Abstract
We examined the functional activity of peripheral blood neutrophils and the complement system activation status in patients with rheumatoid arthritis (RA) undergoing infliximab/methotrexate combined therapy. We studied female RA patients under treatment with infliximab (3–5 mg/kg) and methotrexate (15–25 mg/week) who presented inactive (i-RA; n = 34, DAS-28 ≤ 2.6) or at least moderately active disease (a-RA; n = 29, DAS-28 > 3.2), and age-matched healthy women (n = 38). We measured the levels of reactive oxygen species (ROS) generation (chemiluminescence assay) and membrane expression of FcγRIIa/CD32, FcγRIIIb/CD16, CR1/CD35, and CR3/CD11b receptors (ELISA assay) in neutrophils. We also determined the hemolytic activity of the alternative and classical pathways of the complement system (spectrophotometry), serum levels of C5a and Bb (ELISA assay), and serum chemotactic activity (Boyden chamber). Compared with the control group, i-RA and a-RA patients exhibited: (1) increased neutrophil ROS production and membrane expression of FcγRIIa/CD32, FcγRIIIb/CD16, and CR1/CD35, indicating neutrophil activation; and (2) increased serum chemotactic activity and decreased activity of the alternative complement pathway, indicating systemic complement system activation. The levels of C-reactive protein in a-RA patients were augmented, compared with i-RA patients. Although infliximab/methotrexate combined therapy induced disease remission according to the DAS-28 criteria, both i-RA and a-RA patients still exhibited significant levels of systemic activation of neutrophils and the complement system.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rheumatoid arthritis (RA) is a systemic autoimmune disease that causes joint damage and long-term disability. Pathogenesis of RA involves a complex interplay among environmental, genetic, and immunologic factors, including dysregulation of innate and adaptive immune function and loss of tolerance to self-antigens [1, 2]. Some key features of RA are swollen joints and the growth of an invasive and inflammatory tissue or pannus across the surface of synovial joints, consisting of activated fibroblasts, macrophages, lymphocytes, and neutrophils. Synovial neutrophils secrete collagen-degrading enzymes and inflammatory mediators that activate osteoclasts to resorb bone and cause irreversible joint destruction [3].
The early diagnosis of RA and early onset of treatment are essential to control disease activity and prevent joint disability [1]. The current drug therapy for RA relies on non-steroidal anti-inflammatory drugs, glucocorticoids, and disease-modifying anti-rheumatic drugs (DMARD) that should be prescribed to the patients as soon as the disease is diagnosed. Methotrexate (MTX) is the gold-standard DMARD for treating RA and is usually well-tolerated, reduces disease signs and symptoms, and improves joint function. When MTX monotherapy fails, rheumatologists associate other DMARD or biological agents, especially anti-tumor necrosis factor-α (TNF-α) antibodies [4,5,6,7,8]. The combined therapy with MTX and anti-TNF-α antibodies has revolutionized the treatment of RA by providing significant clinical, structural, and functional improvement [9]. Three TNF-α-blocking agents are under clinical use in Brazil: infliximab (IFM; Remicade®, an anti-TNF-α chimeric monoclonal antibody), etanercept (Enbrel®, a soluble TNF-α receptor), and adalimumab (Humira®, an anti-TNF-α humanized monoclonal antibody) [10]. The anti-TNF-α therapy effectively alleviates the inflammation signs and joint damage in about 66% of the patients with RA [11, 12].
TNF-α acts in concert with other chemotactic factors to recruit circulating leukocytes to the synovia. In neutrophils, TNF-α triggers the generation of reactive oxygen species (ROS) that are released to the extracellular milieu together with intracellular proteolytic enzymes and participate in the onset, progression, and perpetuation of joint damage in RA [13]. The active phase of the disease is characterized by increased neutrophil mobilization from bone marrow and a predominant and continuous influx of these cells to the synovia, as demonstrated in animal models and patients with RA [14, 15].
The local and systemic activation of the complement system also contributes to the pathogenesis of RA and other inflammatory human diseases, such as systemic lupus erythematosus [16, 18, 25, 46]. Components of the activated complement system opsonize soluble and tissue-bound immune complexes (ICs), as well as they prime, chemoattract, and activate circulating and synovial neutrophils [18]. Although several studies have reported the complement system activation at the synovial level, just few studies have examined its systemic activation in patients with RA stratified according to disease activity [17].
We have previously reported that neutrophils from MTX-treated patients with active RA exhibit augmented levels of systemic activation, chemotactic capacity, ROS generation, and Fcγ and complement receptor (FcγR and CR, respectively) expression [19]. To continue investigating the impact of drug therapy on patients with RA, we examined the functional responsiveness of neutrophils (respiratory burst, chemotaxis, and expression of membrane receptors) and the activation status of the complement system (classical and alternative pathway) in MTX-non-responder patients treated with combined therapy with IFM and MTX.
Materials and methods
Patients and healthy subjects
This study enrolled 63 women aged 27–58 years (mean = 45 years), with established RA—34 with inactive disease (i-RA) and 29 with active disease (a-RA)—who were followed-up from 2 to 28 years (mean = 7 years) in the Rheumatology Outpatient Clinic at Ribeirão Preto Medical School Hospital of the University of São Paulo (HC-FMRP-USP, Ribeirão Preto, SP, Brazil), from April 2011 to April 2014. The patients were diagnosed according to the American College of Rheumatology criteria [20]. RA duration varied from 2 to 27 years (mean = 8 years) and all patients exhibited some degree of bone erosion in hands and/or feet radiographs. Among the 29 a-RA patients, 9 were double-positive for rheumatoid factor (RF) and anti-cyclic citrullinated peptide (anti-CCP) antibody, 10 were single-positive for RF, 16 were single-positive for anti-CCP, and 12 were double-negative for RF and anti-CCP. Among the 34 i-RA patients, 18 were double-positive for RF and anti-CCP, 22 were single-positive for RF, 24 were single-positive for anti-CCP, and 6 were double-negative for RF and anti-CCP.
All the patients had undergone previous treatment with MTX for at least six months, but they had not reached inactivity or low activity disease status, according to the Disease Activity Score of 28 joints (DAS-28) criteria [4, 21]. At the time of enrollment, the patients were under treatment with IFM (3–5 mg/kg every eight weeks) in combination with MTX (15–25 mg/week) and prednisone (5–10 mg/day); after six months of treatment, they were classified as responders or non-responders to the therapy according to the DAS-28 criteria. The patients treated with this combined therapy for more than six months who remained with active disease (at the time of enrollment) had secondary failure (loss of efficacy) and their therapy was adjusted. However, the further therapeutic adjustments were out of the scope of the present study.
Age- and sex-matched healthy subjects (n = 38) who fulfilled the criteria reported by Paoliello-Paschoalato et al. [22] were also recruited for this study. The Research Ethics Committee of the Ribeirão Preto Medical School Hospital of the University of São Paulo (HC-FMRP-USP, Ribeirão Preto, SP, Brazil) approved the study protocol (HCRP n. 10097/2002). All the patients and healthy subjects signed the informed consent form to participate in this study.
Clinical and laboratory parameters of patients with RA
The level of disease activity was determined using the DAS-28 score [23]. Blood samples were collected and patient assessment was performed just before each IFM infusion. The serum levels of RF, anti-CCP antibody, and C-reactive protein (CRP) were determined using immunological assay kits according to the manufacturer’s instructions, as reported previously [24].
Neutrophil isolation
Venous blood was collected from the antecubital vein of each subject into vacutainer tubes, and immediately diluted into the same volume of Alsever’s solution as anticoagulant. Neutrophils were isolated by the gelatin method as described previously [22].
Preparation of immune complexes (ICs)
Immune complexes composed of ovalbumin and rabbit polyclonal anti-ovalbumin IgG were prepared at equivalence, and further opsonized with sera from patients with RA (RAHS) or healthy control subjects (CHS), as described previously [19]. Non-opsonized IC stimulates neutrophils via FcγR alone, while opsonized IC stimulates neutrophils via FcγR + CR cooperation [25].
Neutrophil ROS generation
The overall neutrophil ROS production was measured as reported by Paoliello-Paschoalato et al. [19]. Briefly, neutrophils (1 × 106 cells/mL) were incubated with luminol (280 µM; Sigma–Aldrich, St. Louis, MO, USA) for 3 min, at 37 °C, and further stimulated with IC (80 µg/mL)—either non-opsonized or opsonized with autologous serum—for 30 min, at 37 °C. The luminol-enhanced chemiluminescence (CL) production was measured using the AutoLumat LB 953 luminometer (EG&G Berthold, Bad Wildbad, Germany).
FcγR and CR expression in neutrophils
The levels of FcγRII (CD32), FcγRIII (CD16), CR1 (CD35), and CR3 (CD11b) expression in neutrophils were measured as reported previously [19], using the following mouse monoclonal antibodies acquired from BD Pharmingen™ (BD Biosciences, San Jose, CA, USA): fluorescein isothiocyanate (FITC)-conjugated anti-human CD32 (clone FLI 8.26, IgG2b,k), FITC-conjugated anti-human CD35 (clone E11, IgG1,k), phycoerythrin (PE)-conjugated anti-human CD16 (clone 3G8; IgG1,k), PE-conjugated anti-human CD11b/Mac-1 (clone ICRF 44; IgG1,k), FITC-conjugated non-specific isotype control (clone 27–35; IgG2b,k), PE-conjugated non-specific isotype control (clone MOCPC-21; IgG1,k), and BD Simultest™ Control γ1/γ1. The cell samples were analyzed using the BD FACSCanto flow cytometer (Becton, Dickinson and Company; Franklin Lakes, NJ, USA). Data from 10,000 events were collected and analyzed using the BD FACSDiva software. Neutrophils were identified on the basis of the forward- and right-angle light scattering, while the relative number of labeled neutrophils and the median channel fluorescence intensity (MFI) for each antibody indicated the receptor density.
Serum chemotactic activity
Neutrophil migration towards RAHS and CHS was assayed following the procedure described by Boyden [26] and modified by Paoliello-Paschoalato et al. [19]. Briefly, the lower compartment of the 48-well modified Boyden chamber (Neuro Probe, Cabin John, MD, USA) was filled with serum, pre-treated or not with zymosan (Sigma–Aldrich, St. Louis, MO, USA), and covered with a polycarbonate membrane filter (13-mm diameter and 3-µm pore size). The upper compartment was filled with neutrophil suspension (6 × 105 cells). After a 30-min incubation at 37 °C in a humidified chamber, the membranes were fixed and stained with appropriate solutions. Neutrophil migration was determined by means of the leading front technique [27].
Hemolytic activity of the complement system
Hemolytic activity of the classical and alternative pathways of the complement system in RAHS and CHS was assessed as reported by Landi-Librandi et al. [28]. Briefly, erythrocytes—rabbit erythrocytes (for the alternative pathway) or sheep erythrocytes sensitized with rabbit anti-sheep erythrocyte antibodies (for the classical pathway)—were suspended in appropriate buffers and the absorbance value of the suspension was adjusted to 0.7–0.8 at 700 nm. Diluted sera were added to the erythrocyte suspension and kinetics of hemolysis was monitored at 700 nm for 10 min, at 37 °C. The time required for sera to lyse 50% of the erythrocyte suspension (T1/2) was calculated.
Analysis of complement activation fragments
The complement activation fragments Bb and C5a were quantified in RAHS and CHS samples using the MicroVue™ Complement Bb Plus Fragment EIA and the MicroVue™ Complement C5a EIA assay kits, respectively, according to the manufacturer’s instructions (Quidel, San Diego, CA, USA).
Data analysis
Experimental data were processed and analyzed with the aid of the GraphPad Prism software (version 5.01 for Windows, GraphPad Software, San Diego, CA, USA).
The normality of distribution of data was assessed using the Shapiro–Wilk test. The groups were compared using the following non-parametric tests: (1) the Mann–Whitney test for two independent variables or (2) the Kruskal–Wallis test followed by the Dunn’s post-hoc test for three independent variables, as indicated in the legends. p < 0.05 was considered significant.
Results
Inflammatory markers
The patients were stratified into two groups according to their disease activity status (a-RA and i-RA) based on their DAS-28 score and serum levels of some inflammatory markers. Compared with i-RA patients, a-RA patients exhibited higher DAS-28 score and serum CRP levels, but similar levels of RF and anti-CCP antibodies (Table 1).
Functional responsiveness of neutrophils
First, we examined the neutrophil ROS-generating capacity elicited via FcγR alone or in combination with CR. The FcγR + CR cooperation elicited ROS generation more strongly than FcγR alone in i-RA and a-RA patients and control subjects (Fig. 1). Compared with the respective controls for stimulation via FcγR or FcγR + CR, neutrophils from both i-RA and a-RA patients produced increased levels of ROS (Fig. 1).
Neutrophil ROS generation in patients with active (a-RA) or inactive (i-RA) rheumatoid arthritis treated with infliximab in combination with methotrexate and prednisolone. The cells were stimulated via FcγR or FcγR + CR with non-opsonized IC and IC opsonized with autologous serum, respectively, and overall ROS generation was measured using the luminol-enhanced chemiluminescence (CL) assay. The control group was composed of age- and sex-matched healthy subjects. Groups not sharing the same letter were significantly different from each other (p < 0.05; Kruskal–Wallis test followed by the Dunn’s multiple comparison test). Bars represent median values
Next, we analyzed the pattern of FcγR and CR expression in neutrophils, especially the FcγRIIa (CD32a), FcγRIIIb (CD16b), CR1 (CD35), and CR3 (CD11b/CD18) expression levels. Compared with the control group, neutrophils from i-RA and a-RA patients exhibited increased levels of FcγRIIa, FcγRIIIb, and CR1 expression; their expression levels in both groups of patients were statistically similar (p > 0.05) (Fig. 2a–c). The level of CR3 expression was similar among the three groups studied (Fig. 2d). Hence, the increased levels of membrane receptors expression were directly associated with the enhanced ROS production in neutrophils from patients with RA treated with IFM + MTX.
Neutrophil FcγRIIa (a), FcγRIIIb (b), CR1 (c), and CR3 (d) expression in patients with active (a-RA) or inactive (i-RA) rheumatoid arthritis treated with infliximab in combination with methotrexate and prednisolone. The control group was composed of age- and sex-matched healthy subjects. MFI median channel fluorescence intensity. Groups not sharing the same letter were significantly different from each other (p < 0.05; Kruskal–Wallis test followed by the Dunn’s multiple comparison test). Bars represent median values
Activation status of the complement system
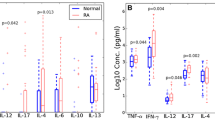
We examined the activation status of the classical and alternative pathways of the complement system in sera from i-RA and a-RA patients. Compared with the control group, both groups of patients displayed slower hemolytic activity of the alternative pathway of the complement system (Fig. 3a), which indicated an increased systemic activation. The three groups also did not differ with respect to the rate of hemolytic activity of the classical pathway and serum levels of the activation fragments Bb and C5a (Fig. 3b–d).
Activation status of the complement system in patients with active (a-RA) or inactive (i-RA) rheumatoid arthritis, treated with infliximab in combination with methotrexate and prednisolone. The control group was composed of age- and sex-matched healthy subjects. Hemolytic activity of the alternative (a) and classical (b) pathways of the complement system was assayed in serum samples diluted 1:40 and 1:12, respectively. Concentration of the complement activation fragments C5a (c) and Bb (d) in serum samples was determined by the ELISA. Groups not sharing the same letter were significantly different from each other (p < 0.05; Kruskal–Wallis test followed by the Dunn’s multiple comparison test). Bars represent median values
We examined the chemotactic activity of sera from patients with RA using neutrophils from healthy subjects to indirectly determine the presence of activated complement system fragments (Fig. 4). Before treatment with zymosan, sera from a-RA patients induced the strongest neutrophil migration, which was significantly different from the migration induced by sera from i-RA patients; both samples induced neutrophil migration over two times more effectively than sera from control subjects. Pre-treatment with zymosan significantly enhanced the chemotactic activity of sera from the three groups studied, indicating that they contained intact complement system proteins.
Neutrophil migration induced by sera from patients with active (a-RA) or inactive (i-RA) rheumatoid arthritis treated with infliximab in combination with methotrexate and prednisolone. Serum samples from each subject were tested before and after treatment with zymosan (treated samples were marked with *). Groups not sharing the same letter were significantly different from each other (p < 0.05; Kruskal–Wallis test followed by the Dunn’s multiple comparison test). Bars represent median values
Influence of autoantibodies on the studied variables
Considering that RF and anti-CCP antibodies are essential elements of the pathogenesis of RA—the former is associated with disease activity while the latter is associated with structural outcomes that not directly impact on disease activity [29]—we further stratified the a-RA and i-RA patients into four subgroups according to the presence of these autoantibodies: double-positive for RF and anti-CCP, single-positive for RF, single-positive for anti-CCP, double-negative for RF and anti-CCP. Although stratification reduced the number of subjects in each group, the results of statistical data analyses for all the variables tested were similar to those found for the whole data set (data not shown), as reported in the previous sections. This finding confirms the lack of influence of the autoantibodies RF and anti-CCP on the neutrophil and complement system parameters assessed in patients with RA.
Discussion
In the present study, we examined the functional responsiveness of neutrophils and the activation status of the complement system in patients with RA treated with IFM + MTX combined therapy. We also compared the main results obtained with those previously reported for patients with RA who responded to MTX monotherapy [19], as summarized in Table 2. We found that neutrophils from the three groups studied—i-RA and a-RA patients and control subjects—stimulated via FcγR + CR produced greater amounts of ROS than those stimulated via FcγR alone, corroborating literature reports [19, 30].
In contrast to neutrophils from patients with RA undergoing MTX monotherapy, which exhibit increased ROS production only when the disease is active [19], we found that neutrophils from both i-RA and a-RA patients treated with IFM + MTX combined therapy produced increased levels of ROS (Fig. 1). In agreement with our findings, peripheral blood neutrophils from patients with RA treated with either the anti-TNF-α monoclonal antibody adalimumab or non-steroidal anti-inflammatory drugs exhibit increased ROS-producing and chemotactic capacity [31]. In addition, treating RA patients with a chimeric TNF-α-blocking monoclonal antibody does not suppress the peripheral blood neutrophil capacity to produce ROS and phagocytose Staphylococcus aureus [32].
Human neutrophils constitutively express the low-affinity FcγRIIa and FcγRIIIb receptors, CR1 (CD35), CR3 (CD11b/CD18, Mac-1), and CR4 receptors [18]. We have previously reported that peripheral blood neutrophils from a-RA but not i-RA patients undergoing MTX monotherapy express increased levels of FcγRIIa, CR1, and CR3 [19]. The levels of FcγRIIa, FcγRIIIb, and CR1 expression were augmented in both i-RA and a-RA patients treated with IFM + MTX (Fig. 2), which may explain, at least in part, their increased neutrophil ROS-producing capacity. A two-week treatment with IFM reverses the FcγRI expression back to the basal levels, but does not change the levels of FcγRIIa and FcγRIIb expression in peripheral blood monocytes from patients with RA [33]. IFM also lowers the levels of FcγRIIa expression after the first and/or second administration cycle without changing the levels of FcγRIIIb expression in neutrophils from patients with RA [34].
In addition to the increased levels of membrane receptors expression, the activation status of the complement system can directly affect neutrophil activation in peripheral blood [18]. The literature reports that synovia of patients with RA usually exhibits low levels of the complement proteins C3, C4, and factor B, associated with increased levels of complement fragments [35]. Hemolytic activity of the classical pathway of the complement system in sera from i-RA and a-RA patients during treatment with MTX alone [19] or in combination with IFM was similar to that detected in sera from control subjects (Fig. 3b). Hemolytic activity of the alternative pathway was slowed down in sera from both i-RA and a-RA patients during treatment with IFM + MTX (Fig. 3a). In contrast, only sera from a-RA patients undergoing MTX monotherapy exhibit slower hemolytic activity [19]. The serum levels of Bb were increased in both a-RA and i-RA patients undergoing MTX monotherapy [19], but they were not significantly different from the levels detected in the control group in a-RA and i-RA patients undergoing IFM + MTX combined therapy (Fig. 3d). a-RA patients under treatment with MTX alone [19] or in combination with IFM (Fig. 3c) displayed increased serum levels of C5a; the difference between the latter group and the control group was near the limit of statistical significance (p = 0.0586). The stronger ability of sera from a-RA patients to induce neutrophil chemotaxis, as compared with sera from i-RA patients (Fig. 4), indicates the presence of activated complement system fragments. The increased neutrophil chemotactic activity of zymosan-treated sera also indicates activation of the complement system. Together, these findings demonstrated that the complement system remained activated in a-RA and iRA patients, probably associated with the alternative pathway, which was not inhibited by IFM + MTX combined therapy.
Circulating and deposited ICs, RFs, anti-CCP antibodies, and CRP are some activators of the complement system in RA [36, 37]. Anti-CCP antibodies from RA patients may activate the complement system via both the classical and alternative pathways [38]. In our study, the serum levels of anti-CCP antibodies and RF did not significantly differ between a-RA and i-RA patients (Table 1). The acute-phase protein CRP activates the complement system both in vitro and in vivo, mainly via the classical pathway [39, 40]. We found that i-RA patients had lower serum levels of CRP than a-RA patients, which was associated with the DAS-28 score (Table 1), and probably mediated systemic activation of the complement system. This finding corroborates literature reports [41,42,43]. Most of the patients with RA display augmented plasma levels of activated complement and CRP-complement complexes, which correlate with the parameters of disease activity [40]. The synovial levels of TNF-α in patients with RA are over twofold greater than in patients with other degenerative joint diseases, and correlate positively with the levels of the complement activation markers C3a and Bb [44]. The plasma levels of the C3 and C4 activation fragments C3b/c and C4b/c, respectively, the DAS-28 score, and the plasma level of CRP markedly lowered two weeks after IFM administration to patients with RA [41].
The most common complication of the long-term therapy with TNF-α-blocking agents is the high incidence of infections, mainly caused by intracellular pathogens [45]. The set of results reported herein suggests that the increased susceptibility to infections is not related to impairment of neutrophil ROS generation and expression of the phagocytic receptors FcγR and CR. In both i-RA and a-RA patients, their levels remained significantly higher than those detected in the control group during the course of IFM + MTX combined therapy (Figs. 1, 2).
Although patients with RA who responded to IFM + MTX combined therapy (i.e. i-RA patients) exhibited decreased disease activity (DAS-28) and lowered levels of inflammatory mediators (CRP) when compared with non-responders (i.e. a-RA patients), both i-RA and a-RA patients presented functional alterations in neutrophils and complement system that indicated that they were activated and the disease activity was not completely subsided. Considering that: (1) neutrophils are one of the major actors of pathogenesis of RA [3]; (2) the main autoantibodies (anti-CCP and RF) detected in sera from patients with RA activate the complement system [35, 37]; and (3) treatment of RA with MTX alone diminishes neutrophil ROS production and complement system activity in MTX-responder patients [19]; further studies are required to examine the efficacy of the IFM + MTX combined therapy for MTX-non-responder patients.
Abbreviations
- Anti-CCP:
-
Anti-cyclic citrullinated peptide antibody
- CHS:
-
Serum from healthy control subject
- CL:
-
Chemiluminescence
- CR:
-
Complement receptor
- CRP:
-
C-reactive protein
- DAS-28:
-
Disease activity score of 28 joints
- DMARD:
-
Disease-modifying anti-rheumatic drug
- EULAR:
-
European League against Rheumatism
- FcγR:
-
Fcγ receptors
- FITC:
-
Fluorescein isothiocyanate
- HA:
-
Hemolytic activity
- IC:
-
Immune complex
- IFM:
-
Infliximab
- MFI:
-
Median fluorescence intensity
- MTX:
-
Methotrexate
- PE:
-
Phycoerythrin
- RA:
-
Rheumatoid arthritis
- a-RA:
-
Active rheumatoid arthritis
- i-RA:
-
Inactive rheumatoid arthritis
- RAHS:
-
Serum from patient with rheumatoid arthritis
- ROS:
-
Reactive oxygen species
- TNF-α:
-
Tumor necrosis factor-α
References
McInnes IB, Schett G (2011) The pathogenesis of rheumatoid arthritis. N Engl J Med 365:2205–2219
McInnes IB, Buckley CD, Isaacs JD (2016) Cytokines in rheumatoid arthritis—shaping the immunological landscape. Nat Rev Rheumatol 12:63–68
Thieblemont N, Wright HL, Edwards SW, Witko-Sarsat V (2016) Human neutrophils in auto-immunity. Semin Immunol 28:159–173
American College of Rheumatology (2002) Guidelines for the management of rheumatoid arthritis—2002 update. Arthritis Rheum 46:328–346
Curtis JR, Singh JA (2011) The use of biologics in rheumatoid arthritis: current and emerging paradigms of care. Clin Ther 33:679–707
Jani M, Barton A, Warren RB, Griffiths CEM, Chinoy H (2014) The role of DMARDs in reducing the immunogenicity of TNF inhibitors in chronic inflammatory diseases. Rheumatology 53:213–222
Dimopoulou D (2015) Infliximab as a treatment option for patients with rheumatoid arthritis and primary biliary cirrhosis. Rheumatol Int 35:1913–1916
Costa JO, Lemos LLP, Machado MAA, Almeida AM, Kakehasi AM, Araújo VE et al (2015) Infliximab, methotrexate and their combination for the treatment of rheumatoid arthritis: a systematic review and meta-analysis. Rev Bras Reumatol 55:146–158
Tanaka Y (2016) Stopping tumour necrosis factor-targeted biological DMARDs in rheumatoid arthritis. Rheumatology 55(suppl 2):ii15–ii22
Mota LM, Cruz BA, Albuquerque CP, Gonçalves DP, Laurindo IM, Pereira IA et al (2015) Update on the 2012 Brazilian Society of Rheumatology Guidelines for the treatment of rheumatoid arthritis: position on the use of tofacitinib. Rev Bras Reumatol 55:512–521
Pomirleanu C, Ancuta C, Miu S, Chirieac R (2013) A predictive model for remission and low disease activity in patients with established rheumatoid arthritis receiving TNF blockers. Clin Rheumatol 32:665–670
Shaffu S, Edwards J, Neame R, Hassan W (2013) Self-assessment of 28-joint disease activity scores by patients with rheumatoid arthritis on anti-TNF therapy. Rheumatology 52:576–578
Cascão R, Rosário HS, Souto-Carneiro MM, Fonseca JE (2010) Neutrophils in rheumatoid arthritis: more than simple final effectors. Autoimmun Rev 9:531–535
Asquith DL, Miller AM, McInnes IB, Liew FY (2009) Animal models of rheumatoid arthritis. Eur J Immunol 39:2040–2044
Gierut A, Perlman H, Pope RM (2010) Innate immunity and rheumatoid arthritis. Rheum Dis Clin N Am 36:271–296
Chen M, Daha MR, Kallenberg CG (2010) The complement system in systemic autoimmune disease. J Autoimmun 34:276–286
Ballanti E, Perricone C, Greco E, Ballanti M, Di Muzio G, Chimenti MS et al (2013) Complement and autoimmunity. Immunol Res 56:477–491
Paoliello-Paschoalato AB, Marchi LF, Andrade MF, Kabeya LM, Donadi EA, Lucisano-Valim YM (2015) Fcγ and complement receptors and complement proteins in neutrophil activation in rheumatoid arthritis: contribution to pathogenesis and progression and modulation by natural products. Evid Based Complement Alternat Med 2015:429878
Paoliello-Paschoalato AB, Moreira MR, Azzolini AECS, Cavenaghi AA, Marzocchi-Machado CM, Donadi EA et al (2011) Activation of complement alternative pathway in rheumatoid arthritis: implications in peripheral neutrophils functions. Open Autoimmun J 3:1–9
Arnett FC, Edworthy SM, Bloch DA, McShane DJ, Frie JF, Cooper NS et al (1988) The American Rheumatism Association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 31:315–324
van Gestel AM, Haagsma CJ, van Riel PL (1998) Validation of rheumatoid arthritis improvement criteria that include simplified joint counts. Arthritis Rheum 41:1845–1850
Paoliello-Paschoalato AB, Azzolini AECS, Cruz MFC, Marchi LF, Kabeya LM, Donadi EA et al (2014) Isolation of healthy individuals’ and rheumatoid arthritis patients’ peripheral blood neutrophils by the gelatin and Ficoll-Hypaque methods: comparative efficiency and impact on the neutrophil oxidative metabolism and Fcγ receptor expression. J Immunol Methods 412:70–77
Dougados M, Aletaha D, van Riel P (2007) Disease activity measures for rheumatoid arthritis. Clin Exp Rheumatol 25(5 Suppl 46):S22–S29
Oliveira RD, Junta CM, Oliveira FR, Silva LM, Donadi EA, Louzada-Junior P (2008) Share epitope, citrullinated cyclic peptide antibodies and smoking in Brazilian rheumatoid arthritis patients. Clinic Rev Allerg Immunol 34:32–35
Marzocchi-Machado CM, Alves CMOS, Azzolini AECS, Polizello ACM, Carvalho IF, Lucisano-Valim YM (2002) Fcγ and complement receptors: expression, role and cooperation to mediate the oxidative burst and degranulation of neutrophil of Brazilian SLE patients. Lupus 11:240–248
Boyden S (1962) The chemotactic effect of mixtures of antibody and antigen on polymorphonuclear leucocytes. J Exp Med 115:453–466
Zigmond SH, Hirsch JG (1973) Leukocyte locomotion and chemotaxis. New methods for evaluation, and demonstration of a cell-derived chemotactic factor. J Exp Med 137:387–410
Landi-Librandi AP, Chrysóstomo TN, Azzolini AECS, Recchia CGV, Uyemura SA, Assis-Pandochi AI (2007) Effect of the extract of the tamarind (Tamarindus indica) fruit on the complement system: studies in vitro and in hamsters submitted to a cholesterol-enriched diet. Food Chem Toxicol 45:1487–1495
Aletaha D, Blüml S (2016) Therapeutic implications of autoantibodies in rheumatoid arthritis. RMD Open 2:e000009
Kundu S, Ghosh P, Datta S, Ghosh A, Chattopadhyay S, Chatterjee M (2012) Oxidative stress as a potential biomarker for determining disease activity in patients with rheumatoid arthritis. Free Radic Res 46:1482–1489
den Broeder AA, Wanten GJ, Oyen WJ, Naber T, van Riel PL, Barrera P (2003) Neutrophil migration and production of reactive oxygen species during treatment with a fully human anti-tumor necrosis factor-alpha monoclonal antibody in patients with rheumatoid arthritis. J Rheumatol 30:232–237
Lorenz HM, Antoni C, Valerius T, Repp R, Grünke M, Schwerdtner N et al (1996) In vivo blockade of TNF-α by intravenous infusion of a chimeric monoclonal TNF-alpha antibody in patients with rheumatoid arthritis. Short term cellular and molecular effects. J Immunol 156:1646–1653
Wijngaarden S, van de Winkel JG, Bijlsma JW, Lafeber FP, van Roon JA (2008) Treatment of rheumatoid arthritis patients with anti-TNF-γ monoclonal antibody is accompanied by down-regulation of the activating Fcγ receptor I on monocytes. Clin Exp Rheumatol 26:89–95
Belostocki K, Pricop L, Redecha PB, Aydin A, Leff L, Harrison MJ et al (2008) Infliximab treatment shifts the balance between stimulatory and inhibitory Fcγ receptor type II isoforms on neutrophils in patients with rheumatoid arthritis. Arthritis Rheum 58:384–388
Neumann E, Barnum SR, Tarner IH, Echols J, Fleck M, Judex M et al (2002) Local production of complement proteins in rheumatoid arthritis synovium. Arthritis Rheum 46:934–945
Happonen KE, Heinegard D, Saxne T, Blom AM (2012) Interactions of the complement system with molecules of extracellular matrix: relevance for joint diseases. Immunobiology 217:1088–1096
Malhotra R, Wormald MR, Rudd PM, Fischer PB, Dwek RA, Sim RB (1995) Glycosylation changes of IgG associated with rheumatoid arthritis can activate complement via the mannose-binding protein. Nat Med 1:237–243
Trouw LA, Haisma EM, Levarht EW, van der Woude D, Ioan-Facsinay A, Daha MR et al (2009) Anti-cyclic citrullinated peptide antibodies from rheumatoid arthritis patients activate complement via both the classical and alternative pathways. Arthritis Rheum 60:1923–1931
Volanakis JE (1982) Complement activation by C-reactive protein complexes. Ann N Y Acad Sci 389:235–250
Molenaar ET, Voskuyl AE, Familian A, van Mierlo GJ, Dijkmans BA, Hack CE (2001) Complement activation in patients with rheumatoid arthritis mediated in part by C-reactive protein. Arthritis Rheum 44:997–1002
Familian A, Voskuyl AE, van Mierlo GJ, Heijst HA, Twisk JW, Dijkmans BA et al (2005) Infliximab treatment reduces complement activation in patients with rheumatoid arthritis. Ann Rheum Dis 64:1003–1008
Wolbink GJ, Brouwer MC, Buysmann S, ten Berge IJ, Hack CE (1996) CRP-mediated activation of complement in vivo: assessment by measuring circulating complement-C-reactive protein complexes. J Immunol 157:473–479
Elliot MJ, Maini RN, Feldmann M, Long-Fox A, Charles P, Katasikis P et al (2008) Treatment of rheumatoid arthritis with chimeric monoclonal antibodies to tumor necrosis factor alpha. Arthritis Rheum 58(Suppl 2):S92–S101
Brodeur JP, Ruddy S, Schwartz LB, Moxley G (1991) Synovial fluid levels of complement SC5b-9 and fragment Bb are elevated in patients with rheumatoid arthritis. Arthritis Rheum 34:1531–1537
Di Franco M, Lucchino B, Spaziante M, Iannuccelli C, Valesini G, Iaiani G (2017) Lung infections in systemic rheumatic disease: focus on opportunistic infections. Int J Mol Sci 18:293
Alves CMOS, Marzocchi-Machado CM, Louzada-Junior P, Azzolini AECS, Polizello ACM, Carvalho IF, Lucisano-Valim YM (2008) Superoxide anion production by neutrophils is associated with prevalent clinical manifestations in systemic lupus erythematosus. Clin Rheumatol 27:701–708
Acknowledgements
The authors are grateful to Dr. Ana Paula Landi Librandi, from the School of Pharmaceutical Sciences of Ribeirão Preto (Ribeirão Preto, SP, Brazil) for helping us to perform the ELISA assays, and to Mrs. Patricia Vianna Bonini Palma and Mrs. Camila Cristina O. M. Bonaldo from the Hemotherapy Center of Ribeirão Preto (FUNDHERP, Ribeirão Preto, SP, Brazil) for their helpful technical assistance in the flow cytometry analyses.
Funding
The Brazilian funding agencies São Paulo Research Foundation (FAPESP; Grants 2013/22331-5, and 2013/20810-7), the National Council for Scientific and Technological Development (CNPq; Grants 151665/2010-9, 308111/2013/3), and the Coordination for the Improvement of Higher Education Personnel (CAPES) provided the fellowships and financial support.
Author information
Authors and Affiliations
Contributions
LFM and ABPP devised the project, designed the study, analyzed and interpreted the results, and drafted the manuscript. LFM conducted all the experiments. RDRO followed-up and selected the patients, and determined their clinical parameters. AECSA assisted in all the experiments, and in data processing and analysis. LMK assisted in statistical data analyses, and drafted and proofread the manuscript. EAD selected the patients and supervised the study. YMLV devised the project, searched for funding, and supervised the study. All authors discussed the results, and revised and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
The Research Ethics Committee of the Ribeirão Preto Medical School Hospital of the University of São Paulo (HC-FMRP-USP, Ribeirão Preto, SP, Brazil) approved the study protocol (HCRP n. 10097/2002). All the procedures conducted in humans complied with Resolution 466/12 of the Brazilian National Health Council, which follows the recommendations of the 1964 Helsinki declaration and its later amendments.
Informed consent
All the patients and healthy subjects signed the informed consent form to participate in this study.
Additional information
This study is part of the post-doctoral research project developed by Dr. Larissa Fávaro Marchi under supervision of Dr. Yara Maria Lucisano-Valim, at School of Pharmaceutical Sciences of Ribeirão Preto. The present institutional address of Dr. Marchi is: Faculdades Integradas Padre Albino, FIPA, Rua dos Estudantes 225, Catanduva, SP, 15809-144 Brazil.
Rights and permissions
About this article
Cite this article
Marchi, L.F., Paoliello-Paschoalato, A.B., Oliveira, R.D.R. et al. Activation status of peripheral blood neutrophils and the complement system in adult rheumatoid arthritis patients undergoing combined therapy with infliximab and methotrexate. Rheumatol Int 38, 1043–1052 (2018). https://doi.org/10.1007/s00296-018-3997-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-018-3997-1