Abstract
Rheumatoid arthritis (RA) is one of the most prevalent inflammatory rheumatic diseases. As it is a chronic and a lifelong destructive disease, the aim of the treatment is to reduce disability and improve quality of life. The Rheumatoid Arthritis Quality of Life (RAQoL) questionnaire is a patient-reported outcome measure, specific to RA. To adapt and validate the RAQoL for use in Serbia, two translation panels were involved to produce the Serbian RAQoL. After successful translation, face and content validity was determined via cognitive debriefing interviews. The psychometric properties of the questionnaire were examined, including reliability and construct validity, by using the Nottingham Health Profile (NHP) as a comparator scale. The RAQoL was translated successfully and rated as applicable, relevant and comprehensive by respondents. The questionnaire had high internal consistency (alpha = 0.94 at both time points) and test–retest reliability (r = 0.92). Moderately high correlations were found between the RAQoL and physical mobility, pain and energy level sections of the NHP, providing evidence of convergent validity. The RAQoL was able to distinguish between patients grouped by perceived general health, incidence of flare-up and disease severity. The Serbian language version of the RAQoL showed strong evidence of reliability and validity and is recommended for use in clinical trials and routine general practice in RA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is a chronic, inflammatory autoimmune disease. Common symptoms include pain, swelling and stiffness in the joints. As a consequence, RA has a detrimental effect on many aspects of life such as mood, hobbies, social life and relationships [1]. The prevalence of RA varies throughout the world. Southern European countries have a marginally lower incidence than Northern European and North American countries [2, 3]. The estimated prevalence of RA in the Serbian population is 0.34%, and the female-to-male ratio is approximately 3:1 [3]. There is no cure for RA, and the main goal of treatment is to reduce the impact of this destructive disease by reducing disability and improving quality of life (QoL) [4].
It is now well established that, in addition to clinical outcomes, the value of interventions from the patient’s perspective should be measured. This requires the use of patient-centric outcome measures—that is, measures that determine impact on those issues that they consider to be important. The Rheumatoid Arthritis Quality of Life (RAQoL) questionnaire is a disease-specific QoL measure that was developed simultaneously in the UK and the Netherlands [1]. The content of the RAQoL was derived from in-depth qualitative interviews with RA patients exploring the impact of RA on their QoL. Unlike other measures, the RAQoL is based on a clear theoretical construct, the needs-based QoL model. This model postulates that QoL is determined by the ability of an individual to satisfy his or her needs. QoL is high when needs are fulfilled and poor when they are not [5]. The model is well established and has been used as the theoretical basis in the development of 30 disease-specific patient-reported outcome (PROs), including measures for rheumatic diseases such as ankylosing spondylitis, osteoarthritis, psoriatic arthritis and systemic lupus erythematosus [6–9]. Since its development, the RAQoL has demonstrated excellent psychometric properties, exhibiting high internal consistency, reproducibility and construct validity across 37 different languages (see for example [10–13]). RAQoL items were generated from the patient’s own words, ensuring good face and content validity. The measure has been used frequently as an endpoint in clinical trials with both pharmaceutical and non-pharmaceutical interventions [14–16].
The aim of the current study was to adapt and validate a Serbian version of the RAQoL for use in international clinical trials and routine clinical practice.
Methods
The RAQoL adaptation involved three stages: translation, cognitive debriefing interviews to assess face and content validity, and psychometric evaluation. This study was approved by the local ethical committee at the Rheumatology Institute, Belgrade.
Translation
The adaptation used the dual-panel translation methodology, which is recommended for translation of needs-based measures [17]. First, a translation of the RAQoL into Serbian was produced by a team of bilingual professionals fluent in English. The translated questionnaire was then presented to a lay panel consisting of monolingual Serbian individuals of an average to low educational level more representative of the general population. The purpose of the second panel was to ensure that the items and instructions were clear and likely to be understood by future respondents and that ‘natural’ language was used.
Face and content validity
Cognitive debriefing interviews were conducted with RA patients to assess the applicability, relevance and comprehensiveness of the questionnaire. Questionnaires were completed in the presence of an experienced rheumatologist, who made note of any obvious difficulties arising during completion. Patients were asked whether they found the items relevant, applicable, comprehensible and whether they believed any important aspects of their experience had been omitted from the questionnaire.
Psychometric evaluation
Patients were selected randomly, as they came to the Institute of Rheumatology in Belgrade for a routine examination. The RAQoL was completed on two occasions, with approximately two weeks between administrations, at the same location. At the same time, participants also completed the Nottingham Health Profile—NHP [18]—which was used as a comparator scale, as well as demographic questionnaire, also comprising perceived general health and the perceived severity of the RA disease questions. The NHP has already been adapted for use in Serbia. Internal consistency was assessed using Cronbach’s alpha coefficient. Alpha measures the extent to which the items in a scale are inter-related. A low alpha (below 0.7) indicates that the items do not work together to form a scale [19].
The test–retest reliability of a measure is an estimate of its reproducibility over time when no change in condition has taken place. It was assessed by correlating RAQoL scores at Times 1 and 2. A minimum value of 0.85 is required to demonstrate low levels of random measurement error [20].
Convergent validity and known-group validity were assessed to determine construct validity. Convergent validity was evaluated by determining the level of association between RAQoL and NHP section scores using Spearman’s rank correlation coefficients. Known-group validity was investigated by testing the ability of the RAQoL to distinguish between groups of patients who differed according to known factors. The factors used for the current study were perceived general health (very good, good, fair or poor), incidence of flare-up (yes or no) and perceived disease severity (mild, moderate or severe) at the time of examination. Nonparametric tests for independent samples (Mann–Whitney U test for two groups or Kruskal–Wallis one-way analysis of variance for three or more groups) were used.
Results
Translation
The bilingual panel consisted of one male and five female Serbian professionals fluent in English. Their ages ranged from 25 to 35 years. The panel had only minor difficulties translating the items and instructions. One item required extended discussion: ‘I often get frustrated.’ Two translations were produced by the panel which translated back into English as ‘I am often irritated’ and ‘I often get nervous.’ Both versions were sent for consideration by the lay panel. The lay panel consisted of two males and three females aged between 28 and 65 years. Regarding the item with two suggested versions, the lay panel decided to create a new version, considered to be more elegant and closer to everyday speech. This had the meaning ‘I often feel irritated.’ Two other items were also modified slightly to produce a more literal translation. Overall, the lay panel agreed with most of the translations provided by the bilingual panel.
Face and content validity
Demographic details for the cognitive debriefing interviews are shown in Table 1. The RAQoL took between 3 and 10 min to complete (mean time = 4.6, SD = 2.1). All interviewees understood clearly the purpose of the interview and the questionnaire instructions. There was no particular item that stood out as being awkwardly worded or difficult to understand. No items were thought to be inappropriate or unacceptable. However, one item (‘I’m unable to join in activities with my family or friends’) was changed in order to avoid a double negative. Overall, the patients found the measure clear and easy to understand.
Psychometric evaluation
Fifty-eight patients completed the RAQoL at both time points. Table 1 shows demographic and disease information for the validation survey participants.
Internal reliability and test–retest reliability
Cronbach’s alpha coefficient was 0.94 at both time points, indicating high levels of internal consistency. Test–retest reliability was also high (r = 0.92), demonstrating that the Serbian RAQoL produces low levels of random measurement error.
Construct validity
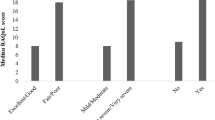
Table 2 shows correlations between RAQoL scores and those on the NHP sections at both time points. High correlations were observed between the RAQoL and physical mobility, pain and energy sections of the NHP, highlighting the importance of these factors for RA patients. Figure 1 shows mean RAQoL scores by perceived general health, incidence of flare-up and perceived disease severity. Significant differences in RAQoL scores were found between patients grouped by these factors, demonstrating the ability of the RAQoL to distinguish between subgroups of patients.
Discussion
This adaptation of the RAQoL into Serbian was successful. The adaptation used the dual-panel translation methodology, recommended for the translation of PROs [17]. Cognitive debriefing interviews confirmed that the Serbian language version was acceptable, relevant and easy to complete.
Sample size for the psychometric properties was found to be adequate enough to establish validity, according to our previous experience. The Serbian RAQoL demonstrated excellent psychometric properties, with high levels of internal consistency, test–retest reliability and construct validity. Moderately high correlations were observed between RAQoL scores and the physical mobility, pain and energy sections of the NHP. These findings mirror those found in the original RAQoL development [1].
Generic PROs such as the SF-36 [21] are often used when measuring QoL in RA. Generic PROs generally have poor psychometric properties [22, 23], and head-to-head comparison studies have shown they are less sensitive to change than disease-specific measures [24, 25]. Furthermore, generic measures contain items that are not well targeted to respondents. This can alienate patients and result in high levels of missing data [26]. A good example relates to difficulties related to physical contact experienced by RA patients. Items in the RAQoL refer to being in crowds, shaking hands and participating in intimate situations. This aspect of RA is not covered by the SF-36 [1]. The RAQoL is a disease-specific PRO and its content was derived from qualitative interviews with RA patients, meaning that all items are directly relevant to the patients concerned.
Given the high psychometric quality of the Serbian RAQoL, its relevance to respondents and the fact that the other RAQoL language versions have been shown to demonstrate differences in impact of alternative interventions, it is recommended that it is used instead of the SF-36 in future studies. It is important to note that different patient-reported measures may assess different types of outcome. For example, the Health Assessment Questionnaire [27] is also frequently included in clinical trials. Rather than assessing QoL, the HAQ is a high-quality pseudo-clinical measure of physical functioning. Consequently, the RAQoL and HAQ can be used as complementary outcome measures.
Limitations of the study should be noted. The scaling properties of the measure could not be assessed. This is because Rasch analysis, a more stringent test of a scale’s properties, requires a larger sample size than was available [28]. Furthermore, because this study did not include an intervention, it was not possible to assess the responsiveness of the Serbian RAQoL. Responsiveness is an important quality for all outcome measures included in clinical trials.
Conclusion
The Serbian RAQoL is a valid and reliable tool for measuring QoL in RA patients. Therefore, it is recommended for use in clinical practice in the future, as well as in research and international clinical trials.
References
Whalley D, McKenna S, De Jong Z, Van der Heijde D (1997) Quality of life in rheumatoid arthritis. Rheumatology 36(8):884–888
Alamanos Y, Voulgari P, Drosos A (2006) Incidence and prevalence of rheumatoid arthritis, based on the 1987 American College of Rheumatology criteria: a systematic review. Semin Arthritis Rheum 36(3):182–188
Zlatkovic-Svenda M, Stojanovic R, Sipetic-Grujicic S, Guillemin F (2014) Prevalence of rheumatoid arthritis in Serbia. Rheumatol Int 34(5):649–658
Pollard L, Choy E, Scott D (2005) The consequences of rheumatoid arthritis: quality of life measures in the individual patient. Clin Exp Rheumatol 23(5):S43
Hunt SM, McKenna SP (1992) The QLDS: a scale for the measurement of quality of life in depression. Health Policy 22(3):307–319
Doward L, Spoorenberg A, Cook S, Whalley D, Helliwell P, Kay L, Chamberlain M (2003) Development of the ASQoL: a quality of life instrument specific to ankylosing spondylitis. Ann Rheum Dis 62(1):20–26
Keenan A, Mckenna S, Doward L, Conaghan P, Emery P, Tennant A (2008) Development and validation of a needs-based quality of life instrument for osteoarthritis. Arthritis Care Res 59(6):841–848
McKenna S, Doward L, Whalley D, Tennant A, Emery P, Veale D (2004) Development of the PsAQoL: a quality of life instrument specific to psoriatic arthritis. Ann Rheum Dis 63(2):162–169
Doward L, McKenna S, Whalley D, Tennant A, Griffiths B, Emery P, Veale D (2009) The development of the L-QoL: a quality-of-life instrument specific to systemic lupus erythematosus. Ann Rheum Dis 68(2):196–200
Neville C, Whalley D, McKenna S, Le Comte M, Fortin P (2001) Adaptation and validation of the rheumatoid arthritis quality of life scale for use in Canada. J Rheumatol 28(7):1505–1510
Tammaru M, McKenna S, Meads D, Maimets K, Hansen E (2006) Adaptation of the rheumatoid arthritis quality of life scale for Estonia. Rheumatol Int 26(7):655–662
Hedin P, McKenna S, Meads D (2006) The rheumatoid arthritis quality of life (RAQoL) for Sweden: adaptation and validation. Scand J Rheumatol 35(2):117–123
Wilburn J, McKenna S, Twiss J, Rouse M, Korkosz M, Jancovic R, Durez P (2015) Further international adaptation and validation of the Rheumatoid Arthritis Quality of Life (RAQoL) questionnaire. Rheumatol Int 35(4):669–675
Bejarano V, Quinn M, Conaghan P, Reece R, Keenan A, Walker D, Jarrett S (2008) Effect of the early use of the anti–tumor necrosis factor adalimumab on the prevention of job loss in patients with early rheumatoid arthritis. Arthritis Care Res 59(10):1467–1474
Herenius M, Hoving J, Sluiter J, Raterman H, Lems W, Dijkmans B, Frings-Dresen M (2010) Improvement of work ability, quality of life, and fatigue in patients with rheumatoid arthritis treated with adalimumab. J Occup Environ Med 52(6):618–621
Willich S, Rossnagel K, Roll S, Wagner A, Mune O, Erlendson J, Winther K (2010) Rose hip herbal remedy in patients with rheumatoid arthritis—a randomised controlled trial. Phytomedicine 17(2):87–93
Hunt SM, Alonso J, Bucquet D, Niero M, Wiklund I, McKenna S (1991) Cross-cultural adaptation of health measures. Health Policy 19(1):33–44
Hunt SM, McKenna SP, McEwen J, Williams J, Papp E (1981) The Nottingham Health Profile: subjective health status and medical consultations. Soc Sci Med Part A Med Psychol Med Sociol 15(3):221–229
Streiner DL, Norman GR (1995) Health measurement scales: a practical guide to their development and use, 2nd edn. Oxford University Press, New York
Weiner EA, Stewart BJ (1984) Assessing individuals. Little Brown, Boston
Ware JE Jr, Sherbourne CD (1992) The MOS 36-item short-form health survey (SF-36): I. Conceptual framework and item selection. Med Care 30(6):473–483
Taylor WJ, McPherson KM (2007) Using Rasch analysis to compare the psychometric properties of the Short Form 36 physical function score and the Health Assessment Questionnaire disability index in patients with psoriatic arthritis and rheumatoid arthritis. Arthritis Care Res 57(5):723–729
Twiss J, Meads DM, Preston EP, Crawford SR, McKenna SP (2012) Can we rely on the Dermatology Life Quality Index as a measure of the impact of psoriasis or atopic dermatitis? J Investig Dermatol 132(1):76–84
Angst F, Verra ML, Lehmann S, Aeschlimann A (2008) Responsiveness of five condition-specific and generic outcome assessment instruments for chronic pain. BMC Med Res Methodol 8(1):1
Jull A, Parag V, Walker N, Rodgers A (2010) Responsiveness of generic and disease-specific health-related quality of life instruments to venous ulcer healing. Wound repair and regeneration 18(1):26–30
McKenna SP (2011) Measuring patient-reported outcomes: moving beyond misplaced common sense to hard science. BMC Med 9(1):1
Fries JF (1991) The hierarchy of quality-of-life assessment, the Health Assessment Questionnaire (HAQ), and issues mandating development of a toxicity index. Control Clin Trials 12(4 Suppl):106S–117S
Linacre JM (1994) Sample size and item calibration stability. Rasch Meas Trans 7(4):328
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that there is no conflict of interest.
Human and animal rights statement
All procedures performed in the study are in accordance with the ethical standards of the national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Zlatkovic-Svenda, M., Rouse, M., Radak-Perovic, M. et al. Adaptation and validation of the Rheumatoid Arthritis Quality of Life (RAQoL) questionnaire for use in Serbia. Rheumatol Int 37, 641–646 (2017). https://doi.org/10.1007/s00296-016-3586-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-016-3586-0