Abstract
Cancer cells are metabolically reprogrammed to support their high rates of proliferation, continuous growth, survival, invasion, metastasis, and resistance to cancer treatments. Among changes in cancer cell bioenergetics, the role of glutamine metabolism has been receiving increasing attention. Increased glutaminolysis in cancer cells is associated with increased expression of membrane transporters that mediate the cellular uptake of glutamine. ASCT2 (Alanine, Serine, Cysteine Transporter 2) is a Na+-dependent transmembrane transporter overexpressed in cancer cells and considered to be the primary transporter for glutamine in these cells. The possibility of inhibiting ASCT2 for antineoplastic therapy is currently under investigation. In this article, we will present the pharmacological agents currently known to act on ASCT2, which have been attracting attention in antineoplastic therapy research. We will also address the impact of ASCT2 inhibition on the prognosis of some cancers. We conclude that ASCT2 inhibition and combination of ASCT2 inhibitors with other anti-tumor therapies may be a promising antineoplastic strategy. However, more research is needed in this area.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cancer is presently a highly prevalent pathology and, despite improvements in diagnostic and medical treatment, including the development of new therapies, the mortality rates associated with this condition are still very high. Therefore, it is fundamental to find new anti-tumor drugs with new mechanisms of action to improve cancer prognosis.
Cancer cells are metabolically reprogrammed to support their high proliferative ratio [1, 2]. This characteristic, which is one of the cancer hallmarks, is essential to provide the energy needs and biosynthetic precursors and for maintenance of redox homeostasis of tumor cells [1]. Indeed, metabolic reprogramming is a mechanism of adaptation of cancer cells, allowing them to develop and evolve, competing with other cells for glucose and other nutrients [1, 2]. Of importance, many of the metabolic changes observed in cancer cells are associated with a poor outcome [1, 2]. Thus, interference with metabolic reprogramming mechanisms of cancer cells constitutes a new hotspot in antineoplastic research.
It is known that glucose metabolism plays a very important role in tumor prognosis, but the role of glutamine metabolism in cancer cells is receiving a crescent attention [3]. Glutamine is a nutritionally non-essential amino acid with a very important role in cancer. It is not only a source of energy production and nucleotide/protein biosynthesis, but also possesses other effects in cancer cells (see below) that establish this nutrient as playing an important role in cancer cells, contributing to malignancy [4,5,6]. Thus, knowledge/modulation of glutamine metabolism can be useful, both in the diagnosis, monitoring, and cancer therapy [5].
To meet their high glutamine needs, cancer cells develop mechanisms that allow them to take up high amounts of this amino acid, namely by increasing the expression of membrane transporters that mediate the cellular uptake of glutamine [7]. ASCT2 (Alanine, Serine, Cysteine Transporter 2; encoded by SLC1A5 gene), a Na+-dependent transmembrane transporter involved in the cellular uptake of neutral amino acids such as glutamine, is a primary transporter of glutamine in cancer cells [3]. There is overexpression of ASCT2 in many cancer cells, and ASCT2 appears to have a role in tumorigenesis and in the outcome of malignant neoplasia [3, 8,9,10,11,12,13]. In agreement with this important role of ASCT2 in glutamine uptake by cancer cells, silencing ASCT2 was shown to interfere with cancer cells proliferation and survival, thus improving prognosis [3]. Furthermore, silencing ASCT2 appears to improve response to other antineoplastic therapies [3]. Therefore, ASCT2 is currently attracting a lot of attention in oncology research, and the possibility of using it as a therapeutic target has been investigated [3, 8,9,10,11,12,13].
In this article, we will describe the pharmacological agents that act on ASCT2, which has been attracting a lot of attention in antineoplastic investigation. We will also address the impact of silencing/inhibition of this glutamine transporter on the prognosis of some cancers.
Glutamine metabolism and its functions in cancer cells
Glutamine is a very important amino acid in our body, being the main amino acid flowing in the bloodstream [14]. This nutrient has been receiving a growing attention in cancer research, due to its role in cancer cells and on cancer initiation and progression. Indeed, cancer cells have a high glutamine demand (they use 10–100 times more glutamine than any other amino acid) and become glutamine “addicted”, as glutamine withdrawal can cause cell death [15]. This amino acid performs numerous important functions in cancer cells. It is an important energy source because, in most cancer cells, less pyruvate is oxidized in the mitochondria and an increase in the conversion of glucose to lactate is observed—the Warburg effect—and so other energy sources, including glutamine, branched-chain amino acids, and fatty acids, are used [4, 11, 16, 17]. Additionally, glutamine is also involved in redox homeostasis, cell signaling, apoptosis inhibition, and in autophagy [4,5,6]. In addition, cancer cell biosynthesis is enhanced to support their increased proliferation [18,19,20], and glutamine is a source of carbon and nitrogen, allowing molecules such as amino acids, proteins, nucleotides, and fatty acids, to be synthesized [18, 21, 22], as next described.
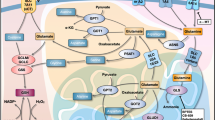
Glutaminolysis, which is increased in cancer cells, involves a set of reactions beginning with glutamine (Fig. 1) [21]. After entering the cells, glutamine is hydrolyzed by glutaminases (GLS/GLS2), within the mitochondria, being converted into ammonia and glutamate [18]. Silencing or inhibition of GLS (whose expression correlates with cancer growth and malignancy in rodents) delays tumor growth in both in vitro and animal models [5], including breast cancer and B lymphoma [23] and glioblastoma [24]. Importantly, the GLS inhibitor telaglenastat improved progression-free survival in patients with advanced renal cell carcinoma and is currently in clinical trials for advanced renal cell carcinoma and for non-small cell lung cancer (NSCLC) with mutations that activate the NRF2/KEAP1 pathway [25].
Overview of glutamine metabolism in cancer cells. Glutamine is converted into ammonia and glutamate by glutaminases (GLS/GLS2) [18]. In turn, from glutamate it is possible to produce serine, which, in turn, allows glycine and cysteine to be synthesized. Alternatively, the combination of glutamate, glycine, and cysteine allows the synthesis of glutathione [19, 28], or glutamate can be converted to α-ketoglutarate, an intermediate of the TCA cycle [18, 21, 26], from which malate or citrate can be obtained. Malate can be expelled from the mitochondria, being converted to pyruvate and lactate (NAPDH is produced during these reactions) [20, 27]. Citrate can also be expelled from the mitochondria and it permits both lipid and α-ketoglutarate (in association with NADPH production) syntheses [20, 27]. So, glutamine is a source of nitrogen, allowing amino acids to be synthesized (alanine, serine, proline, aspartate, among others [19]) through its conversion to glutamate, and by transamination reactions [18, 20]. In addition to amino acids, glutamine also allows nucleotides, proteins, and lipids to be synthesized [18, 21, 22]. Taken from [19]. ACC acetyl-CoA carboxylase, AKG α-ketoglutarate, AS asparagine synthetase, ALT alanine transaminase, Ac-CoA acetyl-coenzyme A, AST aspartate transaminase, CS citrate synthase, ACO aconitase, G6P glucose-6-phosphate, FH fumarate hydratase, GCS gamma-glutamylcysteine synthetase, GFAT glutamine:fructose-6-phosphate aminotransferase, F6P fructose-6-phosphate, Gln glutamine, GLS glutaminase isoenzyme, GLS2 glutaminase 2 isoenzyme, Glc glucose, Gln glutamine, GLUD glutamate dehydrogenase, GS glutamine synthetase, IDH-1 cytosolic isocitrate dehydrogenase, GSH glutathione, HIF-1 hypoxia-inducible factor-1, IDH-2 mitochondrial isocitrate dehydrogenase, KGDH ketoglutarate dehydrogenase, LRH-1 nuclear receptor liver receptor homolog 1, LDH lactate dehydrogenase, ME malic enzyme, MDH malate dehydrogenase, NF-κB nuclear factor-kappa B, OAA oxaloacetate, mTOR mammalian target of rapamycin, OXPHOS oxidative phosphorylation, ODC ornithine decarboxylase, PC pyruvate carboxylase, PDH pyruvate dehydrogenase, PKM2 pyruvate kinase, M2 isoform, ROS reactive oxygen species, PPP pentose phosphate pathway, SDH succinate dehydrogenase, SCS succinyl-coenzyme A-synthetase, STAT1 signal transducer and activator of transcription 1
In turn, from glutamate, it is possible to produce serine, which, in turn, allows glycine and cysteine to be synthesized; glutamate also allows other non-essential amino acids (alanine, proline, aspartate, among others [19]) to be synthesized through transamination reactions [18, 20]. Alternatively, glutamate can be converted to α-ketoglutarate, an intermediate of the Krebs cycle [18, 19, 21, 26], that can be converted into malate and citrate [19, 27, 28]. Malate can be effluxed from the mitochondria, and then converted into pyruvate [20, 27]. From pyruvate, lactate (and NADH) can be produced. Citrate, in turn, can also be removed from the mitochondria, leading to generation of acetyl-CoA (which is essential to form fatty acids and cholesterol) and α-ketoglutarate (in association with NADPH production) in the cytosol [20, 27]. In addition, combination of glutamate, glycine, and cysteine allows the synthesis of glutathione (an antioxidant neutralizing reactive oxygen species, ROS) [19, 28], by the action of glutamate cysteine ligase [18].
So, glutamine metabolism is important for redox balance in cancer cells [18, 21] through glutathione and NADPH synthesis [29]. Consequently, inhibition of glutamine metabolism originates higher ROS levels, increasing cancer cell apoptosis [3]. Moreover, glutamine is also a source of nitrogen, as its γ-nitrogen can be used for the synthesis of aspartate, asparagine, hexosamines, and nucleotides [18]. So, in addition to its role in energy production, glutamine is considered both a carbon source and a nitrogen donor, being involved in the biosynthesis of other amino acids and proteins, nucleotides, fatty acids, cholesterol, and glutathione (Fig. 1) [18, 21, 22]. All of these functions of glutamine are very important for cancer progression. Consequently, interference with its metabolism may constitute a new therapeutic target in cancer.
Glutamine cellular uptake: ASCT2 as a primary transporter of glutamine in cancer cells
Glutamine is a nutrient that has been receiving crescent attention for its role in antineoplastic therapy. As shown in the previous section, glutamine is essential for tumor development, as glutaminolysis is stimulated in cancer cells and cancer cells have an increased demand for this nutrient [4, 7, 30, 31]. This amino acid can be endogenously synthesized by cancer cells [18, 21, 26]. Alternatively, glutamine can be taken up from the extracellular space [4] and, thus, its cellular inlet becomes essential for tumor cells [4]. Glutamine is transported across the plasma membrane using different transporters [30], and there is an overexpression of transporters for this amino acid in cancer cells [7], as shown next.
Amino acid transporters are grouped into transport systems (e.g., system A, ASC, N, and L), depending on the substrates for which they have greater specificity and on their Na+ dependence or Na+ independence, among other differentiating characteristics [32].
In normal (non-cancerous) cells, glutamine transporter expression varies depending on the tissue [33,34,35,36,37,38]. For instance, SNAT1 (SLC38A1) is a glutamine transporter expressed in the neocortex, hippocampus, and neuroepithelium, and it provides glutamine to neurons, contributing to neuronal glutamate synthesis [38]. On the other hand, SNAT3 (SLC38A3) is expressed in astrocytes (which produce glutamine), allowing them to release glutamine [37]. Thus, both SNAT1 and SNAT3 are involved in brain glutamine transport and in glutamine–glutamate cycling: SNAT3 allows the release of glutamine by astrocytes, and SNAT1 allows neurons to take up glutamine [37, 38]. SNAT3 is also expressed in hepatic and renal tissues, allowing periportal cells and proximal renal tubule cells to obtain glutamine, and liver perivenous cells to release glutamine [33, 35, 36]. Additionally, SNAT2 (SLC38A2) is widely expressed in our body, playing very important roles [34]. It is associated with glutamine uptake and mTOR pathway signaling (contributing to cell growth and differentiation) [34, 39, 40].
As previously mentioned, although glutamine transporters exist in normal cells [33,34,35,36,37,38], their expression is increased in some cancers [7]. ASCT2 (Alanine, Serine, Cysteine Transporter 2) is a Na+-dependent transmembrane transporter, transporting glutamine and other neutral amino acids across the plasma membrane [41]. These other neutral amino acids include serine, cysteine, valine, threonine, and alanine [11, 41, 42]. ASCT2 is considered an obligatory exchanger of neutral amino acids and it is associated with both glutamine uptake and glutamine efflux [8, 43]. ASCT2 belongs to the SLC1A family (solute carrier 1A) [8], being encoded by the SLC1A5 gene (solute-linked carrier family A1 member 5), and it is considered a primary transporter for glutamine uptake in cancer cells [41], including breast cancer cells [44]. The SLC1A family of transporters includes ASCT1, ASCT2 (neutral amino acid transporters), and the EAAT1-5 transporters (excitatory amino acid transporters, which are associated with glutamate transport) [8].
ASCT2 (which is overexpressed in cancer cells) accounts for the inlet of glutamine into the cell, favoring tumorigenesis and tumor progression with consequent metastasis [45], and is associated with a poor prognosis [3]. Consequently, inhibition of ASCT2 may prevent tumor proliferation by interfering with glutamine influx [45,46,47,48,49]. So, we will focus on ASCT2 (SLC1A5) as a possible target for the development of new antineoplastic therapies, due to its notable role in glutamine uptake by cancer cells.
However, other transporters are also involved in glutamine transport by cancer cells. One of these transporters is LAT1 [50]. LAT1 (SLC7A5) is a transporter responsible for leucine uptake into the cells [3]. LAT1 allows an exchange of glutamine with leucine, allowing leucine to enter the intracellular space and glutamine, in turn, to pass into the extracellular space (Fig. 1) [3, 26, 50]. LAT1-mediated leucine uptake into the intracellular space allows activation of mTOR, being associated with tumor proliferation and, so, indirectly, glutamine allows mTOR activation [3, 6, 51]. LAT1 and CD98 constitute a heterodimeric complex, but whether CD98 is necessary for the transport function of this complex, or is only responsible for LAT1 membrane insertion, remains controversial [50, 52,53,54], although the activity of mTORC1 appears to be dependent on LAT1, but not CD98 [54]. Since LAT1 action depends on the intracellular concentration of glutamine (because it acts as an exchanger), there is an association between LAT1 and ASCT2 activities [3]. Both ASCT2 and LAT1 expression are independent prognostic factors in different cancers, and according to El Ansari et al., the combined expression of ASCT2 and LAT1 seems to influence breast cancer outcome [55].
Besides ASCT2 and LAT1, glutamine uptake may also involve other transporters. In relation to breast cancer cells, for instance, the following transporters are also able to transport glutamine across the plasma membrane: (a) SLC6A14 (also known as ATB0,+), known to be upregulated in ER-positive breast cancer cell lines [56]; (b) SLC3A1 (also known as rBAT), a Na+-independent transporter of cystine and neutral and dibasic amino acids, reported to be associated with breast cancer tumorigenesis [57]; (c) SLC7A7 (also known as y + LAT1), which mediates the influx of dibasic amino acids in a Na+-independent manner and is highly overexpressed in triple-negative breast cancer cells (MDA-MB-231 cells) [58] and in HER2 positive breast cancer cell lines [59]; (d) SLC7A8, a Na+-independent, large neutral amino acid transporter 2 (LAT2) that was reported as a predictive biomarker of good response to endocrine therapy in ER-positive breast cancer [60], and (e) SLC38A1, also known as Na+-coupled neutral amino acid transporter 1 (SNAT1), which is upregulated in breast cancer cell lines and human breast cancer tissues [61], and which overexpression is related to tumor size, nodal metastasis, and advanced tumor stage [62].
Although other glutamine transporters may be therapeutic targets for some cancers [34], ASCT2 is crucial for its primary and essential role in glutamine transport in several different cancers, and the therapeutic role of ASCT2 inhibition in some cancers has been well documented, as shown below [3, 9,10,11,12,13, 63, 64].
ASCT2 regulation in cancer cells
Tumor cells overexpress ASCT2. However, the mechanisms that regulate this increased expression are yet not completely understood, and more research is needed. Nevertheless, the following mechanisms have been described to regulate ASCT2.
Glutamine
Glutamine regulates the expression of ASCT2 [8]. According to Bungard et al., glutamine deprivation declined both ASCT2 expression, as well as tumor growth in a hepatoma (HepG2) cell line [65]. On the other hand, adding glutamine and, consequently, increasing glutamine availability, increased ASCT2 promoter activity and ASCT2 expression, favoring tumor growth [65]. Similarly, Dolinska et al. verified that in C6 glioma cells, glutamine deprivation was associated with lower ASCT2-mediated glutamine uptake [66]. Glutamine appears to regulate ASCT2 expression in cancer cells either by interfering with its transcription or at post-transcriptional level [8].
C-MYC
The MYC family of transcription factors is associated, in cancer, with nutrient uptake, metabolism, and proliferation. MYC activates the expression of some genes related to the acquisition and metabolism of glutamine, namely ASCT2 [67, 68].
Rb tumor suppressor
Retinoblastoma (Rb) tumor suppressor family can also regulate ASCT2 [68]. The entry of glutamine into the cell can be negatively regulated by Rb tumor suppressor, whose deletion enhances ASCT2-mediated glutamine uptake and upregulates GLS1 (via an E2F-dependent manner) [68].
microRNA-137 (miR-137)
This microRNA acts on ASCT2 mRNA, promoting its degradation or inhibiting its translation, and consequently decreasing ASCT2 levels [69, 70], glutamine uptake, and metabolism. Interestingly, MYC is associated with microRNA downregulation [67].
RNF5
Another regulator of ASCT2 is RNF5 [71]. Paclitaxel is a stress factor for breast cancer, promoting ubiquitination, RNF5 association, and a reduction of ASCT2 levels [71]. Accordingly, RNF5 deletion is associated with greater expression of ASCT2, which is related to a worse prognosis and less response to paclitaxel in breast cancer [71].
Leptin
An in vitro study using a human colon adenocarcinoma cell line (Caco-2 cells) evaluated the impact of leptin on the uptake of glucose and amino acids by these cells [72]. In relation to glutamine, this study demonstrated that, after the addition of leptin, uptake of glutamine rapidly decreased, together with a decrease in membrane ASCT2 protein expression [72]. Thus, leptin appears to inhibit glutamine uptake by downregulating ASCT2 [72]. Similarly, leptin was also found to decrease ASCT2-mediated glutamine uptake by both ER-positive and triple-negative breast cancer cell lines (Silva et al. unpublished).
Insulin
A study using rat adipocytes showed that both insulin and cell swelling stimulate ASCT2-mediated glutamine uptake [73]. In addition, insulin increased glutamine uptake not only directly, but also indirectly, through increased cell swelling [73]. The mechanism by which insulin increased ASCT2-mediated glutamine transport involves activation of the ERK/MAPK cascade pathway, suggesting that activation of the ERK pathway contributes to the regulation of ASCT2 by insulin [73]. In contrast, insulin appears to decrease ASCT2-mediated glutamine uptake by breast cancer cell lines (Silva et al. unpublished).
Interferon-γ and oxidative stress
In breast cancer cell lines, ASCT2-mediated uptake of glutamine was increased by INF-γ and by the oxidative stress inducer tert-butyl hydroperoxide (Silva et al. unpublished). Because higher levels of these parameters are associated with obesity/type 2 diabetes, this effect may contribute to the known higher prevalence of breast cancer in obese/type 2 diabetic women [74].
ASCT2 silencing/inhibition strategies
Currently, glutamine metabolism is seen as a target for cancer therapy development [3]. Glutamine has an essential role, both in the development and progression of the tumor [1, 2]. This amino acid is particularly important for cancer cells, compared to normal cells [4]. It is associated with metabolic reprogramming, allowing cells to adapt to their demands [3]. By interfering with glutamine metabolism, it is possible to inhibit one of the mechanisms of tumor adaptation. Therefore, inhibiting glutamine metabolism seems to be a good therapeutic strategy [3, 9].
Several strategies to inhibit glutamine metabolism have been investigated. One of the strategies consists of inhibiting transmembrane transporters involved in glutamine cellular uptake. In this context, ASCT2 is considered as a primary glutamine transporter and ASCT2 inhibitors have been developed [8], using distinct strategies: neutral amino acid analogs, which act as competitive inhibitors [8]; monoclonal antibodies targeting ASCT2 [9, 12, 13]; non-competitive ASCT2 inhibitors [75]; and strategies causing ASCT2 downregulation [10, 64] (Fig. 2). However, only a few ASCT2 inhibitors exist presently, and those are described below.
Mechanisms to inhibit ASCT2. Several ASCT2 inhibitors have been developed. Nevertheless, most of the existing inhibitory drugs are not sufficiently potent or selective [8]. GPNA inhibits several transporters related to glutamine metabolism, such as ASCT2, LAT1, SNAT1, and SNAT [79, 80]; V-9302 does not act by inhibiting ASCT2, but rather SNAT2, another glutamine transporter [81]; monoclonal antibodies recognizing ASCT2 appear to have a therapeutic role in cancer, slowing tumor growth and proliferation [9, 13]; δ-tocotrienol inhibits not only ASCT2, but also LAT1 and the mTOR pathway [63]; miRNA-137 acts on ASCT2 mRNA, promoting its degradation and inhibiting its translation [69, 70]
Benzyl-serine/benzyl-cysteine derivatives
Benzyl-serine/benzyl-cysteine derivatives are competitive inhibitors of ASCT2 [76]. They bind to ASCT2, competing for the binding site with neutral amino acids, that are transported [76]. Consequently, these drugs prevent the transport of neutral amino acids through ASCT2 [76]. However, benzyl-serine reduces, significantly, both glutamine and leucine uptake in melanoma cells [48] and breast cancer cells (MDA-MB-231, MCF-7, and HCC1806 cells) in vitro [77]. Therefore, benzyl-serine is not a specific ASCT2 inhibitor, as it also blocks the leucine transporter LAT1 [77].
Phenyl-glycine analogs
These compounds are not ASCT2-specific inhibitors, inhibiting also other amino acid transporters such as ASCT1 [78]. They were used for targeting both ASCT1 and ASCT2 in rat visual cortex slices [78].
L-γ-glutamyl-p-nitroanilide (GPNA)
It is an analog of glutamine [8, 79]. GPNA inhibits several transporters related to glutamine metabolism, such as ASCT2, LAT1, SNAT1, and SNAT2, in several human cancer cell lines (Fig. 2) [79, 80]. So, it is not specific for ASCT2.
Monoclonal antibodies (mAb)
Monoclonal antibodies are being created to recognize ASCT2, interfering with its function or promoting its internalization [9, 13]. Hara et al. developed a monoclonal antibody that recognizes ASCT2, trying to understand if resorting to this therapeutic strategy would improve KRAS-mutated cancer prognosis [9]. They verified that it would be a good strategy for KRAS-mutated cancer treatment, improving its outcome and being promising, because cancers with this mutation have few effective pharmacological treatments [9]. Ab 3-8 mAb binds to ASCT2, promoting its internalization in SW1116 and HCT116 colon cancer cell lines harboring KRAS mutation [9]. Consequently, Ab 3-8 mAb reduces glutamine uptake, and inhibits in vivo tumor growth of these cells in athymic mice, by promoting ASCT2 internalization (Fig. 2) [9]. This monoclonal antibody was also found to bind ASCT2 in colon cancer cells without a KRAS mutation (HT29 cells) [9]. However, contrary to what happens in mutated cancer cells, Ab3-8 mAb does not cause ASCT2 internalization in cells not harboring KRAS mutation (HT29 cells) [9]. The reason for non-internalization of ASCT2 in these non-mutated cancer cells is unknown [9]. Additionally, Suzuki et al. also developed mAbs recognizing ASCT2 (Fig. 2) [13]. They isolated KM4008, KM4012, and KM4018 mAb, and found that they bind to cells expressing ASCT2, mainly recognizing EL2 (an extracellular domain of ASCT2) [13]. KM8094 mAb was found to suppress gastric cancer cell growth in vitro [12]. KM8094 appears to decrease tumor growth by inhibiting glutamine uptake, increasing oxidative stress and inducing antibody-dependent cellular cytotoxicity in gastric tumor cells [12]. So, monoclonal antibodies recognizing glutamine transporters appear to have a therapeutic role in cancer, slowing tumor growth and proliferation [9, 13]. Monoclonal antibodies have some advantages, being selective and causing other effects such as antibody-dependent cellular cytotoxicity [12, 13].
microRNA-137 (miR-137)
This compound suppresses tumor development by interfering with ASCT2 [69]. More specifically, decreasing miR-137 transcription increases ASCT2 expression and, consequently, glutamine uptake and metabolism and cancer cell proliferation in vitro and in vivo [69]. Thus, there is an inverse association between miR-137 and ASCT2 [69].
1, 2, 3-Dithiazoles
According to a study carried out on proteoliposomes, these compounds appear to inhibit ASCT2 [75]. It is a non-competitive inhibition with a covalent interaction between these compounds and cysteine thiol groups [75]. This non-competitive inhibition is advantageous because it is associated with a longer duration of inhibition [75].
Others
In addition to these strategies, some drugs already known for other functions also seem to interfere with ASCT2-mediated glutamine transport. These include (a) topotecan, an inhibitor of DNA topoisomerase I (Topo I) which prevents both the replication of DNA and RNA synthesis, causing the death of malignant cells. It is used to treat non-small cell lung cancer and ovarian cancer [64]. Despite its anti-tumor effect acting as a Topo I inhibitor, according to a study that evaluated the impact of this compound on gastric cancer cell metabolism, topotecan also downregulates ASCT2, reducing the entry of glutamine into the cell, decreasing proliferation and increasing oxidative stress and apoptosis in a gastric cancer cell line in vitro [64]; (b) resveratrol, which downregulates ASCT2 in human hepatocellular carcinoma cell lines [10]; and (c) δ-tocotrienol, which inhibits not only ASCT2, but also LAT1 and the mTOR pathway in non-small cell lung cancer cell lines [63] (Fig. 2).
V-9302
This compound was initially presented as an ASCT2 inhibitor, decreasing glutamine uptake with a higher potency than GPNA, both in vitro and in mice [47]. Currently, it is known that this molecule does not act by inhibiting ASCT2, but rather SNAT2, another glutamine transporter, and LAT1 (Fig. 2) [81]. Combining V-0302 with an ASCT2 inhibitor more drastically decreases glutamine uptake in some cancer cell lines, such as head and neck squamous carcinoma cell lines [3]. Nevertheless, caution is needed when combining an ASCT2 inhibitor with V-9302 because non-malignant cells also use glutamine, triggering adverse effects [3].
So, it is necessary to develop new strategies to inhibit ASCT2 since most of the existing inhibitory drugs are not sufficiently potent or selective [8].
Impact of ASCT2 silencing/inhibition in different cancers
As next described, ASCT2 is associated with growth/proliferation, survival, autophagy, and apoptosis of several cancer cell lines in vitro (Table 1), and with tumor development and disease progression and outcome of several cancers (namely gastric cancer, colorectal carcinoma, breast cancer, prostate cancer, non-small cell lung cancer, head and neck squamous cell carcinoma, and liver cancer) in animal models (Table 2) [3, 42, 82,83,84,85,86]. So, ASCT2 inhibition can be a therapeutic strategy, mainly if associated with other therapies, by improving the response to them [3].
Head and neck squamous cell carcinoma (HNSCC)
Several studies have demonstrated an association between glutamine and the development and progression of HNSCC [3, 87, 88].
According to Zhang et al., ASCT2 inhibition reduced intracellular glutamine levels and significantly decreased the survival and growth of HNSCC tumor cells (SCC15 and FaDu cell lines), both in vitro and in vivo (human xenograft that was transfected to mice) (Tables 1 and 2) [3]. Moreover, tumor cells in which ASCT2 was silenced responded more effectively to cetuximab [3]. Cetuximab (an anti-EGFR monoclonal antibody approved for HNSCC treatment) acts on EGFR, which is expressed at high levels in HNSCC [3, 89, 90]. Interestingly, ASCT2 forms a complex with EGFR and AP1G1 [3, 89, 90]. Thus, cetuximab, by acting on EGFR (causing EGFR endocytosis), also targets ASCT2, decreasing glutamine transport into the cells [3, 89, 90]. Zhang et al. used three different strategies to inhibit ASCT2: ASCT2 silencing with shRNA, miR-137, and shRNA combined with V-9302. All strategies decreased, significantly, glutamine uptake. However, combining ASCT2 silencing with V-9302 (a SNAT2 inhibitor) further reduced the entry of glutamine into the cells (Table 1) [3]. It was suggested that ASCT2 is not the unique transporter accounting for glutamine uptake in HNSCC and that SNAT2 (which is inhibited by V-9302) is also involved in this process and, probably, it is induced when ASCT2 is silenced to compensate the entry of glutamine into the cancer cell [3, 30].
Thus, based on the actual knowledge, glutamine cellular uptake and its metabolism appear to be essential for the development and progression of HNSCC [3]. Moreover, inhibiting ASCT2 and combining ASCT2 inhibitors and cetuximab may constitute an effective strategy for HNSCC treatment [3].
Colorectal carcinoma (CRC)
Suzuki et al. developed monoclonal antibodies recognizing ASCT2 (KM4008, KM4012, and KM4018 mAbs). They found that these antibodies inhibited the proliferation of colorectal cancer cells (WiDr cells), in vitro, by blocking glutamine uptake (Table 1) [13]. Therefore, ASCT2 inhibition could have an anti-tumor effect on colorectal carcinoma [13].
CRC is associated with several mutations, including KRAS mutation [9]. KRAS mutation occurs in several cancers (CRC, non-small-cell lung cancer, and pancreatic ductal adenocarcinoma) [91,92,93,94]. KRAS (codified by an oncogene and, when mutated, it contributes to cancer development) is a GTPase, which interferes with MAPK signaling pathway, contributing to cancer development, by propagating the signal from the extracellular environment to the nucleus and increasing the expression of proteins involved in tumorigenesis and cell proliferation [95]. It is necessary to create new treatments for tumors with this mutation since there are a limited number of effective drugs [9]. A very recent work investigated whether ASCT2 inhibition could interfere with the outcome of KRAS-mutated tumors [9]. It was verified that, in CRC cells with KRAS mutation, Ab3-8 mAb (a monoclonal antibody that targets human ASCT2) reduced the entry of glutamine into the cells in vitro (Table 1) and tumor development in vivo (Table 2) [9]. On the other hand, in CRC cells without KRAS mutation, Ab3-8 mAb was not effective in decreasing cell growth (Table 1) [9]. So, interference with the entry of glutamine into the intracellular space seems to be a good strategy to treat KRAS-mutated CRC [9].
Leukemia
The role of ASCT2 in hematopoiesis and in the development of leukemia is being investigated [11]. According to Ni et al., silencing of ASCT2 has a slight impact on hematopoiesis, in the absence of a stress factor, but, in contrast, it decreases the development and progression of leukemia [11]. According to these authors, the role of ASCT2 in tumorigenesis is not entirely justified for its association with glutamine transport, and other amino acids that are transported by ASCT2 are also important [11]. Moreover, although ASCT2 inhibition had a different impact on malignant and non-malignant cells, mainly interfering with the malignant ones, it was concluded that caution is necessary, when combining ASCT2 inhibition with chemotherapy that damages DNA [11]. This is because in the presence of the myelotoxic agent fluorouracil (5-FU), both in in vitro and in vivo models, glutamine starts to play an important role in hematopoiesis, even in non-malignant cells, and, consequently, combining these two therapies can be associated with adverse consequences (Tables 1 and 2) [11].
Gastric cancer
A new monoclonal antibody recognizing ASCT2—KM8094—appears to improve gastric cancer prognostic, causing cytotoxicity and suppressing tumor growth both in vitro (Table 1) and in vivo (using human xenograft-transfected mice) (Table 2) [12]. Moreover, combination of M8094 with docetaxel (an antineoplastic therapy used in gastric cancer) improves tumor outcome in vivo (Table 2) [12]. KM8094 was found to decrease the cellular entry of glutamine and intracellular glutathione levels, resulting in an increase in oxidative stress levels in vitro (Table 1) [12].
Similar results were obtained with the ASCT2 inhibitor GPNA or ASCT2 knockdown [64]. These treatments reduced glutamine uptake, decreasing glutathione production, increasing ROS level and significantly suppressing tumor cell growth, both in vitro (Table 1) and in vivo (Table 2) [64]. ASCT2 knockdown also induced apoptosis via the mitochondrial pathway [64].
Topotecan is a Topo I inhibitor, preventing DNA replication and RNA synthesis and being indicated to treat some cancers [64]. However, topotecan appears to perform anti-tumor effects on gastric cancer cells both by acting as a Topo I inhibitor and by interfering with cancer metabolism [64]. More specifically, it was found to cause ASCT2 downregulation and to reduce glutamine metabolism in vitro (Table 1), and a reduction of gastric cancer growth in vivo (Table 2) [64].
Hepatocellular carcinoma (HCC)
HCC is associated with a poor outcome [96]. Consequently, developing new therapeutic strategies is necessary. ASCT2 expression is increased in HCC cells and ASCT2 upregulation is directly related to tumor size and outcome in humans [96]. So, ASCT2-based therapies may be of interest for this type of cancer.
One of the most effective chemotherapeutic agents for HCC treatment is cisplatin [10]. However, some people with HCC relapse due to resistance to cisplatin [10]. Interestingly, the dietary polyphenol resveratrol was found to decrease ASCT2 expression and cell proliferation, potentiating cisplatin-induced cytotoxicity by causing apoptosis (Table 1) [10].
Non-small cell lung cancer (NSCLC)
ASCT2 expression is increased and directly related to tumor size, disease stage, lymphatic and vascular invasion, metastasis, and prognosis in patients with pulmonary adenocarcinoma [86]. So, ASCT2-based therapies may be also interesting for this type of cancer. In support of this, δ-tocotrienol was recently found to inhibit the glutamine transporters ASCT2 and LAT1, which prevents glutamine uptake into cancer cells, inhibiting the in vitro proliferation of malignant cells and increasing apoptosis, associated with mTOR pathway downregulation (Table 1) [63].
Clear cell renal cell carcinoma (ccRCC)
An increased expression of ASCT2 (SLC1A5) is related to a more advanced TNM (tumor, node, metastases) stage, higher Fuhrman degree and worst outcomes, in patients with ccRCC [85]. So, ASCT2 is an independent prognostic factor in ccRCC [85].
Breast cancer
The expression of glutamine metabolism-related proteins was found to differ according to human breast cancer phenotype, with ASCT2 expression being the highest in HER2 type (hormone-receptor negative (ER- and PR-negative) and HER2 positive) and lowest in luminal A type (ER- and/or PR-positive) and HER2 negative) human breast cancers [83]. However, our group recently verified that GPNA reduced glutamine uptake in both MCF-7 (ER + , PR + , and HER2−) and MDA-MB-231 (ER−, PR−, and HER2−) cells to a similar degree (by ± 70%) (Silva et al. submitted) (Table 1). Nevertheless, ASCT2 inhibition appears a promising strategy also in this type of cancer.
Other cancer types
Besides the cancer types mentioned above, targeting glutamine metabolism improves the prognosis of other cancers, including epithelial ovarian cancer and tongue cancer [97, 98].
Discussion and conclusions
Metabolic reprogramming is one of the hallmarks of cancer and favors tumor progression, being a mechanism for malignant cells adaptation [1, 2]. It is fundamental to provide the energy needs and biosynthetic precursors and to decrease oxidative stress levels in tumor cells [1]. To compensate for these greatest demands, an increase in the expression of some transmembrane transporters, including transporters for the nutrient glutamine, is observed in most cancer cells [3]. ASCT2 is a primary transporter of glutamine, being upregulated in malignant cells [3, 7]. So, the possibility of inhibiting this transporter, and consequently decreasing the entry of glutamine into the cell and its subsequent metabolism, has been investigated, and strategies have been developed targeting ASCT2 [3, 13, 47, 79,80,81].
As reviewed here, inhibition of glutamine cellular uptake, by inhibition/silencing of ASCT2, appears to reduce cancer cell proliferation and survival in vitro and tumor development, progression, disease progression and outcome in vivo. This association was verified for several distinct cancers, namely gastric cancer, colorectal carcinoma, breast cancer, prostate cancer, non-small cell lung cancer, head and neck squamous cell carcinoma, liver cancer, among others [3, 42, 82,83,84,85,86]. Thus, inhibiting glutamine uptake and its metabolism, through ASCT2 inhibition/silencing, appears to be a promising strategy in antineoplastic therapy [3, 9,10,11,12,13, 63, 64].
ASCT2 inhibitors have been developed via different strategies: neutral amino acid analogs, acting as competitive inhibitors [8]; monoclonal antibodies targeting ASCT2 [9, 12, 13]; non-competitive ASCT2 inhibitors [75]; and other compounds causing ASCT2 downregulation [10, 64]. However, several limitations are presently known for research in this area, and these must be overcome in order that the full impact of ASCT2 in cancer therapy may be ascertained. First, the ASCT2 inhibitors currently known are not sufficiently potent or selective for this transporter, blocking not only ASCT2 but also other amino acid transporters [8]. These characteristics embarrass assessment of ASCT2 inhibition effects on cancer because the outcomes obtained do not result from an effective or selective inhibition of this transporter [8]. So, developing ASCT2-targeting therapeutic strategies to treat cancer has been hampered by the lack of effective and selective inhibitors [8]. Second, the development of this therapeutic strategy is troubled by the fact that different membrane transporters carry the same amino acids (in the specific case of glutamine, it is transported not only by ASCT2, but also by other transporters such as LAT1 and ASCT1 [8]). Moreover, and related to this fact, some reports concluded that ablation of ASCT2 resulted in an increased expression of other glutamine transporters in some cancers, namely SNAT1 and SNAT2, which are also able to mediate glutamine cellular uptake [99], and that this can be sufficient to supply the tumor needs, compensating for the silencing of ASCT2 [30]. So, cancer cells which have the ability to increase the expression of other glutamine transporters are less vulnerable to ASCT2 inhibition [99]. Third, in addition to its role in cancer, ASCT2 is also expressed in non-malignant tissues in our body: brain, lung, intestine, kidney, and skeletal muscle [8, 100, 101]. So, inhibition of ASCT2 might result in unwanted negative effects in non-cancer cells. Fourth, the role attributed to glutamine and, consequently, to ASCT2, appears to be different, depending on the tumor type [4, 100]. This may be due to not only the different origins of the tissues undergoing malignancy but also the tumor microenvironment [4, 100].
Despite these limitations, silencing/inhibition of ASCT2 appears to improve the outcome of different cancers by itself [3, 9, 11,12,13]. So, we believe that targeting ASCT2 may have a hopeful effect on the prognosis of different cancers, by interfering with the uptake and consequent metabolism of glutamine [1, 4, 7, 8, 29, 81]. Additionally, the combination of other anti-tumor therapies with ASCT2 inhibitors may be a very effective strategy to treat some cancers, improving cancer cell response to treatment, and this should be further investigated [3, 12]. So, more research is needed in this area, and of particular interest, the development of more effective and selective drugs to inhibit ASCT2 is a crucial topic [8].
Abbreviations
- AKG:
-
α-Ketoglutarate
- ASCT2:
-
Alanine, Serine, Cysteine Transporter 2
- CRC:
-
Colorectal carcinoma
- ccRCC:
-
Clear cell renal cell carcinoma
- ER:
-
Estrogen receptor
- Gln:
-
Glutamine
- GS:
-
Glutamine synthetase
- GLUD:
-
Glutamate dehydrogenase
- GLS:
-
Glutaminase isoenzyme
- GLS2:
-
Glutaminase 2 isoenzyme
- GSH:
-
Glutathione
- HCC:
-
Hepatocellular carcinoma
- HNSCC:
-
Head and neck squamous cell carcinoma
- LAT1:
-
Large amino acid transporter
- mTOR:
-
Mammalian target of rapamycin
- NSCLC:
-
Non-small cell lung cancer
- ROS:
-
Reactive oxygen species
- SNAT:
-
Sodium-coupled neutral amino acid transporter
- SLC1A5:
-
Solute-linked carrier family A1 member 5
References
DeBerardinis RJ, Chandel NS (2016) Fundamentals of cancer metabolism. Sci Adv 2(5):e1600200. https://doi.org/10.1126/sciadv.1600200
Vander Heiden MG, DeBerardinis RJ (2017) Understanding the intersections between metabolism and cancer biology. Cell 168(4):657–669. https://doi.org/10.1016/j.cell.2016.12.039
Zhang Z, Liu R, Shuai Y, Huang Y, Jin R, Wang X, Luo J (2020) ASCT2 (SLC1A5)-dependent glutamine uptake is involved in the progression of head and neck squamous cell carcinoma. Br J Cancer 122(1):82–93. https://doi.org/10.1038/s41416-019-0637-9
Altman BJ, Stine ZE, Dang CV (2016) From Krebs to clinic: glutamine metabolism to cancer therapy. Nat Rev Cancer 16(11):749. https://doi.org/10.1038/nrc.2016.114
Hensley CT, Wasti AT, DeBerardinis RJ (2013) Glutamine and cancer: cell biology, physiology, and clinical opportunities. J Clin Invest 123(9):3678–3684. https://doi.org/10.1172/JCI69600
Villar VH, Merhi F, Djavaheri-Mergny M, Duran RV (2015) Glutaminolysis and autophagy in cancer. Autophagy 11(8):1198–1208. https://doi.org/10.1080/15548627.2015.1053680
Bhutia YD, Babu E, Ramachandran S, Ganapathy V (2015) Amino acid transporters in cancer and their relevance to “glutamine addiction”: novel targets for the design of a new class of anticancer drugs. Cancer Res 75(9):1782–1788. https://doi.org/10.1158/0008-5472.CAN-14-3745
Freidman N, Chen I, Wu Q, Briot C, Holst J, Font J, Vandenberg R, Ryan R (2020) Amino acid transporters and exchangers from the SLC1A family: structure, mechanism and roles in physiology and cancer. Neurochem Res. https://doi.org/10.1007/s11064-019-02934-x
Hara Y, Minami Y, Yoshimoto S, Hayashi N, Yamasaki A, Ueda S, Masuko K, Masuko T (2020) Anti-tumor effects of an antagonistic mAb against the ASCT2 amino acid transporter on KRAS-mutated human colorectal cancer cells. Cancer Med 9(1):302–312. https://doi.org/10.1002/cam4.2689
Liu Z, Peng Q, Li Y, Gao Y (2018) Resveratrol enhances cisplatin-induced apoptosis in human hepatoma cells via glutamine metabolism inhibition. BMB Rep 51(9):474–479
Ni F, Yu WM, Li Z, Graham DK, Jin L, Kang S, Rossi MR, Li S, Broxmeyer HE, Qu CK (2019) Critical role of ASCT2-mediated amino acid metabolism in promoting leukaemia development and progression. Nat Metab 1(3):390–403. https://doi.org/10.1038/s42255-019-0039-6
Osanai-Sasakawa A, Hosomi K, Sumitomo Y, Takizawa T, Tomura-Suruki S, Imaizumi M, Kasai N, Poh TW, Yamano K, Yong WP, Kono K, Nakamura S, Ishii T, Nakai R (2018) An anti-ASCT2 monoclonal antibody suppresses gastric cancer growth by inducing oxidative stress and antibody dependent cellular toxicity in preclinical models. Am J Cancer Res 8(8):1499–1513
Suzuki M, Toki H, Furuya A, Ando H (2017) Establishment of monoclonal antibodies against cell surface domains of ASCT2/SLC1A5 and their inhibition of glutamine-dependent tumor cell growth. Biochem Biophys Res Commun 482(4):651–657. https://doi.org/10.1016/j.bbrc.2016.11.089
Scriver CR, Rosenberg LE (1973) Amino acid metabolism and its disorders. Major Probl Clin Pediatr 10:1–478
Nguyen T-L, Durán RV (2018) Glutamine metabolism in cancer therapy. Cancer Drug Resist 1:126–138. https://doi.org/10.20517/cdr.2018.08
DeNicola GM, Cantley LC (2015) Cancer’s fuel choice: new flavors for a picky eater. Mol Cell 60(4):514–523. https://doi.org/10.1016/j.molcel.2015.10.018
Pavlova NN, Thompson CB (2016) The emerging hallmarks of cancer metabolism. Cell Metab 23(1):27–47. https://doi.org/10.1016/j.cmet.2015.12.006
Still ER, Yuneva MO (2017) Hopefully devoted to Q: targeting glutamine addiction in cancer. Br J Cancer 116(11):1375–1381. https://doi.org/10.1038/bjc.2017.113
Mates JM, Di Paola FJ, Campos-Sandoval JA, Mazurek S, Marquez J (2020) Therapeutic targeting of glutaminolysis as an essential strategy to combat cancer. Semin Cell Dev Biol 98:34–43. https://doi.org/10.1016/j.semcdb.2019.05.012
Li C, Zhang G, Zhao L, Ma Z, Chen H (2016) Metabolic reprogramming in cancer cells: glycolysis, glutaminolysis, and Bcl-2 proteins as novel therapeutic targets for cancer. World J Surg Oncol 14(1):15. https://doi.org/10.1186/s12957-016-0769-9
Chen JQ (1826) Russo J (2012) Dysregulation of glucose transport, glycolysis, TCA cycle and glutaminolysis by oncogenes and tumor suppressors in cancer cells. Biochim Biophys Acta 2:370–384. https://doi.org/10.1016/j.bbcan.2012.06.004
Lane AN, Fan TW (2015) Regulation of mammalian nucleotide metabolism and biosynthesis. Nucleic Acids Res 43(4):2466–2485. https://doi.org/10.1093/nar/gkv047
Wang JB, Erickson JW, Fuji R, Ramachandran S, Gao P, Dinavahi R, Wilson KF, Ambrosio AL, Dias SM, Dang CV, Cerione RA (2010) Targeting mitochondrial glutaminase activity inhibits oncogenic transformation. Cancer Cell 18(3):207–219. https://doi.org/10.1016/j.ccr.2010.08.009
Cheng T, Sudderth J, Yang C, Mullen AR, Jin ES, Mates JM, DeBerardinis RJ (2011) Pyruvate carboxylase is required for glutamine-independent growth of tumor cells. Proc Natl Acad Sci USA 108(21):8674–8679. https://doi.org/10.1073/pnas.1016627108
CALITHERA (2020) Glutaminase Inhibitor Telaglenastat (CB-839). https://www.calithera.com/glutaminase-inhibitor-cb-839/. Accessed 19 Nov 2020
Marchiq I, Pouyssegur J (2016) Hypoxia, cancer metabolism and the therapeutic benefit of targeting lactate/H(+) symporters. J Mol Med (Berl) 94(2):155–171. https://doi.org/10.1007/s00109-015-1307-x
DeBerardinis RJ, Mancuso A, Daikhin E, Nissim I, Yudkoff M, Wehrli S, Thompson CB (2007) Beyond aerobic glycolysis: transformed cells can engage in glutamine metabolism that exceeds the requirement for protein and nucleotide synthesis. Proc Natl Acad Sci U S A 104(49):19345–19350. https://doi.org/10.1073/pnas.0709747104
DeBerardinis RJ, Cheng T (2010) Q’s next: the diverse functions of glutamine in metabolism, cell biology and cancer. Oncogene 29(3):313–324. https://doi.org/10.1038/onc.2009.358
Daye D, Wellen KE (2012) Metabolic reprogramming in cancer: unraveling the role of glutamine in tumorigenesis. Semin Cell Dev Biol 23(4):362–369. https://doi.org/10.1016/j.semcdb.2012.02.002
Zhang J, Pavlova NN, Thompson CB (2017) Cancer cell metabolism: the essential role of the nonessential amino acid, glutamine. EMBO J 36(10):1302–1315. https://doi.org/10.15252/embj.201696151
Jin L, Alesi GN, Kang S (2016) Glutaminolysis as a target for cancer therapy. Oncogene 35(28):3619–3625. https://doi.org/10.1038/onc.2015.447
Christensen HN (1990) Role of amino acid transport and countertransport in nutrition and metabolism. Physiol Rev 70(1):43–77. https://doi.org/10.1152/physrev.1990.70.1.43
Chan K, Busque SM, Sailer M, Stoeger C, Broer S, Daniel H, Rubio-Aliaga I, Wagner CA (2016) Loss of function mutation of the Slc38a3 glutamine transporter reveals its critical role for amino acid metabolism in the liver, brain, and kidney. Pflugers Arch 468(2):213–227. https://doi.org/10.1007/s00424-015-1742-0
Ge Y, Gu Y, Wang J (1860) Zhang Z (2018) Membrane topology of rat sodium-coupled neutral amino acid transporter 2 (SNAT2). Biochim Biophys Acta Biomembr 7:1460–1469. https://doi.org/10.1016/j.bbamem.2018.04.005
Gu S, Roderick HL, Camacho P, Jiang JX (2000) Identification and characterization of an amino acid transporter expressed differentially in liver. Proc Natl Acad Sci USA 97(7):3230–3235. https://doi.org/10.1073/pnas.050318197
Haussinger D, Schliess F (2007) Glutamine metabolism and signaling in the liver. Front Biosci 12:371–391. https://doi.org/10.2741/2070
Todd AC, Marx MC, Hulme SR, Broer S, Billups B (2017) SNAT3-mediated glutamine transport in perisynaptic astrocytes in situ is regulated by intracellular sodium. Glia 65(6):900–916. https://doi.org/10.1002/glia.23133
Weiss MD, Derazi S, Rossignol C, Varoqui H, Erickson JD, Kilberg MS, Anderson KJ (2003) Ontogeny of the neutral amino acid transporter SAT1/ATA1 in rat brain. Brain Res Dev Brain Res 143(2):151–159. https://doi.org/10.1016/s0165-3806(03)00107-x
Evans K, Nasim Z, Brown J, Butler H, Kauser S, Varoqui H, Erickson JD, Herbert TP, Bevington A (2007) Acidosis-sensing glutamine pump SNAT2 determines amino acid levels and mammalian target of rapamycin signalling to protein synthesis in L6 muscle cells. J Am Soc Nephrol 18(5):1426–1436. https://doi.org/10.1681/ASN.2006091014
Pinilla J, Aledo JC, Cwiklinski E, Hyde R, Taylor PM, Hundal HS (2011) SNAT2 transceptor signalling via mTOR: a role in cell growth and proliferation? Front Biosci (Elite Ed) 3:1289–1299. https://doi.org/10.2741/e332
Kekuda R, Prasad PD, Fei YJ, Torres-Zamorano V, Sinha S, Yang-Feng TL, Leibach FH, Ganapathy V (1996) Cloning of the sodium-dependent, broad-scope, neutral amino acid transporter Bo from a human placental choriocarcinoma cell line. J Biol Chem 271(31):18657–18661. https://doi.org/10.1074/jbc.271.31.18657
Fuchs BC, Bode BP (2005) Amino acid transporters ASCT2 and LAT1 in cancer: partners in crime? Semin Cancer Biol 15(4):254–266. https://doi.org/10.1016/j.semcancer.2005.04.005
Broer A, Wagner C, Lang F, Broer S (2000) Neutral amino acid transporter ASCT2 displays substrate-induced Na+ exchange and a substrate-gated anion conductance. Biochem J 346(Pt 3):705–710
Cha YJ, Kim ES, Koo JS (2018) Amino acid transporters and glutamine metabolism in breast cancer. Int J Mol Sci. https://doi.org/10.3390/ijms19030907
Lu J, Chen M, Tao Z, Gao S, Li Y, Cao Y, Lu C, Zou X (2017) Effects of targeting SLC1A5 on inhibiting gastric cancer growth and tumor development in vitro and in vivo. Oncotarget 8(44):76458–76467. https://doi.org/10.18632/oncotarget.19479
Marshall AD, van Geldermalsen M, Otte NJ, Lum T, Vellozzi M, Thoeng A, Pang A, Nagarajah R, Zhang B, Wang Q, Anderson L, Rasko JEJ, Holst J (2017) ASCT2 regulates glutamine uptake and cell growth in endometrial carcinoma. Oncogenesis 6(7):e367. https://doi.org/10.1038/oncsis.2017.70
Schulte ML, Fu A, Zhao P, Li J, Geng L, Smith ST, Kondo J, Coffey RJ, Johnson MO, Rathmell JC, Sharick JT, Skala MC, Smith JA, Berlin J, Washington MK, Nickels ML, Manning HC (2018) Pharmacological blockade of ASCT2-dependent glutamine transport leads to antitumor efficacy in preclinical models. Nat Med 24(2):194–202. https://doi.org/10.1038/nm.4464
Wang Q, Beaumont KA, Otte NJ, Font J, Bailey CG, van Geldermalsen M, Sharp DM, Tiffen JC, Ryan RM, Jormakka M, Haass NK, Rasko JE, Holst J (2014) Targeting glutamine transport to suppress melanoma cell growth. Int J Cancer 135(5):1060–1071. https://doi.org/10.1002/ijc.28749
Wang Q, Hardie RA, Hoy AJ, van Geldermalsen M, Gao D, Fazli L, Sadowski MC, Balaban S, Schreuder M, Nagarajah R, Wong JJ, Metierre C, Pinello N, Otte NJ, Lehman ML, Gleave M, Nelson CC, Bailey CG, Ritchie W, Rasko JE, Holst J (2015) Targeting ASCT2-mediated glutamine uptake blocks prostate cancer growth and tumour development. J Pathol 236(3):278–289. https://doi.org/10.1002/path.4518
Napolitano L, Scalise M, Galluccio M, Pochini L, Albanese LM, Indiveri C (2015) LAT1 is the transport competent unit of the LAT1/CD98 heterodimeric amino acid transporter. Int J Biochem Cell Biol 67:25–33. https://doi.org/10.1016/j.biocel.2015.08.004
Jewell JL, Kim YC, Russell RC, Yu FX, Park HW, Plouffe SW, Tagliabracci VS, Guan KL (2015) Metabolism. Differential regulation of mTORC1 by leucine and glutamine. Science 347(6218):194–198. https://doi.org/10.1126/science.1259472
Fort J, de la Ballina LR, Burghardt HE, Ferrer-Costa C, Turnay J, Ferrer-Orta C, Uson I, Zorzano A, Fernandez-Recio J, Orozco M, Lizarbe MA, Fita I, Palacin M (2007) The structure of human 4F2hc ectodomain provides a model for homodimerization and electrostatic interaction with plasma membrane. J Biol Chem 282(43):31444–31452. https://doi.org/10.1074/jbc.M704524200
Wagner CA, Lang F, Broer S (2001) Function and structure of heterodimeric amino acid transporters. Am J Physiol Cell Physiol 281(4):C1077-1093. https://doi.org/10.1152/ajpcell.2001.281.4.C1077
Salisbury TB, Arthur S (2018) The regulation and function of the L-type amino acid transporter 1 (LAT1) in cancer. Int J Mol Sci. https://doi.org/10.3390/ijms19082373
El-Ansari R, Craze ML, Alfarsi L, Soria D, Diez-Rodriguez M, Nolan CC, Ellis IO, Rakha EA, Green AR (2019) The combined expression of solute carriers is associated with a poor prognosis in highly proliferative ER+ breast cancer. Breast Cancer Res Treat 175(1):27–38. https://doi.org/10.1007/s10549-018-05111-w
Karunakaran S, Ramachandran S, Coothankandaswamy V, Elangovan S, Babu E, Periyasamy-Thandavan S, Gurav A, Gnanaprakasam JP, Singh N, Schoenlein PV, Prasad PD, Thangaraju M, Ganapathy V (2011) SLC6A14 (ATB0,+) protein, a highly concentrative and broad specific amino acid transporter, is a novel and effective drug target for treatment of estrogen receptor-positive breast cancer. J Biol Chem 286(36):31830–31838. https://doi.org/10.1074/jbc.M111.229518
Jiang Y, Cao Y, Wang Y, Li W, Liu X, Lv Y, Li X, Mi J (2017) Cysteine transporter SLC3A1 promotes breast cancer tumorigenesis. Theranostics 7(4):1036–1046. https://doi.org/10.7150/thno.18005
Leo JC, Wang SM, Guo CH, Aw SE, Zhao Y, Li JM, Hui KM, Lin VC (2005) Gene regulation profile reveals consistent anticancer properties of progesterone in hormone-independent breast cancer cells transfected with progesterone receptor. Int J Cancer 117(4):561–568. https://doi.org/10.1002/ijc.21186
Sweet R, Paul A, Zastre J (2010) Hypoxia induced upregulation and function of the thiamine transporter, SLC19A3 in a breast cancer cell line. Cancer Biol Ther 10(11):1101–1111. https://doi.org/10.4161/cbt.10.11.13444
Thakkar A, Raj H, Ravishankar MB, Balakrishnan A, Padigaru M (2015) High expression of three-gene signature improves prediction of relapse-free survival in estrogen receptor-positive and node-positive breast tumors. Biomark Insights 10:103–112. https://doi.org/10.4137/BMI.S30559
Timmerman LA, Holton T, Yuneva M, Louie RJ, Padro M, Daemen A, Hu M, Chan DA, Ethier SP, Van ’t Veer LJ, Polyak K, McCormick F, Gray JW (2013) Glutamine sensitivity analysis identifies the xCT antiporter as a common triple-negative breast tumor therapeutic target. Cancer Cell 24(4):450–465. https://doi.org/10.1016/j.ccr.2013.08.020
Wang K, Cao F, Fang W, Hu Y, Chen Y, Ding H, Yu G (2013) Activation of SNAT1/SLC38A1 in human breast cancer: correlation with p-Akt overexpression. BMC Cancer 13:343. https://doi.org/10.1186/1471-2407-13-343
Rajasinghe LD, Hutchings M, Gupta SV (2019) Delta-tocotrienol modulates glutamine dependence by inhibiting ASCT2 and LAT1 transporters in non-small cell lung cancer (NSCLC) cells: a metabolomic approach. Metabolites. https://doi.org/10.3390/metabo9030050
Wang L, Liu Y, Zhao TL, Li ZZ, He JY, Zhang BJ, Du HZ, Jiang JW, Yuan ST, Sun L (2019) Topotecan induces apoptosis via ASCT2 mediated oxidative stress in gastric cancer. Phytomedicine 57:117–128. https://doi.org/10.1016/j.phymed.2018.12.011
Bungard CI, McGivan JD (2004) Glutamine availability up-regulates expression of the amino acid transporter protein ASCT2 in HepG2 cells and stimulates the ASCT2 promoter. Biochem J 382(Pt 1):27–32. https://doi.org/10.1042/BJ20040487
Dolinska M, Dybel A, Hilgier W, Zielinska M, Zablocka B, Buzanska L, Albrecht J (2001) Glutamine transport in C6 glioma cells: substrate specificity and modulation in a glutamine deprived culture medium. J Neurosci Res 66(5):959–966. https://doi.org/10.1002/jnr.10047
Gao P, Tchernyshyov I, Chang TC, Lee YS, Kita K, Ochi T, Zeller KI, De Marzo AM, Van Eyk JE, Mendell JT, Dang CV (2009) c-Myc suppression of miR-23a/b enhances mitochondrial glutaminase expression and glutamine metabolism. Nature 458(7239):762–765. https://doi.org/10.1038/nature07823
Reynolds MR, Lane AN, Robertson B, Kemp S, Liu Y, Hill BG, Dean DC, Clem BF (2014) Control of glutamine metabolism by the tumor suppressor Rb. Oncogene 33(5):556–566. https://doi.org/10.1038/onc.2012.635
Dong J, Xiao D, Zhao Z, Ren P, Li C, Hu Y, Shi J, Su H, Wang L, Liu H, Li B, Gao P, Qing G (2017) Epigenetic silencing of microRNA-137 enhances ASCT2 expression and tumor glutamine metabolism. Oncogenesis 6(7):e356. https://doi.org/10.1038/oncsis.2017.59
Luo M, Wu L, Zhang K, Wang H, Zhang T, Gutierrez L, O’Connell D, Zhang P, Li Y, Gao T, Ren W, Yang Y (2018) miR-137 regulates ferroptosis by targeting glutamine transporter SLC1A5 in melanoma. Cell Death Differ 25(8):1457–1472. https://doi.org/10.1038/s41418-017-0053-8
Jeon YJ, Khelifa S, Ratnikov B, Scott DA, Feng Y, Parisi F, Ruller C, Lau E, Kim H, Brill LM, Jiang T, Rimm DL, Cardiff RD, Mills GB, Smith JW, Osterman AL, Kluger Y, Ronai ZA (2015) Regulation of glutamine carrier proteins by RNF5 determines breast cancer response to ER stress-inducing chemotherapies. Cancer Cell 27(3):354–369. https://doi.org/10.1016/j.ccell.2015.02.006
Fanjul C, Barrenetxe J, Inigo C, Sakar Y, Ducroc R, Barber A, Lostao MP (2012) Leptin regulates sugar and amino acids transport in the human intestinal cell line Caco-2. Acta Physiol (Oxf) 205(1):82–91. https://doi.org/10.1111/j.1748-1716.2012.02412.x
Ritchie JW, Baird FE, Christie GR, Stewart A, Low SY, Hundal HS, Taylor PM (2001) Mechanisms of glutamine transport in rat adipocytes and acute regulation by cell swelling. Cell Physiol Biochem 11(5):259–270. https://doi.org/10.1159/000047812
Kang C, LeRoith D, Gallagher EJ (2018) Diabetes, obesity, and breast cancer. Endocrinology 159(11):3801–3812. https://doi.org/10.1210/en.2018-00574
Oppedisano F, Catto M, Koutentis PA, Nicolotti O, Pochini L, Koyioni M, Introcaso A, Michaelidou SS, Carotti A, Indiveri C (2012) Inactivation of the glutamine/amino acid transporter ASCT2 by 1,2,3-dithiazoles: proteoliposomes as a tool to gain insights in the molecular mechanism of action and of antitumor activity. Toxicol Appl Pharmacol 265(1):93–102. https://doi.org/10.1016/j.taap.2012.09.011
Grewer C, Grabsch E (2004) New inhibitors for the neutral amino acid transporter ASCT2 reveal its Na+-dependent anion leak. J Physiol 557(Pt 3):747–759. https://doi.org/10.1113/jphysiol.2004.062521
van Geldermalsen M, Quek LE, Turner N, Freidman N, Pang A, Guan YF, Krycer JR, Ryan R, Wang Q, Holst J (2018) Benzylserine inhibits breast cancer cell growth by disrupting intracellular amino acid homeostasis and triggering amino acid response pathways. BMC Cancer 18(1):689. https://doi.org/10.1186/s12885-018-4599-8
Foster AC, Rangel-Diaz N, Staubli U, Yang JY, Penjwini M, Viswanath V, Li YX (2017) Phenylglycine analogs are inhibitors of the neutral amino acid transporters ASCT1 and ASCT2 and enhance NMDA receptor-mediated LTP in rat visual cortex slices. Neuropharmacology 126:70–83. https://doi.org/10.1016/j.neuropharm.2017.08.010
Esslinger CS, Cybulski KA, Rhoderick JF (2005) N-gamma-aryl glutamine analogues as probes of the ASCT2 neutral amino acid transporter binding site. Bioorg Med Chem 13(4):1111–1118. https://doi.org/10.1016/j.bmc.2004.11.028
Chiu M, Sabino C, Taurino G, Bianchi MG, Andreoli R, Giuliani N, Bussolati O (2017) GPNA inhibits the sodium-independent transport system L for neutral amino acids. Amino Acids 49(8):1365–1372. https://doi.org/10.1007/s00726-017-2436-z
Broer A, Fairweather S, Broer S (2018) Disruption of amino acid homeostasis by novel ASCT2 inhibitors involves multiple targets. Front Pharmacol 9:785. https://doi.org/10.3389/fphar.2018.00785
Fuchs BC, Finger RE, Onan MC, Bode BP (2007) ASCT2 silencing regulates mammalian target-of-rapamycin growth and survival signaling in human hepatoma cells. Am J Physiol Cell Physiol 293(1):C55-63. https://doi.org/10.1152/ajpcell.00330.2006
Kim S, Kim DH, Jung WH, Koo JS (2013) Expression of glutamine metabolism-related proteins according to molecular subtype of breast cancer. Endocr Relat Cancer 20(3):339–348. https://doi.org/10.1530/ERC-12-0398
Liu P, Ge M, Hu J, Li X, Che L, Sun K, Cheng L, Huang Y, Pilo MG, Cigliano A, Pes GM, Pascale RM, Brozzetti S, Vidili G, Porcu A, Cossu A, Palmieri G, Sini MC, Ribback S, Dombrowski F, Tao J, Calvisi DF, Chen L, Chen X (2017) A functional mammalian target of rapamycin complex 1 signaling is indispensable for c-Myc-driven hepatocarcinogenesis. Hepatology 66(1):167–181. https://doi.org/10.1002/hep.29183
Liu Y, Yang L, An H, Chang Y, Zhang W, Zhu Y, Xu L, Xu J (2015) High expression of Solute Carrier Family 1, member 5 (SLC1A5) is associated with poor prognosis in clear-cell renal cell carcinoma. Sci Rep 5:16954. https://doi.org/10.1038/srep16954
Shimizu K, Kaira K, Tomizawa Y, Sunaga N, Kawashima O, Oriuchi N, Tominaga H, Nagamori S, Kanai Y, Yamada M, Oyama T, Takeyoshi I (2014) ASC amino-acid transporter 2 (ASCT2) as a novel prognostic marker in non-small cell lung cancer. Br J Cancer 110(8):2030–2039. https://doi.org/10.1038/bjc.2014.88
Kamarajan P, Rajendiran TM, Kinchen J, Bermudez M, Danciu T, Kapila YL (2017) Head and neck squamous cell carcinoma metabolism draws on glutaminolysis, and stemness is specifically regulated by glutaminolysis via aldehyde dehydrogenase. J Proteome Res 16(3):1315–1326. https://doi.org/10.1021/acs.jproteome.6b00936
Yang J, Guo Y, Seo W, Zhang R, Lu C, Wang Y, Luo L, Paul B, Yan W, Saxena D, Li X (2019) Targeting cellular metabolism to reduce head and neck cancer growth. Sci Rep 9(1):4995. https://doi.org/10.1038/s41598-019-41523-4
Lu H, Li X, Lu Y, Qiu S, Fan Z (2016) ASCT2 (SLC1A5) is an EGFR-associated protein that can be co-targeted by cetuximab to sensitize cancer cells to ROS-induced apoptosis. Cancer Lett 381(1):23–30. https://doi.org/10.1016/j.canlet.2016.07.020
Tao X, Lu Y, Qiu S, Wang Y, Qin J, Fan Z (2017) AP1G1 is involved in cetuximab-mediated downregulation of ASCT2-EGFR complex and sensitization of human head and neck squamous cell carcinoma cells to ROS-induced apoptosis. Cancer Lett 408:33–42. https://doi.org/10.1016/j.canlet.2017.08.012
Bryant KL, Mancias JD, Kimmelman AC, Der CJ (2014) KRAS: feeding pancreatic cancer proliferation. Trends Biochem Sci 39(2):91–100. https://doi.org/10.1016/j.tibs.2013.12.004
Karachaliou N, Mayo C, Costa C, Magri I, Gimenez-Capitan A, Molina-Vila MA, Rosell R (2013) KRAS mutations in lung cancer. Clin Lung Cancer 14(3):205–214. https://doi.org/10.1016/j.cllc.2012.09.007
Maus MK, Grimminger PP, Mack PC, Astrow SH, Stephens C, Zeger G, Hsiang J, Brabender J, Friedrich M, Alakus H, Holscher AH, Lara P, Danenberg KD, Lenz HJ, Gandara DR (2014) KRAS mutations in non-small-cell lung cancer and colorectal cancer: implications for EGFR-targeted therapies. Lung Cancer 83(2):163–167. https://doi.org/10.1016/j.lungcan.2013.11.010
Normanno N, Tejpar S, Morgillo F, De Luca A, Van Cutsem E, Ciardiello F (2009) Implications for KRAS status and EGFR-targeted therapies in metastatic CRC. Nat Rev Clin Oncol 6(9):519–527. https://doi.org/10.1038/nrclinonc.2009.111
Mai TT, Lito P (2018) A treatment strategy for KRAS-driven tumors. Nat Med 24(7):902–904. https://doi.org/10.1038/s41591-018-0111-x
Sun HW, Yu XJ, Wu WC, Chen J, Shi M, Zheng L, Xu J (2016) GLUT1 and ASCT2 as predictors for prognosis of hepatocellular carcinoma. PLoS ONE 11(12):e0168907. https://doi.org/10.1371/journal.pone.0168907
Bjersand K, Seidal T, Sundstrom-Poromaa I, Akerud H, Skirnisdottir I (2017) The clinical and prognostic correlation of HRNPM and SLC1A5 in pathogenesis and prognosis in epithelial ovarian cancer. PLoS ONE 12(6):e0179363. https://doi.org/10.1371/journal.pone.0179363
Toyoda M, Kaira K, Ohshima Y, Ishioka NS, Shino M, Sakakura K, Takayasu Y, Takahashi K, Tominaga H, Oriuchi N, Nagamori S, Kanai Y, Oyama T, Chikamatsu K (2014) Prognostic significance of amino-acid transporter expression (LAT1, ASCT2, and xCT) in surgically resected tongue cancer. Br J Cancer 110(10):2506–2513. https://doi.org/10.1038/bjc.2014.178
Broer A, Rahimi F, Broer S (2016) Deletion of amino acid transporter ASCT2 (SLC1A5) reveals an essential role for transporters SNAT1 (SLC38A1) and SNAT2 (SLC38A2) to sustain glutaminolysis in cancer cells. J Biol Chem 291(25):13194–13205. https://doi.org/10.1074/jbc.M115.700534
Cormerais Y, Massard PA, Vucetic M, Giuliano S, Tambutte E, Durivault J, Vial V, Endou H, Wempe MF, Parks SK, Pouyssegur J (2018) The glutamine transporter ASCT2 (SLC1A5) promotes tumor growth independently of the amino acid transporter LAT1 (SLC7A5). J Biol Chem 293(8):2877–2887. https://doi.org/10.1074/jbc.RA117.001342
Hashimoto Y, Sadamoto Y, Konno A, Kon Y, Iwanaga T (2004) Distribution of neutral amino acid transporter ASCT 1 in the non-neuronal tissues of mice. Jpn J Vet Res 52(3):113–124
Funding
F. Martel acknowledges funding from FCT—Fundação para a Ciência e a Tecnologia (Instituto de Investigação e Inovação em Saúde, Universidade do Porto, Portugal (UID/BIM/04293/2013)).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Teixeira, E., Silva, C. & Martel, F. The role of the glutamine transporter ASCT2 in antineoplastic therapy. Cancer Chemother Pharmacol 87, 447–464 (2021). https://doi.org/10.1007/s00280-020-04218-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00280-020-04218-6