Abstract
Background
Bedside percutaneous dilatational tracheostomy (PDT) and percutaneous endoscopic gastrostomy (PEG) are common procedures performed in the intensive care unit (ICU). Venous thromboembolism (VTE) prophylaxis is frequently prescribed to ICU patients and it remains unclear whether pre-procedure discontinuation is necessary.
Methods
This multi-center prospective observational study aimed to describe bleeding rates in patients undergoing bedside PEG or PDT who did or did not have VTE prophylaxis held. Decision to hold prophylaxis was made by the operating physician. The primary endpoint was the rate of peri-procedural bleeding complications. Secondary endpoints included quantification of held doses in the peri-procedural period, rate of venous thromboembolism, and characteristics associated with having prophylaxis held.
Results
91 patients were included over a 2-year period. Patients were on average aged 54 years, 40% female, mostly admitted to the trauma service (59%), and most commonly underwent bedside PDT (59%). Overall, 21% of patients had doses of pre-procedure prophylaxis held. Bleeding events occurred in 1 patient (1.4%) who had prophylaxis continued and in 1 patient (5.0%) who had prophylaxis held, a rate difference of 3.6% (95% CI–9.5%, 16.7%). One bleeding event was managed with bedside surgical repair and one with blood transfusion. There were 10 VTE events, all of whom had prophylaxis continued during the pre-procedure period but 3 had prophylaxis held after the procedure.
Conclusions
Bleeding complications were rare and did not significantly differ depending on whether prophylaxis was held or not. Future research is required to confirm the lack of risk with continuing prophylaxis through bedside procedures.
Similar content being viewed by others

Avoid common mistakes on your manuscript.
Introduction
Percutaneous dilatational tracheostomy (PDT) and percutaneous endoscopic gastrostomy (PEG) are procedures that may be safely completed at the bedside in the intensive care unit (ICU) [1,2,3,4,5]. Bleeding is the most common early complication of tracheostomy [6], occurring in up to 5% of patients, and occurs in up to 2.5% of PEG placements [7,8,9,10,11].While bedside procedures offer shorter operative time, fewer adverse events from anesthesia, and lower resource utilization compared to procedures completed in the operating room (OR), it is unclear whether patients continued on anticoagulation or antiplatelet agents are at increased risk for bleeding during these procedures [12].
Patients admitted to the ICU are frequently placed on anticoagulant or antiplatelet medications for a number of indications. Nearly all ICU patients are indicated for chemical venous thromboembolism (VTE) prophylaxis with subcutaneous low molecular weight heparin (LMWH) or unfractionated heparin (UH) [13]. Variation in practice exists in whether prophylactic anticoagulation requires temporary discontinuation prior to bedside operations. A retrospective review of bleeding complications on prophylactic anticoagulation in patients undergoing bedside PEG or PDT did not reveal increased risk of bleeding complications, but this has yet to be prospectively confirmed [14]. In this prospective cohort study, we aimed to describe the difference in rates of bleeding complications between patients who did and did not have prophylactic anticoagulation held prior to bedside PEG or PDT.
Methods
This was a prospective multicenter observational trial conducted from February 2017 to February 2019 at two Level I trauma centers in the USA. Patients aged 16 years or older undergoing bedside percutaneous endoscopic gastrostomy (PEG) and/or percutaneous dilatational tracheostomy (PDT) who were receiving prophylactically dosed anticoagulation with subcutaneous heparin, low molecular weight heparin, or a direct acting anticoagulant (DOAC) were eligible for inclusion. Patients were identified by investigators who reviewed the surgical and ICU procedural schedules or by receiving direct report from the clinician completing the procedure. Patients were excluded if they were receiving therapeutically dosed anticoagulation, were prisoners, pregnant, were decisionally impaired, or if the procedure was conducted in an open or surgical manner. Eligible patients were asked for informed consent to be included in the cohort and data were collected prospectively after the procedure. Decision to hold or continue prophylactic anticoagulation was made by the individual surgeon completing the procedure and was not influenced by study personnel. The study was reviewed and approved by both participating sites’ Institutional Review Boards.
Data collection
After consent, data were prospectively collected from the electronic medical record into a pre-specified Research Electronic Data Capture (REDCap) form [15, 16]. Demographic information included the age, weight, gender, and admitting service (trauma, medical, surgical, or other) of each patient. The hospital and ICU lengths of stay prior to the procedure, type of prophylactic anticoagulation name, dose, frequency, and whether dose adjustments had been made, and type of antiplatelet agent ordered, if any, were also collected. The procedure performed, number of doses of prophylactic anticoagulation held before the procedure, markers of renal function, including serum creatinine (SCr) and blood urea nitrogen (BUN) on the day of the procedure, and any laboratory coagulation values (international normalized ratio [INR], platelet count, fibrinogen, anti-Xa activity, or activated partial thromboplastin time [aPTT]) on the day of the procedure were also recorded. Outcome variables included any peri-procedure bleeding complications requiring intervention which occurred within 48 h of the procedure, whether an intervention was performed to address the bleeding (blood product transfusion, bedside surgical intervention, open surgical intervention, or other), whether a venous thromboembolism occurred post-procedure during the index hospital admission, and whether any doses of prophylactic anticoagulation were held after the procedure. 48 h was selected to minimize the likelihood of non-operative confounders contributing to bleeding events post-operatively.
Bleeding events were defined as any bleeding during the procedure that required more than direct pressure to manage or any post-procedure bleeding that required any intervention (direct pressure, topical hemostatic agent, suture control, or re-exploration). Thromboembolic events were defined as any venous or arterial thromboembolism detected after the procedure during the index admission. One site performed routine, surveillance venous duplex ultrasounds on hospital day 3 then weekly to screen for VTE while one site did not complete routine Doppler ultrasounds but did perform whole leg sonography when clinical suspicion for lower extremity VTE was high.
Outcomes
The primary objective of this study was to describe the rates of peri-procedural bleeding events after bedside PDT or PEG in patients whose VTE prophylaxis was or was not held. Secondary endpoints included quantification of held doses in the peri-procedural period, predictors of having prophylaxis held, identification of risk factors for bleeding events, and the incidence of VTE events in either group.
Statistical analysis
Baseline characteristics were reported as frequencies and percentages or medians and interquartile ranges (IQR) and continuous and ordinal variables were compared between groups with the Mann–Whitney U test or Student’s t-test as appropriate. The difference in the rates of events was compared using the chi-squared test or Fisher’s exact test as appropriate and 95% confidence intervals for rate differences were calculated using the proportion test. Predictors of bleeding events or whether anticoagulation was held was completed using univariable logistic regression with age, gender, serum creatinine, BUN, platelet count, INR, procedure performed, and prophylactic anticoagulant agent used as variables. Predictors of whether VTE prophylaxis was held included the same predictive variables used for bleeding events. Analysis was completed using R version 4.0.0 (R Core Team, R Foundation for Statistical Computing, Vienna, Austria).
Results
A total of 91 patients were included in the cohort with 48 from site 1 and 43 from site 2. The baseline characteristics of the cohort are listed in Table 1 and were well-matched between sites. Patients who did or did not have prophylactic anticoagulation held prior to PEG or PDT were similar with no major differences between groups. Overall, patients were more commonly male (60%), most likely to be admitted by the trauma service (59%), and had been in the hospital for a median of 12 days and in the ICU for 11 days prior to the procedure. Both procedures were performed within 12 h of each other in 35% of patients, whereas 59% of patients only received a PDT and 6.6% of patients only received a PEG at bedside.
Details on the characteristics of prophylactic anticoagulation and antiplatelet agents prescribed are listed in Table 2. Three patients received higher doses of prophylaxis, all adjusted for body mass index (BMI). None of these patients had prophylaxis held. 21% of patients were receiving low-dose aspirin (81 mg) and there was no difference in the rate of antiplatelet use between patients who did and did not have anticoagulation held (25% vs 20%).
There were two bleeding events in the cohort, one in the continuation group (1.4%) and one in the held group (5%) for a rate difference between continuing and holding prophylaxis of -3.6% (95% CI–9.5%, 16.7%) (Table 3). This was also found to be non-significant via Fisher’s exact test (p = 0.54). In response to the bleeding event, one patient had the bleeding surgically repaired at bedside and one required only blood transfusion. The patient who received a transfusion had cardiovascular comorbidities and received a transfusion based on higher transfusion goals after the procedure. Overall, 18% of patients had at least one dose of prophylaxis held after the procedure, with 33% of patients whose prophylaxis was held pre-procedure having dosing held after the procedure compared to 14% of patients whose prophylaxis was not held (p = 0.07). Specific characteristics of each patient who experienced a bleeding event is listed in Table 4. Both patients had doses of prophylaxis held after the bleeding event in response to the bleeding.
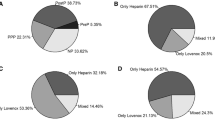
There were 10 thromboembolic events identified in the cohort. None (0% vs. 30%, p = 0.11) of the patients with diagnosed thromboembolism had VTE prophylaxis held prior to their procedure but 3 (30% vs. 17%, p = 0.40) had prophylaxis held after the procedure (Fig. 1). Nine of the thromboembolic events occurred at one site which conducted routine surveillance venous duplex ultrasounds (16.4% event rate) compared to one at one site which did not conduct surveillance ultrasounds (2.7% event rate), a significant difference (p = 0.04). One thromboembolic event was a pulmonary embolism, eight were deep vein thromboses, and one was a middle cerebral artery occlusion.
Factors predicting the decision to hold prophylactic anticoagulation prior to procedure were analyzed and are listed in Table 5. Of the factors evaluated (age, gender, SCr, BUN, platelet count, procedure type, and prophylaxis type), only type of prophylaxis was found to be associated with the decision to hold prophylaxis. Compared to enoxaparin 30 mg every 12 h, patients receiving enoxaparin 40 mg every 24 h were less likely (OR 0.77, 95% CI 0.62–0.97) to have doses held prior to the procedure. Other regimens were not found to confer significantly different odds of having prophylaxis held.
Discussion
In this prospective multicenter observational study of patients undergoing bedside PEG or PDT, the overall rate of bleeding events was found to be 2.2% and was unaffected by whether prophylactic anticoagulation was held pre-procedure. Bleeding events were managed by either bedside intervention or blood transfusion and did not require transition to open procedure. Neither procedure was more likely to result in held prophylactic anticoagulation, nor did any patient-specific factor including age, gender, renal function, or coagulation status. Patients receiving enoxaparin once daily, however, were less likely have doses held than other regimens. These results help confirm prior results suggesting that holding prophylactic anticoagulation is unnecessary prior to bedside PEG or PDT in the ICU [14].
Bedside PEG and PDT have been known to be safe and effective for over two decades and are common practice in many ICUs [5, 17,18,19,20,21]. Among the most serious complications of bedside procedures are peri- and post-operative bleeding events. While variably defined in studies comparing bedside to open procedures, rates of bleeding range from 0 to 10% [7, 8, 22]. Bleeding events described include stoma site bleeding or other minor bleeding and can often be managed with minimally invasive surgical repair. While the risks of the procedures are low, concern still remains over whether it is safe to continue prophylactic anticoagulation in the periprocedural time period. Patients receiving anticoagulation have been described as being high-risk candidates for bedside procedures [7], but the distinction between therapeutic and prophylactic doses of anticoagulation is not always made. Prior single-center retrospective analyses of prophylactic anticoagulation use prior to bedside procedures have failed to detect a significant signal of harm [14, 23].
Holding prophylactic anticoagulation in patients undergoing bedside PEG or PDT is not without risk. Patients undergoing bedside procedures are most commonly admitted to the ICU and likely have substantial risk factors for thromboembolic events [24, 25]. A prospective observational study evaluating the effect of missing prophylactic anticoagulation doses in a surgical ICU reported a linear relationship between missed doses of prophylaxis and risk of venous thromboembolism, with patients who missed between 2 and 4 doses of prophylaxis being at an 8.49 times increased odds of experiencing deep vein thrombosis compared to patients without interruption [26]. A different cohort study of two national registries of patients undergoing colostomy found that in high-risk patients missing at least 1 dose of VTE prophylaxis significantly increased the odds of experiencing a VTE (OR 2.41, 95% CI 1.27–4.57) [27]. Similarly, PDT has been demonstrated to be safe even in patients with coagulopathy (INR > 1.3 or platelet count < 80,000), with rates of bleeding complications occurring in 1.22% of coagulopathic patients compared to 0.94% of noncoagulopathic patients (P > 0.99) [28]. While our study did not reveal an increased risk of VTE events with holding pre-procedure prophylaxis, patients with prophylaxis post-procedure tended to develop more VTEs, although this was not significant. One arterial thrombus was detected in our sample (an MCA stroke); while this is not exclusively related to held prophylaxis dosing, it demonstrates the pro-thrombotic state of these patients. A larger sample size would be needed to accurately estimate the risk of developing a VTE when prophylaxis is held before or after bedside procedures.
The decision to hold prophylactic anticoagulation was left to the discretion of the surgeons in this study. An attempt to identify factors which led surgeons to hold anticoagulation did not identify any significant patient-specific factor, including age, gender, renal function, BUN, or platelet count. Not enough patients had an INR available at the time of the procedure to make accurate predictions. Neither procedure individually nor combined was found to be associated with the decision to hold doses. The only regimen found to have been held less often was enoxaparin 40 mg once daily (OR 0.70, 95% CI 0.62–0.96). Antiplatelet agents (exclusively aspirin 81 mg) were commonly used in this cohort (21%), but none of these patients developed bleeding events after their procedure.
This study has several limitations. First, while patients were included prospectively, they were not randomized to either group, thus introducing the potential that patients at higher risk of bleeding may have been ordered to have prophylaxis held. This introduces the potential that rates of bleeding did not differ between groups because of external confounding. Second, manual patient identification limited the number of patients available for screening, which led to a slower than usual enrollment rate and lower sample size than was expected. This may introduce selection bias, as patients undergoing bedside procedures who were not identified for data collection may not have randomly occurred. We also did not retrospectively review all PEG/PDT cases that were not included in prospective data collection, so a true proportion of procedures included could not be reported. The small sample size reduced the power of the study to detect an uncommon event (bleeding). Third, specific factors which led to the decision to hold prophylaxis revealed enoxaparin 40 mg once daily was less likely to be held than other regimens. Patients also may have been less likely to have this held because the wide dosing interval may be less worrisome to operating physicians. This may lead to patients who otherwise would have had prophylaxis held if the interval was shorter to be allocated to the not held group independent of patient-specific risk factors for bleeding. Fourth, we did not have an open surgical group to compare rates of bleeding to the bedside group. This may have helped further elucidate differences in the safety of these two operative techniques. Screening for VTE occurred differently between sites, as well, with one site conducting routine VTE surveillance scans and one not, which is reflected in the significantly higher rate of VTE in the site 1 patients compared to site 2.
This study does have several strengths, however. The prospective, multi-center nature of the design helps to address practice variation between sites and also allowed investigators to actively screen for bleeding events. The broad inclusion criteria also produced an externally valid, generalizable population to both medical and surgical practitioners. This study also described specific prophylaxis regimens which are often missing from similar reports. While few patients had their prophylaxis regimens adjusted for patient-specific factors, we did collect these variables as well in case this influenced the primary outcome.
Conclusion
Our prospective cohort study found that rates of bleeding between groups did not differ significantly. We conclude that holding prophylactic anticoagulation is likely not necessary before bedside PEG or PDT, although randomized studies would serve to validate this conclusion. Additional research is needed to make conclusions on the impact of antiplatelet therapy or treatment anticoagulation and the risk of bleeding for these procedures.
References
Silvester W, Goldsmith D, Uchino S et al (2006) Percutaneous versus surgical tracheostomy: a randomized controlled study with long-term follow-up. Crit Care Med 34:2145–2152. https://doi.org/10.1097/01.CCM.0000229882.09677.FD
Barbetti JK, Nichol AD, Choate KR et al (2009) Prospective observational study of postoperative complications after percutaneous dilatational or surgical tracheostomy in critically ill patients. Crit Care Resusc 11:244–249
Carrillo EH, Heniford BT, Osborne DL et al (1997) Bedside percutaneous endoscopic gastrostomy: a safe alternative for early nutritional support in critically ill trauma patients. Surg Endosc 11:1068–1071. https://doi.org/10.1007/s004649900531
Stiegmann GV, Goff JS, Silas D et al (1990) Endoscopic versus operative gastrostomy: final results of a prospective randomized trial. Gastrointest Endosc 36:1–5. https://doi.org/10.1016/S0016-5107(90)70911-X
Kornblith LZ, Burlew CC, Moore EE et al (2011) One thousand bedside percutaneous tracheostomies in the surgical intensive care unit: Time to change the gold standard. J Am Coll Surg 212:163–170. https://doi.org/10.1016/j.jamcollsurg.2010.09.024
Simon M, Metschke M, Braune SA et al (2013) Death after percutaneous dilatational tracheostomy: a systematic review and analysis of risk factors. Crit Care. https://doi.org/10.1186/cc13085
Kidane B, Pierre AF (2018) From open to bedside percutaneous tracheostomy. Thorac Cardiovasc Surg 28:263–276. https://doi.org/10.1016/j.thorsurg.2018.03.001
Delaney A, Bagshaw SM, Nalos M (2006) Percutaneous dilatational tracheostomy versus surgical tracheostomy in critically ill patients: a systematic review and meta-analysis. Crit Care 10:1–13. https://doi.org/10.1186/cc4887
de Leyn P, Bedert L, Delcroix M et al (2007) Tracheotomy: clinical review and guidelines. Eur J Cardiothorac Surg 32:412–421
Larson DE, Burton DD, Schroeder KW, DiMagno EP (1987) Percutaneous endoscopic gastrostomy. Indications, success, complications, and mortality in 314 consecutive patients. Gastroenterology 93:48–52. https://doi.org/10.1016/0016-5085(87)90312-X
Schapiro GD, Edmundowicz SA (1996) Complications of percutaneous endoscopic gastrostomy. Gastrointest Endosc Clin N Am 6:409–422. https://doi.org/10.1016/S1052-5157(18)30369-6
Ljungdahl M, Sundbom M (2006) Complication rate lower after percutaneous endoscopic gastrostomy than after surgical gastrostomy: a prospective, randomized trial. Surg Endosc 20:1248–1251. https://doi.org/10.1007/s00464-005-0757-6
Schünemann HJ, Cushman M, Burnett AE et al (2018) American society of hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood Adv 2:3198–3225
Barton CA, McMillian WD, Osler T et al (2012) Anticoagulation management around percutaneous bedside procedures. J Trauma Inj Infect Crit Care 72:815–820. https://doi.org/10.1097/TA.0b013e31824fbadf
Harris PA, Taylor R, Minor BL et al (2019) The REDCap consortium: building an international community of software platform partners. J Biomed Inform 95:103208
Harris PA, Taylor R, Thielke R et al (2009) Research electronic data capture (REDCap)-a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform 42:377–381. https://doi.org/10.1016/j.jbi.2008.08.010
Massick DD, Yao S, Powell DM et al (2001) Bedside tracheostomy in the intensive care unit: a prospective randomized trial comparing open surgical tracheostomy with endoscopically guided percutaneous dilational tracheotomy. Laryngoscope 111:494–500. https://doi.org/10.1097/00005537-200103000-00021
Wease GL, Frikker M, Villalba M (1996) Bedside tracheostomy in the intensive care unit. Arch Surg 131:552. https://doi.org/10.1001/archsurg.1996.01430170098018
Beltrame F, Zussino M, Martinez B et al (2008) Percutaneous versus surgical bedside tracheostomy in the intensive care unit: a cohort study. Minerva Anestesiol 74:529–535
Eisen GM, Baron TH, Dominitz JA et al (2002) Role of endoscopy in enteral feeding. Gastrointest Endosc 55:794–797. https://doi.org/10.1016/S0016-5107(02)70405-7
Porter JM, Ivatury RR (1999) Preferred route of tracheostomy–percutaneous versus open at the bedside: a randomized, prospective study in the surgical intensive care unit. Am Surg Am Surg 65:142–146
Lee CG, Kang HW, Lim YJ et al (2013) Comparison of complications between endoscopic and percutaneous replacement of percutaneous endoscopic gastrostomy tubes. J Korean Med Sci 28:1781. https://doi.org/10.3346/jkms.2013.28.12.1781
Sordo S, Holloway T, Harrison H (2012) Holding anticoagulation for bedside procedures in the ICU: Is it necessary? Crit Care Med 40:1–328. https://doi.org/10.1097/01.ccm.0000424967.84278.86
Geerts WH, Jay RM, Code KI et al (1996) A comparison of low-dose heparin with low-molecular-weight heparin as prophylaxis against venous thromboembolism after major trauma. N Engl J Med 335:701–707. https://doi.org/10.1056/nejm199609053351003
Meade M, Guyatt G, Walter S et al (2011) Dalteparin versus unfractionated heparin in critically Ill patients. N Engl J Med 364:1305–1314. https://doi.org/10.1056/nejmoa1014475
Louis SG, Sato M, Geraci T et al (2014) Correlation of missed doses of enoxaparin with increased incidence of deep vein thrombosis in trauma and general surgery patients. JAMA Surg 149:365. https://doi.org/10.1001/jamasurg.2013.3963
Khorfan R, Kreutzer L, Love R et al (2021) Association between missed doses of chemoprophylaxis and VTE incidence in a statewide colectomy cohort. Ann Surg 273:e151–e152. https://doi.org/10.1097/SLA.0000000000004349
Pandian V, Vaswani RS, Mirski MA et al (2010) Safety of percutaneous dilational tracheostomy in coagulopathic patients. Ear Nose Throat J 89:387–395
Acknowledgements
We would like to thank Dr. Vijay Jayaraman for his invaluable assistance with data collection for this project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interests
The authors have no conflicts of interest to disclose.
Ethical approval
This study was approved by the IRB at each participating institution.
Consent informed
Informed consent was obtained for all participants in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Barton, C.A., Shapiro, D.S., Webb, A.J. et al. Safety of Prophylactic Anticoagulation During Bedside Procedures: A Prospective Multicenter Observational Study. World J Surg 46, 2625–2631 (2022). https://doi.org/10.1007/s00268-022-06662-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-022-06662-2