Abstract
Interstitial lung disease (ILD) is the most common cause of death in dermatomyositis (DM). Cyclosporine A (CsA) has shown to be effective in DM-associated ILD (DM-ILD). This study aimed to define the optimal time of CsA administration. A total of 47 patients with DM-ILD, who were treated with CsA at Seoul National University Hospital between January 1998 and June 2013, were enrolled. ILD was diagnosed based on typical chest high-resolution computed tomography (HRCT) findings. Patients with early and delayed CsA treatment were compared in regard to the mortality and ILD progression on HRCT. The early (n = 16) and the delayed treatment group (n = 31) did not differ in regard to baseline clinical characteristics including HRCT scores and pulmonary function. Patients with clinically amyopathic DM (CADM) were more common in the early treatment group. The mortality rate was significantly lower in the early treatment group than in the delayed treatment group (p = 0.009). The survival benefit of early CsA treatment remained significant even after adjusting for age, degree of dyspnea, CADM status, and the year of CsA treatment (hazard ratio 0.057, 95 % confidence interval 0.007–0.472). CsA stabilized disease progression on HRCT in the early treatment group (p = 0.738). Delay in CsA treatment is associated with a worse survival in patients with DM-ILD. Early CsA treatment should be considered at DM-ILD diagnosis especially in patients at a higher risk of developing a rapidly progressive ILD.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Dermatomyositis (DM) is a connective tissue disease of unknown etiology that causes inflammation in skeletal muscle and skin. It can affect various organs including lungs as interstitial lung disease (ILD). ILD develops in 21–78 % of DM patients, and it is the most common cause of death in patients with DM with a 5-year mortality rate ranging between 7.5 and 55 % [1–5]. Clinically amyopathic dermatomyositis (CADM) is a clinical subtype of DM that features skin involvement with no to little muscle weakness [6]. Patients with CADM have a higher risk of developing rapidly progressive ILD that is often refractory to corticosteroids treatment [6–8]. CADM status, baseline low forced vital capacity (FVC) <60 %, and rapid progression of ILD were associated with a higher mortality in DM-associated ILD (DM-ILD) [8].
Administration of high-dose corticosteroids belongs to the initial standard treatment of DM-ILD [9]. However, a significant number of patients with DM-ILD do not respond to corticosteroid alone and require additional immunosuppressive treatment [10, 11]. Recent studies showed that patients who were treated intensively with the combination of immunosuppressive drugs (ISDs) at DM-ILD diagnosis had a better survival outcome compared to those who received ISDs in a sequential (i.e., step-up) approach after failure of the initial treatment [12, 13].
Cyclosporine A (CsA) is a calcineurin inhibitor that preferentially targets T cell activation during inflammatory response. CsA has been reported to be effective in DM-ILD [10, 13–16]. However, the optimal time of CsA commencement that yields its maximal clinical efficacy remains unknown. In the present study, we sought to determine whether patients that receive early CsA treatment have a better survival benefit than those in whom CsA administration is delayed.
Methods
Patients
In this study, 47 patients with DM-ILD, who received CsA during their clinical care at Seoul National University Hospital between January 1998 and June 2013, were enrolled. Classic DM and CADM were diagnosed according to Bohan and Peter’s classification criteria [17] and Sontheimer’s definition [6], respectively. The demographic and clinical characteristics, laboratory values, and pulmonary function test (PFT) results were obtained from the medical records. The survival status of all patients was confirmed using the national database of Statistics Korea. This study was approved by the Institutional Review Board of Seoul National University Hospital (IRB No. 1307-063-503). Obtaining an informed consent was waived as a retrospective nature of the study with minimal risk to study patients.
HRCT scoring and pulmonary function test
ILD was defined as the presence of typical ILD findings in high-resolution computed tomography (HRCT) of chest. Those findings include ground glass opacities (GGO), linear and reticular opacities, consolidations, and traction bronchiectasis [4, 18, 19]. Severity of ILD on HRCT scan was scored as previously described [20]. Briefly, CT images of both lung lobes (i.e., the right upper, middle and lower lobe and the left upper and lower lobe) at mid-arch of the aorta, tracheal carina, and 1 cm above the diaphragm were scored on a scale of 0–5 in a semi-quantitative manner and were added together, yielding a total score between 0 and 25.
PFT was performed in most patients at ILD diagnosis. FVC and diffusing capacity for carbon monoxide (DLCO) were expressed as a percentage of the predicted values on the basis of a patient’s age, sex, height, and weight.
Study design
This is a retrospective cohort study. Enrolled patients were divided into two groups according to the time of CsA treatment. The patients in the early treatment group (n = 16) received CsA treatment within 2 weeks of the diagnosis of ILD (between 2003 and 2013), whereas the patients in the delayed treatment group (n = 31) received CsA after the trial of other ISDs (between 1998 and 2013). Decision on CsA administration was made solely at the discretion of the treating physician. To ascertain the comparability of the study groups, major risk factors affecting survival of the ILD patients were compared. They included age, duration of disease, presence of malignancy, CADM status, oxygen saturation, functional reserve of lung measured by PFT, the extent of ILD measured by HRCT at baseline, which was defined as the time of diagnosis of ILD, and the treatment year. Survival of the patients from the baseline was compared between the study groups.
Statistical methods
Group comparisons were made using Student’s t test, Chi–square test, or Fisher’s exact test, as appropriate. Survival was analyzed by using Kaplan–Meier curves and the log-rank test. The survival benefit of the early versus the delayed CsA treatment was assessed using the Cox regression hazard model. The paired t test was used to evaluate the changes in HRCT scores before and after CsA treatment. A p value of <0.05 was considered to indicate statistical significance. Data analyses were performed using SPSS software version 18.0 (SPSS, Inc., Chicago, IL, USA).
Results
Clinical characteristics of patients
All patients received corticosteroid therapy (0.5–1.0 mg/kg prednisolone equivalent daily) at DM-ILD diagnosis. Patients in the delayed treatment group received CsA on average 5.3 ± 8.8 months after ILD diagnosis. The ISDs in the delayed treatment group included corticosteroid alone (n = 16) and combination of corticosteroids with methotrexate (n = 6), azathioprine (n = 6), or intravenous cyclophosphamide (n = 3). CsA was orally administered in both groups except in 3 patients who received CsA intravenously. Dosage of CsA was not different between the early and the delayed treatment groups (167.2 ± 37.3 vs. 157.9 ± 77.5 mg/day, p = 0.654).
Overall, the patients in the early and delayed treatment groups were similar in terms of their baseline demographic and clinical characteristics. The mean duration of CsA treatment was 16.0 ± 14.4 months in the early treatment group and 10.3 ± 21.8 months in the delayed treatment group (p = 0.350). At ILD diagnosis, 4 (25.0 %) patients in the early treatment group and 11 (35.5 %) patients in the delayed treatment group had oxygen saturation of 90 % or less at room air (p = 0.527). The groups did not differ in regard to baseline FVC, DLCO, and HRCT scores. However, the early treatment group included more patients with CADM than the delayed treatment group (62.5 vs. 29.0 %, p = 0.034) (Table 1). When the number of patients in both groups was compared according to the treatment year, the early treatment group also had a higher proportion of patients treated with CsA after year 2010 (median treatment time of the total patients) (75.0 vs. 38.7 %, p = 0.030).
Survival outcome with CsA treatment
During the follow-up period (mean 30.6 ± 37.2 months), 1 (6.3 %) of the 16 patients in the early treatment group and 13 (41.9 %) of the 31 patients in the delayed treatment group progressed to hypoxemia status despite oxygen supplement. They all received mechanical ventilation in intensive care unit (ICU), and average duration of ICU stay was 19.3 ± 11.7 days. However, all of these patients, including 4 patients who received lung transplantation (TPL), died. Causes of death included respiratory failure due to ILD aggravation, septic shock, tension pneumothorax, ventricular arrhythmia, and complications associated with lung TPL. Lung TPL was performed in 1 patient in the early and 3 patients in the delayed treatment group after receiving CsA treatment and mechanical ventilation. Three patients died of pneumonia within 12 months after TPL, and the other 1 patient died of pneumonia which complicates with graft rejection 5 years after TPL. Additionally, 1 patient in the delayed treatment group died suddenly due to diffuse alveolar hemorrhage (Table 2).
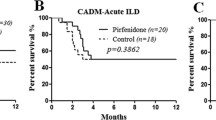
As a result, only 1 (6.3 %) of the 16 patients in the early treatment group died. By contrast, 14 (45.2 %) of the 31 patients in the delayed treatment group died. The early treatment group had a significantly lower all-cause mortality than the delayed treatment group (p = 0.009 by log-rank test) (Fig. 1). The survival benefit of the early CsA treatment remained significant even after adjusting for age, CADM status, the presence of low oxygen saturation at baseline (SpO2 < 90 %), and the year of CsA treatment (hazard ratio 0.057, 95 % confidence interval 0.007–0.472, p = 0.008). In patients with CADM, the early CsA treatment also significantly improved the survival (p = 0.007 by log-rank test) (Fig. 2).
Survival outcomes of subgroup patients with classic dermatomyositis or clinically amyopathic dermatomyositis. Cyclosporine A treatment starting at ILD diagnosis resulted in significantly better survival outcomes in both classic DM (a) and clinically amyopathic dermatomyositis (CADM) (b) as compared to the delayed treatment
Changes in HRCT scores with CsA treatment
Serial assessments of HRCT were available in 12 (75.0 %) patients of the early and in 18 (58.1 %) patients of the delayed treatment group. Among the patients who died, 9 patients did not have a follow-up HRCT scan after CsA treatment. Baseline HRCT scores at ILD diagnosis for patients in whom serial HRCT scans were available were similar in both groups (8.8 ± 2.6 vs. 9.4 ± 3.7, p = 0.620). In the early treatment group, HRCT before CsA treatment was considered as baseline because CsA treatment was done shortly after ILD diagnosis. In the delayed treatment group, HRCTs before CsA treatment were evaluated on average of 4.6 ± 10.6 months after baseline HRCTs. Interval of HRCTs between before and after CsA treatment was mean 16.3 ± 22.3 months in the early treatment group and 13.7 ± 15.5 months in the delayed treatment group, respectively. Two groups had comparable time interval (p = 0.709).
After introduction of CsA, the mean HRCT score did not change in the early treatment group (8.8 ± 2.6 vs. 8.4 ± 3.9, p = 0.738). In the delayed treatment group, the HRCT scores tended to worsen during the initial treatment period with corticosteroids with or without ISDs (9.4 ± 3.7 vs. 10.8 ± 4.5, p = 0.070). ILD progression was stabilized after CsA introduction (10.8 ± 4.5 vs. 11.8 ± 6.0, p = 0.273) (Fig. 3). Overall, there was a significant deterioration in HRCT scores in the delayed treatment group relative to baseline HRCT (9.4 ± 3.7 vs. 11.8 ± 6.0, p = 0.029). The early CsA treatment showed a favorable radiologic response compared to the delayed CsA treatment (HRCT score difference between baseline and after CsA in each group, −0.4 ± 4.2 vs. 2.3 ± 4.2, p = 0.088).
HRCT scores before and after cyclosporine A treatment. In patients with serial HRCT assessment (12 in the early CsA group and 18 in the delayed CsA group), the baseline (i.e., at ILD diagnosis) HRCT scores of the two groups did not differ (8.8 ± 2.6 vs. 9.4 ± 3.7, p = 0.620). In the early treatment group, HRCT remained stable after CsA treatment (p = 0.738). In the delayed treatment group, no further progression was observed (10.8 ± 4.5 vs. 11.8 ± 6.0, p = 0.273) after CsA. However, HRCT significantly deteriorated compared to baseline (9.4 ± 3.7 vs. 11.8 ± 6.0, p = 0.029). Dead patients were indicated as asterisk (*)
Adverse events during CsA treatment
During CsA treatment, adverse events were observed in 10 (62.5 %) of the early treatment group and 11 (35.5 %) of the delayed treatment group (p = 0.116). Most of the adverse events including infection (n = 5), nephrotoxicity (n = 5), and leukopenia (n = 2) were mild and reversible. No life-threatening event was observed. CsA was discontinued in 5 patients due to skin eruption (n = 1), hand tremor (n = 1), chest pain (n = 1), upper abdominal pain (n = 1), and possible tuberculosis infection (n = 1).
Discussion
The present study showed that patients with DM-ILD who received CsA early after the diagnosis had better survival outcomes as compared to those patients whose CsA treatment was delayed or started as a rescue after the failure of other ISDs. In addition, the early CsA treatment slowed down the ILD progression on HRCT.
T cells are abundant in the lung tissue of patients with DM-ILD and play an important role in its pathogenesis [21–23]. CsA, which preferentially inhibits T cell activation, has been suggested as a rescue medication for corticosteroid-resistant DM-ILD. Previous studies have confirmed that concomitant CsA treatment improved survival in DM-ILD patients [10, 13, 16, 24, 26]. However, to date, the optimal time of CsA administration for DM-ILD has not been fully elucidated.
The present study clearly demonstrates that early administration of CsA is associated with a better survival of patients with DM-ILD and CsA might slow down or even reverse, at least in some cases, ILD progression. Conversely, a delay in CsA treatment was associated with a higher mortality and ILD progression. This finding is consistent with the prior report that in patients with acute/subacute DM-ILD, the early combination of corticosteroids with CsA and intravenous pulse cyclophosphamide improved the survival outcome, whereas all patients who were treated with corticosteroid alone died [24].
A previous study showed that rapidly progressive DM-ILD, which develops into respiratory failure within 3 months, was associated with a higher mortality rate despite extensive immunosuppressive treatment after the initial treatment failure [8]. However, a significant subset of DM-ILD patients with slowly progressive ILD responded to corticosteroid monotherapy and might not require other additional ISDs. Until this subset of corticosteroid-responsive DM-ILD is better characterized, the early combination therapy with corticosteroids and CsA should be the treatment of choice, considering the high mortality of rapidly progressive DM-ILD.
CADM-associated ILD has been reported to be a poor prognostic factor, especially among Asian patients [7, 8, 10]. In the present study, 19 (40.4 %) patients with CADM-associated ILD (10 patients in the early treatment group and 9 patients in the delayed treatment group) received CsA. Survival benefit of early introduction of CsA was observed in this subgroup of poor prognosis (Fig. 2).
Kotani et al. [25] demonstrated that the combination therapy with CsA and corticosteroids at DM-ILD diagnosis improved the HRCT scores over the 1-year follow-up period. Similarly, CsA slowed down or stabilized disease progression on HRCT in our study (Fig. 3). A higher mortality in the delayed treatment group could be associated with ongoing ILD progression before CsA administration. This suggests that when ILD progression passes the “point of no return”, CsA might not be able to positively alter the clinical outcome. Therefore, it is important that DM-ILD should be treated aggressively early in the disease course.
As a retrospective design, this study has also several limitations. First, the number of enrolled patients in the current study was rather small despite being one of the largest studies in this field [10, 13, 24, 26]. Second, HRCT scans were not performed at predefined time points, and the data were not available for all of the patients. Treatment effect of CsA might be overestimated in the delayed treatment group since the follow-up HRCT scans could not be often available in dead patients, who were more prevalent in the delayed treatment group. For a similar reason, follow-up PFT could not be performed in most of the patients with severe dyspnea. Third, myositis-specific antibodies (MSAs) except anti-Jo1, which are associated with the outcome of DM, were not fully evaluated [27–29].
In conclusion, our findings suggest that the early treatment with CsA improves the survival and stabilizes disease progression in patients with DM-ILD. To confirm the therapeutic benefit of this strategy, a large-scale randomized controlled study is warranted.
References
Marie I, Hatron P, Dominique S, Cherin P, Mouthon L, Menard JF (2011) Short-term and long-term outcomes of interstitial lung disease in polymyositis and dermatomyositis: a series of 107 patients. Arthritis Rheum 63:3439–3447
Fathi M, Vikgren J, Boijsen M, Tylen U, Jorfeldt L, Tornling G et al (2008) Interstitial lung disease in polymyositis and dermatomyositis: longitudinal evaluation by pulmonary function and radiology. Arthritis Rheum 59:677–685
Chen I-J, Wu Y-JJ, Lin C-W, Fan K-W, Luo S-F, Ho H-H et al (2009) Interstitial lung disease in polymyositis and dermatomyositis. Clin Rheumatol 28:639–646
Cottin V, Thivolet-Bejui F, Reynaud-Gaubert M, Cadranel J, Delaval P, Ternamian P et al (2003) Interstitial lung disease in amyopathic dermatomyositis, dermatomyositis and polymyositis. Eur Respir J 22:245–250
Vij R, Strek ME (2013) Diagnosis and treatment of connective tissue disease-associated interstitial lung disease connective tissue-interstitial lung disease. Chest 143:814–824
Sontheimer RD (2002) Would a new name hasten the acceptance of amyopathic dermatomyositis (dermatomyositis sine myositis) as a distinctive subset within the idiopathic inflammatory dermatomyopathies spectrum of clinical illness? J Am Acad Dermatol 46:626–636
Suda T, Fujisawa T, Enomoto N, Nakamura Y, Inui N, Naito T et al (2006) Interstitial lung diseases associated with amyopathic dermatomyositis. Eur Respir J 28:1005–1012
Kang E, Lee E, Shin K, Im C, Chung D, Han S et al (2005) Interstitial lung disease in patients with polymyositis, dermatomyositis and amyopathic dermatomyositis. Rheumatology 44:1282–1286
Marie I, Hachulla E, Cherin P, Dominique S, Hatron PY, Hellot MF et al (2002) Interstitial lung disease in polymyositis and dermatomyositis. Arthritis Rheum 47:614–622
Nawata Y, Kurasawa K, Takabayashi K, Miike S, Watanabe N, Hiraguri M et al (1999) Corticosteroid resistant interstitial pneumonitis in dermatomyositis/polymyositis: prediction and treatment with cyclosporine treatment with cyclosporine. J Rheumatol 26:1527–1533
Fujisawa T, Suda T, Nakamura Y, Enomoto N, Ide K, Toyoshima M et al (2005) Differences in clinical features and prognosis of interstitial lung diseases between polymyositis and dermatomyositis. J Rheumatol 32:58–64
Takada K, Kishi J, Miyasaka N (2007) Step-up versus primary intensive approach to the treatment of interstitial pneumonia associated with dermatomyositis/polymyositis: a retrospective study. Mod Rheumatol 17:123–130
Kotani T, Makino S, Takeuchi T, Kagitani M, Shoda T, Hata A et al (2008) Early intervention with corticosteroids and cyclosporin A and 2-hour postdose blood concentration monitoring improves the prognosis of acute/subacute interstitial pneumonia in dermatomyositis. J Rheumatol 35:254–259
Takada K, Nagasaka K, Miyasaka N (2005) Polymyositis/dermatomyositis and interstitial lung disease: a new therapeutic approach with T-cell-specific immunosuppressants. Autoimmunity 38:383–392
Maeda K, Kimura R, Komuta K, Igarashi T (1997) Cyclosporine treatment for polymyositis/dermatomyositis: is it possible to rescue the deteriorating cases with interstitial pneumonitis? Scand J Rheumatol 26:24–29
Labirua-Iturburu A, Selva-O’Callaghan A, Martinez-Gomez X, Trallero-Araguás E, Labrador-Horrillo M, Vilardell-Tarrés M (2012) Calcineurin inhibitors in a cohort of patients with antisynthetase-associated interstitial lung disease. Clin Exp Rheumatol 31:436–439
Bohan A, Peter JB, Bowman RL, Pearson CM (1977) A computer-assisted analysis of 153 patients with polymyositis and dermatomyositis. Medicine 56:255–286
Mino M, Noma S, Taguchi Y, Tomii K, Kohri Y, Oida K (1997) Pulmonary involvement in polymyositis and dermatomyositis: sequential evaluation with CT. AJR Am J Roentgenol 169:83–87
Arakawa H, Yamada H, Kurihara Y, Nakajima Y, Takeda A, Fukushima Y et al (2003) Nonspecific interstitial pneumonia associated with polymyositis and dermatomyositisserial high-resolution CT findings and functional correlation. Chest 123:1096–1103
Kazerooni EA, Martinez FJ, Flint A, Jamadar DA, Gross BH, Spizarny DL et al (1997) Thin-section CT obtained at 10-mm increments versus limited three-level thin-section CT for idiopathic pulmonary fibrosis: correlation with pathologic scoring. AJR Am J Roentgenol 169:977–983
Kurasawa K, Nawata Y, Takabayashi K, Kumano K, Kita Y, Takiguchi Y et al (2002) Activation of pulmonary T cells in corticosteroid-resistant and-sensitive interstitial pneumonitis in dermatomyositis/polymyositis. Clin Exp Immunol 129:541–548
Yamadori I, Fujita J, Kajitani H, Bandoh S, Tokuda M, Ohtsuki Y et al (2001) Lymphocyte subsets in lung tissues of interstitial pneumonia associated with untreated polymyositis/dermatomyositis. Rheumatol Int 21:89–93
Connors GR, Christopher-Stine L, Oddis CV, Danoff SK (2010) Interstitial lung disease associated with the idiopathic inflammatory myopathies what progress has been made in the past 35 years? Chest 138:1464–1474
Kameda H, Nagasawa H, Ogawa H, Sekiguchi N, Takei H, Tokuhira M et al (2005) Combination therapy with corticosteroids, cyclosporin A, and intravenous pulse cyclophosphamide for acute/subacute interstitial pneumonia in patients with dermatomyositis. J Rheumatol 32:1719–1726
Kotani T, Takeuchi T, Makino S, Hata K, Yoshida S, Nagai K et al (2011) Combination with corticosteroids and cyclosporin-A improves pulmonary function test results and chest HRCT findings in dermatomyositis patients with acute/subacute interstitial pneumonia. Clin Rheumatol 30:1021–1028
Nagasaka K, Harigai M, Tateishi M, Hara M, Yoshizawa Y, Koike T et al (2003) Efficacy of combination treatment with cyclosporin A and corticosteroids for acute interstitial pneumonitis associated with dermatomyositis. Mod Rheumatol 13:231–238
Marie I, Hatron PY, Dominique S, Cherin P, Mouthon L, Menard J-F et al (2012) Short-term and long-term outcome of anti-Jo1-positive patients with anti-Ro52 antibody. Semin Arthritis Rheum 41:890–899
Hamaguchi Y, Kuwana M, Hoshino K, Hasegawa M, Kaji K, Matsushita T et al (2011) Clinical correlations with dermatomyositis-specific autoantibodies in adult Japanese patients with dermatomyositis: a multicenter cross-sectional study. Arch Dermatol 147:391–398
Mimori T, Nakashima R, Hosono Y (2012) Interstitial lung disease in myositis: clinical subsets, biomarkers, and treatment. Curr Rheumatol Rep 14:264–274
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Eun Bong Lee has acted as a consultant to Pfizer. Other authors have no conflicts of interest to declare.
Rights and permissions
About this article
Cite this article
Go, D.J., Park, J.K., Kang, E.H. et al. Survival benefit associated with early cyclosporine treatment for dermatomyositis-associated interstitial lung disease. Rheumatol Int 36, 125–131 (2016). https://doi.org/10.1007/s00296-015-3328-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00296-015-3328-8