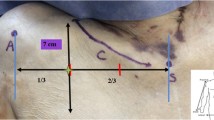
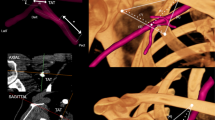
Abstract
Background
The acromial arterial rete (AAR) is the junction between the skin blood supply of the cervical side and that of the upper arm, and it is the only site crossed by the trans-regional blood supply of the cervico-humeral flap (CHF). The aim of this study was to explore the structures of AAR to optimizing flap design.
Methods
A body arteriography and spiral CT scan were performed on 33 whole adult corpses. The 3D reconstruction was used to perform continuous digital layered anatomy of the shoulder and upper chest; the acromion and acromioclavicular joint were used as the center to observe the source, route and distribution characteristics of a perforating branch and their anastomosis.
Results
The perforating branches were separated from an acromial branch of the transverse cervical artery (97%), posterior humeral circumflex artery (95%), a deltoid branch of the thoracoacromial artery (95%), and the acromial branch of the thoracoacromial artery (93%). The diameter of the acromial branch of the transverse cervical artery at its initial location was 1.18 ± 0.37 mm; the trunk length was 12.53 ± 3.83 cm, and it was anastomosed with other blood vessels in three forms.
Conclusion
Deep fascia should be included in the flap design. Three kinds of pedicled transfer flaps can be designed with the acromial branch of transverse carotid artery as the vascular pedicle. Free flaps can be designed with the acromial branch of thoracoacromial artery as the vascular pedicle.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The application of cervico-humeral flap (CHF) has a long history. As early as 1949, Kazanjian [13] reported the clinical application of this flap, naming it acromial flap. In 1958, Kirschbaum suggested the use of acromial flap for the treatment of Mentosternal contracture. In 1978, Mathes [18] first reported the anatomical study of cervico-humeral flap, which was used to repair head and neck defects. In 1979, Lamberty [15] defined the flap obtained by superficial branch of the transverse cervical artery as “supra-clavicular axial patterned flap” rather than CHF. This flap not only provides trans-regional blood supply between the double vascular territories, but also overcomes the restriction of the traditional length–width ratio. However, in 1983, Lamberty and Cormack [16] used the name “cervico-humeral flap” again. It was also found that the use of this flap for the head and neck reconstruction was limited, especially when treating malignant diseases. In 2009, Cordova [5] conducted a more detailed anatomical study of the lateral cervical triangle but did not include the acromion and the upper arm. Chan [3] harvested ten supraclavicular artery island flap from fresh cadavers. All flaps were injected with a contrast agent and examined using a real-time CT scan and three-dimensional (3D) reconstruction and perfusion flow (4D) analyses. The results showed that the average length of the effective perfused flap was 24.2 cm, the average width 8.7 cm, and the average area 152.8 cm2. Since then, clinical applications of CHF have gradually increased [8, 10, 11, 20, 25, 30]. According to Karabulut (2020) [12] and Chiu (2009) [4], supraclavicular island flap, which has a reliable pedicle, can be easily harvested. This kind of flap is commonly used for postoperative reconstruction of head and neck tumors [21,22,23,24]. Yet, early reports by Blevins [1] have shown that the distal necrosis rate tends to be as high as 40%. Moreover, in 2013, Kokot [14] reported that the incidence of partial or total necrosis was 22%. Their results showed that the length of the flap was positively correlated with necrosis when the length was longer than 22 cm. However, in 2018, Sheriff [26] reported that the distal survival of the flap is reliable up to 22 cm. From the anatomical point of view, we believe that the acromial artery rete is the critical factor for the successful application of CHF flap. The rete refers to anastomosis among the vascular territories around the acromion. Therefore, we performed three-dimensional reconstruction of the acromial arterial rete, using the CT angiographic data of 33 cadavers. Based on these 3D models, we observed the distribution of acromial branch of the transverse cervical artery (Branches of superior clavicular branch of transverse cervical artery), posterior humeral circumflex artery, deltoid branch of the thoracoacromial artery, acromial branch of the thoracoacromial artery and suprascapular artery, to get the detailed anatomy basis to optimize the flap design. This study is one of our series of studies on the morphological distribution of perforator vessels.
Materials and methods
A total of 33 fresh whole adult corpses (25 males and 8 females; 28 from Anatomy Department of Wenzhou Medical University, and 5 from Applied Anatomy Department of Xiangya Hospital of Central South University) were obtained through the body donation process (Table 1).
Through unilateral femoral artery, the one-time gelatin–lead oxide whole-body perfusion arteriography [2], and spiral CT scan were performed. The technical parameters of the CT scan varied according to somatotype, location, and contrast. The scanned data were inputted into the MIMICS image workstation (Materialise, Leuven) in DICOM format. A 3D reconstruction [6] of the whole corpse was performed first, after which, the dissection (chest and thigh) was made as required. On this basis, the sagittal, coronal, and horizontal segmentations were performed so as to reduce the scope, speed-up the construction and increase the accuracy of 3D visualization.
By determining the locations, it was possible to observe the chest, neck, and shoulder above the midpoint plane of the humerus. With the acromion as the center, the acromial branch of the transverse cervical artery, the deltoid branch of the thoracoacromial artery, supraspinous branch of the suprascapular artery, as well as the anterior and posterior humeral circumflex arteries, were mainly observed. First, the rapid direct volume rendering (VR) method was used to continuously perform 3D reconstruction from shallow to deep and the source and distribution characteristics of the main perforating branches around the acromion were tracked and observed throughout the whole process, as well as the anastomosis among perforating branches (Fig. 1). If necessary, other MIMICS tools, such as Thresholding, Region growing, Edit mask in 3D and similar were used to perform 3D reconstruction and integration of bones, tendons, muscles, and arteries, thus constructing 3D visualization models [28]. Scan data exclusion criteria: (1) The data of head and neck were missing; (2) After three-dimensional reconstruction, the thoracic acromion arterial network cannot be displayed.
A 3D visualization layered anatomy of AAR (No. 25 corpse, male, right). a The superficial layer mainly shows the distribution of perforating branches around the acromion (white dotted circle). b The deeper layer shows the perforating vascular trunk, and the clavicle is faintly visible (white arrow). c The layer gradually deepens, tracking the source artery of perforating branches. The two blue arrows show that the acromial branch of the transverse cervical artery sends a branch forward anastomosed with the acromial branch of the thoracoacromial artery through the acromioclavicular joint, which is faintly visible due to deeper position. d A close-up of the acromion; the black arrow indicates the perforating branch originating from the transverse cervical artery of the thyrocervical trunk; the four green arrows indicate a rectangular artery circle, which is formed by the anastomosis between the acromial branch of the transverse cervical artery and an acromial branch of the thoracoacromial artery. 1 acromial branch of the transverse cervical artery; 2 acromial branch of the thoracoacromial artery; 3 deltoid branch of the thoracoacromial artery; 4 musculocutaneous perforating branch of posterior humeral circumflex artery; 4’ perforating branch of intermuscular space of posterior humeral circumflex artery; 5 suprascapular artery; 5’ infraspinous perforating branch of suprascapular artery; 6 prethoracic branch of thoracoacromial artery; 7 the 3rd intercostal perforating branch of internal thoracic artery
Results
Arteriography (66 sides) was performed on 33 fresh-whole adult corpses. Due to vascular thrombosis in some locations, which resulted in poor local angiography or damage or loss of local CT scan files, valid statistical data of various locations of the body were different. The final valid data of upper chest, neck, and shoulder, were mainly obtained from 57 sides (n = 57) (Table 1).
Acromial arterial rete (AAR)
A 3D reconstruction was performed from shallow to deeper layers centering on the acromion, thus showing that the superficial surfaces of the acromion and acromioclavicular joint had rich distribution of perforating branches, mutually anastomosing (Fig. 1). The perforating branches were originated from an acromial branch of the transverse cervical artery (55/57, 97%), posterior humeral circumflex artery (54/57, 95%), a deltoid branch of the thoracoacromial artery (54/57, 95%), an acromial branch of the thoracoacromial artery (53/57, 93%), and suprascapular artery (36/57, 63%) (Table 2). The diameter for the initial location of the acromial branch of the transverse cervical artery, which passed laterally across the acromion, was 1.18 ± 0.37 mm; the trunk length was 12.53 ± 3.83 cm. There were three distribution patterns: (1) anastomosed posterolaterally and inferiorly with the ascending branch of posterior humeral circumflex artery (Fig. 2A), with the occurrence frequency of up to 96% (53/55); (2) anastomosed anterolaterally and inferiorly with a deltoid branch of the thoracoacromial artery and/or anterior humeral circumflex artery (Fig. 2B), accounting for 36% (20/55); (3) nine sides (16%) were double branches, which were separately anastomosed with the thoracoacromial artery and posterior humeral circumflex artery (Figs. 1C, 1D, 2B). The diameters of the perforator branches of the posterior humeral circumflex artery, deltoid branch of the thoracoacromial artery, acromial branch of the thoracoacromial artery, and suprascapular artery were 0.81 ± 0.32 mm, 0.9 ± 0.35 mm, 0.6 ± 0.42 mm, and 0.7 ± 0.29 mm, respectively (Table 2).
The anastomosis of AAR with peripheral blood vessels from 3 male corpses. a, b No. 25 corpse; c No. 33 corpse; d, e No. 26 corspe. a Posterior superior view of the right shoulder, shows that the acromial branch of the transverse carotid artery is anastomosed with the ascending branch of the posterior circumflex humeral artery. The blue arrow shows the acromioclavicular joint, the green arrow shows the inferior branch of the supraspinatus artery; the white arrow shows the acromial branch of the transverse carotid artery that is anastomosed with the perforating branch of the posterior circumflex humeral artery and the inferior branch of the supraspinatus artery. b Anterior superior view of the left shoulder, shows that the acromial branches of the transverse carotid artery were anastomosed with the acromial branches and deltoid branches of the thoracoacromial artery, respectively. c Anterior superior view of right shoulder, shows that the acromial branches of the thoracoacromial artery reach to the acromial peak, and there was no true coincidence with other perforating branches. d Anterior superior view of the right shoulder, shows the supraspinatus branch of the suprascapular artery replacing acromial branch of transverse carotid artery. e The superior posterior view of the left shoulder, shows that the supraspinatus perforating branch of the suprascapular artery forms anastomotic network with the acromial branch of the transverse carotid artery and the thoracoacromial artery on the surface of the acromial. 1 Acromial branch of the transverse carotid artery; 2 Acromial branch of thoracoacromial artery; 3 Deltoid branch of the thoracoacromial artery; 4 Anterior circumflex brachial artery; 5 perforating branch of intermuscular space of posterior humeral circumflex artery; 5’ Muscular perforator of posterior circumflex brachial artery; 6 Suprascapular artery; 6’ Supraspinatus perforator of the suprascapular artery
Anastomosis of thoracoacromial artery rete (AAR) with peripheral blood vessels
The acromial branch of the transverse cervical artery is the main blood vessel of the AAR. There are abundant perforating branches converging toward the anteroposterior and lateral shoulder. The occurrence frequency of the deltoid branch of the thoracoacromial artery was 95%, and that of the acromial branch was 93% (Table 2). However, only five sides (9%) of the thoracoacromial perforators (acromial or deltoid branches) had a true anastomosis with the acromial branches of the transverse carotid artery and/or anterior and posterior humeral circumflex arteries (Fig. 1D). The other 50 sides had no true anastomosis with other perforating branches at the acromion. The occurrence frequency of the suprascapular artery was 63% (Table 2), and 17% of them sent supraspinatus perforating branches, which were inserted between the acromial branch of the transverse cervical artery and the posterior humeral circumflex artery, or replaced the acromial branch of the transverse cervical artery (Fig. 2D, E). The occurrence frequency of infraspinous perforating branch was 11%, anastomosed posterosuperiorly with an acromial branch of the transverse cervical artery (Fig. 2A). The intercostal perforating branch of the posterior humeral circumflex artery was close to the posteromedial side, and the musculocutaneous-perforating branch was close to the anterolateral side (Fig. 2A). The occurrence frequency of the anterior humeral circumflex artery was 30%, and its branches were thin and small. There were no thick and large perforating branches involved in the composition of the AAR in this study.
Discussion
There are two main types of supraclavicular artery (perforating branch) flaps: (1) acromion branch used as the vascular pedicle, harvested from the acromion to the upper arm [21], (2) thoracic branch used as a vascular pedicle, harvested forward across the clavicle to the chest [9, 17]. The axial vessels and flap positions of these two types of flaps are different. In this paper, we only discuss the former. To avoid confusion with the latter, we think that it is more appropriate to use the “cervical humeral flap” named by Lamberty and Cormack [16].
The cervico-humeral flap(CHF) is a double-vascular trans-regional blood supply flap. When pedicled transposition is used for the repair of head and neck defects, the blood supply secured through acromial branch of the transverse cervical artery (anatomical donor area) plus ascending branch of posterior humeral circumflex artery (dynamic donor area) [15]; there is only one choke vessel area in the flap, which is the junction of the double vascular territories (Fig. 3B), located around the acromion. According to the analysis of the “trans-regional flap hemodynamic model” [29], the acromion is the blood flow resistance zone within this flap. Therefore, AAR was the focus of this study.
Schematic diagram of flap design (from 3 corpses, a, d No. 6, male; b No. 25, male; c No. 5, female). a A right anterior view; the flap is designed with the acromial branch of the transverse cervical artery as the vascular pedicle towards the anterior shoulder; the light green filling area shows a true anastomosis between an acromial branch of the transverse cervical artery and acromial deltoid branches of the thoracoacromial artery, which can be regarded as a vascular body, and if necessary, it can be expanded downward to cut a random flap. b The left posterior view; the flap is designed with the acromial branch of the transverse cervical artery as the vascular pedicle towards the posterior shoulder; the light yellow filling area shows the choke blood vessel area between the two vascular bodies. c In the left anterior lateral view, the flap is designed with the acromial branch of the transverse cervical artery as the vascular pedicle towards the lateral shoulder; retaining the branching area of an acromial branch of the thoracoacromial artery should be noted; if necessary, it can be expanded downward to cut a random flap. d The left anterior view; the free flap is designed with the acromial branch of the transverse cervical artery as the vascular pedicle towards the anterior shoulder. Notes: the marked rectangular area in the figure is a deeper 3D reconstruction that shows the source of the perforating branch. 1 Acromial branch of the transverse carotid artery; 2 Acromial branch of thoracoacromial artery; 3 Deltoid branch of the thoracoacromial artery; 4 Anterior circumflex brachial artery; 5 perforating branch of intermuscular space of posterior humeral circumflex artery
Clinical significance of acromial arterial rete (AAR)
Due to the thin skin and subcutaneous tissue in the shoulder, the 3D reconstruction of the subcutaneous vascular network is not complicated. It is mainly formed by anastomosis among acromial branches of the transverse cervical artery, thoracoacromial artery, posterior humeral circumflex artery, and suprascapular artery around the acromion and acromioclavicular joint (Table 2, Figs. 1, 2). However, most of the blood vessels are choke anastomosis, while true anastomosis is rare (Figs. 1D, 2E, and 3C). This may be related to the function of the shoulder. For example, a manual worker who often handling heavy loads should have more true anastomoses in this region.
The acromion branch of transverse carotid artery crosses acromion end or acromion of clavicle and is anastomosed around the acromion with acromial branches of the thoracoacromial artery, posterior humeral circumflex artery, and suprascapular artery (Figs. 1, 2, 3). After crossing the acromion, the acromial branch of the transverse cervical artery is mainly posterolaterally and inferiorly anastomosed with the ascending branch of posterior humeral circumflex artery (2A, Fig. 3B), or anterolaterally and inferiorly anastomosed with a deltoid branch of the thoracoacromial artery and/or anterior humeral circumflex artery (Figs. 2B, 3A, C). In addition, the anastomosis with the thoracoacromial artery and posterior humeral circumflex artery can be seen (Figs. 1, 2B, and 3C). Therefore, the island flap can be designed with the acromial branch of the transverse cervical artery as the vascular pedicle towards the anterior, posterior, and lateral sides of the shoulder (Fig. 3).
-
1.
Acromial branch of the transverse cervical artery (anatomical donor area) plus the acromial branch and/or deltoid branch (dynamic donor area) of the thoracoacromial artery is a double-vascular territory trans-regional blood supply flap. Because the acromial branch of the thoracoacromial artery originates from the deltoid branch, the two are homologous and true anastomosis commonly occurs. If the true anastomosis with the acromial branch of the transverse cervical artery reappears, it can be regarded as an anatomical donor area (Fig. 3A). On this basis, it can also be expanded downward to harvest the random flap, i.e., the axial pattern flap plus random pattern flap proposed by McGregor and Morgan [19].
-
2.
Acromial branch of the transverse cervical artery (anatomical donor area) plus ascending branch of posterior humeral circumflex artery (dynamic donor area) is a double-vascular territory trans-regional blood supply flap. The choke vascular anastomosis is common among vascular territories (Fig. 3B); however, true anastomosis is rarely occurs (Fig. 2A).
-
3.
The acromial branch of the transverse cervical artery (anatomical donor area) is anastomosed with the thoracoacromial artery and posterior humeral circumflex artery. The flap can be designed towards anterior and posterior shoulder joint, or the trans-regional blood supply flap can be laterally designed (Fig. 3A–C), which is simple, practical, and flexible.
-
4.
The occurrence frequency of the acromial branch of the thoracoacromial artery is as high as 93%, and it directly inserts in the acromion. It is located between the deltoid branch and the acromial branch of the transverse cervical artery. Since the acromial branch originates from the deltoid branch, the two are homologous, and the true anastomosis between the perforating branches is commonly occurs (Figs. 2B, 3A, C). If the acromial branch of the thoracoacromial artery is used as a vascular pedicle to harvest the free flap, the acromial branch of the thoracoacromial artery becomes the anatomical donor area. Both ends of the flap are the dynamic donor areas, and the blood flow resistance becomes obviously reduced, maximizing the survival area of the flap [29].
-
5.
The occurrence frequency of supraspinatus perforating branch of the suprascapular artery is 17%. When it appears, the acromial branch of the transverse cervical artery is relatively small and the route is short or even absent (Fig. 2D). However, it may also function as a gas station; the acromial branch of the transverse cervical artery will be dilated after anastomosis (Fig. 2E). This phenomenon can also be seen after the anastomosis between the acromial branch of the thoracoacromial artery and the acromial branch of the transverse cervical artery (Fig. 1D). When the supraspinatus perforating branch of the suprascapular artery appears and leads to the absence of the acromial branch of the transverse cervical artery, the thoracic branch of the supraclavicular artery flaps should be selected [27].
Essentials of applied anatomy
A 3D visualization layered anatomy, which can display unlimited layers of anatomy, fluoroscopy and rotation observation of the human body [28], can clearly show the location of the perforating branch and its adjacent structures, track and observe the route and source of the perforating vascular trunk. Thus, it has many advantages in the basic morphological research of skin flaps.
-
1.
The acromion is the key location of the survival of “cervico-humeral flap”
There is no obvious anastomosis of the perforating branches in the subcutaneous tissue of the acromion (Fig. 1A, B). In this study, the subcutaneous vascular network was observed in one case only (Fig. 3C). The position of the AAR is deep, which is close to the surfaces of the acromion and the acromioclavicular joint and belongs to the deep fascia vascular network (Figs. 1C, D, 2A, B, E). Thus, no matter what type of blood vessel is used as the pedicle to harvest the “cervico-humeral flap”, when dissecting the flap around the acromion, the flap should be closely attached to the surfaces of the acromion and acromioclavicular joint, and the deep fascia should be harvested. The deep fascia vascular network is the only way for the flap to supply blood across the region.
-
2.
The flexible design of flap vascular pedicle
Given the acromial branch of the thoracoacromial artery is directly inserted into the acromion, it has multiple function. First, if the branch is truly anastomosed with the acromial branch of a transverse cervical artery, posterior humeral circumflex artery or deltoid branch of the thoracoacromial artery, the “cervico-humeral flap” with double donor areas can be considered as a large donor area (Figs. 3A, C), and the flap is easy to survive. Second, if there is a true anastomosis with only one side, it can still be regarded as a double-donor area flap. Third, if, however, the acromial branch of the thoracoacromial artery has no true anastomosis with the arteries on both sides, it will become a true three-donor area flap. If the acromial branch of the transverse cervical artery is still used as the vascular pedicle for transposition, the distal part of the flap becomes the potential donor area, and it must cross the two choke vascular regions for blood perfusion to the distal end from the acromial branch of transverse cervical artery. In this case, it is recommended to perform free transplantation with the acromial branch of thoracoacromial artery as the vascular pedicle (Fig. 3D), after which the blood flow would only need to cross one choke vascular area from the pedicle to both sides, thus significantly reducing the blood flow resistance and enabling the flap to easily survive[7, 29], without the need to pressurize or super-drain the distal end.
-
3.
During surgery, retaining the branching area of the perforating branch, such as that of the acromial branch of the thoracoacromial artery should be noted (Figs. 3C, D).
Limitations
As this research done on cadavers, the pressure of the perfusion, the elasticity of the blood vessel and whether there is blockage, may impact the results. Meanwhile, when this method applied on the living individual, the possibility of vasoconstriction from the contrast, room temperature, position during the operation, and sympathetic over-activity might change the size of the vessels and appearance of the anastomosis.
Conclusion
The “cervico-humeral flap” with the acromial branch of the transverse cervical artery as the vascular pedicle is reliable in relation to blood supply; however, special attention should be paid to retain the deep fascia when the flap is harvested at the acromion. Moreover, when the acromial branch of the thoracoacromial artery is distributed to the acromion, but it has no true anastomosis with the acromial branch of the transverse cervical artery, or posterior humeral circumflex artery, or deltoid branch of the thoracoacromial artery, it is recommended to use the acromial branch of the thoracoacromial artery as the vascular pedicle for free transplantation. When the supraspinatus-perforating branch of the suprascapular artery appears and leads to the absence of the acromial branch of the transverse cervical artery, the use of thoracic branch flap of the supraclavicular artery can be considered. In summary, we hope that our 3D layered anatomy of the acromion artery rete could provide a detailed anatomical basis for plastic surgeons to optimize the design of acromion flaps.
Abbreviations
- AAR:
-
Acromial arterial rete
- CHF:
-
Cervico-humeral flap
References
Blevins PK, Luce EA, Mathes SJ (1980) Limitations of the cervicohumeral flap in head and neck reconstruction. Plast Reconstr Surg 66:225. https://doi.org/10.1097/00006534-198008000-00008
Blondeel PN, Morris SF, Hallock GG et al (2013) Perforator flaps: anatomy, technique, & clinical applications. CRC Press
Chan JW, Wong C, Ward K et al (2010) Three- and four-dimensional computed tomographic angiography studies of the supraclavicular artery island flap. Plast Reconstr Surg 125:525–531. https://doi.org/10.1097/PRS.0b013e3181c82e37
Chiu ES, Liu PH, Friedlander PL (2009) Supraclavicular artery island flap for head and neck oncologic reconstruction: indications, complications, and outcomes. Plast Reconstr Surg 124:115–123. https://doi.org/10.1097/PRS.0b013e3181aa0e5d
Cordova A, D’arpa S, Pirrello R et al (2009) Anatomic study on the transverse cervical vessels perforators in the lateral triangle of the neck and harvest of a new flap: the free supraclavicular transverse cervical artery perforator flap. Surg Radiol Anat 31:93–100. https://doi.org/10.1007/s00276-008-0410-x
Cui H, Ding M, Mao Y et al (2020) Three-dimensional visualization for extended deep inferior epigastric perforator flaps. Ann Plast Surg 85:e48–e53. https://doi.org/10.1097/SAP.0000000000002379
Ding J, Feng X, Gao W et al (2017) Comparison of a small central versus a large peripheral perforator in a rat model of extended dorsal three-vascular territory perforator flap. Plast Reconstr Surg 139:434e–443e. https://doi.org/10.1097/prs.0000000000002991
Fernandes R (2014) Local and regional flaps in head and neck reconstruction: a practical approach. John Wiley & Sons
Gao Y, Yuan Y, Li H et al (2016) Preoperative imaging for thoracic branch of supraclavicular artery flap: a comparative study of contrast-enhanced ultrasound with three-dimensional reconstruction and color duplex ultrasound. Ann Plast Surg 77:201–205. https://doi.org/10.1097/SAP.0000000000000601
Granzow JW, Suliman A, Roostaeian J et al (2013) The supraclavicular artery island flap (SCAIF) for head and neck reconstruction: surgical technique and refinements. Otolaryngol Head Neck Surg 148:933–940. https://doi.org/10.1177/0194599813484288
Granzow JW, Suliman A, Roostaeian J et al (2013) Supraclavicular artery island flap (SCAIF) vs free fasciocutaneous flaps for head and neck reconstruction. Otolaryngol Head Neck Surg 148:941–948. https://doi.org/10.1177/0194599813476670
Karabulut B (2020) Supraclavicular flap reconstruction in head and neck oncologic surgery. J Craniofac Surg 31:e372–e375. https://doi.org/10.1097/SCS.0000000000006315
Kirschbaum S (1958) Mentosternal contracture. Plast Reconstr Surg 21:131–138
Kokot N, Mazhar K, Reder LS et al (2013) The supraclavicular artery island flap in head and neck reconstruction: applications and limitations. JAMA Otolaryngol Head Neck Surg 139:1247–1255. https://doi.org/10.1001/jamaoto.2013.5057
Lamberty B (1979) The supra-clavicular axial patterned flap. Br J Plast Surg 32:207–212. https://doi.org/10.1016/s0007-1226(79)90033-x
Lamberty BG, Cormack GC (1983) Misconceptions regarding the cervico-humeral flap. Br J Plast Surg 36:60–63. https://doi.org/10.1016/0007-1226(83)90013-9
Ma X, Zheng Y, Xia W et al (2009) An anatomical study with clinical application of one branch of the supraclavicular artery. Clin Anat 22:215–220. https://doi.org/10.1002/ca.20742
Mathes SJ, Vasconez LO, Mathes SJ (1978) The cervicohumeral flap. Plast Reconstr Surg 61:7–12. https://doi.org/10.1097/00006534-197801000-00002
Mcgregor IA, Morgan G (1973) Axial and random pattern flaps. Br J Plast Surg 26:202–213. https://doi.org/10.1016/0007-1226(73)90003-9
Nthumba PM (2012) The supraclavicular artery flap: a versatile flap for neck and orofacial reconstruction. J Oral Maxillofac Surg 70:1997–2004. https://doi.org/10.1016/j.joms.2011.08.043
Pallua N, Kim B-S (2017) Pre-expanded supraclavicular artery perforator flap. Clin Plast Surg 44:49–63. https://doi.org/10.1016/j.cps.2016.08.005
Pallua N, Machens H-G, Rennekampff O et al (1997) The fasciocutaneous supraclavicular artery island flap for releasing postburn mentosternal contractures. Plast Reconstr Surg 99:1878–1884. https://doi.org/10.1097/00006534-199706000-00011
Pallua N, Noah EM (2000) The tunneled supraclavicular island flap: an optimized technique for head and neck reconstruction. Plast Reconstr Surg 105:842–851. https://doi.org/10.1097/00006534-200003000-00003
Pallua N, Wolter TP (2013) Moving forwards: the anterior supraclavicular artery perforator (a-SAP) flap: a new pedicled or free perforator flap based on the anterior supraclavicular vessels. J Plast Reconstr Aesthet Surg 66:489–496. https://doi.org/10.1016/j.bjps.2012.11.013
Ramirez CA, Fernandes RP (2014) The supraclavicular artery island and trapezius myocutaneous flaps in head and neck reconstruction. Oral Maxillofac Surg Clin North Am 26:411–420. https://doi.org/10.1016/j.coms.2014.05.009
Sheriff H, Garcia CV, Jaber S et al (2018) Supraclavicular artery island flap: relation between length and distal end necrosis. Int Microsurg J. https://doi.org/10.24983/scitemed.imj.2018.00089
Suzuki Y, Shimizu Y, Kasai S et al (2019) Indocyanine green fluorescence videoangiography for reliable variations of supraclavicular artery flaps. Arch Plast Surg 46:318. https://doi.org/10.5999/aps.2018.01536
Tang M, Yin Z, Morris SF (2008) A pilot study on three-dimensional visualization of perforator flaps by using angiography in cadavers. Plast Reconstr Surg 122:429–437. https://doi.org/10.1097/PRS.0b013e31817d625a
Tao Y, Ding M, Wang A et al (2016) Basic perforator flap hemodynamic mathematical model. Plast Reconstr Surg Glob Open. https://doi.org/10.1097/GOX.0000000000000689
Wong S, Brennan M, Nishikawa S et al (2019) Supraclavicular artery island flap in head and neck reconstruction: a case series and literature review. Perm J. https://doi.org/10.7812/TPP/19.006
Funding
This study was supported by the National Natural Science Foundation of China (NSFC 81871577, 81974290, 81472104).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies were in accordance with the guideline of the ethical committee of Wenzhou Medical University and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jiang, Y., Ding, M., Zhou, Z. et al. A 3D visualization layered anatomy for acromial arterial rete and flap design. Surg Radiol Anat 43, 1559–1567 (2021). https://doi.org/10.1007/s00276-021-02781-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00276-021-02781-y