Abstract
Though climate change is widely known to negatively affect the distribution and abundance of many species, few studies have focused on species that may benefit. Gentoo Penguin (Pygoscelis papua) populations have grown along the Western Antarctic Peninsula (WAP), a region accounting for ~ 30% of their global population. These trends of population growth in Gentoo Penguins are in stark contrast to those of Adélie and Chinstrap Penguins, which have experienced considerable population declines along the WAP attributed to environmental changes. The recent discovery of previously unknown Gentoo Penguin colonies along the WAP and evidence for southern range expansion since the last global assessment in 2013 motivates this review of the abundance and distribution of this species. We compiled and collated all available recent data for every known Gentoo Penguin colony in the world and report on previously unknown Gentoo Penguin colonies along the Northwestern section of the WAP. We estimate the global population of Gentoo Penguins to be 432,144 (95th CI 338,059 – 534,114) breeding pairs, with approximately 364,359 (95th CI 324,052 – 405,132) breeding pairs (85% of the population) living in the Atlantic sector. Our estimates suggest that the global population has increased by approximately 11% since 2013, with even greater increases (23%) along the WAP. The Falkland Islands population, which comprises 30% of the global population, has remained stable, though only a subset of colonies have been surveyed since the last comprehensive survey in 2010. Our assessment identifies South Georgia and sub-Antarctic islands in the Indian Ocean as being the most critical data gaps for this species.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Climate change is widely known to affect the distribution and abundance of species, with some taxa experiencing range retractions and extinctions and others experiencing latitudinal shifts in response to warming conditions (Thomas et al. 2006; Hickling et al. 2006; Chen et al. 2011; Pecl et al. 2017). While attention has focused largely on species at risk due to the impacts of climate change, less attention has focused on species that may benefit. One such species is the Gentoo Penguin (Pygoscelis papua), whose populations along the rapidly warming Antarctic Peninsula (Vaughan et al. 2003) have not only undergone population growth since the early 2000s, but have expanded their breeding range southwards with the establishment of many new breeding colonies (Lynch et al. 2013).
These trends stand in stark contrast to their sister species, the Adélie Penguin (P. adeliae) and the Chinstrap Penguin (P. antarcticus), which have experienced considerable population declines on the Western Antarctic Peninsula (WAP), the principle drivers of which are still being debated (Forcada and Trathan 2009; Trivelpiece et al. 2011; Lynch et al. 2012). Current hypotheses for population increases and range expansion of Gentoo Penguins in this region include plasticity in their breeding phenology (Lynch et al. 2011), generalist foraging strategies and a flexible trophic niche (Polito et al. 2015; Herman et al. 2017; McMahon et al. 2019), and breeding habitat preferences (Cimino et al. 2013) vis a vis warming temperatures and declines of sea ice (Stammerjohn et al. 2008) throughout the waters around the WAP.
Gentoo Penguins are one of the most widespread of the penguin species, with a circumpolar breeding distribution and a wide latitudinal range stretching from 46°00′ S in the Crozet Islands (2,350 km SSE of Madagascar) to 65°16′ S on the Antarctic Peninsula (Lynch 2013). Current taxonomy of Gentoo Penguins identifies two subspecies, P. papua papua and P. papua ellsworthi, found north and south of 60°S, respectively (de Dinechin 2012; Clements et al. 2017). However, recent genomic evidence suggests Gentoo Penguins found on South Georgia are more closely related to the southern subspecies and that there is a deep divergence of Indian Ocean populations compared to other Gentoo Penguin colonies in the Atlantic sector, such that taxonomic revisions may be required at the subspecies level (Levy et al. 2016; Vianna et al. 2017; Clucas et al. 2018; Pertierra et al. 2020).
Since the last global assessment of approximately 387,000 breeding pairs (Lynch 2013), many new data have been collected across the Gentoo Penguin’s distributional range. While the WAP population has been generally increasing, these trends are not homogenous, as some colonies have experienced recent declines (Petry et al. 2018; Dunn et al. 2019). In addition, the population in the Falkland Islands, where approximately one third of all Gentoo Penguins nest, increased overall by 105% between 2005 and 2010 (Baylis et al. 2013), though a subset of annually monitored colonies have since declined (Crofts and Stanworth 2019). In contrast, the majority of Indian Ocean colonies (approximately 16% of the global population) are rarely surveyed and represent areas of significant uncertainty for both abundance and trend.
To update our understanding of Gentoo Penguin abundance and distribution, we compiled all census data available to estimate the current global abundance and distribution of breeding Gentoo Penguins including new ground-count survey data from previously unknown colonies along the northwestern section of the WAP and on the Danger Islands. This updated population assessment allows us to identify gaps and associated priorities for future research and forms the basis for our forecasts of continued range expansion along the WAP.
Methods
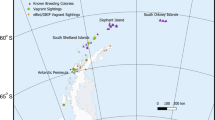
The majority of the Gentoo Penguin distribution falls within the area managed by the Convention for the Conservation of Antarctic Marine Living Resources (CCAMLR), which is divided into three statistical areas: Area 48 (Atlantic Ocean sector), Area 58 (Indian Ocean sector) and Area 88 (Pacific Ocean sector). Gentoo Penguins are found throughout Area 48 (including the Antarctic Peninsula, the South Shetland Islands, the Danger Islands, the South Sandwich Islands, the South Orkney Islands, and South Georgia) and Area 58 (including Marion-Prince Edward Islands, Crozet Island, Heard Island, and Kerguelen Islands) (Fig. 1). Outside the CCAMLR area, Gentoo Penguins have a large breeding population on the Falkland Islands. Smaller populations also exist on Martillo Island in Tierra Del Fuego, Argentina, and Macquarie Island in the southwestern Pacific Ocean.
Global Gentoo Penguin (Pygoscelis papua) breeding distribution (triangles) and CCAMLR statistical sections (bold numbers). Figure created using QGIS Development Team (2018)
Much of the census data from the Antarctic Peninsula and South Shetland Islands are collected via opportunistic vessel-based field surveys (Lynch et al. 2013), though several additional surveys near permanent stations have been published (Petry et al. 2018; Smagol et al. 2018; Dunn et al. 2019). Most surveys were conducted via manual ground counts of individual nests, counting individual nests in panoramic photos taken from the ground or from an offshore vessel, or counting individual penguins from aerial photographs captured by unmanned aerial vehicles (Borowicz et al. 2018). The majority of surveys used in this assessment are precise to within 5% (i.e., they are Accuracy N1 counts, following Croxall and Kirkwood 1979).
The South Sandwich Island census data were either collected via direct ground counts or estimated from the aerial extent of the colony derived from satellite imagery (as described in Lynch et al. 2016). The Falkland Island data are from the most recent island-wide census from Baylis et al. (2013) and from an annual census of a subset of colonies by the Falkland Islands Seabird Monitoring Program (Crofts and Stanworth 2019). Although some sites on South Georgia have been recently surveyed during short landings at popular tourist landing spots, the majority of Gentoo Penguin colonies have not been surveyed recently. The last published comprehensive census data for many South Georgia colonies derive from Trathan et al. (1996), which were based on field data collected between 1985 and 1987 by P.A. Prince and S. Poncet (Unpublished). Many CCAMLR Area 58 populations (Indian Ocean) have not been surveyed comprehensively since the 1980s and 1990s (Weimerskirch et al. 1988; Woehler 1993; Jouventin 1994) with the exception of Marion Island and Prince Edward Island, which were last surveyed in 2008 (Crawford et al. 2009). We include a subset of site-specific census data from Lescroël and Bost (2006) and Weimerskirch (pers. comm.) in Online Resource 1, but chose to use the most recent comprehensive survey data for the Kerguelen and Crozet Island Archipelagos for our global population estimates (Weimerskirch et al. 1988; Jouventin 1994). We also collated the most recent count data for Heard Island (Woehler 1993), Marion Island and Prince Edward Island (Crawford et al. 2009), Macquarie Island (Parks and Wildlife Service 2006), and Martillo Island (Ray et al. 2014).
We used a method for denoting census accuracy used in many other penguin assessments, denoting five levels of accuracy: (1) accurate to better than ± 5%; (2) accurate to 5–10%; (3) accurate to 10–15%; (4) accurate to 25–50%; (5) accurate to the nearest order of magnitude (Croxall and Kirkwood 1979; Naveen et al. 2012; Lynch et al. 2016; Borowicz et al. 2018). We assumed a truncated [0,\(\infty\)) Normal distribution for census errors and propagated observation error to total and regional abundances by drawing from the corresponding Normal distribution (n = 1000) centered on each individual count with a standard deviation appropriate for count accuracy (See Online Resource 1). We grouped sites into regions, and attained population estimates for that region by summing across draws for each site within that region. This allows us to extract a population estimate (the mean of the distribution for the sum) and 95% confidence intervals for the total abundance within that region of interest. Recognizing that our uncertainty on current abundance reflects both the uncertainty of the original survey and the time elapsed since the most recent survey of a colony, we have downgraded (for the purposes of summing up abundances at the regional scale and propagating our uncertainty in those regional abundances) the precision of counts older than 2015 by either one step (e.g., from accuracy = 2 to accuracy = 3) for counts 2005–2014, by two steps for counts 1995–2004, by three steps for counts from 1985–1994, and by four steps for counts prior to 1985, noting that the accuracy code saturates at 5. All analyses were performed in the R computing environment (R Development Core Team 2017).
To explore deglaciation as a possible mechanism for range expansion and colonization in Gentoo Penguins (Cole et al. 2019), we analyzed high-resolution satellite imagery provided through Google Earth for eight of 14 recently colonized areas and compared it with digitized aerial photography taken in December, 1956 by the Falkland Islands and Dependencies Aerial Survey (Mott and Wiggins 1965).
Results
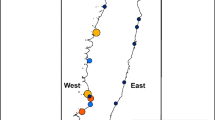
We estimate the global population of Gentoo Penguins to be 432,144 (95th CI 338,059 – 534,114) (Table 1). Colony-specific census data, where available, are detailed in Online Resource 1. Approximately 364,359 (95th CI 324,052 – 405,132) breeding pairs (85% of the population) live in the Atlantic Sector. We report on 14 colonies in the Danger Islands and along the northern coast of Joinville Island unknown at the time of the last global review of Gentoo Penguin abundance and distribution (Lynch 2013; see Online Resource 1 for full details). Including these new and previously unreported populations, the total abundance of Gentoo Penguin breeding pairs on the Antarctic Peninsula is 127,320 (95th CI 118,199 -137,208). In addition to widespread population growth on the WAP, Gentoo Penguins have colonized at least nine new locations since 1994, with six of those locations contributing to a recent southward expansion of this species’ range by approximately 60 km (Fig. 2). One of these newly established colonies is on Green Island, part of the Berthelot Islands, as identified as an area for range expansion by Lynch et al. (2012). There has been rapid population growth at these sites (Fig. 3) and there are currently 1789 breeding pairs in this expansion zone as of 2018. These colonization events have occurred in a cascading southward pattern such that the most recent colonies are at the southernmost end of the species current range.
Locations of all Gentoo Penguin (Pygoscelis papua) breeding colonies on the Antarctic Peninsula (purple circles). Newest colonies formed after 1994 are in orange triangles. Figure created using QGIS Development Team (2018)
Population trends for six new Gentoo Penguin (Pygoscelis papua) breeding colonies of interest (data from MAPPPD; Humphries et al. 2017; see Online Resource 1)
Comparing our assessment of the global Gentoo Penguin breeding population with Lynch (2013), we estimate that the global Gentoo Penguin breeding population has increased by 11% since 2013. The WAP population (excluding the Joinville and Danger Island colonies) estimated to be 116,521 (95th CI 107,692 – 126,331) has increased by approximately 23% since 2013. The current Falkland Islands breeding population estimate is 131,059 pairs (95th CI 129,036 – 133,123). This estimate combines new colony census data and the most recent comprehensive survey in 2010.
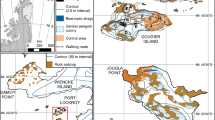
Of the eight newly established colonies in the Danger Islands and Joinville Island region for which historic aerial imagery were available, two sites appear to have recently experienced glacial retreat, exposing land that is now occupied by breeding penguins (Fig. 4).
Glacial retreat at newly colonized Gentoo Penguin (Pygoscelis papua) breeding sites: a Falkland Islands and Dependencies Aerial Survey Expedition (FIDASE) of Moot Point (Mott and Wiggins 1965); b Recent high-resolution satellite imagery taken of Moot Point (2016; Google, CNES/Airbus); c Falkland Islands and Dependencies Aerial Survey Expedition (FIDASE) of Noble Rocks (Mott and Wiggins 1965); d Recent satellite imagery taken of Noble Rocks (2012; Google Earth, Maxar Technologies)
Discussion
Population growth and range expansion on the Antarctic Peninsula
The estimated 11% increase in the total global Gentoo Penguin population – to 432,144 breeding pairs since the last global estimate published in 2013 – can be attributed largely to increases on the Antarctic Peninsula, which has experienced warming air temperatures; increasing precipitation; and declines in the extent, seasonal duration, and thickness of sea ice (Trivelpiece et al. 2011; Turner et al. 2017; Shepherd et al. 2018). These trends are interesting in the context of Gentoo Penguin population growth and range expansion on the Antarctic Peninsula as they are in direct contrast to population trends of Adélie and Chinstrap Penguins in the same region. Since the 1980s, both Adélie and Chinstrap Penguin populations have been declining throughout the Antarctic Peninsula, even as Gentoo Penguin populations have grown steadily. While the drivers of decline for Adélie and Chinstrap Penguins are debated (Forcada and Trathan 2009; Trivelpiece et al. 2011; Lynch et al. 2012) there is considerable evidence that Gentoo Penguin life history provides some relative advantages. In particular, Gentoo Penguins have more flexibility in diet, foraging behavior, and breeding phenology, which suggests they may be more resilient to environmental change and better able to colonize newly exposed territory (Lynch et al. 2011; Masello et al. 2017; McMahon 2019). Moreover, Gentoo Penguins, unlike their congeners, can relay if the brood is lost early during incubation (Bost and Clobert 1992) adding considerable benefit in the ability to buffer population growth from early season events.
In some colonial-nesting bird species, the major contribution to rapid population growth at newly established breeding colonies has been the large-scale immigration of individuals (Oro and Ruxton 2001; Dunlop 2009; Santoro et al. 2016). Given their fast growth rates, it is unlikely that the growth of newly established Gentoo Penguin colonies can be explained by reproductive success and subsequent offspring recruitment alone; continued immigration from other locations over a period of several years would also be required.
Intraspecific competition is a possible explanation for the range expansion exhibited by Gentoo Penguins. A density-dependent decline in the habitat quality of current breeding locations could result in individuals relocating to new, unused habitat and establishing new colonies. Newly available habitat – the result of increased snow melt or glacial retreat (Trathan et al. 2013)–could drive emigration from established colonies or provide habitat to individuals previously unable to recruit into established colonies due to density-dependent processes. It is possible that sites beyond the former southern range margin were previously inaccessible due to heavy winter sea ice conditions, which would preclude foraging in the overwinter period. Declines in the extent or concentration of winter sea ice might make these sites available for breeding and overwinter residency and explain the Gentoo Penguin’s southern range expansion. Historically, deglaciation and terrestrial ice sheet retreat has led to the expansion and proliferation of Antarctic and sub-Antarctic penguin populations since the last glacial maximum (LaRue et al. 2013; Clucas et al. 2014; Younger et al. 2015; Cole et al. 2019). The WAP has continued to experience rapid thinning of its ice shelves and their tributaries since the 1990s, which in part is attributed to increases in upper ocean heat content (Pritchard et al. 2012; Paulo et al. 2015). While Gentoo Penguins are expanding their range further south, they have also colonized two areas that appear to have become available due to very recent deglaciation. Aerial photographs of Noble Rocks in the Neumayer Channel and Moot Point in the Penola Strait, both taken in 1956 (Mott and Wiggins 1965), show these regions as previously covered by terrestrial ice sheets (Fig. 4). For Noble Rocks, the advent of Landsat satellite imagery corresponds to the period in time in which the ice covering this small island began disintegrating, resulting in its full exposure probably early in the 1990s. These sites now have exposed bare rock and are physically separated from the glaciers by ocean and both have been colonized by Gentoo Penguins.
While we do not have data to determine the pace of colonization at these sites, the imagery for Noble Rocks indicates that a colony of 40 nests can establish in as little as 25 years post exposure. Moreover, this colony, situated on a space-limited small rock, seems likely to be at carrying capacity and may be serving as a source population for other bare rock patches in the vicinity. As glacial retreat continues to occur on the Antarctic Peninsula, and perhaps especially on its ice sheet-covered islands whose ice masses are no longer connected to the continental ice sheets, it is likely that expanding Gentoo Penguins may continue to seek out newly exposed areas of the rocky coastline as potential breeding habitat.
Population changes in Indian Ocean Sector
Though the last comprehensive survey of the breeding Gentoo Penguins on the Kerguelen Archipelago was conducted in 1985, the colonies on Courbet Peninsula, Penn Island, and Longue Island, have been regularly monitored since the 1980s. Lescroël and Bost (2006) reported an approximately 30% drop in breeding numbers from the mid-1980s to the mid-2000s on the Courbet Peninsula, which they suggested was the result of inshore food shortages where Gentoo Penguins typically forage (Lescroël and Bost 2005). However, a 2018 survey found that the colony had recovered, surpassing its 1985 breeding population size by 9% (Weimerskirch pers. comm.). In contrast, Possession Island, part of the Crozet Archipelago, was also surveyed in 2018 and found to have declined in breeding population size by 32% (Weimerskirch pers. comm). Similarly, the breeding population at Marion Island has declined by 52% since 1994 (Crawford et al. 2014). With the exception of Courbet Peninsula, these population trends suggest an overall decline among colonies found in the Indian Ocean sector, likely related to declines in food availability due to environmental changes.
Notable data gaps and future work
South Georgia’s last population estimate was in the mid-1980s (P.A. Prince and S. Poncet, Unpublished) and estimated at 98,867 breeding pairs (Trathan et al. 1996), which constitutes over 25% of the last global Gentoo Penguin census. As this population is in close proximity to the Antarctic Peninsula, where populations have increased, and the Falkland Islands, where they appear in decline, it is essential that future efforts be made to fully survey this region. Similarly, the Indian Ocean Gentoo Penguin breeding colonies found on Kerguelen and Crozet Island Archipelagos and those on Heard Island, approximately 14% of the global population, have not been comprehensively surveyed since the early 1970s to late 1980s. Given recent genetic evidence for at least one distinct subspecies in the Indian Ocean (Levy et al. 2016; Vianna et al. 2017; Clucas et al. 2018), updated census data for these regions are particularly urgent.
Monitoring of changes in marine predators is critical to our understanding of marine ecosystem responses to climate change and other environmental perturbations. Predators are sensitive to shifts in prey resources and foraging habitat and can therefore act as proxies for identifying environmental perturbations (Weimerskirch et al. 2003; Sandvick et al. 2005; Cherel et al. 2007; Baylis et al. 2015). While much research is focused on species undergoing declines, few studies focus on species that are robust to – or even benefit from – environmental change (Somero et al. 2010; Fulton et al. 2011; Clucas et al. 2014). Gentoo Penguins are one of the few examples of a marine predator that appears to be adaptive and resilient to environmental change, resulting in continued population growth and range expansion. Future work should focus on the mechanisms of population growth and range expansion in Gentoo Penguins, as well as the creation of habitat suitability models to identify future potential habitat. Updated census data for large populations on South Georgia and the colonies of the sub-Antarctic islands in the Indian Ocean should be considered of high priority.
References
Baylis AM, Crofts S, Wolfaardt AC (2013) Population trends of Gentoo Penguins Pygoscelis papua breeding at the Falkland Islands. Mar Ornithol 41:1–5
Baylis AM, Orben RA, Arnould JP, Christiansen F, Hays GC, Staniland IJ (2015) Disentangling the cause of a catastrophic population decline in a large marine mammal. Ecology 96:2834–2847. https://doi.org/10.1890/14-1948.1
Borowicz A, McDowall P, Youngflesh C, Sayre-McCord T, Clucas G, Herman R, Forrest S, Rider M, Schwaller M, Hart T, Jenouvrier S, Polito MJ, Singh H, Lynch HJ (2018) Multi-modal survey of Adélie Penguin mega-colonies reveals the Danger Islands as a seabird hotspot. Sci Reports 8:1–9. https://doi.org/10.1038/s41598-018-22313-w
Bost CA, Clobert J (1992) Gentoo penguin Pygoscelis papua: Factors affecting the process of laying a replacement clutch. Acta Oecol 13:593–605
Chen IC, Hill JK, Ohlemüller R, Roy DB, Thomas CD (2011) Rapid range shifts of species associated with high levels of climate warming. Science 333:1024–1026. https://doi.org/10.1126/science.1202702
Cherel Y, Hobson KA, Guinet C, Vanpe C (2007) Stable isotopes document seasonal changes in trophic niches and winter foraging individual specialization in diving predators from the Southern Ocean. J Anim Ecol 76:826–836. https://doi.org/10.1111/j.13652656.2007.01238.x
Cimino MA, Fraser WR, Irwin AJ, Oliver MJ (2013) Satellite data identify decadal trends in the quality of Pygoscelis Penguin chick-rearing habitat. Glob Change Biol 19:136–148. https://doi.org/10.1111/gcb.12016
Clements JF, Schulenberg TS, Iliff MJ, Roberson D, Fredericks TA, Sullivan BL, Wood CL (2017) The eBird/Clements checklist of birds of the world: v2016. Retrieved from https://www.birds.cornell.edu/clementschecklist/download/
Clucas GV, Dunn MJ, Dyke G, Emslie SD, Levy H, Naveen R, Polito MJ, Pybus OG, Rogers AD, Hart T (2014) A reversal of fortunes: climate change ‘winners’ and ‘losers’ in Antarctic Peninsula penguins. Sci Reports 4:5024. https://doi.org/10.1038/srep05024
Clucas GV, Younger JL, Kao D, Emmerson L, Southwell C, Wienecke B, Lelliott P (2018) Comparative population genomics reveals key barriers to dispersal in Southern Ocean penguins. Mol Ecol 27:4680–4697. https://doi.org/10.1111/mec.14896
Cole TL, Dutoit L, Dussex N, Hart T, Alexander A, Younger JL, Clucas GV, Frugone MJ, Cherel Y, Cuthbert R, Ellenberg U (2019) Receding ice drove parallel expansions in Southern Ocean penguins. Proc Nat Acad Sci USA 116:26690–26696. https://doi.org/10.1073/pnas.1904048116
Crawford RJ, Whittington PA, Upfold L, Ryan PG, Petersen SL, Dyer BM, Cooper J (2009) Recent trends in numbers of four species of penguins at the Prince Edward Islands. Afr J Mar Sci 31:419–426. https://doi.org/10.2989/AJMS.2009.31.3.14.1002
Crawford RJ, Dyer BM, Upfold L, Makhado AB (2014) Congruent, decreasing trends of gentoo penguins and Crozet shags at sub-Antarctic Marion Island suggest food limitation through common environmental forcing. Afr J Mar Sci 36:225–231. https://doi.org/10.2989/1814232X.2014.926293
Crofts S, Stanworth A (2019) Falkland Islands Seabird Monitoring Programme - Annual Report 2018/2019 (SMP26). Falklands Conservation, Stanley
Croxall JP, Kirkwood ED (1979) The distribution of penguins on the Antarctic Peninsula and islands of the Scotia Sea. British Antarctic Survey, Cambridge, U.K.
de Dinechin M, Dobson FS, Zehtindjiev P, Metcheva R, Couchoux C, Martin A, Quillfeldt P, Jouventin P (2012) The biogeography of Gentoo Penguins (Pygoscelis papua). Can J Zool 90:352–360. https://doi.org/10.1139/z2012-016
Dunlop JN (2009) The population dynamics of tropical seabirds establishing frontier colonies on islands off south-western Australia. Mar Ornithol 37:99–105
Dunn MJ, Forcada J, Jackson JA, Waluda CM, Nichol C, Trathan PN (2019) A long-term study of Gentoo Penguin (Pygoscelis papua) population trends at a major Antarctic tourist site, Goudier Island, Port Lockroy. Biodivers Conserv 28:37–53. https://doi.org/10.1007/s10531-018-1635-6
Forcada J, Trathan PN (2009) Penguin responses to climate change in the Southern Ocean. Glob Change Biol 15:1618–1630. https://doi.org/10.1111/j.1365-2486.2009.01909.x
Fulton EA (2011) Interesting times: winners, losers, and system shifts under climate change around Australia. ICES J Mar Sci 68:1329–1342. https://doi.org/10.1093/icesjms/fsr032
Herman R, Valls FCL, Hart T, Petry MV, Trivelpiece W, Polito MJ (2017) Seasonal consistency and individual variation in foraging strategies differ between and within Pygoscelis Penguin populations in the Antarctic Peninsula region. Mar Biol 164:115. https://doi.org/10.1007/s00227-017-3142-9
Hickling R, Roy DB, Hill JK, Fox R, Thomas CD (2006) The distributions of a wide range of taxonomic groups are expanding polewards. Glob Change Biol 12:450–455. https://doi.org/10.1111/j.1365-2486.2006.01116.x
Humphries GRW, Naveen R, Schwaller M, Che-Castaldo C, McDowall P, Schrimpf M, Lynch HJ (2017) Mapping application for penguin populations and projected dynamics (MAPPPD): data and tools for dynamic management and decision support. Polar Rec 53:160–166. https://doi.org/10.1017/S0032247417000055
Jouventin P (1994) Les populations d’oiseaux marins des T.A.A.F.: Résumé de 20 années de recherché. Aluada 62:44–47
LaRue MA, Ainley DG, Swanson M, Dugger KM, Lyver PO’B, Barton K, Ballard G, (2013) Climate change winners: Receding ice fields facilitate colony expansion and altered dynamics in an Adélie Penguin metapopulation. PLoS ONE 8:e60568. https://doi.org/10.1371/2Fjournal.pone.0060568
Lescroël A, Bost CA (2005) Foraging under contrasting oceanographic conditions: the gentoo penguin at Kerguelen Archipelago. Mar Ecol Prog Ser 302:245–261. https://doi.org/10.3354/meps302245
Lescroël A, Bost CA (2006) Recent decrease in Gentoo Penguin populations at Iles Kerguelen. Antarct Sci 18:171–174. https://doi.org/10.1017/S0954102006000198nt
Levy H, Clucas GV, Rogers AD, Leaché AD, Ciborowski KL, Polito MJ, Lynch HJ, Dunn MJ, Hart T (2016) Population structure and phylogeography of the Gentoo Penguin (Pygoscelis papua) across the Scotia Arc. Ecol Evol 6:1834–1853. https://doi.org/10.1002/2Fece3.1929
Lynch HJ, Fagan WF, Naveen R, Trivelpiece SG, Trivelpiece WZ (2011) Differential advancement of breeding phenology in response to climate may alter staggered breeding among sympatric pygoscelid penguins. Mar Ecol Prog Ser 454:135–145. https://doi.org/10.3354/meps09252
Lynch HJ, Trathan NR (2012) Spatially integrated assessment reveals widespread changes in penguin populations on the Antarctic Peninsula. Ecology 93:1367–1377. https://doi.org/10.1890/11-1588.1
Lynch HJ (2013) Gentoo Penguins (Pygoscelis papua) In: Borboroglu PG, Boersma PD (ed) Penguins: Natural History and Conservation, 1st ed. Seattle: University of Washington Press, pp 75–88
Lynch HJ, Naveen R, Casanovas PV (2013) Antarctic Site Inventory breeding bird survey data 1994/95-2012/13. Ecology 94:2653. https://doi.org/10.1890/13-1108.1
Lynch HJ, White R, Naveen R, Black A, Meixler MS, Fagan WF (2016) In stark contrast to widespread declines along the Scotia Arc, a survey of the South Sandwich Islands finds a robust seabird community. Polar Biol 39:1615–1625. https://doi.org/10.1007/s00300-015-1886-6
Masello JF, Kato A, Sommerfeld J, Mattern T, Quillfeldt P (2017) How animals distribute themselves in space: variable energy landscapes. Front Zool 14(1):33. https://doi.org/10.1186/s12983-017-0219-8
McMahon KW, Michelson CI, Hart T, McCarthy MD, Patterson WP, Polito MJ (2019) Divergent trophic responses of sympatric penguin species to historic anthropogenic exploitation and recent climate change. P Nat Acad Sci USA 116:25721–25727. https://doi.org/10.1073/pnas.1913093116
Mott PG, Wiggins WD (1965) Falkland Islands and Dependencies Aerial Survey Expedition 1955–57. Geogr J 131:430–432
Naveen R, Lynch HJ, Forrest S, Mueller T, Polito M (2012) First direct, site-wide penguin survey at Deception Island, Antarctica, suggests significant declines in breeding Chinstrap Penguins. Polar Biol 35:1879–1888. https://doi.org/10.1007/s00300-012-1230-3
Oro D, Ruxton GD (2001) The formation and growth of seabird colonies: Audouin’s gull as a case study. J Anim Ecol 70:527–535. https://doi.org/10.1046/j.1365-2656.2001.00511.x
Parks and Wildlife Service (2006) Macquarie Island Nature Reserve and World Heritage Area Management Plan. Parks and Wildlife Service, Department of Tourism, Arts, and the Environment, Hobart
Pecl GT, Araújo MB, Bell JD, Blanchard J, Bonebrake TC, Chen IC, Clark TD, Colwell RK, Danielsen F, Evengård B, Falconi L (2017) Biodiversity redistribution under climate change: Impacts on ecosystems and human well-being. Science. https://doi.org/10.1126/science.aai9214
Pertierra LR, Segovia NI, Noll D, Martinez PA, Pliscoff P, Barbosa A, Aragón P, Raya Rey A, Pistorius P, Trathan P, Polanowski A (2020) Cryptic speciation in gentoo penguins is driven by geographic isolation and regional marine conditions: Unforeseen vulnerabilities to global change. Divers Distrib (No volume assigned yet). https://doi.org/10.1111/ddi.13072
Petry MV, Valls FC, Petersen ES, Finger JV, Krüger L (2018) Population trends of seabirds at Stinker Point, Elephant Island, Maritime Antarctica. Antarct Sci 30:220–226. https://doi.org/10.1594/PANGAEA.887844
Pritchard H, Ligtenberg SRM, Fricker HA, Vaughan DG, Van den Broeke MR, Padman L (2012) Antarctic ice-sheet loss driven by basal melting of ice shelves. Nature 484:502–505. https://doi.org/10.1038/nature10968
Paolo FS, Fricker HA, Padman L (2015) Volume loss from Antarctic ice shelves is accelerating. Science 348:327–331. https://doi.org/10.1126/science.aaa0940
Polito MJ, Trivelpiece WZ, Patterson WP, Karnovsky NJ, Reiss CS, Emslie SD (2015) Contrasting specialist and generalist patterns facilitate foraging niche partitioning in sympatric populations of Pygoscelis Penguins. Mar Ecol Prog Ser 519:221–237. https://doi.org/10.3354/meps11095
QGIS Development Team (2018) QGIS Geographic Information System. Open Source Geospatial Foundation Project. https://qgis.osgeo.org.
R Development Core Team (2017) R: a language and environment for statistical computing. R Foundation for Statistical Computing. www.R-project.org/
Rey AR, Rosciano N, Liljesthröm M, Samaniego RS, Schiavini A (2014) Species-specific population trends detected for penguins, gulls and cormorants over 20 years in sub-Antarctic Fuegian Archipelago. Polar Biol 37:1343–1360. https://doi.org/10.1007/s00300-014-15266
Santoro S, Green AJ, Figuerola J (2016) Immigration enhances fast growth of a newly-established source population. Ecology 97:1048–1057. https://doi.org/10.1890/14-2462.1
Sandvik H, Erikstad KE, Barrett RT, Yoccoz NG (2005) The effect of climate on adult survival in five species of North Atlantic seabirds. J Anim Ecol 74:817–831. https://doi.org/10.1111/j.1365-2656.2005.00981.x
Shepherd A, Fricker HA, Farrell SL (2018) Trends and connections across the Antarctic cryosphere. Nature 558:223–232. https://doi.org/10.1038/s41586-018-0171-6
Smagol VM, Dzhulay AO, Dykyy IV, Milinevsky GP, Dykyy EO (2018) Pygoscelis penguin census in the Vernadsky Antarctic station area. Working Paper SC-CAMLR-XXXVII/BG/06 for the Commission for the Conservation of Antarctic Marine Living Resources
Somero GN (2010) The physiology of climate change: how potentials for acclimatization and genetic adaptation will determine ‘winners’ and ‘losers.’ J Exp Biol 213:912–920. https://doi.org/10.1242/jeb.037473
Stammerjohn SE, Martinson DG, Smith RC, Iannuzzi RA (2008) Sea ice in the western Antarctic Peninsula region: Spatio-temporal variability from ecological and climate change perspectives. Deep Sea Res Part II: Top Stud Oceanogr 55:2041–2058. https://doi.org/10.1016/j.dsr2.2008.04.026
Thomas CD, Franco AM, Hill JK (2006) Range retractions and extinction in the face of climate warming. Trends Ecol Evol 21:415–416. https://doi.org/10.1016/j.tree.2006.05.012
Trathan PN, Daunt FJH, Murphy EJ (1996) South Georgia: an ecological atlas. Br Antarct Surv
Trathan PN, Grant SM, Siegel V, Kock KH (2013) Precautionary spatial protection to facilitate the scientific study of habitats and communities under ice shelves in the context of recent, rapid, regional climate change. CCAMLR Science 20:139–151
Trivelpiece WZ, Hinke JT, Miller AK, Reiss CS, Trivelpiece SG, Watters GM (2011) Variability in krill biomass links harvesting and climate warming to penguin population changes in Antarctica. P Nat Acad Sci USA 108:7625–7628. https://doi.org/10.1073/pnas.1016560108
Turner J, Phillips T, Marshall GJ, Hosking JS, Pope JO, Bracegirdle TJ, Deb P (2017) Unprecedented springtime retreat of Antarctic sea ice in 2016. Geophys Res Lett 44:6868–6875. https://doi.org/10.1002/2017GL073656
Vaughan DG, Marshall GJ, Connolley WM, Parkinson C, Mulvaney R, Hodgson DA, King JC, Pudsey CJ, Turner J (2003) Recent rapid regional climate warming on the Antarctic Peninsula. Clim Change 60:243–274. https://doi.org/10.1023/A:1026021217991
Vianna JA, Noll D, Dantas GPM, Virginia Petry M, Barbosa A, González-Acuña D, Le Bohec C, Bonadonna F, Poulin E (2017) Marked phylogeographic structure of Gentoo Penguin reveals an ongoing diversification process along the Southern Ocean. Mol Phylogenet Evol 107:486–498. https://doi.org/10.1016/j.ympev.2016.12.003
Weimerskirch H, Zotier R, Jouventin P (1988) The avifauna of the Kerguelen islands. Emu 89:15–29. https://doi.org/10.1071/MU9890015
Weimerskirch H, Inchausti P, Guinet C, Barbraud C (2003) Trends in bird and seal populations as indicators of a system shift in the Southern Ocean. Antarct Sci 15:249–256. https://doi.org/10.1017/S0954102003001202
Woehler EJ (1993) The distribution and abundance of Antarctic and Subantarctic penguins. Scientific Commission on Antarctic Research, Cambridge, UK, p 2017
Younger J, Emmerson L, Southwell C, Lelliott P, Miller K (2015) Proliferation of East Antarctic Adélie Penguins in response to historical deglaciation. BMC Evol Biol 15:236. https://doi.org/10.1186/s12862-015-0502-2
Acknowledgements
We would like to thank Ron Naveen and IAATO member cruise companies for logistical support of opportunistic field surveys, as well as the Dalio Foundation for funding the Danger Islands Expedition. We would like to thank Alastair Baylis and Henri Weimerskirch for providing information on Gentoo Penguins on the Falkland Islands and several other sub-Antarctic islands. We also thank reviewers Melanie Massaro, Charles-André Bost, and Thomas Mattern for their constructive and insightful feedback to improve this manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Herman, R., Borowicz, A., Lynch, M. et al. Update on the global abundance and distribution of breeding Gentoo Penguins (Pygoscelis papua). Polar Biol 43, 1947–1956 (2020). https://doi.org/10.1007/s00300-020-02759-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00300-020-02759-3