Abstract
Purpose
To report an unusual case of combined Lie’s types A and D of internal carotid artery (ICA) agenesis, diagnosed by magnetic resonance angiography (MRA).
Methods
A 60-year-old woman with dizziness underwent cranial magnetic resonance imaging (MRI) and MRA of the intracranial region for the evaluation of brain and vascular lesions. The magnetic resonance machine was a 3.0-T scanner.
Results
MRI showed no abnormalities, except for multiple small white matter lesions. MRA showed that the left ICA was absent, except for the supraclinoid segment, and an anastomotic vessel was present between the paraclinoid segments of the bilateral ICAs, indicating Lie’s type D ICA agenesis. The left posterior communicating artery (PCoA) was also present. Thus, there were also features of type A ICA agenesis. The anastomotic vessels between the bilateral ICAs and ipsilateral PCoA were relatively small in caliber.
Conclusion
Lie’s type D ICA agenesis usually does not communicate with the anterior and posterior circulations. We encountered a case of combined type D and type A ICA agenesis. To our knowledge, no similar case has been reported in the English literature. This is the second case of type D ICA agenesis with patent ipsilateral PCoA. We speculate that in case of type A ICA agenesis, when the development of the PCoA is insufficient to support collateral blood flow, an anastomotic vessel between bilateral ICAs may develop.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Internal carotid artery (ICA) agenesis is a rare congenital abnormality. Lie [5] classified agenesis of the ICA into six types based on the dysplastic segment and collateral circulation of the ICA. However, ICA agenesis that does not fit Lie’s classification has been reported [3, 6, 9,10,11]. In this study, we present the case of an unusual patient with two types of collateral blood flow: anastomoses from the contralateral ICA and ipsilateral posterior communicating artery (PCoA) incidentally diagnosed by magnetic resonance angiography (MRA).
Case report
A 60-year-old woman presented with dizziness. Cranial magnetic resonance imaging (MRI) and MRA were performed to investigate the brain and cerebral vascular lesions.
The MR machine was a 3-Tesla scanner (Magnetom Skyra; Siemens Healthineers, Erlangen, Germany). MRA was performed using a standard three-dimensional time-of-flight technique. The imaging parameters were as follows: flip angle, 18°; repetition time, 22.0 s; echo time, 3.69 s; slice thickness, 0.6 mm; number of slices, 5 × 36 = 180; slab group thickness, 79.2 mm; field of view, 20.0 × 18.1 cm; and imaging matrix, 384 × 278.
MRI showed no abnormalities, except for multiple small white matter lesions. MRA revealed that the left ICA was not visualized, except for the supraclinoid segment. The left ICA supraclinoid segment receives blood flow from the right ICA paraclinoid segment via an anastomotic vessel. The left PCoA was also present, and blood flow from the left posterior cerebral artery to the left ICA was observed. The A1 segment of the left anterior cerebral artery (ACA) was absent. MRA source images confirmed the absence of the left carotid canal (Figs. 1, 2 and 3).
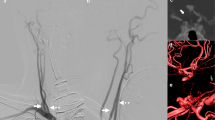
Infero-superior (a) and left lateral (b) projections of maximum-intensity-projection (MIP) images of magnetic resonance angiography (MRA). The left internal carotid artery (ICA) is absent, excluding the supraclinoid segment. There is an anastomotic channel between bilateral ICAs at the paraclinoid and supraclinoid segments (long arrows). The left posterior communicating artery (PCoA) is also present (short arrows)
Anteroinferior-posterosuperior projection of a partial MIP image of the ICA system. The anastomotic channel between bilateral ICAs at the paraclinoid and supraclinoid segments is clearly seen (long arrow), indicative of Lie’s type D ICA agenesis. The left PCoA is also present (short arrow), indicative of type A ICA agenesis. Thus, this patient has combined type D and type A ICA agenesis
The patient was treated conservatively and showed a good clinical course.
Discussion
ICA agenesis is extremely rare. However, the exact prevalence is unknown. Most patients with ICA agenesis are asymptomatic. However, this anomaly can cause transient ischemic attacks (TIAs) owing to blood flow or increase the risk of aneurysm formation, which can lead to stroke or aneurysm-related symptoms. TIA symptoms are common in patients over 40 years old, while subarachnoid hemorrhaging symptoms are common in those 0–20 years old [12].
Traditionally, Lie [5] classified ICA agenesis into six types based on the agenesis segments of the ICA and type of collateral circulation. Type A is the most common type. ICA is absent except for the terminal segment, and the terminal segments of the ICA and middle cerebral artery (MCA) are supplied via the large PCoA (Fig. 4a). Zhang et al. [12] reported a high incidence of aneurysms in patients with type A. In type B, the MCA is supplied by the anterior communicating artery. Type C is bilateral ICA agenesis, in which the anterior circulation is supplied via bilateral large PCoAs [5]. Thus, the collateral blood flow of types A-C is formed via the circle of Willis. In contrast, type D is an intercavernous anastomosis type with no relation to the circle of Willis (Fig. 4b). In type D ICA agenesis, there is a risk of injury to the collateral blood vessels during transsphenoidal pituitary surgery, as the collateral blood vessels are located near the sella. In addition, some cases mimic pituitary microadenomas [2], and care should be taken to avoid misdiagnosing anastomotic vessels as adenomas or carotid cavernous fistulas. Type E is ICA hypoplasia, and type F is rete mirabile (collaterals from the external carotid artery) [5].
Illustrations of agenesis of the ICA, modified from Reference [7]. (a) Lie type A: The terminal segment of the ICA and middle cerebral artery is supplied by the PCoA, the most prevalent type of collateral circulation in cases of ICA agenesis. (b) Lie type D: An anastomotic artery exists between the cavernous segments of the bilateral ICAs. There was no PCoA on the side of agenesis of the ICA. (c) Illustration of the patient. The long arrow indicates the ICA anastomosis between the paraclinoid and supraclinoid segments of the bilateral ICAs. The short arrow indicates the patent ipsilateral PCoA. Both anastomotic vessels were relatively small in caliber. AnaA: anastomotic artery between cavernous segment of bilateral ICAs, BA: basilar artery, LPCoA: left posterior communicating artery, RACA: right anterior cerebral artery, RICA: right internal carotid artery
Recently, several types of ICA agenesis that do not fit Lie’s classifications have been reported [3, 6, 9,10,11]. Lie’s original type D is an intercavernous anastomosis between the cavernous-cavernous segments of the ICAs. Yamashita et al. [11] reported a case of paraclinoid-paraclinoid anastomosis associated with carotid-ACA anastomosis. Uchino et al. [10] reported a case of paraclinoid-supraclinoid anastomosis associated with basilar artery-PCoA anastomosis and a patent ipsilateral PCoA. Hong et al. [3] reported a case of supraclinoid-supraclinoid anastomosis. Okawa et al. [6] reported a case of types D ICA agenesis associated with the right aortic arch. Park et al. [9] reported a case of type D and contralateral PCoA supplying the ipsilateral MCA, resulting in double interinternal carotid artery communication. Our patient had paraclinoid-supraclinoid anastomosis with patent PCoA. Oz et al. [7] reviewed 35 cases of type D ICA agenesis and found that there was no laterality or sex predominance. Chaudhary et al. [2] recently reviewed 50 cases of ICA agenesis with an anastomotic vessel between bilateral ICAs and found 31 cases (62%) of cavernous-cavernous anastomosis (classical Lie’s type D). If type D includes anastomosis between the bilateral ICAs distal to the cavernous segment, i.e. paraclinoid and supraclinoid segments, then our patient likely had combined type D and type A ICA agenesis (Fig. 4c). To our knowledge, no similar case has been reported in the English literature. This is the second case of type D ICA agenesis with patent ipsilateral PCoA [10]. In these two patients, the patent PCoAs and the anastomotic vessels between the bilateral ICAs were relatively small in caliber.
Bonasia et al. [1] recently proposed a new classification system based on embryological aspects. According to this classification, anastomosis between the bilateral ICAs (Lie type D) is classified as type IIIB. This type of ICA agenesis is secondary to the agenesis of segments V or VI of the ICA, with proximal regression of other segments. The flow compensation of ICA territories is provided by anastomosis and enlargement of the side-to-side cavernous or supraclinoid branches of both ICAs. Thus, the terminal segment of the ICA and MCA is supplied by an anastomotic vessel originating from the contralateral ICA. Bonasia et al. [1] reported 34 cases of this type.
In the case of ICA agenesis, it is thought that the vessel that anastomoses with the ICA depends on the segment and timing of ICA impairment. According to Lasjaunias et al. [4], the ICA comprises six segments from origin to termination: cervical, petrous, vertical cavernous, horizontal cavernous, clinoid, and cisternal segments. Each segment is independent and may show agenesis. According to Padget [8], embryologically, the ICA is derived from the dorsal aorta and the third aortic arch at the 3-mm embryonic stage, while the circle of Willis is formed around the 7- to 24-mm embryonic stage. Therefore, if ICA injury occurs after the 24-mm embryonic stage, it is likely to anastomose with the circle of Willis or with other primitive arteries, such as the primitive trigeminal artery, if it is destroyed before that time. Since the formation of the skull base occurs after the development of the ICA, and the presence of the ICA is essential for the formation of the bony carotid canal, the absence of the carotid canal implies that the ICA was damaged before embryonic life. Computed tomography of the bone window setting is particularly useful in the evaluation of carotid canal dysplasia and can distinguish between acquired obstruction and absence of the ICA. We were able to detect the absence of the carotid canal using source images of the MRA (Fig. 3a).
In type D ICA agenesis, the anterior circulation usually does not anastomose with the posterior circulation (Fig. 4b), because the PCoA does not form on the side where the ICA defect occurs. This may be because the segment damaged by the mutation in type D ICA agenesis is also involved in the formation of the artery necessary to connect with the posterior circulation. Lie [5] stated that the remnant of the primitive trigeminal artery that loses its connection with the posterior circulation becomes a type D anastomotic vessel. However, our patient and the case reported by Uchino et al. [10] had a patent ipsilateral PCoA. As mentioned above, the patent PCoAs and the anastomotic vessels between the bilateral ICAs were relatively small in caliber. We speculate that in case of type A ICA agenesis, when the development of the PCoA is insufficient to support collateral blood flow, an anastomotic vessel between bilateral ICAs may develop.
Conclusions
Lie’s type D ICA agenesis usually does not communicate with the anterior and posterior circulation. However, we found a case of type D combined with type A. This is the second case of type D ICA agenesis with patent ipsilateral PCoA. In these two cases of patent ipsilateral PCoA, the PCoA and the anastomotic vessels between the bilateral ICAs were relatively small in caliber. We speculate that in case of type A ICA agenesis, when the development of the PCoA is insufficient to support collateral blood flow, an anastomotic vessel between bilateral ICAs may develop.
Data availability
No datasets were generated or analysed during the current study.
References
Bonasia S, Smajda S, Ciccio G, Bojanowski MW, Robert T (2023) Proposed new classification for internal carotid artery segmental agenesis based on embryologic and angiographic correlation. Surg Radiol Anat 45:375–387. https://doi.org/10.1007/s00276-023-03102-1
Chaudhary RK, Groskreutz D, Gupta E, Baghdadi Y, Smith S, Sapire J (2023) ICA agenesis with transcavernous anastomosis: a systematic review. Surg Radiol Anat 45:777–786. https://doi.org/10.1007/s00276-023-03117-8
Hong JH, Chang HW, Kim JM, Cho HC, Kim SH, Hong JH, Kim SJ, Jeong HW (2016) Internal carotid artery agenesis associated with a rare collateral pathway: supraclinoid-supraclinoid anastomosis. A case report. Surg Radiol Anat 38:261–263. https://doi.org/10.1007/s00276-015-1531-7
Lasjaunias P, Santoyo-Vazquez A A (1984) Segmental agenesis of the internal carotid artery: angiographic aspects with embryological discussion. Anat Clin 6:133–141. https://doi.org/10.1007/BF01773165
Lie TA (1968) The congenital anomalies of the carotid arteries. Excerpta Medica Foundation, Amsterdam, New York, pp 35–51
Okawa M, Higashi T, Komiyama M, Fukuda K, Abe H, Inoue T (2015) Left internal carotid artery agenesis with trans-sellar collateral and a right aortic arch: Case report. Interv Neuroradiol 21:759–764. https://doi.org/10.1177/1591019915609130
Oz II, Serifoglu I, Yazgan O, Erdem R (2016) Congenital absence of internal carotid artery with intercavernous anastomosis: case report and systematic review of the literature. Interv Neuroradiol 22:473–480. https://doi.org/10.1177/1591019916641317
Padget DH (1948) The development of cranial arteries in the human embryo. Contrib Embryol 32:205–262
Park YW, Yoo J, Kim DJ (2018) Double inter-internal carotid artery communication through intercavernous anastomosis and posterior communicating artery associated with multiple intracranial artery segmental agenesis/aplasia. Surg Radiol Anat 40:227–231. https://doi.org/10.1007/s00276-017-1897-9
Uchino A, Saito N, Kohyama S (2015) Agenesis of the internal carotid artery with paraclinoid-supraclinoid anastomosis and basilar artery-posterior communicating artery anastomosis diagnosed by magnetic resonance angiography. Surg Radiol Anat 37:685–687. https://doi.org/10.1007/s00276-014-1388-1
Yamashita K, Yasaka M, Uchino A, Noguchi T (2022) Unilateral agenesis of internal carotid artery with interparaclinoid and contralateral carotid-anterior cerebral artery anastomoses diagnosed by magnetic resonance angiography: a case report. Surg Radiol Anat 44:289–292. https://doi.org/10.1007/s00276-021-02844-0
Zhang P, Wang Z, Yu FX, Lv H, Liu XH, Feng WH, Ma J, Yang ZH, Wang ZC (2018) The clinical presentation and collateral pathway development of congenital absence of the internal carotid artery. J Vasc Surg 68:1054–1061. https://doi.org/10.1016/j.jvs.2018.01.043
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
TM and AU carried out the study design and drafted the manuscript. TM, AU, MS, KM and YB reviewed the manuscript critically, and have read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Consent for publication
The patient gave her written informed consent regarding publishing her data and associated figures.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Maki, T., Uchino, A., Sugiyama, M. et al. Combined lie’s type D and type A agenesis of the left internal carotid artery diagnosed by magnetic resonance angiography. Surg Radiol Anat 46, 871–875 (2024). https://doi.org/10.1007/s00276-024-03366-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-024-03366-1