Abstract
Background
The short- and long-term efficacy of laparoscopic surgery for elderly patients with gastric cancer has not been evaluated. We aimed to use propensity score matching to clarify the efficacy of laparoscopic gastrectomy (LG) for elderly patients with gastric cancer aged ≥80 years.
Methods
We retrospectively collected data from 159 consecutive patients with gastric cancer aged ≥80 years who underwent gastrectomy with curative intent at our institution between 2004 and 2015. Propensity score matching was applied to compare the open gastrectomy (OG) and LG. Short- and long-term outcomes were evaluated between the propensity-matched groups.
Results
Patients’ backgrounds and surgical factors were similar in both groups except for blood loss. The median time to first flatus was significantly shorter in the LG group than in the OG group (P = 0.002). The postoperative hospital stay was significantly shorter in the LG group (P = 0.014). The complication rate of Clavien–Dindo grade III or higher was significantly lower in the LG group (3% vs. 23%, P = 0.023). The 5-year overall survival and 5-year disease-specific survival rates were better in the LG group than in the OG group, but the differences were not significant (45% vs. 42% and 67% vs 57%, respectively).
Conclusion
LG was associated with good short-term outcomes and acceptable oncologic outcomes compared with OG in these propensity-matched patients aged ≥80 years.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Gastric cancer (GC) remains a major cause of cancer-related death and is the fifth most common cancer in the world [1]. Because of the rapidly aging global population, the number of elderly patients with gastric cancer has been increasing steadily, especially in Japan [2, 3].
Surgical resection remains the mainstay treatment for patients with GC; therefore, the number of elderly patients undergoing gastrectomy for GC has also increased [4]. The natural life expectancy of older people is apparently shorter than that of younger people, and they generally have various comorbidities, leading to a limited capacity to tolerate surgical procedures [5, 6]. In addition, surgeons need to overcome perioperative issues specific to elderly patients, such as postoperative delirium, sarcopenia, and frailty [7,8,9]. Therefore, surgeons have some difficulties in making decisions on treatment strategies for elderly patients with malignancies. Considering their limited remaining lifetime, their postoperative quality of life is as valuable as is the need to cure or remove their cancers.
For the reasons described above, minimally invasive approaches are considered when performing operations on elderly patients. Laparoscopic gastrectomy (LG) has been widely accepted as a minimally invasive surgery for GC, especially early GCs [10]. The short-term advantages of LG have also been proved in randomized controlled trials (RCTs) and a large cohort study based on a nationwide registry database comparing LG with open gastrectomy (OG) [11,12,13,14,15,16,17]. However, few studies on the feasibility of using LG in elderly patients, especially in those aged ≥80 years, have been reported.
Here, we aimed to clarify short- and long-term outcomes in patients with GC aged ≥80 years who underwent LG with curative intent, compared with a propensity-matched group of patients who underwent OG.
Patients and methods
Patients
Patients who underwent gastrectomy for GC were selected from the prospective database of the Osaka Medical College Hospital (OMCH) between January 2004 and December 2015. Written informed consent was obtained from all patients. This study was approved by the institutional review board of OMCH.
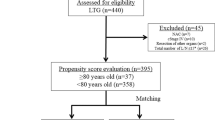
The patient eligible criteria were as follows: age ≥80 years, histologically confirmed gastric adenocarcinoma, gastrectomy as a primary treatment without distant metastases, and R0 resection. The exclusion criteria were as follows: any other concomitant advanced malignancy, emergency operations, thoracotomies, and simultaneous resection of the colon or pancreas. A total of 159 patients were enrolled, of whom 41 patients underwent OG, and 118 underwent LG. We decided whether to treat with OG or LG depending on the tumor stage based on the Japanese treatment guidelines [18], which recommend LG for clinical stage I. Since 2013, in the hope of minimizing invasiveness, we have adopted laparoscopic procedures in patients aged ≥80 years with clinically advanced GC after obtaining their well-informed consent.
Data collection
The patients’ background, including age, gender, body mass index (BMI), and number of comorbidities, was collected. To assess the patient status objectively, Onodera’s prognostic nutritional index [19] (10 × serum albumin [g/dL] + 0.005 × peripheral lymphocyte count [per mm3]) and the American Society of Anesthesiologists (ASA) Physical Status were also examined. The seventh UICC/AJCC TNM classification was used for clinicopathological evaluations [20]. Intraoperative findings, such as time and blood loss, and postoperative course including complications based on the Clavien–Dindo (C–D) classification [21] were collected retrospectively and analyzed statistically. We also divided our investigation into three periods: early, 2004–2009; middle, 2010–2012; and late, 2013–2015.
Follow-up schedule
All patients were received follow-up for at least 5 years after surgery. Follow-up surveillance were scheduled at 3–6-month intervals for the first 3 years. After this, the patients received a follow-up surveillance every 6–12 months by outpatient visits or telephone until death caused by a recurrence of GC or up to December 31, 2020. Based on this surveillance, data on the 5-year overall survival (OS) and disease-specific survival (DSS) were collected. The OS time was defined as the time from the surgery to death for any reason or to interruption of the follow-up. The DSS time was defined as the time from surgery to death from GC, including operative mortality, or to interruption of the follow-up.
Propensity score matching
We performed propensity score matching (PSM) to minimize bias between the OG and LG groups. Propensity scores were calculated using a logistic regression model and the following variables: ASA score, BMI, clinical T factor, clinical N factor, type of gastrectomy (total or not), and lymph node dissection (D2 or not). Nearest neighbor matching without replacement within a caliper was used. The size of the caliper was set to 0.2 of the standard deviation of the logit of the estimated propensity score. Patients whose scores were found to be outside the caliper and unmatched patients were excluded. Finally, 62 patients (31 who underwent OG and 31 who underwent LG) were selected by this method.
Statistical analyses
The χ2 test or Fisher’s exact test was applied to evaluate any dichotomous variables and the Wilcoxon rank–sum test was applied for continuous variables. Survival rates were estimated by the Kaplan–Meier method. P values <0.05 were considered significant. We used JMP® software (v. 13; SAS Institute Inc., Cary, NC, USA).
Results
A total of 1772 patients with GC who underwent gastrectomy in OMCH between 2004 and 2015 were analyzed. Figure 1 shows the study diagram. Using one-to-one PSM, 31 pairs of patients subjected to OG or LG were matched for final analysis. The C-statistic for the goodness-of-fit was 0.875 in the PSM model.
Short-term outcomes
Table 1 shows the patient’s background and clinical characteristics of unmatched patients (n = 159) and of the propensity-matched patients (n = 62). After PSM, the patient distributions were similar among the OG and LG groups. However, the distribution of surgery periods differed between the two groups even after PSM. The median operative time showed no significant difference between the two groups, while the estimated blood loss was very significantly less in the LG group than in the OG group (P < 0.0001). Table 2 lists the postoperative courses of both groups. The median time to first flatus was significantly shorter in the LG group than in the OG group (P = 0.002). The postoperative hospital stay was significantly shorter in the LG group (P = 0.014). The complication rate of C–D grade III or higher was significantly lower in the LG group than in the OG group (P = 0.023). One patient in the OG group died on day 3 because of nonocclusive mesenteric ischemia and one unmatched patient in the LG group died on day 8 because of acute heart failure. Table 3 represents the detail of the postoperative complications in both groups. There were no significant differences in each type of complication between the groups.
Long-term outcomes
The survival data were investigated until December 2020, with a median follow-up of 57 months (interquartile range, 19–76 months). Tumors recurred in 24 of the 159 unmatched patients (15.1%), with peritoneal and lymph node metastases being the most common. Among them, no patients—except for one—received adjuvant chemotherapy, which is regarded as the gold standard treatment in patients <80 years with stage II and III [22, 23]. Chemotherapy was administered for only four patients to control tumor recurrence. Of the 159 elderly patients, 99 (62.3%) died during the follow-up period, including 72 (45.3%) who died of other diseases (Fig. 2), 23 (14.5%) who died of GC, and four (2.5%) who died of postoperative complications. As shown in Fig. 2, the cumulative incidence of death due to causes other than GC was significantly higher in the OG group than in the LG group.
In the unmatched analysis, the 3-year OS was 46.3% in the OG group and 70.3% in the LG group, and the 5-year OS was 38.9% and 57.9%, respectively (Fig. 3a). In the propensity-matched analysis, the 3-year and 5-year OS did not differ between the two groups (Fig. 3b). Regarding DSS, the 3-year DSS rates were 58.9% in the OG group and 88.0% in the LG group; the 5-year DSS rates were 58.9% and 84.0%, respectively (Fig. 4a; P = 0.0002). In the matched analysis, the Kaplan–Meier curves showed no significant difference in DSS between the two groups (Fig. 4b).
Comparison of overall survival rates calculated using the Kaplan–Meier method between the OG and LG groups in the unmatched analysis (a) and the matched analysis (b). In the unmatched analysis, the overall survival rate in the LG group was significantly better than that in the OG group (P = 0.014 by log-rank test). However, in the matched analysis, the overall survival rates showed no significant difference between the two groups
Comparison of disease-specific survival rates between the OG and LG groups calculated using the Kaplan–Meier method in the unmatched (a) and matched (b) analyses. In the unmatched analysis, the disease-specific survival rate in the LG group was significantly higher than in the OG group (P = 0.0002 by log-rank test). However, in the matched analysis, the overall survival rates showed no significant difference between the two groups
Discussion
Despite being a globally adopted procedure, the efficacy of LG for elderly patients remains unclear. Guidelines on the appropriate use of laparoscopic procedures to be performed in this ever-expanding age group are lacking and need to be provided. Here, we verified that short-term outcomes were markedly better with LG than with OG. The postoperative complication rates were also significantly lower with LG, while the long-term survival outcomes were almost similar between the two groups. To our knowledge, this is the first PSM study to demonstrate better short- and long-term outcomes with LG than with OG in elderly patients with GC aged ≥80 years. These results help the widespread use of LG for GC in elderly patients.
Studies of GC surgery in such patients have been used different age criteria: e.g., the commonly defined ages for “elderly” have been ≥70 years [24,25,26], ≥75 years [27,28,29,30], and ≥80 years [31,32,33]. In general, older people frequently have age-related physiological difficulties such as decreased organ function, various comorbidities, and mental imbalance. Surgeons are sometimes reluctant to perform operations on such patients because of the high incidence of age-associated morbidities and mortalities. In our institution, the proportion of patients with GC aged >80 years has also increased to over 15% during the past 5 years. Many previous RCTs of LG have targeted patients ≥80 years of age [11, 13, 34,35,36]. As a result, there are currently insufficient data on LG for the elderly. Therefore, we decided that the subjects of this study would be limited to patients aged ≥80 years.
In unmatched comparative analysis, LG was superior to OG in both short- and long-term outcomes. Moreover, we proved that LG was superior to OG in short-term outcomes even after PSM to eliminate bias due to patient characteristics and oncological factors. Here, we confirmed the benefits of LG for elderly patients in terms of intraoperative blood loss, postoperative recovery of bowel movement, hospital stay, and morbidity compared with those of OG. To ensure low complication rates in these patients, surgeons must strike the right balance between operative morbidity and oncological survival. According to a previous report on colorectal cancer surgery, low blood loss could reduce the surgical stress and further remarkably decrease the postoperative complications. For this reason, the reduction in blood loss could credibly enhance the postoperative recovery for patients [37]. Moreover, because elderly patients usually have a limited functional capacity, any acute massive blood loss can lead to inadequate perfusion of vulnerable organs, resulting in fatal organ damage. In RCTs on nonelderly patients with GC, LG was found to produce a rapid recovery of bowel movements, fewer postoperative complications, and shorter hospital stays than OG [11, 14, 36]. However, to date, no RCTs on focusing on the feasibility and efficacy of LG in the elderly have been conducted. Laparoscopic surgery with minimal destruction of the body wall might be a beneficial approach for the elderly because such people with reduced organ function are more susceptible to surgical invasion. From this point of view, we have developed our technique over the years to adopt a totally intracorporeal approach and previously reported our techniques for intracorporeal reconstruction following LG [10, 38,39,40]. In addition, we have used careful perioperative management to enhance postoperative recovery, such as the enhanced recovery after surgery protocol [41]. We believe that elderly patients are most likely to benefit from such innovative multidisciplinary treatments.
In terms of long-term outcomes, our study using PSM showed that the OS and DSS of patients treated with LG were equivalent to those treated with OG. In general, elderly patients show poorer OS and DSS rates, for every stage of the disease. Katai et al. reported that the 5-year OS of patients aged ≥80 years with stage IA, IB, II, IIIA, IIIB, and IV GC were 72.8%, 63.3%, 50.0%, 33.0%, 18.7%, and 7.8%, respectively, based on the nationwide registry of the Japanese Gastric Cancer Association [4]. Despite differences in patient background and the edition of classification guidelines used, our long-term outcome for the LG group was roughly equivalent to these findings. Elderly patients have a short life expectancy and a high rate of death from other diseases [42]. In fact, our data show that about half of the patients died from other diseases. Of the 159 patients, 19 from the OG group and 53 from the LG group died from other causes during the investigation. Three patients (15.8%) from the OG group died from pneumonia, where the average onset time after surgery was 15.7 months. Meanwhile, 9 patients (17.0%) from the LG group died from pneumonia, with the average onset time of 47.2 months after surgery. We interpreted that because LG had few postoperative severe complications in elderly patients, i.e., it is a minimally invasive technique, it may be less likely to result in fatal pneumonia early after surgery. Therefore, we should first focus on how to overcome the perioperative period. Totally laparoscopic approaches could be associated with maintaining postoperative quality of life and reducing the risk of death from other diseases such as pneumonia in the early period after surgery. In addition, better short-term outcomes for patients with stage II or III GC could improve compliance with adjuvant chemotherapy.
Our study had several limitations. First and most important, this was not an RCT and the inherent selection biases could be adjusted but not completely eliminated by using PSM. We believe that a multi-institutional setting would be better for obtaining more persuasive conclusions. Second, this was a small sample size study conducted at a single center in Asia. Therefore, our results might not be applicable directly to Western populations. However, in spite of this limitation, considering the difficulties of applying RCTs to elderly cohorts, our study using PSM was inexpensive and valuable. We believe strongly that our research could serve as helpful information for future RCTs investigating LG in elderly patients with GC. Third, this study was conducted over a rather long period between 2004 and 2015, which could have been associated with historical biases in terms of treatment strategy and perioperative management, which might dictate the short-term and prognostic outcomes after gastrectomy.
Conclusions
In this cohort of elderly patients, LG was safe and showed advantages such as a lower complication rate and faster recovery than OG. Our results suggest that widening our indications for laparoscopic surgery to include patients aged ≥80 years was not associated with any adverse surgical or oncological outcomes. LG has the potential to provide a balance between oncological clearance and quality of life issues.
References
Bray F, Ferlay J, Soerjomataram I et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424
Ministry Health, Labor and Welfare (2018) Vital Statistics in Japan—The latest trend [in Japanese]. https://ganjoho.jp/reg_stat/statistics/dl/index.html. Accessed 10 June 2018
Nashimoto A, Akazawa K, Isobe Y et al (2013) Gastric cancer treated in 2002 in Japan: 2009 annual report of the JGCA nationwide registry. Gastric Cancer 16:1–27
Katai H, Ishikawa T, Akazawa K et al (2018) Five-year survival analysis of surgically resected gastric cancer cases in Japan: a retrospective analysis of more than 100,000 patients from the nationwide registry of the Japanese Gastric Cancer Association (2001–2007). Gastric Cancer 21:144–154
Kenig J, Mastalerz K, Mitus J et al (2018) The surgical apgar score combined with comprehensive geriatric assessment improves short- but not long-term outcome prediction in older patients undergoing abdominal cancer surgery. J Geriatr Oncol 9:642–648
Endo S, Dousei T, Yoshikawa Y et al (2013) Prognosis of gastric carcinoma patients aged 85 years or older who underwent surgery or who received best supportive care only. Int J Clin Oncol 18:1014–1019
Lieffers JR, Bathe OF, Fassbender K et al (2012) Sarcopenia is associated with postoperative infection and delayed recovery from colorectal cancer resection surgery. Br J Cancer 107:931–936
Shen Y, Hao Q, Zhou J et al (2017) The impact of frailty and sarcopenia on postoperative outcomes in older patients undergoing gastrectomy surgery: a systematic review and meta-analysis. BMC Geriatr 17:188. https://doi.org/10.1186/s12877-017-0569-2
Tanaka T, Suda K, Inaba K et al (2019) Impact of frailty on postoperative outcomes for laparoscopic gastrectomy in patients older than 80 years. Ann Surg Oncol 26:4016–4026
Lee SW, Nomura E, Bouras G et al (2010) Long-term oncologic outcomes from laparoscopic gastrectomy for gastric cancer: a single-center experience of 601 consecutive resections. J Am Coll Surg 211:33–40
Katai H, Mizusawa J, Katayama H et al (2017) Short-term surgical outcomes from a phase III study of laparoscopy-assisted versus open distal gastrectomy with nodal dissection for clinical stage IA/IB gastric cancer: Japan clinical oncology group study JCOG0912. Gastric Cancer 20:699–708
Hu Y, Huang C, Sun Y et al (2016) Morbidity and mortality of laparoscopic versus open D2 distal gastrectomy for advanced gastric cancer: a randomized controlled trial. J Clin Oncol 34:1350–1357
Inaki N, Etoh T, Ohyama T et al (2015) A multi-institutional, prospective, phase II feasibility study of laparoscopy-assisted distal gastrectomy with D2 lymph node dissection for locally advanced gastric cancer (JLSSG0901). World J Surg 39:2734–2741. https://doi.org/10.1007/s00268-015-3160-z
Kim W, Kim HH, Han SU et al (2016) Decreased morbidity of laparoscopic distal gastrectomy compared with open distal gastrectomy for stage I gastric cancer: short-term outcomes from a multicenter randomized controlled trial (KLASS-01). Ann Surg 263:28–35
Haverkamp L, Brenkman HJ, Seesing MF et al (2015) Laparoscopic versus open gastrectomy for gastric cancer, a multicenter prospectively randomized controlled trial (LOGICA-trial). BMC Cancer 15:556. https://doi.org/10.1186/s12885-015-1551-z
Etoh T, Honda M, Kumamaru H et al (2018) Morbidity and mortality from a propensity score-matched, prospective cohort study of laparoscopic versus open total gastrectomy for gastric cancer: data from a nationwide web-based database. Surg Endosc 32:2766–2773
Yoshida K, Honda M, Kumamaru H et al (2018) Surgical outcomes of laparoscopic distal gastrectomy compared to open distal gastrectomy: a retrospective cohort study based on a nationwide registry database in Japan. Ann Gastroenterol Surg 2:55–64
Japanese Gastric Cancer Association (2011) Japanese gastric cancer treatment guidelines 2010 (ver. 3). Gastric Cancer 14:113–123
Onodera T, Goseki N, Kosaki G (1984) Prognostic nutritional index in gastrointestinal surgery of malnourished cancer patients. Nihon Geka Gakkai Zasshi 85:1001–1005
Edge SB, Compton CC (2010) The American Joint Committee on Cancer the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol 17:1471–1474
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
Sakuramoto S, Sasako M, Yamaguchi T et al (2007) Adjuvant chemotherapy for gastric cancer with S-1, an oral fluoropyrimidine. New Engl J Med 357:1810–1820
Noh SH, Park SR, Yang HK et al (2014) Adjuvant capecitabine plus oxaliplatin for gastric cancer after D2 gastrectomy (CLASSIC): 5-year follow-up of an open-label, randomised phase 3 trial. Lancet Oncol 15:1389–1396
Cho GS, Kim W, Kim HH et al (2009) Multicentre study of the safety of laparoscopic subtotal gastrectomy for gastric cancer in the elderly. Br J Surg 96:1437–1442
Hwang SH, Park DJ, Jee YS et al (2009) Risk factors for operative complications in elderly patients during laparoscopy-assisted gastrectomy. J Am Coll Surg 208:186–192
Kumagai K, Hiki N, Nunobe S et al (2014) Potentially fatal complications for elderly patients after laparoscopy-assisted distal gastrectomy. Gastric Cancer 17:548–555
Honda M, Kumamaru H, Etoh T et al (2019) Surgical risk and benefits of laparoscopic surgery for elderly patients with gastric cancer: a multicenter prospective cohort study. Gastric Cancer 22:845–852
Kim KH, Kim MC, Jung GJ (2014) Is the rate of postoperative complications following laparoscopy-assisted gastrectomy higher in elderly patients than in younger patients? World J Surg Oncol 12:97. https://doi.org/10.1186/1477-7819-12-97
Yamamoto M, Shimokawa M, Kawano H et al (2019) Benefits of laparoscopic surgery compared to open standard surgery for gastric carcinoma in elderly patients: propensity score-matching analysis. Surg Endosc 33:510–519
Shimada S, Sawada N, Oae S et al (2018) Safety and curability of laparoscopic gastrectomy in elderly patients with gastric cancer. Surg Endosc 32:4277–4283
Takeshita H, Ichikawa D, Komatsu S et al (2013) Surgical outcomes of gastrectomy for elderly patients with gastric cancer. World J Surg 37:2891–2898. https://doi.org/10.1007/s00268-013-2210-7
Yoshida M, Koga S, Ishimaru K et al (2017) Laparoscopy-assisted distal gastrectomy is feasible also for elderly patients aged 80 years and over: effectiveness and long-term prognosis. Surg Endosc 31:4431–4437
Kwon IG, Cho I, Guner A et al (2015) Minimally invasive surgery as a treatment option for gastric cancer in the elderly: comparison with open surgery for patients 80 years and older. Surg Endosc 29:2321–2330
Katai H, Mizusawa J, Katayama H et al (2019) Single-arm confirmatory trial of laparoscopy-assisted total or proximal gastrectomy with nodal dissection for clinical stage I gastric cancer: Japan clinical oncology group study JCOG1401. Gastric Cancer 22:999–1008
Kim HH, Han SU, Kim MC et al (2019) Effect of laparoscopic distal gastrectomy vs open distal gastrectomy on long-term survival among patients with stage I gastric cancer: the KLASS-01 randomized clinical trial. JAMA Oncol 5:506–513
Lee HJ, Hyung WJ, Yang HK et al (2019) Short-term outcomes of a multicenter randomized controlled trial comparing laparoscopic distal gastrectomy with D2 lymphadenectomy to open distal gastrectomy for locally advanced gastric cancer (KLASS-02-RCT). Ann Surg 270:983–991
Zhou S, Wang X, Zhao C et al (2019) Laparoscopic vs open colorectal cancer surgery in elderly patients: short- and long-term outcomes and predictors for overall and disease-free survival. BMC Surg 19:137
Lee SW, Tanigawa N, Nomura E et al (2012) Benefits of intracorporeal gastrointestinal anastomosis following laparoscopic distal gastrectomy. World J Surg Oncol 10:267. https://doi.org/10.1186/1477-7819-10-267
Lee SW, Kawai M, Tashiro K et al (2016) Laparoscopic gastrointestinal anastomoses using knotless barbed absorbable sutures are safe and reproducible: a single-center experience with 242 patients. Jpn J Clin Oncol 46:329–335
Lee SW, Kawai M, Tashiro K et al (2019) The crossover technique for intracorporeal esophagojejunostomy following laparoscopic total gastrectomy: a simple and safe technique using a linear stapler and two barbed sutures. Surg Endosc 33:1386–1393
Tanaka R, Lee SW, Kawai M et al (2017) Protocol for enhanced recovery after surgery improves short-term outcomes for patients with gastric cancer: a randomized clinical trial. Gastric Cancer 20:861–871
Nunobe S, Oda I, Ishikawa T et al (2020) Surgical outcomes of elderly patients with stage I gastric cancer from the nationwide registry of the Japanese gastric cancer association. Gastric Cancer 23:328–338
Acknowledgements
The authors had no financial support for the research reported.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical Approval and Consent to Participate
This study was approved by the Institutional Review Board of Osaka Medical College Hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tanaka, R., Lee, SW., Imai, Y. et al. Advantages of Laparoscopic Surgery for Gastric Cancer in Elderly Patients Aged Over 80 Years: A Propensity Score Matching Analysis. World J Surg 45, 2830–2839 (2021). https://doi.org/10.1007/s00268-021-06157-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-021-06157-6







