Abstract
Background
Lipoblastoma is a rare benign neoplasm of immature fat cells in children. Imaging appearances are frequently complex, sometimes simulating liposarcoma.
Objective
To characterize features of lipoblastoma on MRI and CT in comparison with recurrence risk.
Materials and methods
We identified cases via retrospective review of histopathology-proven lipoblastoma cases in a large referral database and a pediatric medical center. Two radiologists scored CT and MRI on the basis of lesion features.
Results
We included a total of 56 children (32 boys and 24 girls) with a mean age of 2.6 years (range 0.1–13 years). Extremity lesions were most common (27%), followed by neck (19%), gluteal region (18%), chest (14%) and mesentery (14%). Children most commonly presented with painless masses (73%), followed by dyspnea (9%), distension (9%) and pain (7%). Non-adipose soft-tissue components were identified on CT and MRI in 78% of cases. Significant (moderate or marked) septations were noted in 59% and enhancement in 35%. Compartmental invasion was present in 43% of cases. Of paraspinal cases, 38% involved the neural foramina or central canal. Lesion complexity did not significantly correlate with age. Recurrence was observed in 9% of cases and was significantly correlated with compartmental invasion (correlation: 0.303, P=0.009) and septation complexity (correlation: 0.227, P=0.038) on initial imaging.
Conclusion
Although lipoblastoma is a fat-containing entity, many lesions demonstrate marked complexity and local infiltration that resemble liposarcoma, which is exceedingly rare in younger children. Compartmental invasion and thicker septations appear to confer greater risk of recurrence following resection.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Lipoblastoma is a rare benign neoplasm of embryonal fat cells that usually occurs within the first 3 years of age [1,2,3]. It appears that these tumors result from the clonal expansion of mesenchymal pre-adipocytes from alterations in the PLAG1 gene on Chromosome 8 causing transcriptional upregulation that promotes lipoblast proliferation [4]. Lipoblastoma most commonly occurs at sites with the largest amount of immature fat in the neonate, with cases reported in the subcutaneous tissues of the extremities as well as the cervical, paravertebral, mediastinal and peritoneal regions [1]. When lipoblastoma exhibits a more diffuse appearance, it is often referred to as lipoblastomatosis, although there is no pathological distinction [1]. Because lipoblastoma is a benign soft-tissue tumor, some have proposed renaming it as infantile lipoma to reflect its benign behavior without metastatic potential and ability to mature into lipoma [5]. Although benign, these lesions can grow and cause significant mass effect. Conventional management of lipoblastoma (or lipoblastomatosis) generally consists of non-mutilating wide local excision wherever possible [6, 7].
Lipoblastoma contains predominantly immature fat cells with thin septations; as such, imaging features generally mirror those of fat with lucency on radiographs, hyperechogenicity on US, hypodensity on CT, and T1-weighted hyperintense signal and hypointense fat-suppressed signal on MRI [8, 9]. The presence of complex components and atypical features in some lipoblastomas has raised concern for liposarcoma despite the rarity of this diagnosis in children, especially during the first decade of life [10,11,12]. Lack of appropriate imaging or inadequate understanding of imaging features can lead to inaccurate preoperative diagnoses, as observed in one study in which less than 20% of lipoblastoma cases were correctly diagnosed preoperatively [6].
Leveraging a large database of pathology-validated cases and cases at a quaternary academic pediatric medical center, we provide a comprehensive assessment of advanced imaging features and clinical behavior of this rare tumor. We posit that complex features are observed in many cases and that complex imaging features might be more prevalent in younger patients.
Materials and methods
Case identification
We identified cases of lipoblastoma from retrospective review with two sources: (1) the referral case database of the American Institute for Radiologic Pathology (n=49) and (2) a medical record search from a quaternary pediatric medical center, the Children’s Hospital of Philadelphia (n=7), including cases reported between 2011 and 2019. We reviewed all available imaging studies and relevant clinical information and anonymized them in accordance with our institutional review board protocol. The review board approved the study, and the study complied with the Health Insurance Portability and Accountability Act. We also reviewed pathology reports, including testing for PLAG1 mutations when it was performed. Inclusion criteria consisted of histopathological confirmation of lipoblastoma or lipoblastomatosis, availability of cross-sectional imaging consisting of CT or MRI prior to resection, and documentation of clinical presentation. Cases were excluded if pathology was discordant or inconclusive for lipoblastoma, if CT or MRI was not performed prior to resection, or if insufficient diagnostic images were available for review. Imaging protocols were heterogeneous because of the referral nature of the included database. At least T1-weighted non-fat-suppressed and either T2-weighted or short tau inversion recovery (STIR) acquisitions were required for MRI inclusion in this study.
Imaging review
All available imaging was reviewed in consensus by a musculoskeletal radiologist (M.D.M., 34 years of experience) and a board-certified pediatric radiologist (A.J.D., 2 years of experience). In the event of disagreement, an additional musculoskeletal radiologist (J.S.J., 25 years of experience) served as the tiebreaker. CT and MR images were scored on the basis of septation complexity (compared against unaffected subcutaneous fat); presence of non-adipose components (areas with hyperdensity on CT, T1-weighted hypointense signal or T2-weighted hyperintense signal on MRI); and enhancement (subjective magnitude relative to subcutaneous fat). Scoring for each imaging feature was conducted on a four-point scale — none, mild (1–33%), moderate (34–66%) and marked (67–100%). Compartmental invasion was defined as presence of tumor beyond the primary anatomical space of lesion origin and scored as: mild = transfascial spread, muscle infiltration; moderate = bone remodeling, bowel encasement; and marked = neurovascular encasement, intraspinal extension. In the event of both CT and MRI being performed for an individual case, scoring was based on MRI. When available, US images were reviewed for general features but study availability and images were insufficient for formal scoring.
Statistical analysis
We provide descriptive statistics for clinical characteristics and advanced imaging features. Continuous variables are summarized using the mean ± standard deviation (SD). Categorical variables are presented as counts or percentages. We performed bivariate non-parametric Kendall’s tau correlation analyses to assess associations of imaging features with age as well as clinical and imaging features with recurrence. Statistical significance was defined at the 0.05 level. All analyses conducted in this study were performed using SPSS Statistics for Macintosh, version 23.0 (IBM, Armonk, NY).
Results
Patient characteristics
Fifty-six children were included in this study. Case characteristics are detailed in Table 1. Children with lipoblastoma had a mean age of 2.6 years at initial diagnosis with 82% being 3 years or younger. A male predilection of 1.3:1.0 was observed in this series. Most commonly, lipoblastoma presented as an asymptomatic mass (73%), with the extremities as the most commonly involved site followed by the neck, gluteal region, chest and abdomen (Table 1). Deep location (intramuscular, intermuscular or intracavitary) was approximately three times more common (77%) than subcutaneous location (23%). All of the cases in this series tested for PLAG1 (n=10/10, 100%) were positive for mutations.
Advanced imaging features
Magnetic resonance imaging was available for 71% of patients and CT for 45% of patients. US was additionally used in a smaller proportion of cases (29%) and was of insufficient quality to evaluate in detail. Cross-sectional imaging consisting of CT and MRI demonstrated fat components to some extent in all lesions (100%) in this study.
A summary of the combined imaging features from CT and MRI for all cases is provided in Table 2. Septations were moderate or markedly complex in 59% of cases (Fig. 1). While some cases showed predominantly adipose appearance (Fig. 2), non-adipose components were present to some extent in 79% of cases. Non-lipomatous appearance generally corresponded predominantly to myxoid areas on histopathology (Fig. 3). Significant (moderate or marked) enhancement was observed in 35% of cases. Compartmental invasion was noted to some extent in 43% of cases, with moderate to marked compartmental invasion in 22% of cases.
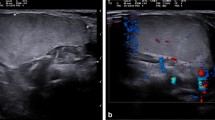
Lipoblastoma with septations in a boy age 1 year 7 months with growing thigh mass and restricted flexion. a Sagittal post-contrast T1-weighted fat-saturated MR image of the knee shows a complex fat-containing mass in the deep popliteal fossa with moderate enhancement of the marked septations within this lesion. (In an adult, these imaging features would suggest the diagnosis of a well-differentiated or myxoid liposarcoma.) b Gray-scale US image shows a predominantly hyperechoic lesion with interspersed hypoechoic septations (arrowheads). c Histopathology (hematoxylin and eosin stain, 40x magnification) shows predominantly fat-containing mass in keeping with lipoblastoma with septations (arrowheads) and myxoid areas (asterisks)
Lipoblastoma in a 2-year-old girl with palpable gluteal mass. a Axial T1-weighted MR image delineates a predominantly fat-containing lesion within the intermuscular gluteal region with moderate septations. (In an adult, this appearance might suggest a well-differentiated liposarcoma.) b Gross pathology photograph shows a primarily adipose lesion, with histopathology confirming lipoblastoma
Lipoblastoma in a 3-year-old girl with 1-year history of painless increasing abdominal girth. a Coronal abdominal CT image emphasizes fat (arrow) and non-adipose components (asterisk) of a large abdominal mass as well as substantial mass effect of this lesion with pelvicaliectasis of the right kidney (arrowhead) from compression of the right ureter. (In an adult, this imaging appearance would raise concern for myxoid liposarcoma.) b Photomicrograph of complex area with hematoxylin and eosin stain (10x) confirms dominant myxoid component of this lipoblastoma
Specific findings of compartmental invasion included transfascial involvement and muscle infiltration (n=14, 25%, Fig. 4); neurovascular encasement (n=4, 7%, Fig. 5); intraspinal extension (n=3, 5%, Fig. 6); mediastinal and tracheal displacement (n=1, 2%, Fig. 6); rib remodeling (n=1, 2%); and bowel encasement (n=1, 2%), which required partial bowel resection. Of lesions located within the paraspinal soft tissues near the vertebral column (n=8, Figs. 6 and 7), 38% (n=3) entered the neural foramen or spinal canal (Fig. 6).
Lipoblastoma with diffuse muscle infiltration in a 3-year-old boy with an asymptomatic foot mass. a Initial lateral foot radiograph demonstrates a large deep plantar-based soft-tissue mass (arrows) with lucent areas, mostly proximally (asterisk), suggesting a fat-containing lesion. b Short-axis T2-weighted MRI highlights diffuse muscle infiltration with hyperintense signal interspersed with suppressed fat involving the intrinsic foot muscles including the plantar interossei (arrows). Histopathology following resection confirmed the diagnosis of lipoblastoma, and genetic analysis demonstrated a PLAG1 locus interruption
Lipoblastoma with neurovascular encasement in a 21-month-old boy with abdominal distension. a Axial contrast-enhanced CT of the abdomen and pelvis shows encasement of the superior mesenteric artery and vein (arrow) by a mostly fat-content mass with heterogeneous areas of moderate non-adipose soft-tissue components (asterisks). b Intraoperative photograph during laparotomy demonstrates the mass to be based in the small bowel mesentery and encase the superior mesenteric artery, precluding complete resection. Histopathology showed typical findings of lipoblastoma, including fat cells of various sizes including lipoblasts with intervening myxoid stroma
Lipoblastoma with intraspinal extension in a 3-year-old girl presenting with dyspnea. a Axial contrast-enhanced CT of the neck shows intracanalicular extension (arrow) of this complex fat-containing mass. b Coronal T1-weighted MR image demonstrated leftward mediastinal shift with tracheal deviation (not pictured) and shows transcompartmental extension of a fat-containing lesion through the thoracic inlet (arrow) and intraspinal extension (arrowhead). Pathology confirmed lipoblastoma
Paraspinal lipoblastoma in a 3-year-old boy with mass in the upper back. a Post-contrast sagittal T1-weighted fat-saturated MRI of the upper thoracic spine region demonstrates a large, deep intermuscular paraspinal mass with fat-containing component superiorly, with marked septations and marked enhancement within the thick septations (arrow) but absent enhancement in the cystic-appearing area inferiorly (asterisk). b Gross specimen photograph highlights a fat-rich component superiorly (arrow) consistent with lipoblastoma and a myxoid-appearing well-defined cyst (asterisk)
Ultrasound imaging features
All of the lesions imaged with US in this study were hyperechoic with septations (Fig. 1). US demonstrated relatively limited utility for invasive manifestations of lipoblastoma because deeper components could not be well-assessed due to technical limits of sonographic penetration. We performed no additional analysis of US data because of the limited nature of US images available in this database.
Lesion complexity and age
Complexity of lipoblastoma on imaging was not significantly correlated with age. Specifically, the correlation coefficients of imaging variables with age were 0.081 (P=0.23) for septation complexity, −0.044 (P=0.34) for non-adipose soft-tissue components, −0.037 (P=0.38) for enhancement magnitude and 0.095 (P=0.19) for deep extension.
Recurrence risk
Recurrence was observed in five cases (9%) with an average follow-up of 3.0 years and 7% of cases lost to follow-up. Recurrence occurred at an average of 2.3 years following initial resection (range: 6 months to 4 years). Recurrence risk was positively correlated with magnitude of deep extension, or compartmental invasion (correlation: 0.303, P=0.01) and complexity of septations (correlation: 0.227, P=0.04) on initial imaging but not with other imaging factors, lesion size or patient age (Table 3).
Discussion
This large case series of lipoblastoma included a broad range of clinical presentations and lesion locations, with extremity lesions and deep soft-tissue involvement noted most frequently. In keeping with the literature, most cases in this study occurred in children younger than 3 years (82%); our oldest case was 13 years. We observed a slight male predilection with an overall male-to-female ratio of 1.3:1.0, concordant with the 1.4:1.0 ratio reported in the literature [13].
Despite exhibiting fat components of various extents in all lesions on imaging, lipoblastoma encountered in this series often exhibited complex features including thick septations, non-adipose soft-tissue areas and enhancement on contrast-enhanced imaging. Others have reported that lipoblasts inherently exhibit slightly lower intensity compared with mature fat on T1-weighted imaging [8]; complexity on MRI including marked enhancement, nodular soft-tissue regions and cystic change have also been reported [9, 11, 14]. These complex non-adipose components have also been correlated with the presence of myxoid tissue, cystic degeneration and tumoral infarction [15, 16]. We observed similar findings with lesions that were predominantly T2 hyperintense on fat-suppressed MR imaging, demonstrating mostly myxoid and other non-lipomatous tissue on histopathology. We hypothesized that lesion complexity decreases with age, but this analysis did not find any significant correlation between age and imaging features, implying that lipoblastoma can be equally complex at any age of presentation.
Complex fat-containing lesions in young children should not be confused for malignant entities such as well-differentiated or myxoid liposarcoma, which are exceedingly rare in young children, especially infants [17]. Other nonmalignant entities to be considered include hibernoma and spindle cell lipoma, although both occur in older adults with more restricted anatomical distributions than lipoblastoma [3, 17]. As a general rule, the primary diagnostic consideration of fat-containing soft-tissue masses in children should be lipoblastoma.
While lipoblastoma is a benign tumor, several cases exhibited locally aggressive-appearing behavior in this case series with compartmental invasion. Lipoblastoma can extend through fascial planes and into skeletal muscle (Fig. 4), as seen in 20% of cases in this series and consistent with prior reports [13]. Previous reports have deemed such intraspinal extension rare, although a few cases exist in the lipoblastoma literature [18,19,20,21]. Of the eight cases of deep paraspinal lipoblastoma in this study, three (38%) demonstrated intraspinal extension (Fig. 6). These observations suggest that when a lipoblastoma is paraspinal in location and demonstrates diffuse infiltration, intraspinal involvement should be carefully considered and inspected for on MRI, especially because these deeper components are difficult to resect completely without morbidity and likely portend higher recurrence risk.
Lipoblastoma arising within the neck, chest and abdomen can have significant growth, resulting in clinically significant mass effect. Mediastinal and neck lesions can result in respiratory compromise, and dyspnea was common in children with lipoblastoma in these locations in our study, with tracheal deviation in one case (Fig. 6), as previously reported particularly in infants [22, 23]. We also observed four cases of neurovascular encasement in the neck and superior mediastinum where tumor surrounded major vessels and nerves. A few cases of such neurovascular encasement have been reported in which lipoblastoma surrounded but did not invade vessel walls [23]. One such case in this study precluded complete resection of an intra-abdominal mass because of encasement of the superior mesenteric artery (Fig. 5).
Conflicting reports exist regarding the frequency of recurrence of lipoblastoma following surgical resection, with estimates varying widely from 0% to 33% [1, 6, 9, 13, 24,25,26]. While our data might be less representative because of the heterogeneous availability of follow-up, including 7% without follow-up, we observed a modest but relatively low rate of recurrence at 9%. Recurrence occurred predominantly in lesions in which tumoral extension led to deep components not amenable to complete resection, with deep extension significantly correlated with recurrence; septation complexity was also associated with recurrence, albeit to a lesser extent. Others have observed that focal lesions do not tend to recur, whereas more invasive, diffuse lipoblastoma (often referred to as lipoblastomatosis) is much more likely to require re-excision [24]. These deeper tumors are particularly problematic at surgery because of the absence of discrete lesion features to discern margins intraoperatively [24]. MRI is uniquely suited, with its superior soft-tissue contrast, for delineating total lesion extent prior to resection and is essential in the evaluation of any deep lipoblastoma [8]. US has relatively limited utility for follow-up, aside from well-encapsulated superficial lesions, because it underestimates deep extent and complex components [8, 9]. There is at least one documented case of spontaneous resolution of lipoblastoma on follow-up MRI, with those authors proposing an alternative wait-and-see approach [27]. None of the cases in the current study was imaged serially prior to resection to comment on the possibility of spontaneous involution, and further investigation of the natural history of these rare lesions is merited to better inform clinical management.
This was a retrospective case series of lipoblastoma cases from both a large referral database and a single tertiary pediatric institution, and availability of follow-up data was limited for some cases. Lack of long-term follow-up confines our understanding of the natural history of this benign tumor and might lead to underestimation of recurrence risk, as discussed. As a referral database, surgical reports, when available, were inadequate to evaluate the effect of incomplete initial resection on recurrence risk. In addition, this study potentially overlooked older children with lipoblastoma because pathology reports might diagnose maturing lipoblastoma as lipoma instead. While our study included only cases with histopathology concordant with lipoblastoma, only a minority of lesions were tested for causative PLAG1 mutations, precluding complete assessment of gene mutation analysis compared with imaging findings. In particular, PLAG1 cytogenetic testing might aid in the differentiation of lipoblastoma from myxoid liposarcoma in older children or lesions with more complex appearances on imaging. Nevertheless, all of the cases in this series tested for PLAG1 were positive for mutations, emphasizing both the consistency of imaging diagnosis in lipoblastoma and the utility of PLAG1 genetic analysis in confirming lipoblastoma [4, 28].
Guidance regarding the follow-up of lipoblastoma post-resection is poorly defined, with authors suggesting follow-up 2–10 years after surgery [6, 7, 24]. One of the recurrent cases in this study occurred 4 years after initial resection, supporting longer follow-up duration. The widespread availability of MRI in developed countries has facilitated imaging follow-up for these lesions and should also be considered after the resection of any deep tumor, especially those without well-defined margins [6]. Generally, our findings and contemporary consensus recommendations support the need for non-mutilating complete resection and serial follow-up MRI of children with deep lesions for several years, perhaps up to 10 years [6, 7]. Superficial lipoblastoma with satisfactory excisional margins likely does not require serial imaging.
Conclusion
Moderate complexity is frequent in lipoblastoma on CT and MRI, with septations, non-adipose components, contrast enhancement and compartmental invasion commonly encountered, and these features should not be confused with liposarcoma in young children, especially during the first decade of life. Recurrence risk is significantly associated with compartmental invasion and complex septations of the initial lipoblastoma.
References
Chung EB, Enzinger FM (1973) Benign lipoblastomatosis. An analysis of 35 cases. Cancer 32:482–492
Stringel G, Shandling B, Mancer K et al (1982) Lipoblastoma in infants and children. J Pediatr Surg 17:277–280
Kransdorf MJ (1995) Benign soft-tissue tumors in a large referral population: distribution of specific diagnoses by age, sex, and location. AJR Am J Roentgenol 164:395–402
Choi J, Bouron Dal Soglio D, Fortier A et al (2014) Diagnostic utility of molecular and cytogenetic analysis in lipoblastoma: a study of two cases and review of the literature. Histopathology 64:731–740
O'Donnell KA, Caty MG, Allen JE et al (2000) Lipoblastoma: better termed infantile lipoma? Pediatr Surg Int 16:458–461
McVay MR, Keller JE, Wagner CW et al (2006) Surgical management of lipoblastoma. J Pediatr Surg 41:1067–1071
Seguier-Lipszyc E, Baazov A, Fichman S et al (2018) Current management of lipoblastoma. Eur J Pediatr 177:237–241
Reiseter T, Nordshus T, Borthne A et al (1999) Lipoblastoma: MRI appearances of a rare paediatric soft tissue tumour. Pediatr Radiol 29:542–545
Moholkar S, Sebire NJ, Roebuck DJ (2006) Radiological–pathological correlation in lipoblastoma and lipoblastomatosis. Pediatr Radiol 36:851–856
Cacciaguerra S, Lebet M, Di Cataldo A et al (1995) An unusual intrathoracic tumor: giant lipoblastoma. Eur J Pediatr Surg 5:40–42
Nagano A, Ohno T, Nishimoto Y et al (2011) Lipoblastoma mimicking myxoid liposarcoma: a clinical report and literature review. Tohoku J Exp Med 223:75–78
Huh WW, Yuen C, Munsell M et al (2011) Liposarcoma in children and young adults: a multi-institutional experience. Pediatr Blood Cancer 57:1142–1146
Bourelle S, Viehweger E, Launay F et al (2006) Lipoblastoma and lipoblastomatosis. J Pediatr Orthop B 15:356–361
Chen CW, Chang WC, Lee HS et al (2010) MRI features of lipoblastoma: differentiating from other palpable lipomatous tumor in pediatric patients. Clin Imaging 34:453–457
Ha TV, Kleinman PK, Fraire A et al (1994) MR imaging of benign fatty tumors in children: report of four cases and review of the literature. Skelet Radiol 23:361–367
Schultz E, Rosenblatt R, Mitsudo S et al (1993) Detection of a deep lipoblastoma by MRI and ultrasound. Pediatr Radiol 23:409–410
Murphey MD, Carroll JF, Flemming DJ et al (2004) From the archives of the AFIP: benign musculoskeletal lipomatous lesions. Radiographics 24:1433–1466
Duhaime AC, Chatten J, Schut L et al (1987) Cervical lipoblastomatosis with intraspinal extension and transformation to mature fat in a child. Childs Nerv Syst 3:304–306
Ko SF, Shieh CS, Shih TY et al (1998) Mediastinal lipoblastoma with intraspinal extension: MRI demonstration. Magn Reson Imaging 16:445–448
Choi SW, Song SH (2007) Intrapelvic lipoblastoma with massive spinal canal invasion. Childs Nerv Syst 23:581–585
Gupta G, Garg R, Wadhwa C et al (2018) A rare primary dumbbell lipoblastoma. Asian J Neurosurg 13:83–85
Whyte AM, Powell N (1990) Mediastinal lipoblastoma of infancy. Clin Radiol 42:205–206
Irgau I, McNicholas KW (1998) Mediastinal lipoblastoma involving the left innominate vein and the left phrenic nerve. J Pediatr Surg 33:1540–1542
Dilley AV, Patel DL, Hicks MJ et al (2001) Lipoblastoma: pathophysiology and surgical management. J Pediatr Surg 36:229–231
Hicks J, Dilley A, Patel D et al (2001) Lipoblastoma and lipoblastomatosis in infancy and childhood: histopathologic, ultrastructural, and cytogenetic features. Ultrastruct Pathol 25:321–333
Kok KY, Telisinghe PU (2010) Lipoblastoma: clinical features, treatment, and outcome. World J Surg 34:1517–1522
Mognato G, Cecchetto G, Carli M et al (2000) Is surgical treatment of lipoblastoma always necessary? J Pediatr Surg 35:1511–1513
Gisselsson D, Hibbard MK, Dal Cin P et al (2001) PLAG1 alterations in lipoblastoma: involvement in varied mesenchymal cell types and evidence for alternative oncogenic mechanisms. Am J Pathol 159:955–962
Acknowledgments
The authors gratefully acknowledge the attendees of the American Institute for Radiologic Pathology courses for their contribution to our series of patients and without whom this project would not have been possible.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 17 kb)
Rights and permissions
About this article
Cite this article
Degnan, A.J., Jelinek, J.S. & Murphey, M.D. Lipoblastoma: computed tomographic and magnetic resonance imaging features correlate with tumor behavior and pathology. Pediatr Radiol 51, 614–621 (2021). https://doi.org/10.1007/s00247-020-04882-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00247-020-04882-z