Abstract
Background
Various types of dressing materials are available for wound care following hip and knee arthroplasty. However, it is unclear if one material is more beneficial than the others in terms of wound complications and fluid handling capacity.
Research questions
We performed a meta-analysis of randomized controlled trials comparing alternative wound dressing materials for the post-operative management of wounds following THA and TKA with respect to (1) incidence of wound complications including infection and (2) fluid handling capacity.
Methods
Randomized controlled trials comparing alternative dressing materials, for post-operative management of wounds following TKA and THA were included in the review. Databases searched included the MEDLINE and the EMBASE from inception to February 2017. Two authors performed study selection, risk of bias assessment and data extraction. Where levels of clinical and statistical heterogeneity permitted, data were pooled for meta-analysis.
Results
Twelve randomized trials with data for the primary outcome were identified. Data were available for meta-analysis for two comparisons. Wounds managed with film dressings (odds ratio, 0.35; 95% confidence interval [CI], 0.21–0.57) or with hydrofiber dressings (odds ratio, 0.28; 95% confidence interval [CI], 0.20–0.40) were significantly less likely to have wound complications than those managed with passive dressings. There was no evidence that any dressing significantly reduced surgical-site infection rates compared with any other dressing. Hydrofibre dressings showed better fluid handling capacity than passive dressings in terms of mean number of dressing changes (mean difference 1.36; 95% confidence interval [CI], 0.15–2.57) and number of patients requiring early dressing change (odds ratio, 8.60; 95% confidence interval [CI], 4.68–15.83).
Conclusion
The evidence available in the current literature suggests that advanced dressings such as film and Hydrofibre dressings have fewer wound complications and better fluid handling capacity. However, insufficient evidence is available to determine whether the use of these advanced dressings reduce periprosthetic joint infection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Arthroplasty wounds are different from other surgical wounds in several aspects, and these unique characteristics should be considered when selecting dressing materials after surgery. First, hip and knee arthroplasty wounds can be highly exuding, with persistent dressing leakage [1,2,3]. An ideal dressing should therefore be able to handle excess exudate, while also maintaining a barrier to prevent bacterial entry. Second, as lower limb arthroplasty is often performed in the elderly with fragile skin, there is a higher chance of wound complications such as blistering and skin injury [4, 5]. Thus, the dressing should ideally be associated with atraumatic dressing changes or allow for wound inspection without requiring removal [6]. Third, as these wounds are located over joints, dressings should allow for freedom of movement and should be able to accommodate changes in wound dimensions accompanied with flexion, especially in the knee [7, 8]. Fourth, as there is an underlying prosthesis, any wound complication impairing skin integrity (such as blistering) should be avoided for prevention of periprosthetic joint infection (PJI) [9].
A large number of wound dressing products are currently available and this creates much confusion in classifying dressing materials. We found it useful to classify dressing materials into three categories based on their interaction with the wound environment: passive, active, and interactive dressings (Table 1). While passive dressings serve a protective function, active dressings actually promote healing through the creation of a moist wound environment. Interactive dressings not only create a moist wound environment but also interact with the wound bed components to further enhance wound healing. An overview of the classification and properties of the various dressing materials and their photographs is provided in the Electronic supplementary material 1. In recent years, various new dressing materials have been developed, all with claimed advantages. Unfortunately, little useful information on the effects of these new dressing materials on wound healing, infection prevention, and fluid handling capacity is currently available to help surgeons decide the most optimal dressing material following THA and TKA.
Therefore, we performed a systematic review and meta-analysis of trials comparing alternative wound dressing materials for the post-operative management of wounds following THA and TKA with respect to (1) incidence of wound complications including surgical site infection and (2) fluid handling capacity.
Methods
Study selection criteria and literature searches/data sources and searches
We searched the PubMed and the EMBASE databases in February 2017. No restrictions were placed on the search engines. The following search terms were used for the literature search: (dressing or post-operative dressing or wound dressing) and (knee arthroplasty or hip arthroplasty or knee replacement or hip replacement or lower limb arthroplasty). The bibliographies of all retrieved and relevant publications identified by these strategies were searched for further studies. We did not contact manufacturers regarding studies for inclusion. All randomized controlled trials, comparing two or more types of dressing materials following THA or TKA, and providing information on any of the following parameters: fluid handling capacity (wear time or frequency of dressing change), wound complications, and SSI were included for review. Participants were required to have dressings applied in the operating theatre, immediately after closure of the skin. Articles which studied negative pressure wound therapy (NPWT) were excluded from the review, thereby concentrating only on dressing materials. Case control studies, case reports, letter to editors, case series and articles including procedures other than THA and TKA were also excluded. In case of doubt whether to include a particular article in the review, the final decision was made by the senior authors.
Types of outcome measures
The primary outcome measures were composite wound complications (including blisters, erythaema, maceration, leakage etc.) and SSI. Occurrence of post-operative SSI was defined by the CDC criteria [10], or the authors’ definition of SSI. The secondary outcome measure was: fluid handling capacity. Fluid handling capacity was expressed as either wear time (in days), or mean number of dressing changes (number) or percentage of patients which required early dressing change (%).
Data extraction
Two reviewers extracted information independently with use of a pre-designed data extraction form from all eligible randomized controlled trials. Any differences in opinion were resolved by discussion. The following data were extracted: first author, year of publication, country where the study was performed, study type, sample size, duration of follow up, and the primary and secondary outcomes.
Assessment of risk of bias in included studies
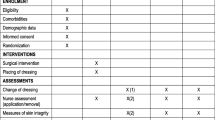
Two review authors independently assessed each included study. Assessment was undertaken using the Cochrane Collaboration tool for assessing risk of bias [11]. The risk of bias tool considers six domains: sequence generation, allocation concealment, blinding, incomplete outcome data, freedom from selective reporting, and other issues (i.e., serious baseline imbalance). A risk of bias table was completed for each eligible study; these data were combined into a risk of bias summary figure where judgments for each domain are tabulated by study.
Statistical analysis
Details of included studies were combined in narrative review according to dressing type. Both clinical and statistical heterogeneity were explored. Where appropriate, data were pooled using meta-analysis (conducted using RevMan 5). Statistical heterogeneity was assessed using the chi-squared test (a significance level of P value less than 0.05 was considered to indicate heterogeneity), and the I2 test. The I2 test examines the percentage of total variation across studies due to heterogeneity rather than to chance. Values of I2 over 50% may represent substantial heterogeneity. In the absence of clinical heterogeneity, and in the presence of statistical heterogeneity (I2 over 50%), a random-effects model was used. Where there was no clinical or statistical heterogeneity, a fixed-effect model was applied.
Results
A systematic search generated 312 abstracts from MEDLINE. A similar protocol was undertaken in EMBASE revealing 35 abstracts. A total of 12 RCTs [3, 5, 6, 12,13,14,15,16,17,18,19,20] met the inclusion criteria (Fig. 1); their study characteristics are shown in Table 2. See Online supplementary material 2 for the list of excluded studies and reasons for exclusion. One of the 12 trials was a three-armed trial [3] while another was a four-armed trial [14]. The three-armed trial was treated as a two-armed trial by combining the results from the two arms that randomized two different brands of film dressing.
Risk of bias assessment
The methodological quality of the eligible trials was limited, as shown in Fig. 2. Even though all studies were randomized controlled trials, concealment of allocation was unclear in six studies, no study was double blind, and outcome assessors were blinded in only one study. Attrition bias was low in all studies as the duration of follow up was less.
Effects of interventions
All 12 trials investigated whether the choice of dressing material affects wound complication rate following THA and TKA. The wound complications assessed varied between studies. While all authors reported on blistering, others also evaluated strike-through across the dressing, erythaema, maceration, skin injury, and dehiscence as wound complications (Table 3). Fluid handling capacity was expressed as either wear time (in days), mean number of dressing changes (number) or percentage of patients which required early dressing change (%).
Comparisons between different passive dressings
There were three trials [12, 13, 15], which compared two different passive dressings in 399 participants (Table 2). Because the three trials involved different types of dressing materials, data from these studies could not be pooled.
Paraffin tulle dressing versus a non-adherent dressing (primary dressings)
One trial (50 participants) compared a paraffin tulle dressing with a non-adherent dressing (25 in each arm) following hip surgery [13]. A statistically significant difference (P = 0.003) between the two dressing groups was observed with the non-adherent group developing blisters in 17 (64%) patients and the Paraffin tulle group in only two (8%) of the patients.
There was no statistically significant difference in the number of SSIs in the non-adherent dressing group (3/25; 12%) compared with the paraffin tulle dressing group (0/25; 0%) (relative risk (RR) = 0.14; 95% CI 0.01 to 2.63). Fluid handling capacity was not assessed in this trial.
Nonstretchable silk tape versus perforated, stretchable cloth tape (secondary dressings)
One trial (99 participants) compared a silk tape versus a cloth tape after hip surgery to determine whether the type of tape influences the rate of blister formation [12]. The incidence of blister formation was 41% (20 of 49 patients) when the nonstretchable silk tape was used and 10% (5 of 50 patients) when the perforated cloth tape was used (RR = 4.08, 95% CI 1.53 to 10.87, p = 0.005). SSI was not evaluated in this trial. Fluid handling capacity was not compared between the two groups.
Perforated, stretchable cloth tape versus elastic Spica bandage (secondary dressings)
One trial (99 participants) compared a cloth tape versus an elastic Spica bandage after hip surgery to determine whether the Spica bandage could reduce the rate of blister formation [15].
The incidence of blister formation was 10% (15 of 150 hips) when the cloth tape was used and 4.7% (7 of 150 hips) when the elastic Spica bandage was used (RR = 2.14, 95% CI 0.90 to 5.11, p = 0.09). SSI was not evaluated in this trial.
Rates of dressing changes done before day three were not significantly different between the two dressing types (8% for spica bandage group versus 10% for cloth tape group; P = 0.55).
Passive dressings compared with film dressings
Two trials involving 356 participants [3, 14] compared passive dressings to film dressings following THA and TKA (Table 2).
Primary outcome: wound complications and superficial SSI
Wounds managed with film dressings were significantly less likely to develop wound complications than those managed with passive dressings (odds ratio (OR) = 0.35; 95% CI, 0.21–0.57) (Fig. 3). These wound complications included blisters, persistent ooze and redness. Pooled data showed there was no significant difference in the number of SSIs between film-dressed groups and passive-dressed groups (OR = 0.90; 95% CI 0.29–2.75) (see figure in Online supplementary material 3).
Secondary outcome: fluid handling capacity
One trial (300 participants) comparing passive dressings to film dressings reported that the wear time was not different between the two groups (2–3 days for both groups) [3]. Another trial (56 participants) comparing passive dressings to film dressings reported fewer mean number of dressing changes in the film dressing group as compared to the passive dressing group (0.3 vs. 1.9, p < 0.001) [14]. As different variables were used to report on the fluid handling capacity, pooling of data was not possible.
Passive dressings compared with hydrofibre dressings
Seven trials [5, 6, 14, 16,17,18, 20] involving 870 participants compared passive dressings to hydrofibre dressings following THA and TKA (Table 2).
Primary outcome: wound complications and superficial SSI
All seven trials which compared passive dressings to a hydrofibre dressing, reported on wound complications. Wounds managed with hydrofibre dressings were significantly less likely to have wound complications than those managed with passive dressings (OR = 0.28; 95% CI 0.20–0.40), Fig. 4. Data was pooled separately for blister (OR = 0.24, 95% CI, 0.13, 0.44) and erythaema (OR = 0.29, 95% CI 0.13, 0.67), both of which were less likely to develop in wounds managed with hydrofibre dressings (see figures in Online supplementary material 4). Of the seven trials investigating the effect of a passive dressing compared with a hydrofibre dressing, only four reported SSI data. None of the patients in either group had SSI in any of these studies.
Secondary outcome: fluid handling capacity
Hydrofibre dressings showed better fluid handling capacity than passive dressings in terms of mean number of dressing changes (mean difference 1.36; 95% CI, 0.15–2.57) and number of patients requiring early dressing change (OR = 8.60; 95% CI, 4.68–15.83) (Fig. 5).
Forest plot showing number of dressing changes (a) as well as requirement for early dressing change (b) comparing passive dressing versus hydrofibre dressing. *The standard deviation for the study by Dobbelaere et al. [14] has been imputed by calculating the mean standard deviation of the other three studies in this analysis
Passive dressings compared with foam based dressings
One trial [14] compared passive dressings to foam based dressings following THA and TKA as part of a four armed trial (Table 2).
Primary outcome: wound complications and superficial SSI
Wounds managed with foam dressings were significantly less likely to develop wound complications than those managed with passive dressings. These wound complications included irritation/redness. There was no significant difference in the number of SSIs between the two groups.
Secondary outcome: fluid handling capacity
There were fewer mean number of dressing changes in the foam dressing group as compared to the passive dressing group (0.3 versus 1.9, p < 0.0001).
Comparison between active dressings
Two trials [14, 19] involving a total of 280 participants compared active dressing materials following THA and TKA (Table 3).
Alginate versus hydrofibre dressings
Primary outcome: wound complications and superficial SSI
One trial (200 participants) compared alginate with hydrofibre dressings following hip replacement surgeries [19]. Wound complications assessed included erythaema, blisters, and skin damage. In the alginate group, there were fewer blisters in the wound area compared with the hydrofibre group (7% versus 18%, p = 0.03). The incidence of erythaema and skin injury did not differ between the two groups. SSI was not evaluated in this trial.
Secondary outcome: fluid handling capacity
Compared to an alginate dressing (Tegagen, 3 M), the hydrofiber dressing (Aquacel, Convatac) was found to have a better wear time; 6.1 days (± 2.8) in the alginate group versus 7.2 days (±3.2) in the hydrofibre group (p = 0.01).
Film dressing versus hydrofibre dressing versus foam dressing
Primary outcome: wound complications and superficial SSI
One trial (included three arms, in a four-arm trial; 80 participants) compared three active dressings with regard to occurrence and frequency of wound complications and mean number of dressing changes [14]. Five wound complications were reported: (1) blistering, (2) stripping, (3) maceration, (4) redness/irritation, and (5) infection. Wound complications were not statistically different between the three active dressings, although Mepilex Border®, foam based silicone dressing had the least wound complications. No SSI was reported in either arm.
Secondary outcome: fluid handling capacity
The mean number of dressing changes was similar for the three active dressings.
Discussion
The primary aim of this systematic review was to present and appraise all existing evidence regarding the relative effectiveness of various surgical dressings on the development of wound complications and SSIs in surgical wounds following THA and TKA. Patients requiring early surgical treatment for wound-healing problems after primary arthroplasty are at significantly increased risk for further complications, including deep infection [21]. The importance of obtaining primary wound healing after THA and TKA therefore cannot be over-emphasized. One of the interventions that may improve wound healing and reduce wound complications is the use of proper wound dressing materials. To our knowledge, there have been no published meta-analyses to date comparing various dressing materials following THA and TKA, although three review articles have been published [22,23,24]. In our meta-analysis, we found lower incidence of wound complications with use of active dressings such as film and hydrofiber dressings over gauze based dressings. The rate of SSI was however not different.
Limitations
This meta-analysis has several limitations. Various dressing materials have been compared to each other, often from different manufacturers, resulting in clinical heterogeneity. A number of trials also had low numbers of participants and low event rates. Their results require cautious interpretation as this low statistical power leaves the equivalent findings at risk of a type II error, where a real difference in a dressing’s effectiveness may exist but has not been demonstrated. Several other variables of importance such as patient comfort and cosmesis, pain during dressing change, and cost effectiveness have been sought by few trials and therefore not included in this meta-analysis.
Our meta-analysis showed that, compared with a passive gauze based dressing, occlusive active dressings such as films and hydrofibre dressings were associated with a significant decrease in overall wound complications. Specifically, the rate of blisters and erythaema were less. Wound complications such as dehiscence, necrosis, and prolonged wound drainage were assessed by only a few authors and the number of these events was very small. Only one author investigated haematoma and found that the incidence of haematoma was more with a gauze dressing than with a hydrofibre dressing following THA [20]. In theory, breakdown of the skin due to blisters or skin injury could lead to an entry portal for wound infection [18]. However, our meta-analysis did not find an increased incidence of SSI with gauze based dressings as compared to the active dressings. The reason for this may be that the numbers of cases of SSI reported were very few. However, given the fact that there is an underlying prosthesis, any wound complication impairing skin integrity (such as blistering) should be avoided for prevention of periprosthetic joint infection (PJI). Apart from SSI, blisters result in morbidity, require additional treatment and are associated with delayed discharge from the hospital. One author reported that on average, the length of hospital stay for patients who developed blisters was eight days compared with seven days for patients who did not develop blisters [16]. It is believed that wound blisters are caused by friction between the dressing and skin surface, leading to separation of the epidermis from the dermis. The skin stretches approximately 20% during knee flexion, and that dressings with greater extensibility are more able to accommodate changes in wound length [25]. It suggested that lower dressing extensibility may explain the increased rates of wound blistering seen with traditional gauze-based dressings.
An optimal dressing should be able to handle excess exudate (fluid handling capacity), while also maintaining a barrier to prevent bacterial entry. Frequent dressing changes are a potential risk factor for SSI as exogenous bacteria may contaminate the wound during the procedure. A study by Clarke and colleagues demonstrated higher skin colonization rates for patients who had dressings changed on POD 1 than for patients who had their first dressing change on POD 6 [26]. In addition, the rate of miotic cell division and leucocyte activity, which is necessary for wound healing and bacterial defence, is disrupted every time the dressing is changed and it takes three to four hours for this biological activity to resume. Furthermore, nursing time is taken up for the dressing changes [27]. We found that hydrofibre dressings showed better fluid handling capacity than passive dressings in terms of mean number of dressing changes and number of patients requiring early dressing change. This may be because the hydrofiber dressing is able to lock-in the fluid in the fibres, which then swell up and is therefore able to handle the excess exudate better.
Apart from wound complications and fluid handling capacity, other variables such as patient satisfaction, length of hospital stay and cost effectiveness are important in choosing the dressing material following THA and TKA. However, these variables were not included in this meta-analysis because patient satisfaction is not measured using rigorous tools in most studies that report on it, length of stay is expected to be influenced by many factors apart from dressing, and robust cost-effectiveness data has not been undertaken by most studies.
Future direction
While this uncertainty regarding optimal dressings for surgical wounds following THA and TKA remains, any investment in future research must maximize its value to clinicians. Given the large number of dressing options, future trials should focus on evaluating dressings that surgical teams use most often. Future studies should use standardized outcomes measures to report subjective and objective results. Means and standard deviations should be reported for all continuous variables. It may be worthwhile to include raw data, perhaps in the form of an appendix. This would allow appropriate pooling of data for meta-analysis.
Conclusion
Various types of dressing materials are available for wound care following hip and knee arthroplasty. The evidence available in the current literature suggests that advanced dressings such as film and hydrofiber dressings have fewer wound complications and better fluid handling capacity. However, insufficient evidence is available to determine whether the use of these advanced dressings reduce periprosthetic joint infection. Furthermore, few studies have compared the different advanced dressings to each other following THA and TKA. Well-designed future studies are warranted to establish optimal wound dressing materials following total joint arthroplasty.
References
Sweeney IR, Miraftab M, Collyer G (2012) A critical review of modern and emerging absorbent dressings used to treat exuding wounds. Int Wound J 9(6):601–612. doi:10.1111/j.1742-481X.2011.00923.x
Hopper GP, Deakin AH, Crane EO, Clarke JV (2012) Enhancing patient recovery following lower limb arthroplasty with a modern wound dressing: a prospective, comparative audit. J Wound Care 21(4):200–203. doi:10.12968/jowc.2012.21.4.200
Cosker T, Elsayed S, Gupta S, Mendonca AD, Tayton KJ (2005) Choice of dressing has a major impact on blistering and healing outcomes in orthopaedic patients. J Wound Care 14(1):27–29. doi:10.12968/jowc.2005.14.1.26722
Bredow J, Oppermann J, Hoffmann K, Hellmich M, Wenk B, Simons M, Eysel P, Zarghooni K (2015) Clinical trial to evaluate the performance of a flexible self-adherent absorbent dressing coated with a soft silicone layer compared to a standard wound dressing after orthopedic or spinal surgery: study protocol for a randomized controlled trial. Trials 16:81. doi:10.1186/s13063-015-0599-z
Ravenscroft MJ, Harker J, Buch KA (2006) A prospective, randomised, controlled trial comparing wound dressings used in hip and knee surgery: Aquacel and Tegaderm versus Cutiplast. Ann R Coll Surg Engl 88(1):18–22. doi:10.1308/003588406x82989
Langlois J, Zaoui A, Ozil C, Courpied JP, Anract P, Hamadouche M (2015) Randomized controlled trial of conventional versus modern surgical dressings following primary total hip and knee replacement. Int Orthop 39(7):1315–1319. doi:10.1007/s00264-015-2726-6
Wong KL, Peter L, Liang S, Shah S, Johandi F, Wang W (2015) Changes in dimensions of total knee arthroplasty anterior knee dressings during flexion: preliminary findings. Int J Orthop Trauma Nurs 19(4):179–183. doi:10.1016/j.ijotn.2015.06.002
Dillon JM, Clarke JV, Deakin AH (2007) Correlation of total knee replacement surgery wound dynamic morphology and dressing material properties. J Biomech 40(Suppl. 2):S61
Gupta SK, Lee S, Moseley LG (2002) Postoperative wound blistering: is there a link with dressing usage? J Wound Care 11(7):271–273. doi:10.12968/jowc.2002.11.7.26413
Horan TC, Andrus M, Dudeck MA (2008) CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control 36(5):309–332. doi:10.1016/j.ajic.2008.03.002
Higgins JP, Altman DG, Gotzsche PC, Juni P, Moher D, Oxman AD, Savovic J, Schulz KF, Weeks L, Sterne JA (2011) The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 343:d5928. doi:10.1136/bmj.d5928
Koval KJ, Egol KA, Polatsch DB, Baskies MA, Homman JP, Hiebert RN (2003) Tape blisters following hip surgery. A prospective, randomized study of two types of tape. J Bone Joint Surg Am 85-a(10):1884–1887
Lawrentschuk N, Falkenberg MP, Pirpiris M (2002) Wound blisters post hip surgery: a prospective trial comparing dressings. ANZ J Surg 72(10):716–719
Dobbelaere A, Schuermans N, Smet S, Van Der Straeten C, Victor J (2015) Comparative study of innovative postoperative wound dressings after total knee arthroplasty. Acta Orthop Belg 81(3):454–461
Koval KJ, Egol KA, Hiebert R, Spratt KF (2007) Tape blisters after hip surgery: can they be eliminated completely? Am J Orthop (Belle Mead, NJ) 36(5):261–265
Abuzakuk TM, Coward P, Shenava Y, Kumar VS, Skinner JA (2006) The management of wounds following primary lower limb arthroplasty: a prospective, randomised study comparing hydrofibre and central pad dressings. Int Wound J 3(2):133–137
Burke NG, Green C, McHugh G, McGolderick N, Kilcoyne C, Kenny P (2012) A prospective randomised study comparing the jubilee dressing method to a standard adhesive dressing for total hip and knee replacements. J Tissue Viability 21(3):84–87. doi:10.1016/j.jtv.2012.04.002
Springer BD, Beaver WB, Griffin WL, Mason JB, Odum SM (2015) Role of surgical dressings in total joint arthroplasty: a randomized controlled trial. Am J Orthop (Belle Mead, NJ) 44(9):415–420
Ravnskog FA, Espehaug B, Indrekvam K (2011) Randomised clinical trial comparing Hydrofiber and alginate dressings post-hip replacement. J Wound Care 20(3):136–142. doi:10.12968/jowc.2011.20.3.136
Harle S, Korhonen A, Kettunen JA, Seitsalo S (2005) A randomised clinical trial of two different wound dressing materials for hip replacement patients. J Orthop Nurs 9(4):205–210. doi:10.1016/j.joon.2005.09.003
Galat DD, McGovern SC, Larson DR, Harrington JR, Hanssen AD, Clarke HD (2009) Surgical treatment of early wound complications following primary total knee arthroplasty. J Bone Joint Surg Am 91(1):48–54. doi:10.2106/JBJS.G.01371
Collins A (2011) Does the postoperative dressing regime affect wound healing after hip or knee arthroplasty? J Wound Care 20(1):11–16. doi:10.12968/jowc.2011.20.1.11
Tustanowski J (2009) Effect of dressing choice on outcomes after hip and knee arthroplasty: a literature review. J Wound Care 18(11):449–450, 452, 454, passim. doi:10.12968/jowc.2009.18.11.44985
Chowdhry M, Chen AF (2015) Wound dressings for primary and revision total joint arthroplasty. Ann Transl Med 3(18):268. doi:10.3978/j.issn.2305-5839.2015.09.25
Dillon JM, Clarke JV, Deakin AH, Nicol AC, Kinninmonth AWG (2007) Correlation of total knee replacement surgery wound dynamic morphology and dressing material properties. J Biomech 40:S570. doi:10.1016/S0021-9290(07)70560-2
Clarke JV, Deakin AH, Dillon JM, Emmerson S, Kinninmonth AW (2009) A prospective clinical audit of a new dressing design for lower limb arthroplasty wounds. J Wound Care 18(1):5–8, 10-11. doi:10.12968/jowc.2009.18.1.32128
Kloeters O, Schierle C, Tandara A, Mustoe TA (2008) The use of a semiocclusive dressing reduces epidermal inflammatory cytokine expression and mitigates dermal proliferation and inflammation in a rat incisional model. Wound Repair Regen 16(4):568–575. doi:10.1111/j.1524-475X.2008.00404.x
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest statement
Gaurav Sharma: No conflict of interest with this work.
Sang Wook Lee: No conflict of interest with this work.
Oliver Atanacio: No conflict of interest with this work.
T.K. Kim: Consultant for: Smith & Nephew, B.Braun-Aesculap.
J. Parvizi: Board membership Journal of Arthroplasty, Journal of Bone and Joint Surgery, Bone and Joint Journal, Philadelphia Orthopaedic Society, Eastern Orthopaedic Association, CD Diagnostics, Muller Foundation, 3 M; Consultant for Zimmer, Smith & Nephew, ConvaTec, TissueGene, CeramTec, Medtronic; Royalties from Elsevier, Wolters Kluwer, Slack, Datatrace, Jaypee Publishers; Stock options - Intellectual properties: Hip Innovation Technologies, CD Diagnostics, PRN.
This work was performed at the Seoul National University Bundang Hospital.
Rights and permissions
About this article
Cite this article
Sharma, G., Lee, S.W., Atanacio, O. et al. In search of the optimal wound dressing material following total hip and knee arthroplasty: a systematic review and meta-analysis. International Orthopaedics (SICOT) 41, 1295–1305 (2017). https://doi.org/10.1007/s00264-017-3484-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-017-3484-4