Abstract
Background
Open abdomen treatment (OAT) is a significant burden for patients and is associated with considerable mortality. The primary aim of this study was to report survival and cause of mortality after OAT. Secondary aims were to evaluate length of stay (LOS) in intensive care unit (ICU) and in hospital, time to abdominal closure and major complications.
Methods
Retrospective review of prospectively registered patients undergoing OAT between October 2006 and June 2014 at Trondheim University Hospital, Norway.
Results
The 118 patients with OAT had a median age of 63 (20–88) years. OAT indications were abdominal compartment syndrome (ACS) (n = 53), prophylactic (n = 29), abdominal contamination/second look laparotomy (n = 22), necrotizing fasciitis (n = 7), hemorrhage packing (n = 4) and full-thickness wound dehiscence (n = 3). Eight percent were trauma patients. Vacuum-assisted wound closure (VAWC) with mesh-mediated traction (VAWCM) was used in 92 (78 %) patients, the remaining 26 (22 %) had VAWC only. Per-protocol primary fascial closure rate was 84 %. Median time to abdominal closure was 12 days (1–143). LOS in the ICU was 15 (1–89), and in hospital 29 (1–246) days. Eighty-one (68 %) patients survived the hospital stay. Renal failure requiring renal replacement therapy (RRT) (OR 3.9, 95 % CI 1.37–11.11), ACS (OR 3.1, 95 % CI 1.19–8.29) and advanced age (OR 1.045, 95 % CI 1.004–1.088) were independent predictors of mortality in multivariate analysis. The nine patients with an entero-atmospheric fistula (EAF) survived.
Conclusion
Two-thirds of the patients treated with OAT survived. Renal failure with RRT, ACS and advanced age were predictors of mortality, whereas EAF was not associated with increased mortality.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
In the updated consensus definitions from the World Society of the Abdominal Compartment Syndrome (WSACS), abdominal compartment syndrome (ACS) is defined as sustained intra-abdominal pressure (IAP) ≥20 mmHg combined with a new organ dysfunction or failure [1]. The elevated IAP results in reduced intra-abdominal blood flow, venous return and cardiac output resulting in organ failure [1]. The incidence of ACS has been reported to be 1 % in trauma patients [2], 1–30 % after aortic surgery [3–6] and 4–23 % in a mixed surgical intensive care unit (ICU) population [7, 8]. If untreated, the mortality associated with ACS is high; 80 % without decompression [7], compared to 36–49 % with decompression [9, 10]. Standard treatment for ACS is decompressive laparotomy (DL) and subsequent open abdomen treatment (OAT) to maintain an acceptable IAP [9]. Clinical and experimental studies on ACS have reported that time from start of ACS to surgical decompression is crucial to re-establish organ perfusion and reverse organ failure(s), but the impact of time to OAT on survival remains controversial [9–13].
When the skin and fascia are not closed after laparotomy, the open abdomen (OA) requires a temporary abdominal closure (TAC) [1]. Several methods are described [14–23], but only two small randomized controlled studies have compared TAC methods on outcomes for patients with OA. One study compared negative pressure wound therapy (NPWT) and the use of a polyglactin-910 mesh and observed no difference in the closure rates (NPWT 31 %, polyglactin-910 mesh 26 %) [24]. The other randomized study compared NPWT and retention sutures with NPWT only and achieved closure rates of 93 and 40 %, respectively, p = 0.005 [25]. Finally, observational prospective and retrospective studies have demonstrated delayed primary fascial closure rates of approximately 90 % with a combination of NPWT and mesh-mediated fascial traction [26–29].
The development of enteric leak and entero-atmospheric fistula (EAF) in the OA is challenging for the patient and the team taking care of the patient, and a multidisciplinary approach is necessary. Several different treatment strategies have been described in the literature [30]; however, the mortality has been reported to be high when this feared complication accompanies OAT [24, 28, 31, 32].
The primary aim of this study was to evaluate in-hospital survival and factors associated with mortality for patients treated with open abdomen. Secondary aims were to assess primary closure rate, length of stay (LOS) in hospital and ICU, time to abdominal closure, and major complications.
Patients and methods
Consecutive patients treated with OA at St. Olavs Hospital, Trondheim University Hospital, between October 2006 and June 2014, were identified in two prospective databases, the hospital’s patient administrative system and the Norwegian vascular registry (NORKAR). Data were gathered from patients’ records and the surgical procedure registry. Information about mechanical ventilator support, renal replacement therapy (RRT), organ failure and Simplified Acute Physiology Score II (SAPS lI) [33] were collected from the ICU-registry. SAPS II uses all observations, the first 24 h of the ICU stay—the worst physiological value during this time is used. For patients who die or who are transferred during the first 24 h, we have used the values observed at admission to the ICU to estimate SAPS II [33]. SOFA scores were registered once daily applying the worst value during the previous 24 h and assess circulatory, respiratory, renal, liver, coagulation and central nervous system function. Each organ function is scored from 0 (normal) to 4 (severe organ dysfunction); scores of 3 and 4 are defined as organ dysfunction/failure [34].
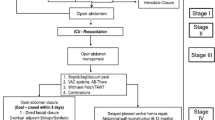
IAP was measured with the intra-vesical technique [35, 36] using the Foley Manometer LV monitoring kit (Holtech Medical, Charlottenlund, Denmark) [37]. At the start of the study period, mechanically ventilated and sedated surgical patients in the ICU had their IAP measured as decided for each patient. From 2012 and onwards, all surgical patients in the ICU had their IAP measured at least every 4 h. If ACS was diagnosed, surgical decompression was performed. For some patients, OAT was decided at the end of the primary surgery to prevent ACS. In patients with OAT, V.A.C. ®therapy (KCI, San Antonio, Texas, USA) with a continuous negative pressure between 25 and 125 mmHg was applied. For patients who needed more than one dressing change, the routine care was vacuum-assisted wound closure and mesh-mediated fascial traction (VAWCM) (Fig. 1) [23, 28, 29]. Despite these recommendations, some patients were treated with NPWT without mesh-mediated fascial traction, i.e., only by vacuum-assisted wound closure (VAWC). After the index operation for OA, the dressing was changed every 48–72 h in the operating room (OR) or in the ICU [38].
Patients who developed an entero-atmospheric fistula (EAF) were treated either with negative pressure therapy mediated through a chimney made by V.A.C. WhiteFoam™ (KCI, San Antonio, Texas, USA) over the opening in the intestine, preventing intestinal fluid contamination of the abdominal cavity [39], by deviation of intestinal fluid through an ostomy bag over the fistula opening and NPWT around the fistula [40], or with a Nipple VAC [30].
At the end of the OAT, the fascia was closed with interrupted sutures using polyglactin-910 or in a running fashion with polydioxanone suture. If tension-free closure was impossible due to loss of domain, reconstruction with mesh was performed with Permacol™ (Medtronic) in a sub-lay position or in an intraperitoneal position [41]. If mesh was not used, the fascia was closed with a free fascia lata transplant, or the intestine was just covered with skin flaps.
Classification of the OAs was done according to the amended Björck classification [1, 42, 43]. The study was approved by the Regional Ethics Committee in Mid-Norway (2014/957). Preliminary observations for OAT in nine patients are previously reported [29].
Statistics
Data were analyzed in Excel, Windows 2010 and IBM SPSS software, version 21. Continuous data are presented as median with range. Between groups, comparisons of continuous variables were made either with Student’s t test or Mann–Whitney test. Categorical variables were compared using Pearson’s Chi-square test or Fischer’s exact test, where appropriate. The statistical significance level was set to p < 0.05, two-tailed. Clinical variables included in univariate analyses for survival were age, SAPS II, sex, ACS, RRT and presence of cardiovascular disease. The variables associated with survival with a p value <0.05 in univariate analyses were included in a multivariable logistic regression analysis and were expressed as odds ratios with 95 % confidence intervals (CI). The variables age, ACS, RRT, SAPS II and preexisting cardiovascular disease were entered as a forward stepwise regression in the multivariate analysis, and variables with p < 0.05 were kept in the final model, excluding SAPS II and preexisting cardiovascular disease.
Results
Patient characteristics
The cohort consisted of all 118 patients identified with OAT between October 2006 and June 2014. Eighty-two (69 %) patients were men, median age was 63 (20–88) years, and 53 (44 %) patients had ACS (Table 1). Abdominal aortic aneurysm was the most common diagnosis (38 %), and aortic repair and bowel resections were the most common surgical procedures prior to or at the index operation for OAT (Tables 2, 3).
OA-grading
Sixty-four (54 %) patients were classified as grade 1A open abdomen at index operation, 33 (28 %) as 1B, 5 (4 %) as 2A, 12 (10 %) as 2B, 1 (1 %) as 2C, 1 (1 %) as 4 and 2 (2 %) patients were not possible to classify.
ACS
In the 53 patients with ACS, 51 had IAP measured with median peak value of 25 (12–40) mmHg. Types of organ failure present in the ACS patients were renal (n = 13), respiratory (n = 12) and cardiovascular failure (n = 2), combined respiratory and renal failure (n = 25) and combined respiratory and cardiovascular failure (n = 1). Forty-three of the patients had ACS following surgery including 21 after aortic repair, six due to sepsis and four due to pancreatitis. The median time from previous surgery to decompression was 29 (1–307) hours: in survivors 24.5 h (4–191) and in non-survivors 35 h (1–306) (p = 0.457). For patients where ACS was not diagnosed, 29 patients were left open prophylactically at the end of the primary surgery; for the remaining 36 patients, OA was performed at a median of three (0–45) days after previous surgery due to the need of second look (n = 10), symptoms of abdominal sepsis (n = 12), necrotizing fasciitis (n = 7), packing of intra-abdominal bleeding (n = 4) and full-thickness wound dehiscence (n = 3). Ninety-two patients were treated with VAWCM, 26 with VAWC (Fig. 2).
Abdominal closure
Primary fascial closure was achieved in 76 (84 %) of the patients surviving the OA treatment, nine patients had an abdominal wall repair with Permacol™ mesh, four with fascia/skin flaps, and two patients were left with a giant hernia (Fig. 3).
OA therapy
The median number of TAC procedures before closure of the OA or death was five (1–32). Eight patients had their OA closed at the next admission to the OR. The 27 patients who died before closure had a median of four (1–15) dressing changes. Patients with primary fascial closure could be closed after 11.5 (1–88) days, and patients in need of a reconstruction or who ended with a planned incisional hernia were closed after 32 (11–143) days. Sixty-three percentages of the ordinary dressing changes were performed in the OR and 37 % at the ICU.
Hospital stay and ICU treatment
LOS in the hospital was 29 (1–246) days. ICU treatment was required for 113 patients for a median of 15 (1–89) days. Admission to a high-dependency unit (HDU), usually a step down from the ICU, was necessary for 62 patients for a median of six (1–36) days. Median SAPS II for the ICU-treated patients was 43 (14–83). Median SAPS II for patients surviving the hospital stay was 39 (14–83) compared to 53 (19–70) in non-survivors (p < 0.001). Hundred and twelve patients received mechanical ventilation support with a median duration of 13 (0–62) days. Twenty-seven patients (21 %) were in need of RRT, of which 22 were in the ACS group. The median number of days with RRT was 11 (1–40). In addition, acute renal failure without need for RRT was present in 16 patients. In two patients, renal failure became permanent; both had undergone repair for a ruptured suprarenal aortic aneurysm.
Survival
Twenty-seven (23 %) of the patients died before closure of the OA, 10 (9 %) died after closure and 81 (68 %) patients survived the hospital stay. Non-survivors had a median of three (2–5) organ failures and died 13 (0–80) days after established OA (Fig. 4). No deaths were related to the OAT per se, but due to the underlying disease and subsequent organ failure(s). Out of the 53 patients with OA due to ACS, 27 (51 %) survived, compared to 53 (83 %) of those with OA for other reasons (p < 0.001). ACS patients required more mechanically ventilator support, had a higher SAPS II and stayed longer in the ICU than those without ACS (Table 4). In a univariate analysis of predictors for mortality, ACS, increasing age, SAPS II, RRT during OAT, and preexisting cardiovascular disease increased the risk of death. In a multivariable logistic regression analysis, RRT, ACS and age were statistical significant predictors for in-hospital mortality (Table 5).
Bleeding
Nine patients underwent additional surgical procedures due to bleeding. Eight patients presented with bleeding before OAT: two after aortic repair, two due to spontaneous retroperitoneal bleeding from the iliopsoas musculature, two patients with terminal liver failure suffered from diffuse intra-abdominal bleeding, and one patient had secondary bleeding due to damage to the iliac vein during surgery for ovarian cancer. Four patients died due to untreatable hemorrhage despite active surgical intervention. Only one patient was presented with a bleeding after initiating of OAT; she was successfully treated with packing and OAT.
Entero-atmospheric fistulas
The nine patients who developed an EAF had all undergone gastrointestinal surgery. All patients treated for fistula survived the hospital stay. Five patients were treated with Chimney-VAC, three patients were treated with deviation of intestinal fluid through an ostomy bag over the fistula opening and NPWT around the fistula, and one with a Nipple VAC. Patients with fistula were younger than those without, 52 (23–64) vs. 64 (20–88) years (p = 0.019). Days with OA for patients with an entero-atmospheric fistula were 29 (5–88) compared to 11 (1–143) for those without (p = 0.022), and hospital LOS was 102 (20–215) and 28 (1–246) days, respectively (p = 0.001). Patients with an EAF needed a median of 14 (7–32) dressing changes before the abdomen was closed and an entero-cutaneous fistula was established (Table 6).
Discussion
The present study confirms that treating patients with OA is resource-demanding with multiple reoperations and prolonged stay in the ICU and the hospital, and that NPWT is a feasible way of TAC with few serious adverse effects. Two-thirds of the patients treated with OA survived, and acute renal failure with dialysis, ACS and advanced age were predictors of mortality. All patients who developed an EAF survived their hospital stay.
Most patients had their OA closed with primary fascial suture, while a minority was in need of more advanced reconstructions of the abdominal wall. Moreover, it was the same closure rate for the few patients without mesh traction (82 %) as for those with mesh traction (84 %), compared to 59 versus 93 % closure rate in a Finnish study [27]. This difference is probably explained by the fact that in the Finnish study, two different time-periods were compared, before and after introduction of the mesh-mediated traction.
In-hospital survival was 68 %, which is similar to results from multicenter trials: one from Sweden reporting 70 % [28] survival, another from the UK reporting 72 % survival within mixed surgical cohorts [32], and a trial from the USA reporting 50 to 72 % survival in a cohort with mainly trauma patients [28, 32, 44]. In the present study, the reason for OAT was ACS in 44 % of the cases, compared to 20 % in the Swedish study [28] and only 2 % in the study from the UK [32]. In a study by Vidal and colleagues in a mixed ICU population, the overall survival was 57 %, but only 20 % in those with non-surgical treated ACS [7]. In a review by De Waele et al. [9], the survival after decompression of ACS was 51 %, similar to the present results, somewhat higher than the prospective study by De Waele et al. [10] which reported 28-day mortality of 36 %. For patients treated with OA for other reasons than ACS, the survival was 83 % in the present study; this is in line with other publications [26, 45, 46], but the survival in an open abdomen cohort will always be influenced according to the included patients.
The frequency of eight percent EAF in the present study is in line with other publications on OA patients [24, 26–29, 47]. Interestingly, all patients with EAF survived in the current study. Previous studies report mortality rates among patients who develop fistulas during OAT from 24 to 88 % [24, 28, 32]. Interestingly, the (in-hospital) mortality was 100 % in patients with EAF in the study from Bjarnason et al. [31] where the fistulas were re-classified according to the amended classification of OA [43] into enteric leak and EAF, [31] similar to the classification used in the present study. In the present study, EAF was only present after gastrointestinal tract surgery in conjunction with OAT, and the patients were younger and less likely to have ACS as the reason for OA. This could have contributed to the high survival in the EAF patients. Another explanation could be the technique with vacuum suction placed directly over the fistula used in five of the patients. When placing the connector direct over the chimney, we experienced that the sealing between the intestine and the VAC become better, preventing leakage of intestinal content into the OA. Thus, keeping the abdominal cavity free from intestinal content, and likely reducing the inflammatory response. We also used fistula VAC in this cohort, but found it more difficult to achieve sealing between the intestine with the fistula and the stoma-bag compared to the chimney with VAC method. Yet another consideration is when placing the connector in distance from the fistula, you might get drainage away from the fistula toward the connector. This could potentially lead to more intestinal content in the OA if the sealing around the fistula is incompetent. If not a spontaneous closure of the EAF is achieved, the goal in EAF treatment in OA is to create a permanent entero-cutaneous fistula when the OA is gradually closing around the fistula. Others have reported spontaneous closure of the EAF during NPWT [48, 49]; this was not seen among our patients.
This study has limitations. Firstly, some of the patients were identified retrospectively, but in order to minimize the possibility of missing cases, several prospective databases were browsed. Secondly, the data for each patient were obtained from registries and patients records retrospectively with the risk of misclassification. Thirdly, although this cohort represents OA patients at a tertiary referral center during an 8-year period, the number of cases is low and type II statistical error is likely, and the high number of patients with AAA might also be a limitation in generalizing the results to cohorts with mostly trauma patients.
Nevertheless, based on the present results, treating patients with OA is resource-demanding with multiple reoperations and prolonged stay in the ICU and hospital. NPWT seems to be a feasible way of TAC with few serious adverse effects. Most patients had their OA closed with primary fascial suture. Two-thirds survived the open abdomen. Presence of EAF was not associated with poorer outcome as all patients with an EAF survived.
References
Kirkpatrick AW, Roberts DJ, De Waele J et al (2013) Intra-abdominal hypertension and the abdominal compartment syndrome: updated consensus definitions and clinical practice guidelines from the World Society of the Abdominal Compartment Syndrome. Intensive Care Med 39(1190–120):6
Hong JJ, Cohn SM, Perez JM et al (2002) Prospective study of the incidence and outcome of intra-abdominal hypertension and the abdominal compartment syndrome. Br J Surg 89(591–59):6
Djavani Gidlund K, Wanhainen A, Björck M (2011) Intra-abdominal hypertension and abdominal compartment syndrome after endovascular repair of ruptured abdominal aortic aneurysm. Eur J Vasc Endovasc Surg 41:742–747
Mayer D, Rancic Z, Meier C et al (2009) Open abdomen treatment following endovascular repair of ruptured abdominal aortic aneurysms. J Vasc Surg 50:1–7
Björck M, Petersson U, Bjarnason T et al (2011) Intra-abdominal hypertension and abdominal compartment syndrome in nontrauma surgical patients. Am Surg 77(Suppl 1):S62–66
Sörelius K, Wanhainen A, Acosta S et al (2013) Open abdomen treatment after aortic aneurysm repair with vacuum-assisted wound closure and mesh-mediated fascial traction. Eur J Vasc Endovasc Surg 45(588–59):4
Vidal MG, Ruiz Weisser J, Gonzalez F et al (2008) Incidence and clinical effects of intra-abdominal hypertension in critically ill patients. Crit Care Med 36:1823–1831
Malbrain ML, Chiumello D, Pelosi P et al (2005) Incidence and prognosis of intraabdominal hypertension in a mixed population of critically ill patients: a multiple-center epidemiological study. Crit Care Med 33(315–32):2
De Waele JJ, Hoste EA, Malbrain ML (2006) Decompressive laparotomy for abdominal compartment syndrome–a critical analysis. Crit Care 10:R51
De Waele JJ, Kimball E, Malbrain M, et al. (2016) Decompressive laparotomy for abdominal compartment syndrome. Br J Surg
Cothren CC, Moore EE, Johnson JL et al (2007) Outcomes in surgical versus medical patients with the secondary abdominal compartment syndrome. Am J Surg 194(804–80):7 (discussion 807-808)
Mentula P, Hienonen P, Kemppainen E et al (2010) Surgical decompression for abdominal compartment syndrome in severe acute pancreatitis. Arch Surg 145(764–76):9
Skoog P, Hörer TM, Nilsson KF et al (2014) Abdominal hypertension and decompression: the effect on peritoneal metabolism in an experimental porcine study. Eur J Vasc Endovasc Surg 47:402–410
Cothren CC, Moore EE, Johnson JL et al (2006) One hundred percent fascial approximation with sequential abdominal closure of the open abdomen. Am J Surg 192(238–24):2
Perez D, Wildi S, Demartines N et al (2007) Prospective evaluation of vacuum-assisted closure in abdominal compartment syndrome and severe abdominal sepsis. J Am Coll Surg 205(586–59):2
Koss W, Ho HC, Yu M et al (2009) Preventing loss of domain: a management strategy for closure of the “open abdomen” during the initial hospitalization. J Surg Educ 66(89–9):5
Wittmann DH, Aprahamian C, Bergstein JM (1990) Etappenlavage: advanced diffuse peritonitis managed by planned multiple laparotomies utilizing zippers, slide fastener, and Velcro analogue for temporary abdominal closure. World J Surg 14:218–226. doi:10.1007/BF01664876
Schein M, Saadia R, Jamieson JR et al (1986) The ‘sandwich technique’ in the management of the open abdomen. Br J Surg 73:369–370
Fernandez L, Norwood S, Roettger R et al (1996) Temporary intravenous bag silo closure in severe abdominal trauma. J Trauma 40:258–260
Aprahamian C, Wittmann DH, Bergstein JM et al (1990) Temporary abdominal closure (TAC) for planned relaparotomy (etappenlavage) in trauma. J Trauma 30:719–723
Brock WB, Barker DE, Burns RP (1995) Temporary closure of open abdominal wounds: the vacuum pack. Am Surg 61(30–3):5
Hougaard HT, Ellebaek M, Holst UT et al (2014) The open abdomen: temporary closure with a modified negative pressure therapy technique. Int Wound J 11(Suppl 1):13–16
Petersson U, Acosta S, Björck M (2007) Vacuum-assisted wound closure and mesh-mediated fascial traction–a novel technique for late closure of the open abdomen. World J Surg 31:2133–2137. 10.1007/s00268-007-9222-0
Bee TK, Croce MA, Magnotti LJ et al (2008) Temporary abdominal closure techniques: a prospective randomized trial comparing polyglactin 910 mesh and vacuum-assisted closure. J Trauma 65:337–342 (discussion 342-334)
Pliakos I, Papavramidis TS, Mihalopoulos N et al (2010) Vacuum-assisted closure in severe abdominal sepsis with or without retention sutured sequential fascial closure: a clinical trial. Surgery 148(947–95):3
Atema JJ, Gans SL, Boermeester MA (2014) Systematic review and meta-analysis of the open abdomen and temporary abdominal closure techniques in non-trauma patients. World J Surg. doi:10.1007/s00268-014-2883-6
Rasilainen SK, Mentula PJ, Leppäniemi AK (2012) Vacuum and mesh-mediated fascial traction for primary closure of the open abdomen in critically ill surgical patients. Br J Surg 99(1725–173):2
Acosta S, Bjarnason T, Petersson U et al (2011) Multicentre prospective study of fascial closure rate after open abdomen with vacuum and mesh-mediated fascial traction. Br J Surg 98(735–74):3
Seternes A, Myhre HO, Dahl T (2010) Early results after treatment of open abdomen after aortic surgery with mesh traction and vacuum-assisted wound closure. Eur J Vasc Endovasc Surg 40(60–6):4
Di Saverio S, Tarasconi A, Walczak DA et al (2016) Classification, prevention and management of entero-atmospheric fistula: a state-of-the-art review. Langenbecks Arch Surg 401:1–13
Bjarnason T, Montgomery A, Acosta S et al (2014) Evaluation of the open abdomen classification system: a validity and reliability analysis. World J Surg 38(3112–312):4. doi:10.1007/s00268-014-2716-7
Carlson GL, Patrick H, Amin AI et al (2013) Management of the open abdomen: a national study of clinical outcome and safety of negative pressure wound therapy. Ann Surg 257(1154–115):9
Le Gall JR, Lemeshow S, Saulnier F (1993) A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA 270:2957–2963
Vincent JL, Moreno R, Takala J et al (1996) The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. Intensive Care Med 22:707–710
Kron IL, Harman PK, Nolan SP (1984) The measurement of intra-abdominal pressure as a criterion for abdominal re-exploration. Ann Surg 199:28–30
Malbrain ML, Cheatham ML, Kirkpatrick A et al (2006) Results from the international conference of experts on intra-abdominal hypertension and abdominal compartment syndrome. I. Definitions Intensive Care Med 32:1722–1732
Desie N, Willems A, De Laet I et al (2012) Intra-abdominal pressure measurement using the FoleyManometer does not increase the risk for urinary tract infection in critically ill patients. Ann Intensive Care 2(Suppl 1):S10
Seternes A, Fasting S, Klepstad P et al (2016) Bedside dressing changes for open abdomen in the intensive care unit is safe and time and staff efficient. Crit Care 20:164
Rekstad LC, Wasmuth HH, Ystgaard B et al (2013) Topical negative-pressure therapy for small bowel leakage in a frozen abdomen: a technical report. J Trauma Acute Care Surg 75(487–49):1
Goverman J, Yelon JA, Platz JJ et al (2006) The “Fistula VAC,” a technique for management of enterocutaneous fistulae arising within the open abdomen: report of 5 cases. J Trauma 60:428–431 (discussion 431)
Sanders DL, Kingsnorth AN (2012) The modern management of incisional hernias. BMJ 344:e2843
Björck M, Bruhin A, Cheatham M et al (2009) Classification–important step to improve management of patients with an open abdomen. World J Surg 33:1154–1157. doi:10.1007/s00268-009-9996-3
Björck M, Kirkpatrick AW, Cheatham M et al (2016) Amended classification of the open abdomen. Scand J Surg 105:5–10
Cheatham ML, Safcsak K (2010) Is the evolving management of intra-abdominal hypertension and abdominal compartment syndrome improving survival? Crit Care Med 38:402–407
Barker DE, Green JM, Maxwell RA et al (2007) Experience with vacuum-pack temporary abdominal wound closure in 258 trauma and general and vascular surgical patients. J Am Coll Surg 204:784–792 (discussion 792-783)
Cheatham ML, Demetriades D, Fabian TC et al (2013) Prospective study examining clinical outcomes associated with a negative pressure wound therapy system and Barker’s vacuum packing technique. World J Surg 37:2018–2030. doi:10.1007/s00268-013-2080-z
Dubose JJ, Scalea TM, Holcomb JB et al (2013) Open abdominal management after damage-control laparotomy for trauma: a prospective observational American Association for the Surgery of Trauma multicenter study. J Trauma Acute Care Surg 74:113–120 (discussion 1120-1122)
Bobkiewicz A, Walczak D, Smoliński S, et al. (2016) Management of enteroatmospheric fistula with negative pressure wound therapy in open abdomen treatment: a multicentre observational study. Int Wound J
D’Hondt M, Devriendt D, Van Rooy F et al (2011) Treatment of small-bowel fistulae in the open abdomen with topical negative-pressure therapy. Am J Surg 202(e20–2):4
Funding
Main investigator received unrestricted grants from St. Olavs Hospital.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Arne Seternes, Martin Björck, Lars Cato Rekstad, Skule Mo, Pål Klepstad, Dag L. Halvorsen, Torbjørn Dahl and Arne Wibe declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Seternes, A., Rekstad, L.C., Mo, S. et al. Open Abdomen Treated with Negative Pressure Wound Therapy: Indications, Management and Survival. World J Surg 41, 152–161 (2017). https://doi.org/10.1007/s00268-016-3694-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00268-016-3694-8