Abstract
Epidemiological studies have assessed the association between Fc gamma receptor IIIA (FCGR3A) 158 V/F and the response to rituximab-based therapy in patients with non-Hodgkin lymphoma (NHL), but the findings have been inconsistent. We performed this meta-analysis to obtain a better assessment of this relationship. Electronic database searches were conducted for relevant studies. A pooled odds ratio (OR) with a 95 % confidence interval (95 % CI) was used to assess the strength of the association. Analyses of the subgroup and publication bias were conducted. A total of 10 studies involving 1050 patients were analyzed. In all the genetic models, no clear relationship was found between the FCGR3A 158 V/F polymorphism and the response to rituximab-based therapy in NHL patients. When categorized by ethnicity, Asian individuals with the FCGR3A 158 V/V allele (OR = 4.37; 95 % CI = 1.07–17.73; P = 0.039) or the non-F/(FV + VV) (OR = 2.50; 95 % CI = 1.04–5.98; P = 0.040) allele have a significantly higher complete response rate (CR) compared to FF individuals. No obvious heterogeneities were observed. In addition, no statistical evidence for a publication bias was found. Our study suggested that the FCGR3A 158 V/F polymorphism can predict the treatment response to rituximab-based chemotherapy in NHL patients, especially for Asian individuals.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Rituximab has been successfully used for the treatment of patients with non-Hodgkin’s lymphoma (NHL) [1, 2]. Rituximab is a chimeric Ab that was engineered by grafting the murine anti-human CD20 variable regions onto human FCG. Human FCG targets CD20+ B cells and depletes subpopulations of peripheral B cells via several putative mechanisms, including Ab-dependent cell cytotoxicity (ADCC) via NK cells, complement dependent lysis (CDL), and apoptosis via cross-linking membrane CD20 [3]. The Fc gamma receptors (FCGRs) play an important role in the recognition of immune complexes (ICs) [4]. The FCGR3A 158 V/F (rs396991) polymorphism exhibits biologic functions that differ among the different FCGR genotypes [5].
Given the crucial role played by the FCGR3A polymorphism in the pathogenesis of NHL, a number of studies have examined the potential contributions of the FCGR3A 158 V/F polymorphism on non-responsiveness to rituximab [6–15]. However, results remain inconsistent. This could be due to the possibility of small effects or relatively small sample sizes in the early publications. Therefore, the aim of the present study was to use a meta-analysis approach to investigate whether the functional FCGR3A 158 V/F polymorphism is associated with non-responsiveness to rituximab therapy in NHL patients.
Materials and methods
Literature search strategy
MEDLINE, EMBASE, and Cochrane Library searches were conducted using the following search terms: “FCγR” or “FCGR” and “polymorphism” and “Rituximab” (the last search update was on 30 July 2015). All identified studies were obtained and their reference lists were also checked for other relevant studies.
Inclusion and exclusion criteria
Studies published in languages other than English were excluded. Related articles, and also potentially relevant articles, were screened. Studies had to meet the following criteria to be eligible for the analysis: (1) involved patients with NHL that were treated with rituximab-based chemotherapy, (2) investigated the association between the FCGR3A 158 V/F polymorphism and the treatment response, i.e., overall response rate (ORR) (complete responders (CR) and partial responders (PR)) and non-responders (stable disease (SD) or progressive disease (PD)), and (3) was a published study with full text available.
Data extraction
The characteristics of selected studies were extracted using a standardized protocol conducted by two investigators independently (Duo Liu and Yuyang Tian). The following information was extracted: the surname of the first author, the year of publication, and the ethnicity, diagnosis, sample size, age group, gender, stages, chemotherapy, outcomes, and number of cases for each genotype of the FCGR3A 158 V/F polymorphism. For studies including subjects of different ethnic groups, data were extracted separately for each ethnic group whenever possible.
Statistical analysis and publication bias
The treatment response (TR) was used to measure chemotherapy efficacy. To summarize this information, patients were divided into responders (CR or PR) and non-responders (SD or PD) according to the WHO criteria [16] or the Response Evaluation Criteria in Solid Tumors (RECIST) criteria [17]. For FCGR3A 158 V/F, four genetic comparison models were analyzed (A: dominant model, FV + VV vs FF; B: heterozygote model, FV vs FF; C: homozygote model, VV vs FF; D: recessive model, VV vs FV + FF). The pooled odds ratio (OR) and the 95 % confidence interval (CI) were calculated for CR vs SD + PD, PR vs SD + PD, and ORR vs SD + PD. Because this study is a systematic review and meta-analysis, each eligible study had already been approved by local institutional review boards, and each local institution has obtained matching informed consent from their patients.
Heterogeneity was checked using the chi-square test based on the Cochran’s Q test, and a P value >0.1 indicated a lack of heterogeneity. The pooled OR estimation of each study was calculated with a random-effect model using the Der Simonian and Laird method or with a fixed-effect model using the Mantel-Haenszel method. The inter-study variance (I 2) was used to quantify the amount of heterogeneity between studies, and the percentage of I 2 was used to express the extent of explained heterogeneity of the characteristics [18]. The publication bias was checked with Begg’s test and Egger’s asymmetry test, and with visual inspection of the funnel plots, in which the standard error was plotted against the Log (OR) to produce a simple scatter plot. All of the statistical analyses were performed using STATA version 11.0 (Stata Corporation, College Station, TX). The P values were in relation to two-sided tests, and P > 0.05 was considered to be statistically significant.
Results
Characteristics of the studies
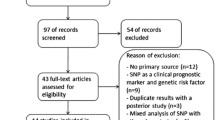
A total of 76 studies on relationship between the FCGR polymorphism and the response to rituximab-based chemotherapy were identified and screened for data retrieval. As shown in Fig. 1, 19 papers that did not involve NHL, 15 studies that did not involve SNP, and 7 studies relating to other FCGR polymorphisms were excluded. Fifteen studies [19–33] were also discarded because they lacked the clinical outcome data, and another three studies were discarded because they only reported rituximab-induced toxicity [34–36]. Furthermore, five articles were reviews [37–41], and two were not in English [42, 43], so these were also excluded. Each group was considered to be a separate study for the analysis.
Response to rituximab-based chemotherapy
There were 10 articles identified that match the criteria chosen for this analysis (Table 1). Three studies were conducted on Asian patients [6, 11, 13], and seven studies were conducted on European patients [7–10, 12, 14, 15]. The data sets that were included in this meta-analysis had a total sample size of 1050 patients. Table 2 shows the genotype frequency of the FCGR3A 158 V/F polymorphism with the response to rituximab-based chemotherapy. Furthermore, Table 3 summarizes the pooled OR estimates and the corresponding 95 % CI of this meta-analysis. When the data were analyzed altogether, no significant associations with the CR, PR rate, or ORR were found for the FF allele compared using all the genetic models.
Considering the potential impact of confounding factors, such as genetic variation in different ethnic groups, we conducted further subgroup analyses based on ethnicity. The data showed that Asian individuals with the FCGR3A 158 V/V (OR = 4.37; 95 % CI = 1.07–17.73; P = 0.039) (Fig. 2) and the non-F/(FV + VV) (OR = 2.50; 95 % CI =1.04–5.98; P = 0.040) (Fig. 3) allele have a significantly higher CR rate compared with the FF allele (Table 3).
Heterogeneity and publication bias
Heterogeneity analysis suggested that there were differences between the meta-analysis results and the actual results. As shown in Table 3, no obvious heterogeneities were found in the overall analysis of the data using all of the genetic models. However, when subgroup analyses based on ethnicity were conducted, we found heterogeneities in the Asian group with the homozygote comparison (P heterogeneity = 0.039, I 2 = 69.2 %).
Begg’s funnel plots were created and Egger’s tests were performed to appraise the publication bias among the studies selected for the meta-analysis. The shape of the funnel plots appeared symmetrical, and Egger’s regression tests showed no evidence of publication bias (P = 0.427 for VV vs FF) in the meta-analysis (Fig. 4).
Discussion
In this meta-analysis, we provided evidence that the FCGR3A FF genotype could be a low-penetrant risk factor in NHL patients. Furthermore, in subgroup analyses based on ethnicity, a significantly higher CR rate was observed for Asian populations under the homozygote comparison and the dominant model. To our knowledge, the present meta-analysis is the first to analyze the relationship between the FCGR3A 158 V/F polymorphism and the response to rituximab-based chemotherapy for NHL patients and uses meta-analysis to analyze data from ten published studies to be able to state a powerful conclusion.
Kim et al. [13] observed a significantly higher CR and ORR rate in the FCGR3A V/V allele, when compared with the FCGR3A V/F or F/F alleles, for Korean individuals. However, Liu et al. [6] suggested that the FCGR3A 158 V/F polymorphism did not affect the CR rate and the ORR of R-CHOP therapy, but there was no subgroup analysis conducted. Furthermore, Table 4 of Liu’s study [6] has little deviation from the included studies, and we have confirmed our findings by checking the data carefully. Another study that also used rituximab with CHOP had a much higher efficacy in patients with the V/V and V/F genotypes [11]. The study by Ahlgrimm et al. [32] did not include the response data, and therefore, it was not included in our study. Based on these studies, we added four additional articles [8, 9, 14, 15] to our analysis. Two French studies [9, 15] found an association between the FCGR3A polymorphism and the clinical and molecular responses to rituximab.
Cartron et al. found a disappearance of the BCL2-JH gene rearrangement in both peripheral blood and marrow was observed at 1 year (12 months) in 5 of 6 of FCGR3A 158VV genotype patients compared with 5 of 17 of FCGR3A 158FF carriers [15]. However, two other reports [8, 14] did not show that the FCGR3A polymorphism influences the response rate and outcome when rituximab is combined with chemotherapy or used as a maintenance treatment.
This meta-analysis also has some limitations that could influence the results slightly. First, in our study, no obvious heterogeneities were observed for the overall analysis, but heterogeneities were found in the analysis of the ethnicity subgroups. These heterogeneities could be due to differences in the frequency of genetic variations between the different groups. In the studies by Liu et al. [6] and Mitroviç et al. [13], the SD or PD rates for the carriers with the FF allele were 14.3 % (10/70) and 10 % (1/10), respectively. However, Zhang et al. [11] found that patients with the FF genotype had a 40 % (2/5) SD or PD rate, which was much higher than the percentage reported by the other two studies. When the number of studies is small, the power to be able to detect bias is low. Therefore, an increase in the number of studies included could improve the validity of the meta-analysis. Second, we only included studies published in English for the analysis, and some relevant reports have been published in other languages and in other electronic databases that we may have missed. However, we did not detect publication bias in our study. Third, our results were based on unadjusted estimates and were also not adjusted for potential confounds such as age, gender, smoking status, and other lifestyle risk factors, as information on potential confounding variables was unavailable.
In summary, the results of this systematic review and meta-analysis suggest that, in patients with non-Hodgkin lymphoma, there is an association in Asian individuals between carrying the FCGR3A 158 FF allele and a poor response to rituximab-based chemotherapy. However, future studies with larger sample sizes and suitable designs are needed to confirm these findings.
References
Feugier P, Van Hoof A, Sebban C, Solal-Celigny P, Bouabdallah R, Fermé C, Christian B, Lepage E, Tilly H, Morschhauser F, Gaulard P, Salles G, Bosly A, Gisselbrecht C, Reyes F, Coiffier B (2005) Long-term results of the R-CHOP study in the treatment of elderly patients with diffuse large B-cell lymphoma: a study by the Groupe d’ Etude des Lymphomes de l’Adulte. J Clin Oncol 23(18):4117–4126
Pfreundschuh M, Trümper L, Osterborg A, Pettengell R, Trneny M, Imrie K, Ma D, Gill D, Walewski J, Zinzani PL, Stahel R, Kvaloy S, Shpilberg O, Jaeger U, Hansen M, Lehtinen T, López-Guillermo A, Corrado C, Scheliga A, Milpied N, Mendila M, Rashford M, Kuhnt E, Loeffler M, MabThera International Trial Group (2006) CHOP-like chemotherapy plus rituximab versus CHOP-like chemotherapy alone in young patients with good-prognosis diffuse large-B cell lymphoma: a randomized controlled trial by the MabThera International Trial (MInT) Group. Lancet Oncol 7(5):379–391
Clynes RA, Towers TL, Presta LG, Ravetch JV (2000) Inhibitory Fc receptors modulate in vivo cytotoxicity against tumor targets. Nat Med 6(4):443–446
Ravetch JV, Bolland S (2001) IgG Fc receptors. Annu Rev Immunol 19:275–290, Review
Koene HR, Kleijer M, Algra J, Roos D, von dem Borne AE, de Haas M (1997) Fc gammaRIIIa-158 V/F polymorphism influences the binding of IgG by natural killer cell Fc gammaRIIIa, independently of the Fc gammaRIIIa-48L/R/H phenotype. Blood 90(3):1109–1114
Liu F, Ding H, Jin X, Ding N, Deng L, He Y, Zhu J, Song Y (2014) FCGR3A 158V/F polymorphism and response to frontline R-CHOP therapy in diffuse large B-cell lymphoma. DNA Cell Biol 33(9):616–623. doi:10.1089/dna.2013.2333
Váróczy L, Zilahi E, Gyetvai A, Kajtár B, Gergely L, Sipka S, Illés A (2012) Fc-gamma-receptor IIIa polymorphism and gene expression profile do not predict the prognosis in diffuse large B-cell lymphoma treated with R-CHOP protocol. Pathol Oncol Res 18(1):43–48. doi:10.1007/s12253-011-9414-7
Ghesquières H, Cartron G, Seymour JF, Delfau-Larue MH, Offner F, Soubeyran P, Perrot A, Brice P, Bouabdallah R, Sonet A, Dupuis J, Casasnovas O, Catalano JV, Delmer A, Jardin F, Verney A, Dartigues P, Salles G (2012) Clinical outcome of patients with follicular lymphoma receiving chemoimmunotherapy in the PRIMA study is not affected by FCGR3A and FCGR2A polymorphisms. Blood 120(13):2650–2657
Cornec D, Tempescul A, Querellou S, Hutin P, Pers JO, Jamin C, Bendaoud B, Berthou C, Renaudineau Y, Youinou P (2012) Identification of patients with indolent B cell lymphoma sensitive to rituximab monotherapy. Ann Hematol 91(5):715–721. doi:10.1007/s00277-011-1369-y
Fabisiewicz A, Paszkiewicz-Kozik E, Osowiecki M, Walewski J, Siedlecki JA (2011) FcγRIIA and FcγRIIIA polymorphisms do not influence survival and response to rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone immunochemotherapy in patients with diffuse large B-cell lymphoma. Leuk Lymphoma 52(8):1604–1606. doi:10.3109/10428194.2011.574760
Zhang W, Wang X, Li J, Duan MH, Zhou DB (2010) Fcgamma receptor IIIA polymorphisms and efficacy of rituximab therapy on Chinese diffuse large B-cell lymphoma. Chin Med J (Engl) 123(2):198–202
Mitroviç Z, Aurer I, Radman I, Ajdukoviç R, Sertiç J, Labar B (2007) FCgammaR IIIA and FCgammaR IIA polymorphisms are not associated with response to rituximaband CHOP in patients with diffuse large B-cell lymphoma. Haematologica 92(7):998–999
Kim DH, Jung HD, Kim JG, Lee JJ, Yang DH, Park YH, Do YR, Shin HJ, Kim MK, Hyun MS, Sohn SK (2006) FCGR3A gene polymorphisms may correlate with response to frontline R-CHOP therapy for diffuse large B-cell lymphoma. Blood 108(8):2720–2725
Galimberti S, Palumbo GA, Caracciolo F, Benedetti E, Pelosini M, Brizzi S, Ciabatti E, Fazzi R, Stelitano C, Quintana G, Conte E, Tibullo D, Di Raimondo F, Petrini M (2007) The efficacy of rituximab plus Hyper-CVAD regimen in mantle cell lymphoma is independent of FCgammaR IIIa and FCgammaR IIa polymorphisms. J Chemother 19(3):315–321
Cartron G, Dacheux L, Salles G, Solal-Celigny P, Bardos P, Colombat P, Watier H (2002) Therapeutic activity of humanized anti-CD20 monoclonal antibody and polymorphism in IgG Fc receptor FcgammaR IIIa gene. Blood 99(3):754–758
Miller AB, Hoogstraten B, Staquet M, Winkler A (1981) Reporting results of cancer treatment. Cancer 47(1):207–214
Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC, Gwyther SG (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92(3):205–216
Mantel N, Haenszel W (1959) Statistical aspects of the analysis of data from retrospective studies of disease. J Nat Cancer Inst 22(4):719–748
Burchard PR, Malhotra S, Kaur P, Tsongalis GJ (2013) Detection of the FCGR3a polymorphism using a real-time polymerase chain reaction assay. Cancer Genet 206(4):130–134. doi:10.1016/j.cancergen.2013.03.002
Mishima Y, Terui Y, Mishima Y, Kuniyoshi R, Matsusaka S, Mikuniya M, Kojima K, Hatake K (2012) High reproducible ADCC analysis revealed a competitive relation between ADCC and CDC and differences between FcγRIIIa polymorphism. Int Immunol 24(8):477–483. doi:10.1093/intimm/dxs048
Veeramani S, Wang SY, Dahle C, Blackwell S, Jacobus L, Knutson T, Button A, Link BK, Weiner GJ (2011) Rituximab infusion induces NK activation in lymphoma patients with the high-affinity CD16 polymorphism. Blood 118(12):3347–3349. doi:10.1182/blood-2011-05-351411
Ternant D, Cartron G, Hénin E, Tod M, Girard P, Paintaud G (2012) Model-based design of rituximab dosage optimization in follicular non-Hodgkin’s lymphoma. Br J Clin Pharmacol 73(4):597–605. doi:10.1111/j.1365-2125.2011.04125.x
Prochazka V, Papajik T, Gazdova J, Divoka M, Rozmanova S, Faber E, Raida L, Kucerova L, Langova K, Jarosova M, Indrak K (2011) FcγRIIIA receptor genotype does not influence an outcome in patients with follicular lymphoma treated with risk-adapted immunochemotherapy. Neoplasma 58(3):263–270
Ternant D, Cartron G, Hénin E, Tod M, Paintaud G, Girard P (2009) Development of a drug-disease simulation model for rituximab in follicular non-Hodgkin’s lymphoma. Br J Clin Pharmacol 68(4):561–573. doi:10.1111/j.1365-2125.2009.03494.x
Weng WK, Levy R (2009) Genetic polymorphism of the inhibitory IgG Fc receptor FcgammaRIIb is not associated with clinical outcome in patients with follicular lymphoma treated with rituximab. Leuk Lymphoma 50(5):723–727. doi:10.1080/10428190902829441
Lejeune J, Thibault G, Ternant D, Cartron G, Watier H, Ohresser M (2008) Evidence for linkage disequilibrium between Fcgamma RIIIa-V158F and Fcgamma RIIa-H131R polymorphisms in white patients, and for an Fcgamma RIIIa-restricted influence on the response to therapeutic antibodies. J Clin Oncol 26(33):5489–5491. doi:10.1200/JCO.2008.19.4118
Hatjiharissi E, Hansen M, Santos DD, Xu L, Leleu X, Dimmock EW, Ho AW, Hunter ZR, Branagan AR, Patterson CJ, Kortsaris A, Verselis S, Fox E, Treon SP (2007) Genetic linkage of Fc gamma RIIa and Fc gamma RIIIa and implications for their use in predicting clinical responses to CD20-directed monoclonal antibody therapy. Clin Lymphoma Myeloma 7(4):286–290
Hatjiharissi E, Xu L, Santos DD, Hunter ZR, Ciccarelli BT, Verselis S, Modica M, Cao Y, Manning RJ, Leleu X, Dimmock EA, Kortsaris A, Mitsiades C, Anderson KC, Fox EA, Treon SP (2007) Increased natural killer cell expression of CD16, augmented binding and ADCC activity to rituximab among individuals expressing the Fc{gamma}RIIIa-158 V/V and V/F polymorphism. Blood 110(7):2561–2564
Dall’Ozzo S, Tartas S, Paintaud G, Cartron G, Colombat P, Bardos P, Watier H, Thibault G (2004) Rituximab-dependent cytotoxicity by natural killer cells: influence of FCGR3A polymorphism on the concentration-effect relationship. Cancer Res 64(13):4664–4669
Weng WK, Levy R (2003) Two immunoglobulin G fragment C receptor polymorphisms independently predict response to rituximab in patients with follicular lymphoma. J Clin Oncol 21(21):3940–3947
Carlotti E, Palumbo GA, Oldani E, Tibullo D, Salmoiraghi S, Rossi A, Golay J, Pulsoni A, Foà R, Rambaldi A (2007) FcgammaRIIIA and FcgammaRIIA polymorphisms do not predict clinical outcome of follicular non-Hodgkin’s lymphoma patients treated with sequential CHOP and rituximab. Haematologica 92(8):1127–1130
Ahlgrimm M, Pfreundschuh M, Kreuz M, Regitz E, Preuss KD, Bittenbring J (2011) The impact of Fc-γ receptor polymorphisms in elderly patients with diffuse large B-cell lymphoma treated with CHOP with or without rituximab. Blood 118(17):4657–4662. doi:10.1182/blood-2011-04-346411
Persky DO, Dornan D, Goldman BH, Braziel RM, Fisher RI, Leblanc M, Maloney DG, Press OW, Miller TP, Rimsza LM (2012) Fc gamma receptor 3a genotype predicts overall survival in follicular lymphoma patients treated on SWOG trials with combined monoclonal antibody plus chemotherapy but not chemotherapy alone. Haematologica 97(6):937–942. doi:10.3324/haematol.2011.050419
Weng WK, Negrin RS, Lavori P, Horning SJ (2010) Immunoglobulin G Fc receptor FcgammaRIIIa 158 V/F polymorphism correlates with rituximab-induced neutropenia after autologous transplantation in patients with non-Hodgkin’s lymphoma. J Clin Oncol 28(2):279–284. doi:10.1200/JCO.2009.25.0274
Keane C, Nourse JP, Crooks P, Nguyen-Van D, Mutsando H, Mollee P, Lea RA, Gandhi MK (2012) Homozygous FCGR3A-158V alleles predispose to late onset neutropenia after CHOP-R for diffuse large B-cell lymphoma. Intern Med J 42(10):1113–1119. doi:10.1111/j.1445-5994.2011.02587.x
Li SC, Chen YC, Evens AM, Lee CC, Liao HF, Yu CC, Tung YT, Su YC (2010) Rituximab-induced late-onset neutropenia in newly diagnosed B-cell lymphoma correlates with Fc receptor FcγRIIIa 158(V/F) polymorphism. Am J Hematol 85(10):810–812. doi:10.1002/ajh.21818
Sarsour K, Greenberg J, Johnston JA, Nelson DR, O’Brien LA, Oddoux C, Ostrer H, Pearlman A, Reed G (2013) The role of the FcGRIIIa polymorphism in modifying the association between treatment and outcome in patients with rheumatoid arthritis treated with rituximab versus TNF-α antagonist therapies. Clin Exp Rheumatol 31(2):189–194
Cartron G (2009) FCGR3A polymorphism story: a new piece of the puzzle. Leuk Lymphoma 50(9):1401–1402. doi:10.1080/10428190903161109
Ternant D, Paintaud G (2005) Pharmacokinetics and concentration-effect relationships of therapeutic monoclonal antibodies and fusion proteins. Expert Opin Biol Ther 5(Suppl 1):S37–S47, Review
Zhuang Y, Xu W, Shen Y, Li J (2010) Fcγ receptor polymorphisms and clinical efficacy of rituximab in non-Hodgkin lymphoma and chronic lymphocytic leukemia. Clin Lymphoma Myeloma Leuk 10(5):347–352. doi:10.3816/CLML.2010.n.067
Treon SP (2010) Fcγ receptor predictive genomic testing and the treatment of indolent non-Hodgkin lymphoma. Clin Lymphoma Myeloma Leuk 10(5):321–322. doi:10.3816/CLML.2010.n.063
Zhuang Y, Qiao C, Yang G, Shen Y, Qian X, Yang L, Xu W, Li J (2014) FcγRIIIa polymorphisms and efficacy of Rituximab combined chemotherapy for diffuse large B-cell lymphoma in Chinese patients. Zhonghua Xue Ye Xue Za Zhi 35(9):816–821. doi:10.3760/cma.j.issn.0253-2727.2014.09.007
Zhang W, Wang X, Duan MH, Li J, Zhou DB (2009) Efficacy of rituximab therapy on diffuse large B-cell lymphoma with different Fcgamma RIIIA gene polymorphisms: a prospective study. Zhonghua Yi Xue Za Zhi 89(6):400–402
Acknowledgments
This work was supported by grants from the Fund of the Outstanding Youth foundation of Heilongjiang Province of China (Grant No. JC201215) and the Science and Technology Research Project of Chinese Ministry of Education (Grant No.213010A), Heilongjiang Province Education Department project (Grant No. 2012CJHB007).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Liu, D., Tian, Y., Sun, D. et al. The FCGR3A polymorphism predicts the response to rituximab-based therapy in patients with non-Hodgkin lymphoma: a meta-analysis. Ann Hematol 95, 1483–1490 (2016). https://doi.org/10.1007/s00277-016-2723-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00277-016-2723-x