Abstract
Background
Guideline recommendations for preoperative chest radiographs vary to the extent that individual patient benefit is unclear. We developed and validated a prediction score for abnormal preoperative chest radiographs in adult patients undergoing elective non-cardiothoracic surgery.
Methods
Our prospective observational study recruited 703 adult patients who underwent elective non-cardiothoracic surgery at Ramathibodi Hospital. We developed a risk prediction score for abnormal preoperative chest radiographs with external validation using data from 411 patients recruited from Thammasat University Hospital. The discriminative performance was assessed by receiver operating curve analysis. In addition, we assessed the contribution of abnormal chest radiographs to perioperative management.
Results
Abnormal preoperative chest radiographs were found in 19.5% of the 703 patients. Age, pulmonary disease, cardiac disease, and diabetes were significant factors. The model showed good performance with a C-statistics of 0.739 (95% CI, 0.691–0.786). We classified patients into four groups based on risk scores. The posttest probabilities in the intermediate-, intermediate-high-, and high-risk groups were 33.2%, 59.8%, and 75.7%, respectively. The model fitted well with the external validation data with a C statistic of 0.731 (95% CI, 0.674–0.789). One (0.4%) abnormal chest radiograph from the low-risk group and three (2.4%) abnormal chest radiographs from the intermediate-to-high-risk group had a major impact on perioperative management.
Conclusions
Four predictors including age, pulmonary disease, cardiac disease, and diabetes were associated with abnormal preoperative chest radiographs. Our risk score demonstrated good performance and may help identify patients at higher risk of chest abnormalities.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Preoperative chest radiographs are commonly used to detect abnormalities requiring perioperative management, identify patients at higher risk of postoperative complications, aid in postoperative chest film interpretation, and screen tuberculosis in high-prevalence areas [1]. In addition, they help identify pulmonary masses or tumors that may impact surgical decisions [2]. Nonetheless, routine chest radiographs are no longer recommended due to risk of radiation-induced cancer, false-positive results, and increased costs [3]. Moreover, most abnormalities are chronic, with more than half detectable through history taking and physical examination [4]. Unexpected abnormalities rarely impact clinical management [5].
It is unclear which patients benefit from preoperative chest radiographs. Previous findings have been inconsistent [4, 6,7,8,9,10], perhaps because the studies were retrospective and interpretation was not standardized [3]. These studies also failed to consider associations between multiple predictors and their efficacy for predicting abnormal chest radiographs. The inconsistent findings have led to a change in guidelines [11,12,13,14] and different ordering practices among surgeons and hospitals [1, 15]. Therefore, this study aimed to develop and validate a risk prediction score for abnormal preoperative chest radiographs in adult patients undergoing elective non-cardiothoracic surgery. We further evaluated the impact of abnormal preoperative chest radiographs in perioperative management and the role of the predictive risk score in identifying abnormal chest radiographs.
Material and methods
Study design and population
Ethical approval was provided by the Human Research Ethics Committee of the Faculty of Medicine Ramathibodi Hospital and Thammasat University. A prospective observational study was conducted consisting of a development phase at Ramathibodi Hospital and a validation phase at Thammasat University Hospital (TUH). This study was conducted and reported according to Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis Or Diagnosis (TRIPOD) and STrengthening the Reporting of OBservational studies in Epidemiology (STROBE) [16, 17]. Study participants included consecutive adult patients aged 18 years or older, who had preoperative chest radiographs and underwent elective non-cardiothoracic operation and anesthesia. Exclusion criteria were pregnancy, cancelation due to administrative reasons, previous cardiothoracic surgery, re-surgery within the study period, chest or cardiac trauma, or unwillingness to participate in the study.
Study outcomes
The primary outcome of the study was preoperative chest radiographs classified as abnormal by a panel of anesthesiologists, radiologists, pulmonologists, and cardiologists. All chest radiographs were interpreted at a resolution of 6 million pixels. Interpretation was guided by comparison to previous radiographs with blinding to clinical information. Radiologists completed all reports at Ramathibodi Hospital, and 15% were re-evaluated by W.S. for standardization. Chest radiographs at TUH were doubly interpreted by both radiologists and anesthesiologists (A.S., A.K., N.S.). Disagreement in reporting was resolved by A.K. The percentages of agreement and kappa values were reported. We also collected data at TUH on how abnormal chest radiographs were used for perioperative management by anesthesiologists or surgeons.
Study predictors
Nineteen potential predictors were selected by considering clinical importance and previous relevant studies.
-
Patient characteristics:
-
age (years) categorized by distribution
-
sex
-
body mass index (BMI) (kg/m2) categorized by the WHO definitions for Asians [18]
-
-
Risk behaviors:
-
Comorbidities:
-
upper respiratory tract infection within two weeks
-
presence of tracheal tube
-
pulmonary diseases categorized into (1) no pulmonary disease, (2) stable airway disease including asthma, chronic obstructive pulmonary disease (COPD), and bronchiectasis, (3) stable non-airway diseases, which were pulmonary diseases affecting parenchyma, interstitium, blood vessels, or pleura, and (4) active pulmonary disorders with respiratory symptoms
-
cardiac diseases categorized into (1) no cardiac disease, (2) cardiac diseases with stable conditions, and (3) cardiac diseases with active conditions [21]
-
hypertension
-
cerebrovascular disease
-
acute kidney injury/chronic kidney disease (AKI/CKD) [22]
-
diabetes mellitus
-
thyroid mass/nodule
-
cancer
-
immunocompromised status
-
-
Types of surgery were classified into low, intermediate, and high cardiac risk [23]
Data were collected from pre-anesthesia evaluation forms or the International Classification of Diseases, Tenth Revision (ICD-10) codes by anesthesiology residents blinded to the reports of chest radiographs.
Sample size estimation
Our sample size was estimated based on the 9% prevalence of abnormal preoperative chest radiographs in Thailand [6], providing 10 potential predictors in the final model, and 20 events per predictor [24]. The resulting estimated sample size was 2223 patients for the development phase. Assuming 10% incomplete data, our sample size target was 2446 patients for the development phase at Ramathibodi Hospital, with 1048 patients (30%) required for external validation at TUH.
Statistical analysis
Multiple imputation by chained equations was used to impute missing data with 20 replications [25]. For model development, a simple logistic regression was performed by fitting each predictor on abnormal preoperative chest radiographs. Predictors with p < 0.10 were considered in the multivariable logistic regression model. A forward selection was manually applied with only significant variables retained within the final model. Risk prediction scores were constructed based on the coefficients of the predictors and then trichotomized into four risk groups corresponding to the positive likelihood ratio. Calibration performance was assessed using the Hosmer–Lemeshow goodness of fit (HL-GOF) test and the ratio of observed and expected values (O/E ratio). The model discrimination was determined by the receiver operating characteristic (ROC) curve analysis and C-statistic estimated [26].
A bootstrap 5000-replication analysis was used for internal validation. The bootstrap-corrected calibration and discrimination coefficients were estimated to assess model performance. External validation was performed by applying the final developed model to data from TUH. Calibration and discrimination performance were also assessed as described above. Recalibration or model revision was undertaken when calibration performance was considered poor [27].
All statistical analyses were performed by STATA software version 16 (StataCorp LLC. 2019, College Station, Texas, USA). A value of p < 0.05 was considered statistically significant.
Results
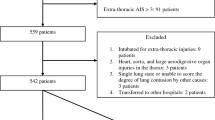
The study was halted prematurely when all elective surgeries in both hospitals were postponed due to the COVID-19 pandemic. A total of 730 and 411 patients were scheduled for elective non-cardiothoracic surgery at Ramathibodi Hospital and TUH, respectively, throughout February and March 2020 (Fig. 1). Seventeen (2.0%) patients at Ramathibodi Hospital and nine (1.7%) patients at TUH were excluded because they did not have preoperative chest radiographs (Fig. 1). Characteristics of the patients included are presented in Table 1. At Ramathibodi Hospital 137 patients (19.5% with a 95% confidence interval [CI] of 16.6–22.6%) had abnormal preoperative chest radiographs. The reliability of radiograph interpretations was 97.4% with kappa statistic of 0.90.
Development phase
Imputation was performed on five predictors in 49 patients. Ten predictors including age, smoking, presence of tracheal tube, pulmonary disease, cardiac disease, diabetes, AKI/CKD, hypertension, cerebrovascular disease, and cancer were associated with abnormal preoperative chest radiographs from the univariate analysis and were simultaneously included in the multivariate model (Table 2). Four predictors including age, pulmonary disease, cardiac disease, and diabetes were significantly associated with abnormal chest radiographs in the final model (Table 3). The risk of abnormal chest radiographs increased 2.4- and 4.0-times higher odds in ages ≥45 and ≥65 years, respectively. Patients with stable non-airway lung and active lung diseases had 6.5- and 26.9-times higher odds of abnormal radiographs compared to patients with stable airways and free from pulmonary diseases. In addition, patients with stable and active-cardiac diseases were approximately 2.2- and 16.9-times more likely to have abnormal radiograph than patients who did not have cardiac diseases. The prediction equation fitted well with the data (HL-GOF χ2 = 2.53, p = 0.469); the O/E ratio was 0.986 (95% CI, 0.965–1.006). The final model showed good discriminative performance with a C statistic of 0.739 (95% CI, 0.691–0.786).
Four risk groups were generated at scores of −1.34, −0.56, and 0.33 (Table 4). Of the 703 patients, 403, 215, 61, and 24 patients were classified within the low, intermediate, intermediate-high, and high-risk groups. Posttest probabilities of having an abnormal preoperative chest radiograph in the intermediate-, intermediate-high-, and high-risk groups were 33.2%, 59.8%, and 75.7%, respectively. A simplified color-coded table classifying the patients’ risk groups is shown in Table 5. The risk increased from low risk to at least intermediate risk in any patients aged 65 years or older. Patients with stable non-airway pulmonary diseases were classified as intermediate risk group when they were younger than 65 years and did not have cardiac disease or diabetes. Otherwise, they were classified as intermediate-high or high risk. All patients with active lung diseases were classified as high risk. Patients with cardiac diseases or diabetes were classified into the intermediate- to high-risk groups, except patients aged less than 45 years who had either diabetes or stable cardiac disease and were free from pulmonary diseases and were therefore classified as low risk.
Validation phase
The predictive score from bootstrap analysis performed well with a mean bootstrap-corrected calibration coefficient of 0.465 (95% CI, 0.463–0.466) relative to the original coefficient of 0.477. The bootstrap-corrected C statistic was 0.732 (95% CI, 0.732–0.733).
External validation was performed using data from patients at TUH. The majority of patient characteristics were similar to Ramathibodi Hospital except for rates of alcohol or drug use, cancer, thyroid mass/nodule, and being immunocompromised (Table 1). Of the 411 patients, 87 had abnormal preoperative chest radiographs, a prevalence of 21.2% (95% CI, 17.3–25.4%). The agreement in chest radiograph interpretation was 95.9% with kappa statistic of 0.87.
The model fitted well with the external validation data (HL-GOF χ2 = 4.70, p = 0.195) with an O/E ratio of 0.987 (95% CI, 0.888–1.085). The C statistic was 0.731 (95% CI, 0.674–0.789). Risk prediction scores were calculated with 230 (60%) and 181 (40%) patients classified in the low- and intermediate-to-high-risk group with abnormal chest radiographs of 10.8% (95% CI, 7.7–14.6%) and 34.3% (95% CI, 21.4-41.7%), respectively.
Abnormal chest radiographs had a major impact on perioperative management in four patients. One patient (0.4%) in the low-risk group postponed surgery and was diagnosed with active tuberculosis. Three patients (2.3%) were in the intermediate-to-high-risk group, with two having surgery cancelled. They were diagnosed with non-tuberculosis mycobacterium and pulmonary metastasis. One patient scheduled for spinal surgery was newly diagnosed with lung cancer.
Discussion
We developed and validated a risk prediction score for abnormal preoperative chest radiographs. The model consisted of four predictors including age, pulmonary disease, cardiac disease, and diabetes. The risk of abnormality increased with age from low to at least intermediate in patients of 65 years and older. Higher rates of abnormality were reported in patients with active cardiopulmonary conditions compared to those with stable conditions. There was little value derived from preoperative chest radiographs in patients with stable airway diseases as the radiographs rarely indicate abnormalities that affect clinical management [28]. The finding that diabetes patients were more likely to have abnormal preoperative chest radiographs suggests increased cardiovascular risk in patients with longer disease duration or the presence of renal dysfunction or microalbuminuria [29]. However, our study did not reveal association with hypertension, AKI/CKD, or cerebrovascular disease.
Our study also investigated associations between abnormal preoperative chest radiographs and other reported predictors. Previous investigations associated with smoking status proved inconclusive [8, 30, 31]. Our univariate analysis revealed an association between smoking and abnormal preoperative chest radiographs. However, the subsequent multivariate analysis failed to demonstrate an association. Our results support previous reports that preoperative chest radiographs should not be recommended in patients with thyroid mass or undergoing high-risk surgery [10, 32]. Weibman [33] recommended performing preoperative chest radiographs in cancer patients based on a high incidence of new findings, but our results failed to demonstrate this association. Furthermore, computed tomography examination, not chest radiograph, is currently used for staging in cancer patients. We were also unable to demonstrate associations between tuberculosis symptoms, alcohol/drug use, or immunocompromised status with abnormal chest radiographs.
Our study supports the opinion of previous researchers that routinely performing preoperative chest radiographs may not be necessary because most abnormalities are chronic and do not affect clinical management [1, 3, 5]. Preoperative chest radiographs in the low-risk group provided less clinical impact compared to the other three groups. The benefit may not justify the risk of radiation-induced cancer, delayed operation, over-investigation, and costs [3]. Our risk prediction score may help practitioners identify patients at higher risk of abnormal chest radiographs. Furthermore, it can reduce medical expenses for patients and workload for healthcare professionals, especially in limited resource settings.
There were several strengths to our study. Firstly, we followed the STROBE and TRIPOD recommendations for conducting observational studies and developing prediction scores [34, 35]. Secondly, we simultaneously considered predictors identified from previous studies and validated the models with a good performance [27]. Thirdly, the small amount of missing data was appropriately imputed. Lastly, our scoring system is easy to use.
Our study had some limitations. Because of the COVID-19 pandemic, our study size was smaller than intended, but it was deemed sufficient. The prevalence was higher than anticipated, and the number of abnormal chest radiographs per parameter was 17.1, within the range of recommendation [24]. The estimated shrinkage was 0.92, higher than the recommendation [36].
The proportion of abnormal preoperative chest radiographs in our study was higher than the proportion in a previous meta-analysis [5]. This can be attributed to different definitions of abnormal radiographs and a variety of patient characteristics. Our study included only adult patients in Thai tertiary hospitals, where tuberculosis is prevalent, while the meta-analysis included only studies from European and North American countries [5]. Although our risk prediction score has not yet been tested in these countries, it may still prove clinically useful. Meanwhile, tuberculosis is endemic in many parts of the world and is becoming more prevalent in more developed countries through political and economic migration [37]. To generalize, further study is needed of the validity, clinical impact, and cost-effectiveness of our scoring system in other settings.
In conclusion, a prediction score with good performance for classifying risk associated with abnormal preoperative chest radiographs was developed. Advanced age, pulmonary disease, cardiac disease, and diabetes were associated with higher risk of abnormalities. Our risk prediction model may help identify patients at higher risk of chest abnormalities.
References
Munro J, Booth A, Nicholl J (1997) Routine preoperative testing: a systematic review of the evidence. Health Technol Assess 1(i–iv):1–62
Schueller G, Herold CJ (2003) Lung metastases. Cancer Imaging 3:126–128
Joo HS, Wong J, Naik VN et al (2005) The value of screening preoperative chest x-rays: a systematic review. Can J Anaesth 52:568–574
Bouillot JL, Fingerhut A, Paquet JC et al (1996) Are routine preoperative chest radiographs useful in general surgery? A prospective, multicentre study in 3959 patients. Association des Chirurgiens de l’Assistance Publique pour les Evaluations medicales. Eur J Surg 162:597–604
Archer C, Levy AR, McGregor M (1993) Value of routine preoperative chest x-rays: a meta-analysis. Can J Anaesth 40:1022–1027
Bhuripanyo K, Prasertchuang C, Chamadol N et al (1990) The impact of routine preoperative chest X-ray in Srinagarind Hospital, Khon Kaen. J Med Assoc Thai 73:21–28
Ali IS, Khan M, Khan MA (2013) Routine preoperative chest x-ray and its impact on decision making in patients undergoing elective surgical procedures. J Ayub Med Coll Abbottabad 25:23–25
Sommerville TE, Murray WB (1992) Information yield from routine pre-operative chest radiography and electrocardiography. S Afr Med J 81:190–196
Rucker L, Frye EB, Staten MA (1983) Usefulness of screening chest roentgenograms in preoperative patients. JAMA 250:3209–3211
Tape TG, Mushlin AI (1988) How useful are routine chest x-rays of preoperative patients at risk for postoperative chest disease? J Gen Intern Med 3:15–20
Apfelbaum JL, Connis RT, Nickinovich DG et al (2012) Practice advisory for preanesthesia evaluation: an updated report by the American Society of Anesthesiologists Task Force on preanesthesia evaluation. Anesthesiology 116:522–538
Qaseem A, Snow V, Fitterman N et al (2006) Risk assessment for and strategies to reduce perioperative pulmonary complications for patients undergoing noncardiothoracic surgery: a guideline from the American College of Physicians. Ann Intern Med 144:575–580
O’Neill F, Carter E, Pink N et al (2016) Routine preoperative tests for elective surgery: summary of updated NICE guidance. BMJ 354:i3292
Card R, Sawyer M, Degnan B et al (2014) Perioperative protocol. Institutes for Clinical Systems Improvement
Young EM, Farmer JD (2017) Preoperative Chest Radiography in Elective Surgery: Review and Update. S D Med 70:81–87
Moons KG, Altman DG, Reitsma JB et al (2015) New Guideline for the Reporting of Studies Developing, Validating, or Updating a Multivariable Clinical Prediction Model: The TRIPOD Statement. Adv Anat Pathol 22:303–305
Vandenbroucke JP, von Elm E, Altman DG et al (2014) Strengthening the Reporting of Observational Studies in Epidemiology (STROBE): explanation and elaboration. Int J Surg 12:1500–1524
WHO Expert Consultation (2004) Appropriate body-mass index for Asian populations and its implications for policy and intervention strategies. Lancet 363:157–163
Miskovic A, Lumb AB (2017) Postoperative pulmonary complications. Br J Anaesth 118:317–334
Department of disease control Systematic screening for active TB and drug-resistant TB, Bangkok, Thailand, Aksorn graphic and design publishing limited, 2018;10.
Freeman WK, Gibbons RJ (2009) Perioperative cardiovascular assessment of patients undergoing noncardiac surgery. Mayo Clin Proc 84:79–90
Levey AS, Eckardt KU, Tsukamoto Y et al (2005) Definition and classification of chronic kidney disease: a position statement from Kidney Disease: Improving Global Outcomes (KDIGO). Kidney Int 67:2089–2100
Kristensen SD, Knuuti J, Saraste A et al (2014) 2014 ESC/ESA Guidelines on non-cardiac surgery: cardiovascular assessment and management: The Joint Task Force on non-cardiac surgery: cardiovascular assessment and management of the European Society of Cardiology (ESC) and the European Society of Anaesthesiology (ESA). Eur Heart J 35:2383–2431
Guyatt GH (2006) Determining prognosis and creating clinical decision rules. In: Haynes RB, Sackett DL, Guyatt GH et al (eds) Clinical epidemiology: how to do clinical practice research. Lippincott Williams & Wilkins (LWW), Philadelphia, pp 323–356
Moons KG, Donders RA, Stijnen T et al (2006) Using the outcome for imputation of missing predictor values was preferred. J Clin Epidemiol 59:1092–1101
Pencina MJ, D’Agostino RB Sr, D’Agostino RB Jr et al (2008) Evaluating the added predictive ability of a new marker: from area under the ROC curve to reclassification and beyond. Stat Med 27:157–172 (discussion 207–112)
Toll DB, Janssen KJ, Vergouwe Y et al (2008) Validation, updating and impact of clinical prediction rules: a review. J Clin Epidemiol 61:1085–1094
Thiboutot J, Yuan W, Park HC et al (2019) Current Advances in COPD Imaging. Acad Radiol 26:335–343
Bertoluci MC, Rocha VZ (2017) Cardiovascular risk assessment in patients with diabetes. Diabetol Metab Syndr 9:25
Garcia-Miguel FJ, Garcia Caballero J, Gomez de Caso-Canto JA (2002) Indications for thoracic radiography in the preoperative evaluation for elective surgery. Rev Esp Anestesiol Reanim 49:80–88
De la Matta MM, Herrera González A, López Conejos JA et al (2011) Utility of preoperative chest radiography in smokers undergoing transurethral resection of urinary bladder tumors. Rev Esp Anestesiol Reanim 58:203–210
Mikhael A, Patell R, Tabet M et al (2018) Chest X-ray prior to thyroidectomy: is it really needed? World J Surg 42:1403–1407
Weibman MD, Shah NK, Bedford RF (1987) Influence of preoperative chest X-rays on the perioperative manakement of cancer patients. Anesthesiology 67:A332–A332
Zamanipoor Najafabadi AH, Ramspek CL, Dekker FW et al (2020) TRIPOD statement: a preliminary pre-post analysis of reporting and methods of prediction models. BMJ Open 10:e041537
Heus P, Damen J, Pajouheshnia R et al (2018) Poor reporting of multivariable prediction model studies: towards a targeted implementation strategy of the TRIPOD statement. BMC Med 16:120
Riley RD, Ensor J, Snell KIE et al (2020) Calculating the sample size required for developing a clinical prediction model. BMJ 368:m441
Mangione CM, Barry MJ, Nicholson WK et al (2023) Screening for latent tuberculosis infection in adults: US preventive services task force recommendation statement. JAMA 329: 1487–1494
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
The study was approved by the Human Research Ethics Committee of the Faculty of Medicine Ramathibodi Hospital and Thammasat University No. 1.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Seangleulur, A., Thakkinstian, A., Supaopaspan, W. et al. Optimizing the Yield of Abnormal Preoperative Chest Radiographs in Elective Non-cardiothoracic Surgery: Development of a Risk Prediction Score and External Validation. World J Surg 47, 2698–2707 (2023). https://doi.org/10.1007/s00268-023-07146-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-023-07146-7