700–800 m.
Image processing
All seabed images were thoroughly examined at different magnifications to determine those images which contained fishes. Each fish was identified to the species level, based on general morphology, colouration and known depth distributions (Gon and Heemstra 1990). The main criteria allowing species identification were summarized as supplementary information (SI). Within each transect, the fish species density was calculated as number of individuals per 100 m2, derived from the total number of specimens counted along the transect and the surveyed area (mean area of single image x total number of images) (La Mesa et al. 2019). Particular attention was devoted to documenting fish behaviour of particular interest, such as typical body posture and nesting or egg guarding activities. In addition, the faunistic composition and relative abundance of mega-epibenthic fauna was assessed for each transect, regarding seven functional or systematic groups: ascidians (colonial and solitary), bryozoans, cnidarians (anthozoans, hydrozoans and gorgonians), echinoderms (asteroids, crinoids, echinoids, holothurians and ophiuroids), hemichordates, polychaetes (mobile), sponges (demosponges and hexactinellids) (Gutt et al. 2019).
Statistical analyses
Stepwise multiple regression analyses were run to determine the proportion of variance in the fish assemblage parameters (i.e., the number of species, S, the Shannon diversity index, H’ and density of the dominant species that could be attributed to the mega-epibenthic epifauna). Rare species (i.e., those with less than 3 specimens overall) were excluded from the analysis. A backward elimination procedure was adopted, whereby all the independent variables were entered into the equation and then sequentially removed, with a probability of F(p) = 0.1 criterion of removal. Histograms and normal probability plots were controlled to ascertain whether the assumption of normality was violated, and the residuals were plotted against the deleted residuals to identify outliers. An inspection of correlation coefficients and Tolerance/Variance Inflation Factor (VIF) values was made to detect multicollinearity. Unless otherwise stated, p < 0.05 was used as a criterion for significance in all tests.
In evaluating the influence of ecoregion, habitat and depth range on the multivariate variation in density of fish assemblages we employed a constrained ordination using a canonical analysis of principal coordinates (CAP), based on Bray–Curtis dissimilarities (Anderson and Willis 2003; Espino et al. 2015; French et al. 2021). The canonical correlations were tested using 4999 random permutations of the raw data, for ecoregion, habitat or depth range as constrained factors. Distinctness of groups was investigated using leave-one-out allocation success (Anderson and Robinson 2003). The product-moment correlations of the 24 species variables with the first two canonical discriminant axes (ρ1 and ρ2) were calculated and only those with an absolute correlation (i.e., square root (ρ12 + ρ22) > 0.50) were considered meaningful and showed in the biplot.
Results
General overview
Overall, 12,715 images from 26 stations were analysed and approximately 26% of these included at least one fish specimen. A total of 4405 notothenioid specimens were counted during the photographic survey and identified as belonging to four families, 24 genera and at least 34 species (Table 2, 3). Other non-notothenioid fishes, such as liparids, macrourids, muraenolepidids, rays and primarily zoarcids were recorded but not identified to higher taxonomic level for their problematic taxonomy, and thus excluded by further statistical analyses. Overall, they represented 9.5% of total specimens recorded. Zoarcids represented the most abundant non-notothenioid fishes, with peaks of abundance in the Drake Passage and in deeper waters (Fig. 3). Similarly, the pelagic nototheniid Pleuragramma antarctica was rarely encountered and, being clearly attracted to light sources, was also excluded from the analyses. Any other notothenioid species apparently was not attracted by the light, although we cannot exclude this behaviour at all. On the contrary, it should be possible that some species might intentionally avoid the light source, thus representing a potential limitation of this methodology. For each species recorded, a detailed description of abundance, morphological and behavioural aspects are reported in the appendix as Supplementary Information (SI).
Spatial patterns in fish diversity
The number of species slightly declined from Joinville to Bransfield Strait, whereas it dropped drastically in the Drake Passage (Fig. 4). A similar, though not as marked, decreasing trend was observed in species diversity. The highest species richness was recorded on banks and deep/canyons, the lowest on slopes. In comparison, the variation in diversity among the four habitats was weak. Species richness did not show substantial variation across the first five strata, but it declined sharply in the deepest one. The pattern of variation in diversity was quite similar, with a less marked decline in the deepest stratum.
Patterns of fish diversity across environmental categories, derived from the analysis of seabed images taken during Polarstern cruise ANT-XXIX/3 (PS81) off the northern Antarctic Peninsula between 26 January and 12 March 2013: a ecoregions, b habitats and c depth strata (1: 100–200 m, 2: 200–300 m, 3: 300–400 m, 4: 400–500 m, 5: 500–600 m, 6: 700–800 m)
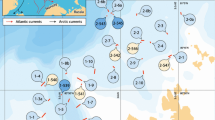
The results of the canonical analysis of principal coordinates (CAP) with ecoregion as the independent variable showed a clear separation among sampling stations from different ecoregions (Fig. 5a). Along the first CAP axis, the stations of Drake Passage segregated from those located at Joinville and Bransfield Strait, which in turn were separated along the second axis (squared canonical correlations δ12 = 0.85 and δ22 = 0.49; p = 0.0002; 84.6% of sampling stations correctly classified). Three groups of species made the largest contribution to the ecoregion-related differences observed in the fish assemblage structure. One group of three species (L. squamifrons, C. dewitti and C. aceratus) showed a positive correlation with the first axis (i.e., arrows pointing to the right side of the plot) and were exclusively or mostly recorded in the Drake Passage. A second group included six species (G. gibberifrons, L. nudifrons, A. orianae, P. evansii, P. macropterus and P. charcoti), plotted in the upper left quadrant of the CAP graphs, indicates their preferential occurrence in the Joinville Island stations. The three species (T. eulepidotus, P. mentella group and C. rastrospinosus) in the third group showed a negative correlation with both axes (i.e., arrows pointing to the lower left quadrant of the plot) and were confined primarily to Bransfield Strait.
Scatter plots of the canonical discriminant analysis on the effects of environmental categories on fish distribution, derived from the analysis of seabed images taken during Polarstern cruise ANT-XXIX/3 (PS81) off the northern Antarctic Peninsula between 26 January and 12 March 2013: a ecoregions, b habitats and c depth strata, with the relevant species contribution indicated by directional vectors. Aori, Artedidraco orianae; Cace, Chaenocephalus aceratus; Cant, Cryodraco antarcticus; Cdew, Chionobathyscus dewitti; Cras, Chionodraco rastrospinosus; Cwil, Chaenodraco wilsoni; Ggib, Gobionotothen gibberifrons; Llar, Lepidonotothen larseni; Lnud, Lepidonotothen nudifrons; Lsqu, Lepidonotothen squamifrons; Pbar, Pogonophryne barsukovi group; Pcha, Parachaenichthys charcoti; Peva, Prionodraco evansii; Pmac, Pagetospis macropterus; Pmen, Pogonophryne mentella group; Tber, Trematomus bernacchii; Teul, Trematomus eulepidotus
The fish assemblage structure was also significantly influenced by seabed habitat (Fig. 5b). In the CAP plot corresponding to habitat effect, the first axis separated the sampling stations located on Slope and Deep/canyon from those on Bank and Upper slope, which in turn were segregated along the second axis (squared canonical correlation δ12 = 0.74 and δ22 = 0.25; p = 0.0002; 73.1% of sampling stations correctly classified). The Leave-one-out allocation procedure showed that the assemblages recorded on Slope overlapped (with a mis-classification error of 43% of points) those inhabiting the Deep/canyon. Four species (C. dewitti, C. antarcticus, P. mentella and P. barsukovi groups) showed a positive correlation with the first axis (their arrows pointed to the right side of the graph) due to their preference for the Slope and/or Deep/canyon. Conversely, several species plotted on the right side of the plot, specifically (T. bernacchii and A. orianae) and primarily (G. gibberifrons, L. larseni, L. nudifrons, T. eulepidotus, P. evansii and P. macropterus) associated with the Bank and Slope stations.
The effects of water depth closely resembled those of the seabed habitat (Fig. 5c). Along the first canonical axis, the stations located in the shallower depth strata (1, 2 and 3) were opposite those sampled deeper (4, 5 and 6), the latter being separated along the second axis (squared canonical correlations δ12 = 0.95 and δ22 = 0.79; p = 0.0004; 65.4% of sampling stations correctly classified). In terms of taxonomic composition, the two groups of species that contributed most to the observed pattern were very similar to those indicated in the plot of seabed habitats. The position of P. mentella and P. barsukovi groups in the upper right quadrant of the plot was noteworthy in relation to their preferences for the deepest strata (5 and 6).
Relationships between fishes and invertebrate epibenthic megafauna
Multiple regression analysis indicated that the density pattern of 13 fish species (out of 24 species examined) was significantly influenced by different combinations of invertebrate mega-epibenthic fauna (Table 4). The largest amount of variation in species density explained by the regression models was obtained in decreasing importance for L. nudifrons (74%), P. macropterus (70%), N. coriiceps (65%), T. bernacchii (60%) and A. orianae (58%). In this group of species, a significant contribution to the total correlation coefficient was provided by bryozoans, ascidians and sponges, which showed positive correlations with fish densities, and by cnidarians and echinoderms, which were conversely characterised by negative relationships. The regression models for T. scotti, G. gibberifrons and T. hansoni explained smaller amounts of density variation (48%, 47% and 41%, respectively), which were mostly attributable to a positive relationship with ascidians and cnidarians, and a negative relationship with bryozoans and/or echinoderms. Significant regression models were also found for density of five species (L. squamifrons, P. charcoti, P. evansii, G. acuticeps and C. dewitti), with percentages of explained variation ranging between 16 and 33%. The variation in density of these species was dependent on a positive correlation with only one macrobenthic taxa (polychaetes, ascidians or cnidarians) and, only in C. dewitti, upon a negative relationship with echinoderms. The analysis performed on the assemblage parameters provided evidence of a significant regression model only for species richness, which showed a positive relationship with sponges and ascidians and a negative relationship with echinoderms (Table 5).
Discussion
Our results confirm that underwater photography is a powerful tool for investigating local fish communities and for identifying also small-sized or fish with different phenotypes at the species level. On the other hand, species exhibiting negative phototaxis behaviour or with benthopelagic habits might be underrepresented. Remotely operated vehicles equipped with still and video cameras provide insights on fish behaviour, information unavailable from traditional trawl surveys. By avoiding the impact of benthic trawling, the use of imaging to study benthic communities would be also a logical choice for sampling and investigations of the recently designated marine protected areas of the Southern Ocean, such as the Ross Sea or the southern shelf of the South Orkney Islands (Brooks et al. 2020).
In agreement with information about species composition and relative abundance of fishes derived from trawl catches in previous studies (Kock and Stransky 2000; Kock and Jones 2005), the benthic fish fauna in the Bransfield Strait and neighbouring areas consisted primarily of notothenioid fishes (90% as number of individuals). The total number of species recorded during the photographic survey was comparable with the species inventory obtained by previous bottom trawl surveys (34 vs 39 species) (Kock et al. 2000). As expected, the few notothenioid species with semi-pelagic or pelagic habits (Eastman 2020) were absent (Dissostichus mawsoni and Vomeridens infuscipinnis) or seldom documented (A. mitopteryx, P. antarcticum, T. loennbergii, D. hunteri, N. ionah, P. georgianus and C. dewitti), preventing a reliable estimate of their abundance.
The taxonomic composition of the benthic fish fauna in the study area clearly indicated the presence of low-Antarctic species, with geographic distributions centered along the southern Scotia Arc (G. gibberifrons, L. nudifrons, L. larseni, P. charcoti, C. rastrospinosus and C. aceratus), and high-Antarctic species with circum-Antarctic distributions, with the tip of the Antarctic Peninsula representing the northern limit (Trematomus spp. and some species of artedidraconids, bathydraconids and channichthyids). Among the low-Antarctic species, Notothenia rossii and Champsocephalus gunnari represented the bulk of catches in benthic trawling activities carried out recently in this area, suggesting a clear indication of recovery compared to a previous survey (Arana et al. 2020). Nevertheless, they were not recorded during the seabed imaging survey, despite the relatively wide area and depth range explored. A possible explanation is the benthopelagic habits of these species, which feed almost exclusively on krill in the water column (Eastman 1993). On the other hand, several relatively rare species, such as A. mitopteryx and D. hunteri, were documented for the first time in underwater images.
Direct field observation of the sea floor also allowed descriptions of certain aspects of fish behaviour, for example regarding fish aggregation, specific body postures and parental care activities. Unlike all other species encountered during the survey, N. coriiceps occurred in a dense aggregation of individuals north of Joinville Island, possibly consisting of adults in prespawning condition, as previously reported from nearby areas (Jones et al. 2003). Although burying behaviour is generally uncommon among notothenioids, it was frequently seen during the photographic survey in G. gibberifrons, likely linked to their ambush feeding strategy (Daniels 1982). As previously reported from the Weddell Sea (Ekau and Gutt 1991; La Mesa et al. 2019), C. antarcticus and C. mawsoni both displayed a unique posture, perching on the extremely long pelvic fins or assuming a defensive or S-shaped position of the trunk, respectively.
The adoption of parental care is a reproductive characteristic shared by species of all five notothenioid families occurring in the Southern Ocean (La Mesa et al. 2021). During the survey, two species of channichthyids (C. wilsoni and P. macropterus) were observed guarding eggs laid on glacial dropstones, as previously reported (Kock et al. 2008; Ziegler et al. 2017; La Mesa et al. 2019). Nesting behaviour of T. hansoni and C. rastrospinosus is documented here for the first time. Both species guarded eggs laid on nests built directly on the sea floor on scattered gravel and small stones.
Fish density and diversity were significantly influenced by the mega-epibenthic communities by low growth forms, such as bryozoans and ascidians, as well as large cup-shaped sponges. These organisms provide topographic and trophic complexity, specifically a three-dimensional habitat that is a suitable environment for settling, foraging, breeding and hiding from predators (Gutt and Ekau 1996). Fish density and species richness exhibited negative relationships with echinoderms, suggesting they possibly compete for food resources (Marina et al. 2018). As documented in the Weddell Sea (Gutt and Ekau 1996; La Mesa et al. 2019), sandy or muddy bottoms with poor epibenthic coverage or completely bare were the preferred habitat of several species, such as T. scotti, T. tokarevi, A. orianae, D. longedorsalis and P. evansii. The relatively low correlation between fish density and the epibenthic fauna likely indicates that further parameters play a role in determining species composition and distribution, such as temperature or food availability (Ekau and Gutt 1991).
Fish diversity showed a decreasing trend from stations off Joinville Island and in the Bransfield Strait to those located in the Drake Passage, where we recorded almost exclusively low-Antarctic species. This is primarily due to the relatively high bottom water temperature regime characterizing the Drake Passage (Huneke et al. 2016), and this is an unsuitable habitat for the cold-adapted high-Antarctic species. Hence, the different water masses and their associated temperature regime observed in the studied area greatly influenced the pattern of distribution of demersal notothenioids, limiting the dispersal of low- and high-Antarctic species towards cold or warm water, respectively. In agreement with the general bathymetric distributions of notothenioid fishes (Eastman 2017), species richness and diversity were consistently high within the depth range of 100–600 m, falling steeply at depths greater than 700 m. The canonical analysis of principal coordinates applied to species composition indicated clear separation of ecoregions, habitats and depth strata.
Although all notothenioids are typically stenotherm (Eastman 1993), low-Antarctic and high-Antarctic species have different physiological requirements linked for water temperature and depth. The benthic fish fauna off the northern Antarctic Peninsula is zoogeographically composite and widespread, as evidenced by the distinctly structured spatial partitioning. Notothenioid fish largely predominate, with relatively few other benthic non-notothenioid competitors, such as zoarcids, liparids and gadiforms. Nevertheless, bearing in mind that the sea water warming is more pronounced in the Peninsula region then elsewhere in Antarctic (Moline et al. 2008), in the next future both faunal composition and spatial distribution shared by species of different origin could be altered in some way. Considering the present results as baseline data, the future planning of similar surveys to monitor species composition and relative abundance in faunistic boundary areas like the Bransfield Strait will enable us to assess potential latitudinal or depth distribution changes in advance.
Data availability
Raw count data of fish recorded in seabed images will be available in the PANGAEA repository.
Code availability
Not applicable.
References
Amsler MO, Eastman JT, Smith KE, McClintock JB, Singh H, Thatje S, Aronson RB (2016) Zonation of demersal fishes off Anvers Island, western Antarctic Peninsula. Antarct Sci 28:44–50. https://doi.org/10.1017/S0954102015000462
Anderson MJ, Robinson J (2003) Generalised discriminant analysis based on distances. Aust N Z J Stat 45:301–318. https://doi.org/10.1111/1467-842X.00285
Anderson MJ, Willis TJ (2003) Canonical analysis of principal coordinates: a useful method of constrained ordination for ecology. Ecology 84:511–525. https://doi.org/10.1890/0012-9658(2003)084[0511:CAOPCA]2.0.CO;2
Arana PM, Jones CD, Alegrìa NA, Sarralde R, Rolleri R (2020) Antarctic demersal finfish around the Elephant and the South Orkney islands: distribution, abundance and biological characteristics. Lat Am J Aquat Res 48:304–322. https://doi.org/10.3856/vol48-issue2-fulltext-2469
Balushkin AV, Eakin RR (1998) A new toad plunderfish Pogonophryne fusca sp. nova (Fam. Artedidraconidae: Notothenioidei) with notes on species composition and species groups in the genus Pogonophryne Regan. J Ichthyol 38:574–579. https://doi.org/10.1134/S003294521506003X
Brasier MJ, Barnes D, Bax N, Brandt A, Christianson AB, Constable AJ, Downey R, Figuerola B, Griffiths H, Gutt J, Lockhart S, Morley SA, Post AL, Van de Putte A, Saeedi H, Stark JS, Sumner M, Waller CL (2021) Responses of Southern Ocean seafloor habitats and communities to global and local drivers of change. Front Mar Sci 8:622721. https://doi.org/10.3389/fmars.2021.622721
Brooks C, Chown SL, Douglass LL, Raymond BP, Shaw JD, Sylvester ZT, Torrens CL (2020) Progress towards a representative network of Southern Ocean protected areas. PLoS ONE 15:e0231361. https://doi.org/10.1371/journal.pone.0231361
Clarke A, Murphy EJ, Meredith MP, King JC, Peck LS, Barnes DKA, Smith RC (2007) Climate change and the marine ecosystem of the western Antarctic Peninsula. Philos Trans R Soc Lond B Biol Sci 362:149–166. https://doi.org/10.1098/rstb.2006.1958
Convey P, Peck LS (2019) Antarctic environmental change and biological responses. Sci Adv 11:eaaz0888. https://doi.org/10.1126/sciadv.aaz0888
Daniels RA (1982) Feeding ecology of some fishes of the Antarctic Peninsula. Fish Bull 80:575–588
DeWitt HH, Heemstra PC, Gon O (1990) Nototheniidae. In: Gon O, Heemstra PC (eds) Fishes of the Southern Ocean. JLB Smith Institute of Ichthyology, Grahamstown, pp 279–331
Dorschel B, Gutt J, Huhn O, Bracher A, Huntemann M, Huneke W, Gebhardt C, Schröder M, Herr H (2016) Environmental information for a marine ecosystem research approach for the northern Antarctic Peninsula (RV Polarstern expedition PS81, ANT-XXIX/3). Polar Biol 39:765–787. https://doi.org/10.1007/s00300-015-1861-2
Eastman JT (1993) Antarctic fish biology: evolution in a unique environment. Academic Press, San Diego
Eastman JT (2017) Bathymetric distributions of notothenioid fishes. Polar Biol 40:2077–2095. https://doi.org/10.1007/s00300-017-2128-x
Eastman JT (2020) The buoyancy-based biotope axis of the evolutionary radiation of Antarctic cryonotothenioid fishes. Polar Biol 43:1217–1231. https://doi.org/10.1007/s00300-020-02702-6
Eastman JT, Amsler MO, Aronson RB, Thatje S, McClintock JB, Vos SC, Kaeli JW, Singh H (2013) Photographic survey of benthos provides insights into the Antarctic fish fauna from the Marguerite Bay slope and the Amundsen Sea. Antarct Sci 25:31–43. https://doi.org/10.1017/S0954102012000697
Ekau W, Gutt J (1991) Notothenioid fishes from the Weddell Sea and their habitat, observed by underwater photography and television. Proc NIPR Symp Polar Biol 4:36–49
Espino F, Brito A, Haroun R, Tuya F (2015) Macroecological analysis of the fish fauna inhabiting Cymodocea nodosa seagrass meadows. J Fish Biol 87:1000–1018. https://doi.org/10.1111/jfb.12771
French B, Wilson S, Holmes T, Kendrick A, Rule M, Ryan N (2021) Comparing five methods for quantifying abundance and diversity of fish assemblages in seagrass habitat. Ecol Indic 124:107415. https://doi.org/10.1016/j.ecolind.2021.107415
Gon O, Heemstra PC (1990) Fishes of the Southern Ocean. JLB Smith Institute of Ichthyology, Grahamstown
Gutt J (2013) The expedition of the research vessel “Polarstern” to the Antarctic in 2013 (ANT-XXIX/3). Ber Polarforsch Meeresforsch 665:1–150
Gutt J, Ekau W (1996) Habitat partitioning of dominant high Antarctic demersal fish in the Weddell Sea and Lazarev Sea. J Exp Mar Biol Ecol 206:25–37. https://doi.org/10.1016/0022-0981(95)00186-7
Gutt J, David B, Isla E, Piepenburg D (2016) High environmental variability and steep biological gradients in the waters off the northern Antarctic Peninsula: Polarstern expedition PS81 (ANT-XXIX/3). Polar Biol 39:761–959. https://doi.org/10.1007/s00300-016-1937-7
Gutt J, Arndt J, Kraan C, Dorschel B, Schröder M, Bracher A, Piepenburg D (2019) Benthic communities and their drivers: a spatial analysis off the Antarctic Peninsula. Limnol Oceanogr 62:2341–2357. https://doi.org/10.1002/lno.11187
Gutt J, Isla E, Xavier J, Adams B, et al (2020) Antarctic ecosystems in transition - life between stresses and opportunities. Biol Rev 96:798–821. https://doi.org/10.1111/brv.12679
Hourigan TF, Radtke RL (1989) Reproduction of the Antarctic fish Nototheniops nudifrons. Mar Biol 100:277–283. https://doi.org/10.1007/BF00391969
Huneke WGC, Huhn O, Schröeder M (2016) Water masses in the Bransfield Strait and adjacent seas, austral summer 2013. Polar Biol 39:789–798. https://doi.org/10.1007/s00300-016-1936-8
Iwami T, Kock K-H (1990) Channichthyidae. In: Gon O, Heemstra PC (eds) Fishes of the Southern Ocean. JLB Smith Institute of Ichthyology, Grahamstown, pp 381–399
Jones CD, Kock K-H, Ashford J, DeVries A, Detrich K, Hanchet S, Near T, Turk T, Wilhelms S (2003) Standing stock, biology, diet and spatial distribution of demersal finfish from the 2003 US AMLR bottom trawl survey of the South Shetland Islands (Subarea 48.1). Document WG-FSA-03/388, CCAMLR, Hobart, Australia
Kock K-H, Jones CD (2005) Fish stocks in the southern Scotia Arc region-a review and prospects for future research. Rev Fish Sci 13:75–108. https://doi.org/10.1080/10641260590953900
Kock K-H, Stransky C (2000) The composition of the coastal fish fauna around Elephant Island (South Shetland Islands). Polar Biol 23:825–832. https://doi.org/10.1007/s003000000159
Kock K-H, Jones CD, Wilhelms S (2000) Biological characteristics of Antarctic fish stocks in the southern Scotia Arc region. CCAMLR Sci 7:1–41
Kock K-H, Pshenichnov L, Jones CD, Gröger J, Riehl R (2008) The biology of the spiny ice fish Chaenodraco wilsoni Regan, 1914. Polar Biol 31:381–393. https://doi.org/10.1007/s00300-007-0366-z
La Mesa M, Piepenburg D, Pineda-Metz S, Riginella E, Eastman JT (2019) Spatial distribution and habitat preferences of demersal fish assemblages in the southeastern Weddell Sea (Southern Ocean). Polar Biol 42:1025–1040. https://doi.org/10.1007/s00300-019-02495-3
La Mesa M, Llompart F, Riginella E, Eastman JT (2021) Parental care and reproductive strategies in notothenioid fishes. Fish Fish 22:356–376. https://doi.org/10.1111/faf.12523
Marina TI, Salinas V, Cordone G, Campana G, Moreira E, Deregibus D, Torre L, Sahade R, Tatián M, Barrera Oro E, De Troch M, Doyle S, Quartino ML, Saravia LA, Momo FR (2018) The Food Web of Potter Cove (Antarctica): complexity, structure and function. Estuar Coast Shelf Sci 200:141–151. https://doi.org/10.1016/j.ecss.2017.10.015
Moline MA, Karnovsky NJ, Brown Z, Divoky GJ, Frazer TR, Jacoby CA, Torres JJ, Fraser WR (2008) High latitude changes in ice dynamics and their impact on polar marine ecosystems. Ann N Y Acad Sci 1134:267–319. https://doi.org/10.1196/annals.1439.010
Peck L (2018) Antarctic Marine Biodiversity: adaptations, environments and responses to change. Oceanogr Mar Biol 56:105–236
Piepenburg D, Buschmann A, Driemel A, Grobe H, Gutt J, Schumacher S, Segelken-Voigt A, Sieger R (2017) Seabed images from Southern Ocean shelf regions off the northern Antarctic Peninsula and in the southeastern Weddell Sea. Earth Syst Sci Data 9:461–469. https://doi.org/10.5194/essd-9-461-2017
Rogers AD, Frinault BAV, Barnes DKA, Bindoff NL, Downie R, Ducklow HW, Friedlaender AS, Hart T, Hill SL, Hofmann EE, Linse K, McMahon CR, Murphy EJ, Pakhomov EA, Reygondeau G, Staniland IJ, Wolf-Gladrow DA, Wright RM (2020) Antarctic futures: an assessment of climate-driven changes in ecosystem structure, function, and service provisioning in the Southern Ocean. Annu Rev Mar Sci 12:87–130. https://doi.org/10.1146/annurev-marine-010419-011028
Siegert M, Atkinson A, Banwell A, Brandon M, Convey P, Davies B, Downie R, Edwards T, Hubbard B, Marshall G, Rogelj J, Rumble J, Stroeve J, Vaughan D (2019) The Antarctic Peninsula Under a 1.5°C Global Warming Scenario. Front Environ Sci 7:102. https://doi.org/10.3389/fenvs.2019.00102
Turner J, Barrand NE, Bracegirdle TJ, Convey P, Hodgson D, Jarvis M, Jenkins A, Marshall G, Meredith MP, Roscoe H, Shanklin J, French J, Goosse H, Gutt J, Jacobs S, Kennicutt MC II, Masson-Delmotte V, Mayewski P, Navarro F, Robinson S, Scambos T, Sparrow M, Summerhayes C, Speer K, Klepikov A (2014) Antarctic climate change and the environment: an update. Polar Rec 50:237–259. https://doi.org/10.1017/S0032247413000296
Ziegler AF, Smith CR, Edwards KF, Vernet M (2017) Glacial dropstones: Islands enhancing seafloor species richness of benthic megafauna in West Antarctic Peninsula fjords. Mar Ecol Progr Ser 583:1–14. https://doi.org/10.3354/meps12363
Acknowledgements
We are grateful to the crew and scientific personnel aboard R/V Polarstern during the cruise ANT-XXIX/3 (PS81) off the Antarctic Peninsula for their valuable support during field activities. We are much indebted to Richard Eakin, who greatly contributed to the taxonomic identification of artedidraconids. The sampling of seabed images during the cruise was supported by grants AWI_PS81_03. JT Eastman was supported by National Science Foundation grant NSF ANT 04-36190.
Funding
The study was financially supported by grants AWI_PS81_03 and NSF ANT 04-36190.
Author information
Authors and Affiliations
Contributions
MLM and GLM conceived the study. DP and JG conducted field and lab work. MLM and JE analyzed images, GLM performed data analyses. The first draft of the manuscript was written by MLM, thus all authors contributed to read, edit and approve the final manuscript.
Corresponding author
Ethics declarations
Conflict interest
The authors declare that they have no conflict or competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
La Mesa, M., La Mesa, G., Piepenburg, D. et al. Spatial patterns and behaviour of notothenioid fishes off the northern Antarctic Peninsula. Polar Biol 45, 971–985 (2022). https://doi.org/10.1007/s00300-022-03047-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-022-03047-y