Abstract
Rheumatoid arthritis (RA) is a chronic disease with an enormous impact on patients’ quality of life. The Rheumatoid Arthritis Quality of Life (RAQoL) questionnaire is a disease-specific measure of QoL for individuals with rheumatoid arthritis. Our aim was to adapt and validate the RAQoL for use in Bulgaria. The development of a new language version of the RAQoL consisted of three stages: translation, field testing and psychometric evaluation. The dual-panel methodology, requiring two independent panels of Bulgarian speakers, was applied to translate the UK English version of the RAQoL into Bulgarian. Face and content validity of the translated questionnaire were assessed through cognitive debriefing interviews. Lastly, the RAQoL was administered on two occasions to a random sample of RA patients to evaluate reliability and validity. At the first occasion, the SF-36 was also administered for use as a comparator scale. The RAQoL was successfully adapted into Bulgarian and considered easy to understand, acceptable and comprehensive by RA patients. A psychometric study demonstrated that the new language version has excellent internal consistency (Cronbach’s alpha coefficients = 0.93 and 0.94) and test–retest reliability (a Spearman’s rank correlation coefficient = 0.97). Convergent validity was established by correlating scores on the RAQoL with a comparator measure, the SF-36. A strong correlation between RAQoL scores and the physical functioning section of the SF-36 was observed. Known group validity was established by the ability of the measure to distinguish between subgroups of patients, who differed according to their perceived general health, disease severity (p < 0.001) and whether they were experiencing a flare-up (p < 0.01). The new language version is recommended for use in future research studies, clinical trials and routine practice with Bulgarian RA patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is a chronic progressive disease, characterised by pain, stiffness, swelling, and tenderness of the synovial joints. The condition ultimately results in joint destruction, disability and impaired quality of life (QoL) [1, 2]. RA affects approximately 0.8–1% of the worldwide population, with approximately 29,711 people (0.48%) in Bulgaria living with the condition [3].
The prevalence and incidence rates of RA are increasing globally, with overall occurrence of RA between two and four times greater in women than men [4, 5]. Approximately, one-third of those affected by RA stop working within 2 years of onset of the disease [6]. Since there is no cure for RA, the aim of treatment is to improve quality of life by slowing the progression of the condition and effectively managing the symptoms. It is essential that appropriate instruments are adopted to accurately measure QoL and, in turn, to assess whether new and existing therapies and treatment strategies are of value to the patient [5]. The Rheumatoid Arthritis Quality of Life scale (RAQoL) is a disease-specific patient-reported outcome measure (PROM) that is capable of measuring the value of treatment from the patient’s perspective. The RAQoL is patient-centric, meaning it only assesses relevant issues that patients consider most pertinent to their experience of living with RA. The measure was developed simultaneously in the UK and the Netherlands. It consists of 30 questions with dichotomous yes/no answers, with the total score ranging from 0 to 30 [7, 8].
The content of the RAQoL was derived from in-depth qualitative interviews with RA patients. The interviews explored the impact of RA and its treatment on day to day life and were guided by the interviewee. The conceptual model underlying the RAQoL is the needs-based model of QoL. This postulates that QoL is determined by the ability of an individual to satisfy their basic human needs [9]. QoL is considered poor where few needs of these needs are met. This model has been used in the development of over 30 disease-specific PROMs, including measures for other rheumatic diseases such as ankylosing spondylitis, osteoarthritis, psoriatic arthritis and systemic lupus erythematosus [10,11,12,13].
The RAQoL is available in 37 languages, including the original UK English and Dutch versions. Twenty-seven of these language versions have been fully validated, all of which demonstrate excellent psychometric properties (see for example 14–17). The measure has been included as an end point in numerous clinical trials to assess the value patients gain from pharmaceutical and non-pharmaceutical interventions [18, 19]. The RAQoL has also been adopted in a wide range of correlational and nonexperimental studies [20].
Until now, a Bulgarian version of the RAQoL has not been available for use in routine clinical practice, research studies or international clinical trials. Therefore, the aim of the current study was to adapt and validate the RAQoL questionnaire for use in Bulgaria.
Methodology
The development of a new language version of the RAQoL consisted of three stages: translation, field testing and psychometric evaluation. All procedures performed in the study are in accordance with standards of the Human Research Ethics Committee of the University Clinical Hospital “St. Ivan Rilski”, (approval reference number No.1/03.06.2019).
Translation
The dual-panel methodology recommended by Hunt et al. was used to translate the UK English version of the RAQoL into Bulgarian. This method does not require back-translation [21]. Two independent panels of native Bulgarian speakers were required. First, a bilingual panel was held that consisted of professional individuals (teacher, writer, engineer, lawyer, biologist, transport manager), who were fluent in English. Their role was to work collaboratively to provide an initial translation into Bulgarian, focusing on conceptual rather than linguistic equivalence. A panel of monolingual individuals of a lower education level were then presented with the translated questionnaire. Their task was to evaluate the items and instructions in terms of comprehension and acceptability. The group was asked to comment on whether the translations were expressed in common, everyday language and to amend the phrasing or choice of words, where necessary. The panels were chaired by the same moderator whose role was to ensure that all panel members had the opportunity to express their opinion and to encourage panel members to reach consensus on the most appropriate translations. A representative from the research company that developed the RAQoL was also present to guide the process and ensure that the precise meaning of the items was retained in the translations.
Cognitive debriefing interviews
Semi-structured interviews were conducted to assess the face and content validity of the translated questionnaire. Individuals with RA were asked to take part in a one-to-one, cognitive debriefing interview (CDI). Participants completed the questionnaire in the presence of an interviewer who noted any difficulties or hesitation. The participants were asked to discuss the reason for any observed difficulties after they had completed the questionnaire. Lastly, participants were invited to comment on whether the items were applicable, relevant and comprehensive, and whether any important aspects of their experience were not covered by the questionnaire. Based on the participants’ feedback, modifications could be made to the questionnaire at this stage.
Psychometric evaluation
A psychometric study was conducted in which a questionnaire pack was administered to relevant respondents on two occasions. The purpose of this was to evaluate the psychometric properties (reliability and validity) of the new language version. Patients with RA were selected randomly and invited to participate as they attended their routine appointments in the Clinic of Rheumatology of the University Hospital “St. Ivan Rilski”, Medical University of Sofia. The RAQoL was completed twice, with approximately 14 days between the administrations. At the first administration, participants also completed a comparator questionnaire, the 36-item Short Form Health Survey (SF-36) [22]. The SF-36 is a generic measure of health status and is comprised of eight sections. Relevant demographic and clinical data was also collected. All participants were allocated an ID number to ensure anonymity.
Statistical analysis
Responses to the completed questionnaires were entered into an electronic database and non-parametric statistical analyses were conducted using SPSS (version 21.0). The distributional properties of the Bulgarian RAQoL questionnaire were investigated through descriptive statistics (median and interquartile range [IQR]). Floor and ceiling effects were also assessed.
Internal consistency was assessed by Cronbach’s alpha coefficients. Alpha measures how closely related a set of items are. A low alpha (below 0.7) suggests that the items are not sufficiently interrelated to create a scale [23].
The test–retest reliability of a measure is an estimate of its reproducibility over time under stable conditions. Scores obtained on two separate occasions were correlated using Spearman’s rank correlation coefficient. A minimum value of 0.85 is required to demonstrate that the measure produces low random measurement error [24].
Construct validity was assessed by determining the convergent validity and known-group validity of the new language version. To examine convergent validity, the level of association between scores on one scale and those on a comparator scale that measure the same or a related construct is assessed. In this study, the SF-36 was used as a comparator instrument. In contrast to the RAQoL, a higher score on the SF-36 is more favourable and indicates better health status. Correlations between scores on the RAQoL and SF-36 sections were calculated using Spearman’s rank correlation coefficients [21].
Known-group validity is demonstrated by the ability of a measure to discriminate between groups of individuals that differ according to some known variable. The factors used for investigation in this study were perceived general health (poor/fair/good/excellent) and perceived RA severity (mild/moderate/quite severe/very severe). RAQoL scores were also compared by age group (above median versus below median) to examine whether any differences in QoL were observed between older and younger patients. To test for differences in RAQoL scores between groups, non-parametric statistical tests for independent samples were applied.
Results
Translation
The bilingual panel consisted of three male and three female Bulgarian professionals, aged between 32 and 54 years, who were fluent in English. On the whole, the panel had no major difficulties in providing translations for the items and instructions. The items that evoked discussion were those in which more than one translation was suggested. It was decided that all alternatives would be presented to the lay panel for consideration. For example, the panel informed that there are two words that can be used for ‘activities’ in the item ‘I am unable to join in activities with family or friends’. The bilingual panel agreed that both translations should be retained for the lay panel to decide on the most natural and appropriate expression.
The lay panel consisted of three male and three female participants, aged between 28 and 51 years. The panel members were able to reach a consensus on all items that had more than one translation. For the remaining items, all except two were easily understood and considered to be clear, natural expressions. The lay panel made changes to two items to simplify the phrasing, resulting in an expression that more closely reflected everyday speech.
Cognitive debriefing interviews
CDIs were conducted with eight female and two male RA patients. The age of the sample ranged between 37 to 59 years and most patients were either in full-time or part-time employment. All individuals had a clear understanding of the purpose of the interview and had no issues completing the questionnaire. The mean time taken to complete the RAQoL was 4.4 min (SD = 2.2; range = 1–9 min). All items except one were well received and considered relevant and appropriate. A literal translation was preferred for the item ‘I am unable to join in activities with my friends or family’ and so the translation was changed from ‘I cannot’ to ‘I am unable’. This decision was made to avoid potential issues with responding arising from the inclusion of the negation ‘I cannot’. Overall, patients considered the questionnaire to be easy to understand, comprehensive and reflective of their experience of RA.
Psychometric evaluation
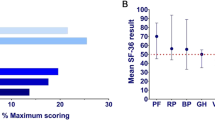
The psychometric study included 51 patients with RA. Demographic and disease information of the sample is presented in Table 1. Descriptive statistics for the RAQoL and SF-36 sections and correlations between RAQoL and SF-36 score are displayed in Table 2. No floor or ceiling effects were observed for the RAQoL, suggesting that the questionnaire is well targeted to RA patients. However, ceiling effects were observed for the physical role limitations, emotional role limitations and social functioning sections of the SF-36—with a quarter of the sample obtaining the maximum possible score on the latter section.
Internal consistency and test–retest reliability
The RAQoL demonstrated high internal consistency at both administrations. Cronbach’s alpha coefficients of 0.93 and 0.94 were obtained at the first and second administration, respectively. A Spearman’s rank correlation coefficient of 0.97 was achieved, indicating high test–retest reliability. This suggests that the questionnaire produces low levels of random measurement error.
Convergent validity
The correlations between scores on the RAQoL and SF-36 sections are shown in Table 2. RAQoL scores were most strongly correlated with the Physical functioning section of the SF-36. Strong correlations were also observed between scores on the RAQoL and the mental health and vitality sections, demonstrating that multiple factors influence QoL in RA.
Known-group validity
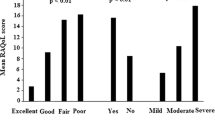
Median RAQoL scores for groups of patients who differed according to their perceived general health, perceived RA severity and presence of a flare-up are illustrated in Fig. 1. Significant differences were observed between patients grouped in this way, such that patients who considered their general health to be “fair/poor” had significantly worse RAQoL scores than patients who rated their health as “excellent/good” (p < 0.001). Patients who rated their disease severity as “quite severe/very severe” also obtained significantly higher scores on the RAQoL (p < 0.001), as did those experiencing a flare-up (p < 0.01).
Descriptive statistics showed that older patients (Mdn = 17; Q1–Q3 = 8.5–21.5) scored higher on the RAQoL than younger patients (Mdn = 10; Q1–Q3 = 5–19). However a Mann–Whitney U test found that this difference was not statistically significant (p = 0.14).
Discussion
The RAQoL was successfully adapted into Bulgarian using the dual-panel approach. This methodology has been adopted in the generation of all official language versions of needs-based QoL measures. The Bulgarian RAQoL was considered to be easy to understand, acceptable and comprehensive by relevant respondents. The study revealed that the Bulgarian RAQoL is a reliable and valid measure. The new language version possesses the appropriate psychometric properties to become a valuable tool in the global RA community for measuring QoL and evaluating treatment strategies in terms of what matters most to patients.
The psychometric properties, including internal consistency, test–retest reliability and construct validity, are in line with the results of previous studies that have validated new language versions of the RAQoL [14,15,16,17]. In the present study, a strong correlation was observed between scores on the RAQoL and the physical functioning section of the SF-36, demonstrating the importance of this factor on QoL in RA. Psychometric studies of the Serbian and Estonian RAQoL both showed similar findings [14, 17], reporting strong correlations between scores on the RAQoL and physical mobility section of the Nottingham Health Profile (NHP; [25]). The presence of ceiling effects for three of the SF-36 sections suggests that the instrument is not well targeted to RA patients, unlike the RAQoL. It is questionable whether the SF-36 should be used in studies of RA patients, considering other psychometric studies have shown the physical role limitations, emotional role limitations and social functioning sections to have large ceiling effects in RA samples [26,27,28]. Known group validity of the Bulgarian RAQoL was established by showing that the new language version was capable of detecting meaningful differences. Comparison of scores by age group revealed that older patients did not report significantly worse QoL compared to younger patients, despite obtaining higher average scores on the RAQoL. Closer inspection of the descriptive statistics showed that there were no marked differences in the variability of scores between the two groups. Therefore, it is not surprising that a statistically significant difference was not found in RAQoL scores. Previous psychometric studies have reported mixed findings on the association between QoL scores on needs-based measures and demographic factors (see for example [29,30,31]). Further research is required to understand what may have contributed to the differences in these results.
The SF-36 is an example of a generic PROM that is still frequently used to measure ‘QoL’ in RA patients. However, researchers still fail to distinguish between Health-Related Quality of Life (HRQL) and needs-based QoL. It is assumed that these outcomes can be used interchangeably, which is problematic considering the different types of outcome are relevant to different audiences. PROMS that measure HRQL predominantly assess symptoms and/or functional limitations and are of primary interest to clinicians, as opposed to the patient.
Therefore, complementary outcome measures are required to assess different types of outcome. The RAQoL is the only available tool for patient-centric QoL measurement in RA. Since the measure was derived from qualitative interviews with RA patients, all items are relevant to respondents. It has been well established that generic PROMs are not as comprehensive and relevant as disease-specific PROMs [32]. Research has also demonstrated that generic PROMs have inferior psychometric properties and are less sensitive to change compared to disease-specific PROMs [33,34,35,36]. A review detailing the use of RAQoL revealed that the measure has been shown to be capable of detecting the impact of clinical and non-clinical interventions [20].
Although the psychometric study sample was representative of the gender differences in the RA population, it was not appropriate to compare RAQoL scores by gender. A larger sample size is required to investigate whether significant differences in QoL between males and females are observed in a Bulgarian RA sample. Another limitation of the current study is that it was not possible to assess the responsiveness of the Bulgarian RAQoL. This is because an intervention study is required to evaluate if the new language version is capable of detecting change over time. Outcome measures should be sensitive to change if they are to be included in clinical trials. It is expected that the RAQoL would be capable of detecting true changes in QoL, given its excellent psychometric properties.
Conclusions
The current study demonstrated that the Bulgarian RAQoL is a valid and reliable measure of QoL in RA patients. The new language version can be used as an outcome measure in international research studies and clinical trials. The RAQoL can also be applied in routine clinical practice to monitor QoL of RA patients in Bulgaria.
References
Taylor PC, Moore A, Vasilescu R, Alvir J, Tarallo M (2016) A structured literature review of the burden of illness and unmet needs in patients with rheumatoid arthritis: a current perspective. Rheumatol Int 36:685–695. https://doi.org/10.1007/s00296-015-3415-x
Studenic P, Smolen JS, Aletaha D (2012) Near misses of ACR/EULAR criteria for remission: effects of patient global assessment in Boolean and index-based definitions. Ann Rheum Dis 71:1702–1705. https://doi.org/10.1136/annrheumdis-2012-201519
Kobelt, G., and Kasteng, F. (2009). Access to innovative treatments in rheumatoid arthritis in Europe, EFPIA report. Available online at: http://www.comparatorreports.se/Access%20to%20RA%20Treatments%20October%202009.pdf. Accessed 5 Aug 2020
Safiri S, Kolahi AA, Hoy D, Smith E, Bettampadi D, Mansournia MA, Almasi- Hashiani A, Ashrafi-Asgarabad A, Moradi-Lakeh M, Qorbani M, Collins G, Woolf AD, March L, Cross M (2019) Global, regional and national burden of rheumatoid arthritis 1990–2017: a systematic analysis of the Global Burden of Disease study 2017. Ann Rheum Dis 78:1463–1471. https://doi.org/10.1136/annrheumdis-2019-215920
Van der Woude D, van der Helm-van Mil AHM (2018) Update on the epidemiology, risk factors, and disease outcomes of rheumatoid arthritis. Best Pract Res Cl Rh 32:174–187. https://doi.org/10.1016/j.berh.2018.10.005
Pollard L, Choy EH, Scott DL (2005) The consequences of rheumatoid arthritis: quality of life measures in the individual patient. Clin Exp Rheumatol 23:S43–S52. www.clinexprheumatol.org/article.asp?a=2685
Whalley D, McKenna S, De Jong Z, Van der Heijde D (1997) Quality of life in rheumatoid arthritis. Rheumatology 36:884–888. https://doi.org/10.1093/rheumatology/36.8.884
De Jong Z, Van der Heijde DM, McKenna SP, Whalley D (1997) The reliability and construct validity of the RAQoL: a rheumatoid arthritis-specific quality of life instrument. Br J Rheumatol 36:878–883. https://doi.org/10.1093/rheumatology/36.8.878
Hunt SM, McKenna SP (1992) The QLDS: a scale for the measurement of quality of life in depression. Health Policy 22:307–319. https://doi.org/10.1016/0168-8510(92)90004-U
Doward L, Spoorenberg A, Cook S, Whalley D, Helliwell P, Kay L, Chamberlain M (2003) Development of the ASQoL: a quality of life instrument specific to ankylosing spondylitis. Ann Rheum Dis 62:20–26. https://doi.org/10.1136/ard.62.1.20
Keenan A, Mckenna S, Doward L, Conaghan P, Emery P, Tennant A (2008) Development and validation of a needs-based quality of life instrument for osteoarthritis. Arthritis Care Res 59:841–848. https://doi.org/10.1002/art.23714
McKenna S, Doward L, Whalley D, Tennant A, Emery P, Veale D (2004) Development of the PsAQoL: a quality of life instrument specific to psoriatic arthritis. Ann Rheum Dis 63:162–169. https://doi.org/10.1136/ard.2003.006296
Doward L, McKenna S, Whalley D, Tennant A, Griffiths B, Emery P, Veale D (2009) The development of the L-QoL: a quality-of-life instrument specific to systemic lupus erythematosus. Ann Rheum Dis 68:196–200. https://doi.org/10.1136/ard.2007.086009
Tammaru M, McKenna S, Meads D, Maimets K, Hansen E (2006) Adaptation of the rheumatoid arthritis quality of life scale for Estonia. Rheumatol Int 26:655–662. https://doi.org/10.1007/s00296-005-0038-7
Hedin P, McKenna S, Meads D (2006) The rheumatoid arthritis quality of life (RAQoL) for Sweden: adaptation and validation. Scand J Rheumatol 35:117–123. https://doi.org/10.1080/03009740500311770
Wilburn J, McKenna SP, Twiss J, Rouse M, Korkosz M, Jancovic R, Nemec P, Pacheco-Tena CF, Saraux A, Westhovens R, Durez P, Martin M, Tammaru M (2015) Further international adaptation and validation of the rheumatoid arthritis quality of life (RAQoL) questionnaire. Rheumatol Int 35:669–675. https://doi.org/10.1007/s00296-014-3138-4
Zlatkovic-Svenda M, Rouse M, Radak-Perovic M, Stojanovic R, Vujasinovic-Stupar N, Lazovic-Popovic B, Wilburn J, McKenna SP (2017) Adaptation and validation of the rheumatoid arthritis quality of life (RAQoL) questionnaire for use in Serbia. Rheumatol Int 37:641–646. https://doi.org/10.1007/s00296-016-3586-0
Huizinga TW, Conaghan PG, Martin-Mola E, Schett G, Amital H, Xavier R, Troum O, Aassi M, Bernasconi C, Dougados M (2015) Clinical and radiographic outcomes at 2 years and the effect of tocilizumab discontinuation following sustained remission in the second and third year of the ACT-RAY study. Ann Rheum Dis 74:35–43. https://doi.org/10.1136/annrheumdis-2014-205752
Adly AS, Adly AS, Adly MS, Serry ZM (2017) Laser acupuncture versus reflexology therapy in elderly with rheumatoid arthritis. Lasers Med Sci 32:1097–1103. https://doi.org/10.1007/s10103-017-2213-y
Heaney A, Stepanous J, Rouse M, McKenna SP (2017) A review of the psychometric properties and use of the rheumatoid arthritis quality of life questionnaire (RAQoL) in clinical research. Curr Rheumatol Rev 13:197–205. https://doi.org/10.2174/1573396313666170615092324
Hunt SM, Alonso J, Bucquet D, Niero M, Wiklund I, McKenna S (1991) Cross-cultural adaptation of health measures. Health Policy 19:33–44. https://doi.org/10.1016/0168-8510(91)90072-6
Ware JE Jr (2000) SF-36 health survey update. Spine 25:3130–3139. https://doi.org/10.1097/00007632-200012150-00008
Bland JM, Altman DG (1997) Statistics notes: Cronbach’s alpha. Br Med J 314:572. https://doi.org/10.1136/bmj.314.7080.572
Weiner EA, Stewart BJ (1984) Assessing individuals. Little Brown, Boston
Hunt SM, McKenna SP, McEwen J, Williams J, Papp E (1981) The Nottingham health profile: subjective health status and medical consultations. Soc Sci Med A 15:221–229. https://doi.org/10.1016/0271-7123(81)90005-5
Linde L, Sorensen J, Ostergaard M,Horslev-Petersen K, Hetland ML (2008) Health- related quality of life: validity reliability, and responsiveness of SF-36, EQ-15D, EQ-5D, RAQoL, and HAQ in patients with rheumatoid arthritis. J Rheumatol 35:1528–1537. www.jrheum.org/content/35/8/1528.short
ten Klooster PM, Vonkeman HE, Taal E, Siemons L, Hendriks L, de Jong AJ, Dutmer EA, van Riel PL, van de Laar MA (2013) Performance of the Dutch SF-36 version 2 as a measure of health-related quality of life in patients with rheumatoid arthritis. Health Qual Life Outcomes 11:77. https://doi.org/10.1186/1477-7525-11-77
Koh ET, Leong KP, Tsou IY, Lim VH, Pong LY, Chong SY, Seow A (2006) The reliability, validity and sensitivity to change of the Chinese version of SF-36 in oriental patients with rheumatoid arthritis. Rheumatology 45:1023–1028. https://doi.org/10.1093/rheumatology/kel051
Heaney Α, Stepanous J, McKenna SP, Athanassiou P, Vidalis AA (2017) Translation and validation of the Greek psoriatic arthritis quality of life scale. Psychiatriki 28:219–225. https://doi.org/10.22365/jpsych.2017.283.219
Wilburn J, McKenna SP, Kutlay Ş, Bender T, Braun J, Castillo-Gallego C, Favero M, Geher P, Kiltz U, Martin-Mola E, Ramonda R (2017) Adaptation of the osteoarthritis-specific quality of life scale (the OAQoL) for use in Germany, Hungary, Italy, Spain and Turkey. Rheumatol Int 37:727–734. https://doi.org/10.1007/s00296-017-3664-y
Gomes JL, Águeda AF, Heaney A, Duarte C, Lopes C, Costa T, Marona J, Rodrigues- Manica S, Maia S, Costa M, Branco JC (2019) Translation, cross-cultural adaptation and validation of the Osteoarthritis Quality of Life (OAQoL) questionnaire for use in Portugal. Rheumatol Int 39:715–722. https://doi.org/10.1007/s00296-018-4197-8
McKenna SP (2011) Measuring patient-reported outcomes: moving beyond misplaced common sense to hard science. BMC Med 9:86. https://doi.org/10.1186/1741-7015-9-86
Taylor WJ, McPherson KM (2007) Using Rasch analysis to compare the psychometric properties of the short form 36 physical function score and the health assessment questionnaire disability index in patients with psoriatic arthritis and rheumatoid arthritis. Arthritis Care Res 57:723–772. https://doi.org/10.1002/art.22770
Twiss J, Meads DM, Preston EP, Crawford SR, McKenna SP (2012) Can we rely on the dermatology life quality index as a measure of the impact of psoriasis or atopic dermatitis? J Investig Dermatol 132:76–84. https://doi.org/10.1038/jid.2011.238
Angst F, Verra ML, Lehmann S, Aeschlimann A (2008) Responsiveness of five condition-specific and generic outcome assessment instruments for chronic pain. BMC Med Res Methodol 8:26. https://doi.org/10.1186/1471-2288-8-26
Jull A, Parag V, Walker N, Rodgers A (2010) Responsiveness of generic and disease-specific health-related quality of life instruments to venous ulcer healing. Wound Rep Reg 18:26–30. https://doi.org/10.1111/j.1524-475X.2009.00556.x
Acknowledgements
The authors would like to thank all participants in the bilingual and lay translation panels and all the patients for their participation.
Funding
This work is supported by the Bulgarian Ministry of Education and Science under the National Program for Research “Young Scientists and Postdoctoral Students”.
Author information
Authors and Affiliations
Contributions
Study concept and design: SPM and AH. Data collection: DTP and SVM. Statistical analysis and data interpretation: SPM and AH. DTP prepared the first version of the manuscript and all authors contributed to critical revision of the manuscript. All authors read and approved the final version.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Ethics approval
All procedures performed in the study are in accordance with the standards of the Human Research Ethics Committee of the University Clinical Hospital “St. Ivan Rilski” (approval reference number No. 1/03.06.2019) and the 1964 Helsinki Declaration and its later amendments.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Pencheva, D.T., Heaney, A., McKenna, S.P. et al. Adaptation and validation of the Rheumatoid Arthritis Quality of Life (RAQoL) questionnaire for use in Bulgaria. Rheumatol Int 40, 2077–2083 (2020). https://doi.org/10.1007/s00296-020-04686-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-020-04686-2