Abstract
Design
This trial is a randomized controlled, patient-blinded, multicentre, superiority trial.
Methods
All patients ≥18 years with a single, symptomatic and primary umbilical or epigastric hernia (<2 fingers) qualified for participation in the study. Flat polypropylene mesh repair was compared to patch repair (PROCEED® Ventral Patch) (PVP). The objective of this trial was to identify a superior method for umbilical and epigastric hernia repair in terms of complication rates.
Results
A total of 352 patients were randomized in this trial; 348 patients received the intervention (n = 177 PVP vs. n = 171 mesh). No peri-operative complications occurred. PVP placement was significantly faster compared to mesh placement (30 min, SD 11 vs. 35 min, SD 11) and was scored as an easier procedure. At 1-month follow-up, 76 patients suffered any kind of complication. There was no significant difference in the proportion of complications (24.9% for PVP and 18.7% for mesh, p = 0.195). A significant difference was seen in re-operation rate within 1 month, significantly less early re-operations in the mesh group (0.0 vs. 2.8%, p = 0.027). After 1-year follow-up, no significant differences are seen in recurrence rates (n = 13, 7.8% PVP vs. n = 5, 3.3% mesh, p = 0.08).
Conclusions
Both mesh and PVP had a comparable amount of reported complications. There was a significantly higher incidence of early re-operations due to early complications in the PVP group. No differences were seen in infection rates and the need for antibiotic treatment. No significant difference was seen in the recurrence rates.
Registration
This trial was registered in the Dutch Trail Registry (NTR) NTR2514NL33995.060.10. [12].
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In hernia surgery and research, for years the primary goal was reducing recurrence rates. Due to the performed research concerning this matter, mesh-based repair became the gold standard in the vast majority of hernia repair. Evidence is accumulating that this also applies to the repair of small primary umbilical and epigastric hernias [1,2,3,4,5,6]. However, primary closure is still performed in a lot of these small hernias. It should be known that mesh repair even for small umbilical and epigastric hernias reduces the recurrence rate from 6 to 2% without increasing the complication rate [2]. The next question to answer is where the mesh should be placed. Intraperitoneal placement is one of the options and can be done by means of a laparoscopic approach. However, in laparoscopic mesh placement, several new fascial defects are created in the abdominal wall due to the use of several trocars. This potentially leads to the development of incisional hernias. This awareness has played an important role in the search for other techniques and possibly attributed to the development of the mesh patches. These mesh patches can be placed intraperitoneal using an open procedure and with a single incision. In theory, open intraperitoneal placement could have advantages in terms of lesser dissection, shorter operation time and maybe even in decreasing costs. This is a comprehensive hypothesis; in the Netherlands, only the treatment of an umbilical or epigastric hernia involves over 6200 patients and costs almost 12.5 million euro per year [7].
In line with other hernia research, the research focusing on epigastric and umbilical hernias is shifting from recurrence as primary outcome, to complications, pain, costs and quality of life (QoL) [8,9,10].
This randomized controlled trial was conducted to identify a superior mesh device in open placement. The primary outcome measure of this trial is the number of complications within 1 year. Secondary endpoints for this report are Verbal Descriptor Scale (VDS) pain score and VDS cosmetic score, operation duration and recurrence.
Methods
Design
The MORPHEUS (Mesh OR Patch for Hernia on Epigastric and Umbilical Sites) trial was designed as a prospective randomized controlled, patient-blinded, regional, multicentre superiority trial with two parallel groups. Primary outcome was the number of complications within 1 year post-operatively. Randomization was performed as block randomization with a 1:1 allocation. During this trial, no changes to the trial design were made. The institutional review board of the participating hospitals approved the trial protocol. The protocol was accepted and published in an open access, peer reviewed journal [11]. This trial was registered in the Dutch Trail Registry (NTR) NTR2514 NL33995.060.10 [12]. For the designing and reporting on this trial, the CONSORT 2010 guidelines and updated guidelines are used [13, 14].
Participants
Adult patients with a single, primary and symptomatic umbilical or epigastric hernia qualified for participation in the study. Patients with incarcerated hernia were also included. Small hernias are frequently considered smaller than 3 cm of width. For the convenience of not needing a sterile ruler, and in accordance with the European Hernia Society (EHS) classification for inguinal hernias also using fingers, size was graded into 1 or 2 fingers width [15]. Therefore, the maximum of the included hernia size was defined as 2 fingers width.
Patients with ascites, patients under 18 years old and those who could not sufficiently understand and/or follow through participation in a trial were excluded.
All included patients gave written informed consent. If a recruited patient did not participate in the trial, the reason was stated [11].
Study setting
Participating patients were recruited from five secondary care hospitals in the southern region of the Netherlands. All hospitals are teaching hospitals varying in size, from 297 to 700 hospital beds. In every participating hospital, a dedicated abdominal wall surgeon guided and supervised the trial process.
Interventions
All procedures took place under general anaesthesia. Administering a local anaesthetic peri-operative was recommended. Prophylactic antibiotics were given only on the surgeon’s opinion, but were not mandatory in this trial. The usage of Steri-Drape™, as well as drains, was not advised. Enlarging the herniation orifice for adequate mesh placement, as well as closing fascia over the mesh, was permitted for both techniques, but had to be noted.
The conventional mesh procedure started with a para-umbilical or median incision across the herniation, followed by dissection of the fascia and mobilization of the hernia sac. Opening of the hernia sac for inspection was permitted. Dissection of the pre-peritoneal area took place after repositioning of the hernia. A flat large pore and lightweight polypropylene mesh with a minimum diameter of 6 cm were placed pre-peritoneal, to ensure 3 cm overlap. Fixation of the mesh was carried out with non-absorbable monofilament sutures.
For the patch or PVP procedure, the hernia sac was opened. Any intraperitoneal adhesions had to be released. The patch was placed underneath the peritoneum, the slips fixed to the fascia. If, in the surgeon’s opinion, the hernia sac could be repositioned without opening it, placement was also permitted in the pre-peritoneal plane. The PROCEED™ Ventral Patch (PVP, Ethicon, Norderstedt, Germany) 6.4 cm was used in all cases in the PVP intervention group, to ensure enough overlap. For the mesh group, the hospital’s standard flat large pore and lightweight polypropylene mesh was chosen for the hernia repair in the mesh-randomized patients [11].
Outcomes
The primary outcome of this trial was the complication rate within 1 year after initial surgery. Complications were described as follows:
-
Extending operation time due to bleeding or other injury during the procedure.
-
Prescribed medication treatment such as antibiotics and painkillers other than paracetamol after discharge.
-
Re-operation due to evacuation of a haematoma, drainage of an abscess, exploration related to pain or intra-abdominal problems, or early recurrence.
-
Wound infection or seroma.
-
Hospitalization longer than scheduled or re-admission.
The secondary endpoints were Verbal Descriptor Scale (VDS) pain scores and VDS cosmetic score 1 and 3 months and 1 year after the operation, operation duration, recurrence after 1 year and costs. No changes were made regarding the outcomes since the trial commenced.
Sample size
The primary outcome was the number of complications within 1 year post-operatively. An occurrence of complications up to 23% is described in mesh-operated patients [16]. Trials involving patch repair report a much lower complication rate, but the number of trials is significantly less. It is assumed that a conventional procedure leads to a complication in 20% of the cases [16, 17]. The hypothesis was that this would reduce to around 9% if a patch is used for repair. At a significance level of 5% and a power of 80%, the random sample size is 157 patients per group in a proportions 2-sample, 2-sided equality test [18]. Taking a minimum of 10% loss-to-follow-up into account as well, the total number of patients to be randomized was 346.
Randomization
Sequence allocation and randomization were performed by computer generation. Twelve random sequences of 1’s and 2’s were generated in blocks of forty and printed on paper cards. Block randomization was used to limit the chance of an imbalance between treatment groups between participating hospitals and decrease the probability of confounding factors due to relatively small number of participants per hospital.
Number 1 corresponded to PVP placement and number 2 to mesh placement. These cards were put in non-see through envelopes by the principal investigators. The envelopes were sequentially numbered to assure the random allocation of the random sequence.
The operating surgeon opened the envelopes just before the time-out procedure, without informing the patient about the outcome of the randomization.
Blinding
This trial was a patient-blinded randomized controlled type. The patient was not informed on the type of mesh device used. Only if desired by the patient, he or she could be informed after the 2-year follow-up. In the data-handling phase of the trial, the analyst was blinded to treatment. The consulted physician during follow-up was not blind for the received treatment.
Statistical method
Data were expressed as mean ± SD (standard deviation) versus median with range. A p value <0.05 was regarded as significant. SPSS statistics 21 (IBM) was used for processing the data. Non-adherent data were not anticipated for this trial. Comparison between the two interventions can be subdivided into three periods: pre-operative, peri-operative and post-operative comparison.
Pre-operative parameters included body mass index (BMI), VDS pain score at rest, pain score while exercising and VDS cosmetic score, comorbidity and daily workload. Differences in baseline characteristics were measured and presented in a baseline characteristics table. Peri-operative findings that were recorded are the presence of incarceration, resection of the hernia protrusion, number of fingers for the width of the herniation orifice, the need of widening the herniation orifice, closure of fascia over mesh, presence of adhesions when placing the device intraperitoneal, operation duration, ease of the procedure, possible reasons protocol violation. Post-operative measurements included the occurrence of complications, pain score at rest and pain score while exercising (VDS), the use of analgesics at that time, cosmetic (VDS) and signs of recurrence (1, 3, 12, 24 months post-operative). Subgroup analysis were performed to further investigate the patients with a complicated postoperative course, including patients with a recurrence. The t test was used for continuous data and the Pearson’s Chi-square/Fisher’s exact test for categorical data.
Results
Flow chart
The trial participant flow chart is shown in Fig. 1.
Recruitment
Patients were recruited for inclusion from 1 February 2011 until 1 July 2015. All patients with a signed informed consent form, in possession of the research group, were included in this trial. After reaching the acquired number of participant’s, enrolment stopped. From the point of inclusion and operation, all patients were followed up for a 2-year period.
Baseline characteristics
No significant differences between groups were seen in gender, BMI, hernia type, incarceration, level of exercise, pre-operative pain at rest and during exercise, appearance, diabetes mellitus, skin disease and pain syndrome diagnosed by a medical doctor. A significant difference was seen in the age between both groups. Detailed baseline characteristics are shown in Table 1.
Numbers analysed
The primary analysis, the pre-, peri- and 1-month post-operative results, involved all 177 and 171 patients who were randomly assigned to both groups. At that time, there were no lost-to-follow-up patients. Four patients, who were initially recruited for the trial, received primary closure of the hernia. This was due to the operating surgeon’s opinion that the hernia was too small to fit in a mesh, or PVP. This was against the trial protocol, and after renewed trial explanation and stressing the importance of including very small hernias, this did not happen again. The four patients who underwent primary closure were excluded from the trial results.
After 3-month follow-up, 7 patients in the PVP group and 10 patients in the mesh group were lost to follow up; thus, data from 170 PVP operated patients and 161 mesh-operated patients were available for the 3 months analysis.
For the 1-year analysis, 12 patients from the PVP group were lost-to-follow-up and 17 from the mesh-operated group. For the 1-year post-operative analysis, 154 from the mesh group are used and 164 patients from the PVP operated group. This makes a lost-to-follow-up rate of 8.3% after 1 year. There was no significant difference in incidence of lost-to-follow-up patients between the groups (p = 0.286).
Outcomes and estimation
Primary outcome
All complications were reported according to the Clavien–Dindo grading system for surgical complications [19, 20]. After 1 year of follow-up, no grade IV or V complications were seen.
There were no peri-operative complications in either group. Within 1-month after the operation, 76 patients suffered a complication, ranging from Clavien–Dindo grade I–IIIb. In the PVP group, 24.9% (n = 44) of the patients suffered from at least one complication, and in the mesh-operated group 18.7% (n = 32) of the patients. No significant difference was seen between groups (p = 0.195). The majority of these complications were wound related, representing superficial surgical site infection (SSI) or a seroma. For the diagnosis of a SSI, the definition of the Centers for Disease Control and Prevention was used [21]. Wound infections requiring antibiotic treatment, Clavien–Dindo II, did not differ significantly between both groups (p = 0.707). There was a significant difference in re-operation rate within 1 month between groups (n = 5 in the PVP group, 3%, and n = 0 in the mesh group, 0%, p = 0.027). Of the 5 PVP operated patients, Clavien–Dindo IIIa–b, one patient was initially operated within 12 h, after reducing an incarcerated hernia. Five days after the operation, this patient suffered from a persistent small bowel obstruction. Eventually, the patient was re-operated, by means of a laparotomy, and a poorly perfused small gut segment was removed. This was seen as a non-device-related complication. One of these 5 patients had a persistent infection without a satisfying effect of intravenous antibiotic treatment. Three weeks after placement, the PVP device was removed. One other patient was re-operated within several days after the initial operation due to a very extensive haematoma, which was evacuated under general anaesthesia. One patient suffered from extensive pain after the operation, exploration under local anaesthesia, within 3 weeks showed no significant problems. The last of these 5 early re-operated patients showed an early recurrence due to device malfunction of insufficient placement; a laparoscopic mesh repair was performed. Three months post-operatively, no significant differences were seen between groups in terms of complications. After 1-year follow-up, no significant differences were seen in recurrence rates and re-operation rates. Thirteen patients in the PVP group showed a recurrence (7.8%) at the 1-year follow-up clinical examination, and 5 patients in the mesh group (3.3%), p = 0.078. The eight re-operated PVP patients include the 5 re-operated patients at 1-month follow-up. The three remaining PVP re-operated patients at 1-year follow-up were operated due to symptomatic recurrence. In the five re-operated patients in the mesh group, three were operated due to persisting pain without clinical and radiological signs of a recurrence. During re-operation (Clavien–Dindo grade III), in two cases performed with local anaesthetics (Clavien–Dindo grade IIIa), rather long stitches were seen very close to the skin. The two remaining mesh-operated patients were operated due to a symptomatic recurrence (grade IIIb). Results are shown in Tables 2, 3 and 4.
Secondary outcome
Several significant differences between groups are seen in the peri-operative analysis shown in Table 2. The hernia sac was resected significantly (p = 0.01) more in case of PVP placement. This outcome is expected due to the intraperitoneal placement possibility of the PVP and the need of the pre-peritoneal mesh placement.
The PVP placement procedure was significantly (p ≤ 0.01) faster, by 5 min on average, compared to mesh placement (30 min, SD 11 vs. 35 min, SD 11). This procedure was also scored as significantly (p ≤ 0.01) easier compared to mesh placement.
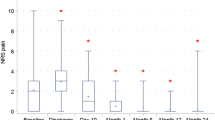
No statistically significant differences were seen in the use of painkillers, VDS pain scores at rest, VDS pain scores during excise, and appearance/VDS cosmetic score at 1 month, 3 months and 1 year post-operatively.
Ancillary analyses
A subgroup analysis was performed to further assess the majority of complications, the wound infection/seroma group, concerning 68 patients. The patients suffering from wound complications have a significantly higher BMI, p = 0.026 (BMI 28.9 SD 4.4 vs. 27.5 SD 4.5). The skin-to-skin time in the wound complication group was significantly longer compared to the non-wound complicated group, p = 0.014 (35.4 min SD 13 vs. 31.5 SD 11). The ease of the operation was not scored as more difficult by the operating surgeon in the wound complicated patient group, p = 0.340 (VDS ease 2.1 SD 0.7 vs. VDS ease 2.2 SD 0.7).
Patients suffering from a wound complication had a significantly higher pain score, at rest (p = 0.003, VDS pain 1.4 SD 0.7 vs. 1.2 SD 0.5) and during exercise (p = 0.017, VDS pain 1.7 SD 0.8 vs. 1.5 SD 0.7), 1 month after the surgical procedure. Three months (p = 0.822, VDS at rest 1.1 SD 0.7 vs. 1.1 SD 0.7 and p = 0.085, VDS pain during exercise 1.4 SD 0.5 vs. 1.2 SD 0.5) and 1 year (p = 0.265, VDS at rest 1.1 SD 0.2 vs. 1.1 SD 0.4 and p = 0.267, VDS during exercise 1.2 SD 0.4 vs. 1.2 SD 0.6) after the operation, the pain scores did not differ between the wound complicated and the non-wound complicated patient groups.
Patients who suffered from any kind of reported complication (Clavien–Dindo I–IIIb) at the 1-year follow-up (n = 59) had a borderline significant risk of the developing of recurrence; n = 7, 10.6% versus n = 11, 4.3%, p = 0.05.
Harms
No important harms or unintended effects occurred in either patient group. The deceased patients in both groups died from causes unrelated to the hernia operation.
Discussion
This is one of the largest trials investigating two mesh devices for small epigastric and umbilical hernia in a randomized setting. Therefore, this trial helps to evaluate mesh repair for small epigastric and umbilical hernia and find a surgical device or method that minimizes the complication rate. At this point, PVP device usage shows an easier and faster operating procedure. Nevertheless, this advantage is outweighed by the significantly higher incidence of early re-operations due to early complications (Clavien–Dindo III). Although it could be stated that some of these early re-operations seem not device related, and at least one of these complications is related to the emergency character of the disease; mesh repair seems superior.
No statistically significant differences were seen in wound infection rates and the need for additional antibiotic treatment. From the patient’s perspective, there are also no statistically significant differences between groups in post-operative evaluation of pain and appearance within the first follow-up year. Nevertheless, a rather high incidence of wound related complications is reported in this trial; 24.9 vs. 18.7% for the PVP and mesh groups are high wound complication rates; many other, although retrospective, studies describe lower rates after mesh repair [22, 23]. It could well be that, due to the prospective design and thorough follow-up, this trial reports a higher wound complication rate. It has to be stated that there is retrospective literature that supports this high wound complication rate [24] and even prospective research finding even higher rates [16, 25]. In this trial, no recommendations are given concerning the use prophylactic antibiotics. There is a possibility that due to the scares use of this prophylaxis a higher wound infection rate is reported. The use of prophylactic antibiotics is still a matter of debate in these small primary hernias and could be the subject of a randomized controlled trial itself. Another difficulty concerning this subject is that for wound infections and seromas, several definitions are used [26]. Moreover, the most frequently used definition for wound infections is multi-interpretable [21].
Because of a relatively short follow-up, 1-year after surgery, no important conclusions can be drawn concerning the secondary outcome measures like recurrence and costs. It is well known that recurrence can occur long after the first operative year [2]. This is one of the reasons that recurrence was not seen as a short-term complication and reported separately as secondary outcome. It could well be that during a longer period of follow-up all complications, short-term, long-term and undesirable outcome will be pooled to find a superior method. That is why longer follow-up of the included patients is necessary for reliable results concerning these secondary outcome parameters including costs.
This trial is a patient-blinded trial; the treating physician was not blinded to the given treatment. The observer could retrieve the operative report, when deciding to start a treatment for a complication. Although in practice did seem not to have influenced decisions and the analysis/data-handling was blinded for the researcher, there still is a bias as the study was not fully blinded.
A loss-to-follow-up rate of 8% after 1-year is undesirable although not uncommon in recent prospective hernia research, investigating patient reported outcome [27]. This loss-to-follow-up was estimated in the published protocol, and even a higher loss-to-follow-up rate was taken into account during calculating the sample size [11].
The conditions under which this trial is performed can be easily replicated in routine clinical practice due to extensive and thorough describing of methods and study design. Treatment of patients was performed in several medium to large size, secondary care and teaching hospitals. Experienced surgeons and residents in training performed the procedures and follow-up, which is very comparable with daily practice in hospitals in the Netherlands. Therefore, this trial consists of good external validity, and results are thereby applicable to the vast majority of patients suffering from primary epigastric and umbilical hernias in the western world.
Subgroup analysis showed that patients suffering from any kind of reported complication (Clavien–Dindo I–IIIb) have a borderline significant risk of the developing of recurrence within 1-year follow-up. It would be very interesting to see whether this difference is consistent after longer follow-up.
Subgroup analysis also confirmed known factors and predictors for wound complications occurrence. Patients in whom there was extensive operation duration and patients with a higher BMI are significantly more prone to wound complications. It is unknown whether the longer operation duration was due to the higher BMI or it was unrelated. Nevertheless, the procedure was not scored more difficult by the operating surgeon in patients with a higher BMI. As estimated, patients with wound complications scored significantly higher pain scores 1 month after the operation. After 3 months and 1 year, these pain scores did not differ between the wound complicated group and the not complicated group.
An often-heard excuse for primary closure of a small hernia is that it is unnatural to widen a small hernia orifice for sufficient mesh placement. In subgroup analysis, this trial shows that there is no increase in post-operative complications, pain or recurrences after widening the hernia orifice. This means that one of the last arguments for primary closure of a small hernia is refuted.
The planned and prolonged follow-up of 2 years is needed to give a better estimation of long-term complications and costs and see whether the recurrence rates are consistent.
References
Christoffersen MM (2015) Clinical outcomes after elective repair for small umbilical and epigastric hernias. Dan Med J 62(11):B5161
Christoffersen MW et al (2013) Lower reoperation rate for recurrence after mesh versus sutured elective repair in small umbilical and epigastric hernias. A nationwide register study. World J Surg 37(11):2548–2552. doi:10.1007/s00268-013-2160-0
Christoffersen MW et al (2015) Long-term recurrence and chronic pain after repair for small umbilical or epigastric hernias: a regional cohort study. Am J Surg 209(4):725–732
Westen M et al (2014) Chronic complaints after simple sutured repair for umbilical or epigastric hernias may be related to recurrence. Langenbecks Arch Surg 399(1):65–69
Ponten JE et al (2015) A consecutive series of 235 epigastric hernias. Hernia 19(5):821–825
Arroyo A et al (2001) Randomized clinical trial comparing suture and mesh repair of umbilical hernia in adults. Br J Surg 88(10):1321–1323
NZa. Open data van de Nederlandse Zorgautoriteit. http://www.opendisdata.nl/msz/zorgproduct/110401049
Nienhuijs S et al (2007) Chronic pain after mesh repair of inguinal hernia: a systematic review. Am J Surg 194(3):394–400
Colavita PD et al (2014) Umbilical hernia repair with mesh: identifying effectors of ideal outcomes. Am J Surg 208(3):342–349
Cox TC et al (2016) The cost of preventable comorbidities on wound complications in open ventral hernia repair. J Surg Res 206(1):214–222
Ponten JE et al (2014) Mesh Or Patch for Hernia on Epigastric and Umbilical Sites (MORPHEUS trial): study protocol for a multi-centre patient blinded randomized controlled trial. BMC Surg 14:33
NTR. Nederlands Trial Register. 2010. http://www.trialregister.nl/trialreg/admin/rctview.asp?TC=2514
Moher D et al (2010) CONSORT 2010 explanation and elaboration: updated guidelines for reporting parallel group randomised trials. BMJ 340:c869
Schulz KF et al (2011) CONSORT 2010 statement: updated guidelines for reporting parallel group randomised trials. Int J Surg 9(8):672–677
Simons MP et al (2009) European Hernia Society guidelines on the treatment of inguinal hernia in adult patients. Hernia 13(4):343–403
Abdel-Baki NA, Bessa SS, Abdel-Razek AH (2007) Comparison of prosthetic mesh repair and tissue repair in the emergency management of incarcerated para-umbilical hernia: a prospective randomized study. Hernia 11(2):163–167
Helgstrand F et al (2013) Outcomes after emergency versus elective ventral hernia repair: a prospective nationwide study. World J Surg 37(10):2273–2279. doi:10.1007/s00268-013-2123-5
Chow S, Shao J, Wang H (2008) Sample size calculations in clinical research, 2nd edn. CRC Press, Boca Raton
Clavien PA et al (2009) The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg 250(2):187–196
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Horan TC, Andrus M, Dudeck MA (2008) CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control 36(5):309–332
Nguyen MT et al (2014) Comparison of outcomes of synthetic mesh vs suture repair of elective primary ventral herniorrhaphy: a systematic review and meta-analysis. JAMA Surg 149(5):415–421
Winsnes A et al (2016) Surgical outcome of mesh and suture repair in primary umbilical hernia: postoperative complications and recurrence. Hernia 20(4):509–516
Berger RL et al (2013) Development and validation of a risk-stratification score for surgical site occurrence and surgical site infection after open ventral hernia repair. J Am Coll Surg 217(6):974–982
Abramov D et al (1996) Antibiotic prophylaxis in umbilical and incisional hernia repair: a prospective randomised study. Eur J Surg 162(12):945–948 discussion 949
Aslani N, Brown CJ (2010) Does mesh offer an advantage over tissue in the open repair of umbilical hernias? A systematic review and meta-analysis. Hernia 14(5):455–462
Muysoms FE et al (2016) A prospective, multicenter, observational study on quality of life after laparoscopic inguinal hernia repair with ProGrip laparoscopic, self-fixating mesh according to the European Registry for Abdominal Wall Hernias Quality of Life Instrument. Surgery 160(5):1344–1357
Acknowledgements
The authors would like to thank T.A. Mulders, MD and Ph.D., for his help and statistical knowledge, but above all, his friendship. Special thanks to Jax Café, New York City, USA, where the first lines of this manuscript were written, and Hotel Armentarola, Alto Adige, Italy, where this manuscript was finished. Both places are ideal writing environments.
Funding
The MORPHEUS trial started as a non-funded trial, because of slow inclusion rate in participating centres due to high device costs; an investigator initiated grant proposal was deposited. An Investigator Sponsored-Studies (ISS) proposal, which is investigator, initiated applied for and granted by the Johnson & Johnson Company. This funding was used to create levelling in costs for participating hospitals between the two mesh devices used. The Johnson & Johnson Company was not involved in the analysis of the data and the drafting of the manuscript.
Protocol
The protocol was approved and published in an open access, peer reviewed journal [11].
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ponten, J.E.H., Leenders, B.J.M., Leclercq, W.K.G. et al. Mesh Versus Patch Repair for Epigastric and Umbilical Hernia (MORPHEUS Trial); One-Year Results of a Randomized Controlled Trial. World J Surg 42, 1312–1320 (2018). https://doi.org/10.1007/s00268-017-4297-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-017-4297-8